Abstract
Background
Ilexsaponin A1 (IA1) is a bioactive triterpene saponin derived from natural medicinal plants. IA1 exhibits anti-inflammatory and proangiogenic activities and improves intestinal barrier function. It has been reported that IA1 could be metabolized into a dominant metabolite, ilexgenin A (IA) by β-glucosidase enzymes in intestinal microflora.
Materials and Methods
Herein, an accurate, sensitive, and selective method based on ultra-performance liquid chromatography coupled with mass spectrometry was established to simultaneously profile the metabolism and pharmacokinetic behaviors of IA1 in normal and antibiotic-treated rat plasma after intragastric administration of IA1. The precursor-to-product ion pairs of IA and IA1 were m/z 501.32↓439.32 and m/z 663.38↓501.32, respectively. For method validation, the specificity, matrix effect, accuracy, precision, and stability of the pharmacokinetic study were measured, and a calibration curve was created. The collaborative pharmacological target pathways of IA1 and its metabolite IA were investigated using network pharmacology tools.
Results
The validