Abstract
Background
Adverse effects of benzene (BZ) exposure toxicities were documented by many researchers worldwide. BZ exposure causes many hematological abnormalities that might be treated with naturally occurring phytocompounds.
Aim
In this experimental study, we evaluated the anti-inflammatory effect of L-carvone and thymoquinone together (LCTQ) against BZ-induced inflammatory toxicities in Sprague-Dawley (SD) rats.
Introduction
BZ exposure could cause an excess formation of immature blood cells to enter the peripheral bloodstream. Phytocompounds namely L-carvone (LC) and thymoquinone (TQ) have confirmed anti-inflammatory effects against various diseases.
Materials and Methods
Rats were divided into four different groups such as control, LCTQ group, BZ pathologic group, and treatment group (LCTQ+BZ). After 10 weeks of the experimental period, body weight changes, hematological parameters, pro-inflammatory cytokines, oxidative stress, RBC antioxidants, bone marrow cellular abnormalities, and bone marrow DNA fragmentation were evaluated.
Results
BZ toxicity showed abnormal loss of body weight, altered hematological parameters, increased pro-inflammatory cytokines, abnormal cellular oxidative status, and DNA damage. LCTQ treatment showed significant (p < 0.05) increase in body weight, normalized hematological parameters such as red blood cells, hemoglobin, white blood cells (lymphocytes and eosinophils), platelets (PLT), and hematocrit with RBC parameters, reduction of pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6), controlled oxidative stress, normalized enzymatic antioxidants in the RBC cells, normalized nucleated cells, megakaryocyte cells, and controlled DNA fragmentation were observed.
Conclusion
The current study showed an anti-inflammatory effect of LCTQ through the control of inflammation against benzene-induced toxicities in rats.
Introduction
Acute myeloid leukemia (AML) is a blood cancer that affects young people (Hwang, 2020). Bone marrow, the soft internal component of your bones, is where it all starts. According to the American Cancer Society, there are going to be roughly 60,650 new instances of leukemia and 24,000 people were dead in the United States in 2022, including a total of 20,050 new cases of AML have been reported (Sung et al., 2021). About 11,540 people died from AML, the majority of whom were adults. Despite this, AML is a relatively uncommon malignancy, accounting for only around 1% of all cancers (De Kouchkovsky & Abdul-Hay, 2016). Most types of AML are treated with chemotherapy, which is occasionally combined with a targeted therapy medicine and followed by a stem cell transplant. Alternative approaches to traditional chemotherapeutic medicine may be utilized to treat people with acute promyelocytic leukemia (Mi et al., 2012). Treatment has certain initial negative effects, including fatigue in cancer patients. Fatigue (tiredness) is the most prevalent side effect of cancer treatment, as well as low resistance to infection, anemia, bruising, and bleeding risks, feeling or being ill, a sore mouth, taste changes, and heart muscle alterations (Narayanan & Koshy, 2009). Therefore, novel therapeutic techniques are needed to treat this type of cancer.
The organic solvent benzene (BZ) is commonly utilized as a forerunner in the manufacture of a wide range of products, including rubbers, dyes, pesticides, and pharmaceuticals (Li et al., 2014). Because it is used as a fuel additive and is found in cigarette smoke nevertheless BZ is also an environmental chemical. Chronic exposure to BZ has been linked to hematotoxicity and a variety of blood disorders, AML, aplastic anemia, and myelodysplastic syndrome (MDS) are only a few examples according to research (He et al., 2020). By deregulating the immune system, BZ may cause bone marrow immunotoxicity or inhibit T lymphocyte activity and cytokine production, which can lead to bone marrow immunotoxicity. A study revealed that employees who were exposed had decreased levels of CD80+ T-cell and CD86+ T-cell expression in their peripheral blood cells to BZ indicating the immunological effects of BZ (Trzupek et al., 2020). BZ exposure also led to immunological suppression, as well as the number of CD3+ and CD4+ T cells decreased significantly, according to clinical and animal experimental trials (Macpherson et al., 2020). These findings imply that BZ-induced toxicity is caused by a reduction in T-cell-mediated immunological activity. Therefore, immunological modulation may be a viable method for reducing hematotoxicity caused by BZ (He et al., 2020). Higher reactive oxygen species (ROS) accumulation causes a deteriorative effect on hematopoietic cells (Valko et al., 2006). Earlier research proved an association between oxidative stress and leukemia (Al-Gayyar et al., 2007). At the moment, there are no viable treatments for protecting BZ-affected workers in the workplace from AML, as well as the present therapy options have short- and long-term negative effects (Crossnohere et al., 2019). However, new therapeutic approaches are needed, as well as the usage of innovative protective agents, to prevent and treat the disease.
The essential oil from the spearmint plant contains 50%−65% L-carvone (LC), which has a minty scent (Mentha spicata) (Snoussi et al., 2015). D-limonene can also be used to make it synthetically. LC has a long history of use in toothpaste and mouthwash, as well as in a range of foods and beverages. It is also utilized in consumer products like air fresheners and as a fragrance in personal care products (Steinemann, 2021). The technical grade active ingredient (TGAI) is Bedoukian L-Carvone, which is indicated for use in the production of a mosquito and biting fly repellent (Hazarika et al., 2022). Thymoquinone (TQ) is the most active component in black seeds’ volatile oil (2-isopropyl-5-methylbenzo-1,4-quinone) (Ahmad et al., 2019). TQ is the primary bioactive ingredient, and it has anti-fungal, anti-cancer, anti-convulsant, antioxidant, anti-bacterial, and anti-inflammatory properties (Ahmad et al., 2013). El-Dakhakhny was the first to extract TQ and it is one of the many active ingredients that have been discovered thus far. Due to its wide variety of therapeutic effects, TQ remains the most important bioactive principle that is isolated from plant sources (Badary et al., 2021). TQ and black seed fixed oil anti-viral efficacy against a murine cytomegalovirus infection model have also been discussed in further detail (Badary et al., 2021).
In this research investigation, we used BZ exposure to successfully induce abnormalities that had been treated with LC with TQ in combination (LCTQ). For determining the protective advantages of LCTQ against BZ-induced animal toxicity, different degrees of hematotoxicities were analyzed and documented to substantiate the knowledge of natural protective drugs and BZ toxicities in vivo.
Materials and Methods
Materials
Merck delivered purified benzene (C6H6) that was 99.7% pure (Germany, NO K8008T383540). Xi’an-Ciyuan Company (China) supplied L-carvone and thymoquinone (LCTQ) (>98% purity by HPLC). Beijing Chemical Works provided all additional compounds with the highest commercial grade or analytical grade quality (Beijing, China).
Experimental Design
Animal experimental protocols adopting ARRIVE guidelines were approved by the institutional animal experiment and care committee (Approval number: 20220603) prior to the start of the research experiments. Further, 24 male Sprague-Dawley (SD) rats, 12 weeks old and weighed 220 ± 10 g were grouped into four groups (six animals in each group) and maintained as the same until the end of the experimental period of 10 weeks. The animals were given a seven-day acclimatization period. The rats were kept at ambient room temperature and given a 12 h light/dark cycle, as well as unlimited access to water and standard rat feed. Also, 0.2 mL of diluted benzene solution (1:10 diluted using a solution of equal volumes of deionized water and 2-propanol) was used for intravenous injections in experimental rats to induce leukemia (AML)-like toxicities (Akanni et al., 2014). In Group 1, control animals were administered normal saline in the tail vein. Group 2 animals received LCTQ (dissolved in 80% ethanol) for 10 weeks. Group 3 received intravenous injections of diluted BZ 0.2 mL for 4 weeks. Group 4 animals received LCTQ and BZ injections as in Groups 2 and 3, respectively, for a specified time as mentioned earlier.
After the completion of the experimental period of 10 weeks, all animals were sacrificed by cervical decapitation under anesthesia for collecting blood and bone marrow samples as published earlier (Percival et al., 2017). Blood samples were processed to collect serum samples and EDTA-added blood for hematological analyses. Bone marrow samples were prepared for cells count and DNA fragmentation analyses.
Morphological Study and Blood Parameters
Body weight, smears of peripheral blood cells, biopsies of the femur, and counts of peripheral blood cells were used to evaluate the efficacy of LCTQ in combination. The body weights of all the rats were measured with an electronic balance. The number of RBC/erythrocytes, WBC/leukocytes, and platelets (PLT) in each animal were counted using an automated hematology analyzer (Mindray BC-2800 Vet, China).
Antioxidant Parameter Analysis
Superoxide dismutase (SOD) and catalase (CAT) antioxidant activities in RBC were evaluated using earlier methods (Liu et al., 2019). The activity of glutathione peroxidase (GPx) in RBC was measured using the method published earlier (Jablonska et al., 2015). Determination of the quantity of GSH in RBC using the earlier published methods (Mehdi et al., 2018). The activity of glutathione-S-transferase (GST) in RBC was determined by an earlier method (Pour et al., 2014) and glutathione reductase (GR) activity in RBC was measured using an earlier method (Sarker & Oba, 2018).
Estimation of Cytokines
Using commercial ELISA kits, the cytokines tumor necrosis factor-alpha (TNF-α) (Catalog # MBS9711597), interleukin-6 (IL-6) (Catalog # MBS269892), and interlekin-1β (IL-1β) (Catalog # MBS825017) were evaluated according to the manufacturers’ protocol and recommendations (MyBioSource Company, CA, USA).
Cellular Oxidant Parameters
A conventional approach was used to measure oxidant parameters such as malondialdehyde (MDA) (Atmaca et al., 2008) and oxidized glutathione (GSSG) (Nur et al., 2011).
Isolation and Determination of Rat Bone Marrow Cells
As previously described, bone marrow cells were isolated from the tibia and femur of the experimental rats (Alhadlaq & Mao, 2004). MGG stain (May Grunwald Giemsa) was used to mark and identify the cells as described in the previous publication (Provan et al., 2009). Cell counts were done microscopically using hematology counting chamber for the determination of bone marrow nucleated cells and bone marrow megakaryocyte cells.
Qualitative DNA Fragmentation Assay
The standard procedure was used to determine DNA fragmentation (Saadat et al., 2015). Bone marrow cells were suspended for 20 min on ice in a 100-µL lysis buffer containing 10 mM Tris-HCl (pH 7.4), 10 mM NaCl, 10 mM EDTA, and 0.2% triton-100, then incubated overnight at 56°C with 50 µg/mL proteinase K, centrifuged at 10,000 rpm for 30 min, and finally precipitated in 0.1 volume of 5 M NaCl. Soluble DNA in the supernatant was precipitated overnight in 600 µL isopropyl alcohol and stored at −20°C. After that, the DNA pellet was washed in 70% ethanol, dissolved in TE buffer, and treated with 100 µg/mL RNAse-A for 1 h at 56°C. A 1.5% agarose gel containing ethidium bromide was electrophoresed at 80 V (Power supply Bio-Rad, Hercules, CA, USA) and photographed with a UV-light transilluminator (Strata gene, La Jolla, CA, USA) to see DNA fragmentation (laddering) to assess the sign of apoptosis in the bone marrow cells.
Statistical Analysis
The data was presented as a mean ± standard deviation (SD). A one-way analysis of variance (ANOVA) test was used to calculate the significance of variations in the collected data. The level of significance was set at p < 0.05 and the same was considered statistically significant.
Results
Body Weight Changes of Rats Exposed to BZ
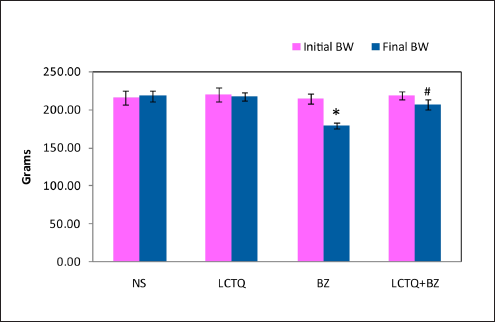
Figure 1 summarizes the body weights of rats from each group. As compared to their initial body weights, no significant differences were observed between groups. Group-1 animals showed an increase in body weight during the experimental period. Although Group-2, Group-3, and Group-4 animals showed alterations in body weight, a significant decrease in the body weight was noticed in Group-3 (BZ). This alteration in the body weight at the end of the experimental period made the BZ group of animals have a significant decrease in the body weight when compared to the control group. Also, after treatment with LCTQ in BZ toxicity, a significant improvement in final body weight was noticed at the level of p < 0.05.
BZ-induced Blood Parameter Changes
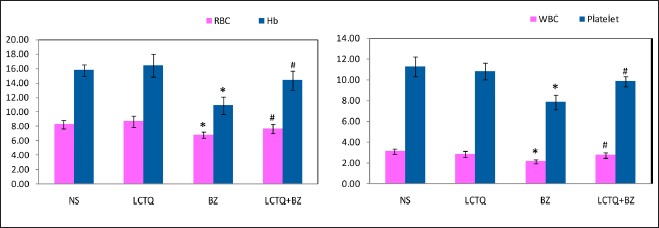
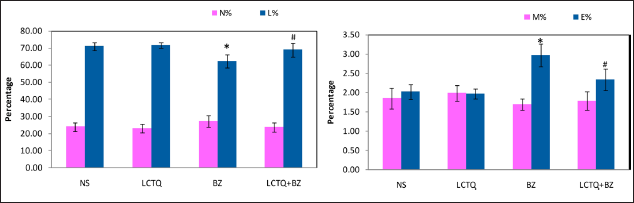
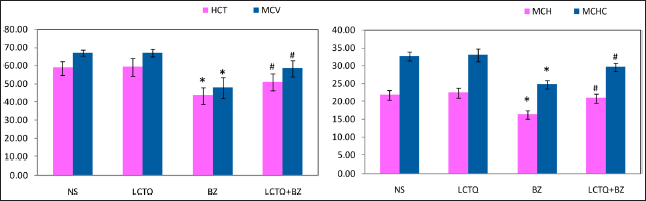
After 10 weeks of treatment with LCTQ from BZ-induced AML, peripheral blood cell counts and hemoglobin levels in the rat from all groups were assessed. Figure 2 showed a substantial reduction in RBC, Hb, WBC, and PLT counts in the BZ group (p < 0.05) when compared to the control group. In contrast, Figure 3 showed no significant alterations in the levels of neutrophils and monocyte percentage. Alternatively, significant alterations (p < 0.05) were noticed in the levels of lymphocytes (decreased) and eosinophils (increased) percentage in BZ toxicity. However, alterations were controlled by the treatment of LCTQ in Group 4, at a significant level, of lymphocytes and eosinophils only. Figure 4 showed a significant decrease in the levels of HCT%, MCV, MCH, and MCHC in BZ toxicity group. Surprisingly, LCTQ treatment controlled these losses and normalized these parameter levels at a significant (p < 0.05) levels.
Pro-inflammatory Cytokine Levels in BZ Toxicity
Figure 5 showed an increase in the inflammatory mediator and pro-inflammatory cytokines, as well as an inflammatory response. The BZ-induced group of rats showed increased levels of cytokines such as TNF-α, IL-6, and IL-1β than the control group of rats. LCTQ treatment significantly (P < 0.05) lowered these cytokines when compared to the BZ toxicity group of rats.
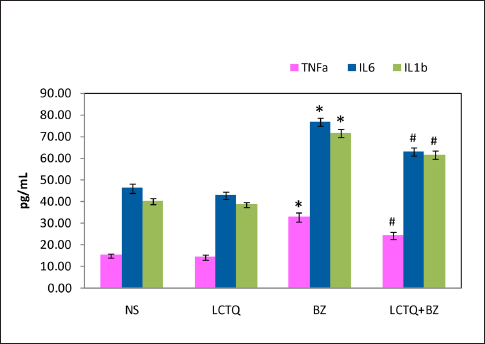
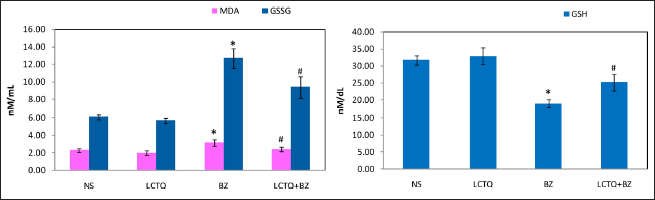
Cellular Oxidant Status in BZ Toxicity
Figure 6A showed the effect of BZ on the oxidant indicators in BZ leukemic rats. At the end of the experimental period, BZ-induced leukemia group had higher levels of MDA and GSSG in the serum. LCTQ treatment controlled these abnormalities and decreased the elevated oxidants in the serum at significant levels (p < 0.05). Similarly, GSH level was decreased BZ toxicity, which was shown in the Figure 6B. In the same way as earlier, LCTQ treatment significantly increased the GSH level in the experimental treatment.
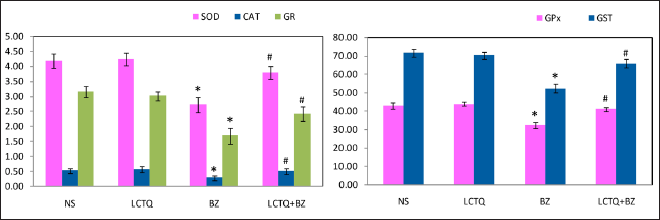
Enzymic Antioxidant Status in RBC
The status of serum RBC enzymatic antioxidants was depicted in Figure 7. Activities of antioxidant enzymes in the cell such as SOD, CAT, GPx, GST, and GR activity were significantly (p < 0.05) lowered in BZ toxicity and were significantly normalized during LCTQ treatment.
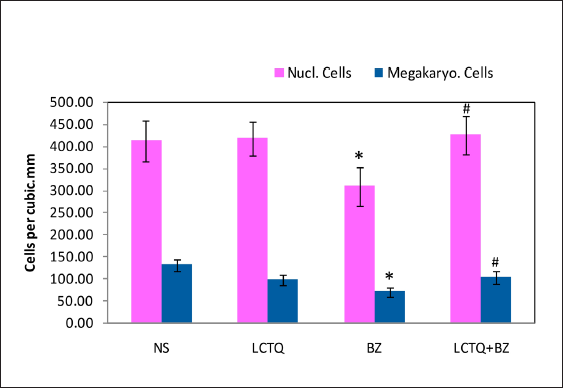
Megakaryocyte Cells Level
Figure 8 showed the levels of bone marrow nucleated cells and megakaryocyte cells in the experimental group of rats. Exposure to BZ significantly lowered the counts of these cells, which were significantly raised during LCTQ treatment (p < 0.05).
DNA Fragmentation Assay
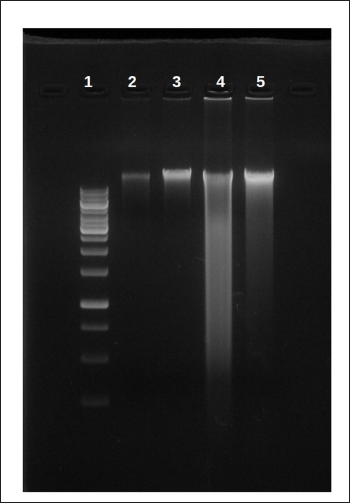
A characteristic of late-stage of apoptosis is DNA fragmentation. The NS group DNA did not show any DNA fragmentation and there was no DNA ladder or smear on the agarose gel in Figure 9. On the other hand, the DNA fragmentation of bone marrow cells in the toxicity group (BZ) demonstrated an appearance of fragmented DNA segments that were smeared and laddered. Treatment of LCTQ showed decreased impact on DNA damage and demonstrated almost no smearing of DNA.
Discussion
Although numerous investigations on leukemogenicity and toxicity caused by benzene have been conducted, the mechanism that drives it has yet to be thoroughly described (Strese et al., 2017). Lymphoblastic leukemia, erythroblastic stem cell leukemia, myelogenous leukemia, and thymic leukemia are all known to be caused by benzene (Snyder, 2012). Although BZ toxicity caused a decreased body weight due to loss or damage to bone marrow cells, recovery was supporting the beneficial effect of the treatment. In humans and animals, blood is an important indicator of pathological and physiological condition, with RBC, WBC, and PL total leukocyte counts being the most commonly measured (Koscielniak et al., 2017). Benzene-induced leukemia is defined by a decrease in erythromyelopoiesis, which leads to a decrease in the number of erythrocytes and leukocytes (particularly lymphocytes) in the peripheral blood (Roland et al., 2014). Earlier studies documented that altered blood parameters in benzene toxicity were modulated in a dose-dependent concentration of phytocompound therapy and significantly recovered the blood parameters (Muhammad et al., 2012). Parallel to the previous studies, decreased hematological parameters were normalized during treatment. As described earlier, decreased lymphocytes percentage was also significantly normalized during treatment with LCTQ. However, an insignificant alteration in the levels of neutrophils and monocyte percentage was controversial in earlier research (Roland et al., 2014).
Inflammatory responses due to toxicities could cause significantly elevated levels of pro-inflammatory cytokines (Li et al., 2018), which was evident in the present study that documented a significant elevation in BZ toxicity and recovery during LCTQ treatment. Furthermore, ROS cause necrosis and cellular harm through a variety of mechanisms, including protein, DNA, and lipid peroxidation (Nita & Grzybowski, 2016). In the present investigation, we discovered that BZ-induced leukemia group rats had higher levels of lipid peroxidation, as measured by elevated MDA and GSSG, indicating an increase in lipid peroxidation. Antioxidant activities were found reduced during increased cellular oxidative stress (Li et al., 2015). We noticed an increased amount of MDA and GSSG in the current experimental investigation and the same were reduced as a result of LCTQ treatment that depicted the interaction in controlling cellular stress and macromolecular peroxidation. This could be important evidence and support the antioxidant role of the treatment compound.
Increased lipid peroxidation and reduced enzymatic antioxidant activities paralleled the oxidative stress found after benzene intoxication (Dumanović et al., 2020). The conditions of oxidative stress, which was coupled with decreased hepatic glutathione level and increased peroxidation were proved earlier (Ahmed et al., 2021). In line with earlier findings, which demonstrated the same, enzymatic antioxidant genes were down-regulated in BZ toxicity. The cell’s functional and structural integrity were harmed by a chain of events triggered by GSH depletion, which increased oxidative stress and the formation of ROS (Guo et al., 2013). CAT and SOD are antioxidative enzymes that work together to give protection against reactive oxygen species (Ighodaro & Akinloye, 2018). Glutathione is available in 90%–95% of healthy human RBCs in the reduced form as GSH (Zitka et al., 2012), which can be used to reduce ascorbate, oxidized proteins, and oxidized lipids (Suh et al., 2003). GST and GPx are the enzymes that use GSH reducing equivalence for reduction reactions. GR is the enzyme responsible for GSH recycling. It consumes NADPH to reduce the GSSG back to reduced GSH. After treatment with LCTQ, the levels of all the antioxidant enzymes in RBC were significantly improved, as evidenced by an earlier report (Senthilkumar et al., 2006).
Previous research has suggested that benzene-induced leukemia triggers apoptosis and caspase responses (He et al., 2020; Guo et al., 2019; McHale et al., 2012). Phytocompounds have been shown to be effective in reducing apoptosis caused by oxidative stress by exploring ROS, improving inhibiting lysosome rupturing, and maintaining redox state by chelating intraliposomal iron. Antioxidant-altering enzymes and GSH have been found to have a major role in the detoxification of ROS and other contaminants either directly by interacting with ROS or by decreasing the disulfide linkage in various proteins, protecting cells from oxidative damage (McHale et al., 2012). However, in the present research, abnormalities in the bone marrow nucleated and megakaryocyte cells have been normalized during treatment, which showed the beneficial effect of LCTQ treatment.
Cellular DNA damage and its repair mechanisms are time-dependent and steady-state processes (Chatterjee & Walker, 2017). BZ-induced DNA degradation was observed by smearing of isolated DNA and LCTQ had a protective effect and was evidenced by decreased intensity of DNA smearing during the electrophoresis. Also, the protective effect of LCTQ could be speculated that chromatin accessibility to DNA repair enzymes, as well as DNA excision repair enzymes, were enhanced, as documented in earlier reports (Luijsterburg & van Attikum, 2011). This evidenced the possible protective role of LCTQ towards bone marrow DNA that could avoid the DNA from damage.
Conclusion
Our findings demonstrated that three weeks of BZ exposure in rats can produce toxicities, presumably related to the BZs production of ROS, resulting in peripheral blood, cellular oxidative stress, and increased oxidative stress in bone marrow cells. Antioxidative characteristics of LCTQ could also contribute to its protection against BZ-induced toxicities. These findings suggested that LCTQ could have therapeutic effects in relation to the treatment of hematological disorders during BZ exposure. Therefore, our findings could help prevent and avoid BZ-induced hematological toxicity in experimental rats. More future research could warrant learning about the role of LCTQ in vivo.
Footnotes
Acknowledgments
All authors of this manuscript thankfully acknowledge for the infrastructure facilities provided by the institutions.
Abbreviations
BZ: Bezene LC: L-Carvone TQ: Thymoquinone; SD rat: Sprague-Dawley rat; RBC: Red blood cells; WBC: White blood cells; PLT: Platelets; DNA: Deoxy ribonucleic acid; TNF-α: Tumor necrosis factor-alpha; IL-1β: Interleukin-1β; IL-6 Interleukin 6; AML: Acute myeloid leukemia; SOD: Superoxide dismutase CAT: Catalase; GST: Glutathione-S-transferase; GPx: Glutathione peroxidase; GR: Glutathione reductase; GSH: Reduced glutathione; GSSG: Oxidized glutathione; MDA: Malondialdehyde; ROS: Reactive oxygen species.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animals used in this study were reviewed and approved by the institutional animal experiment and care committee (Approval number: 20220603).
Funding
This project was supported by the Researchers Supporting Project of King Saud University, Riyadh, Saudi Arabia (RSP2023R230).
Informed Consent
Not applicable.
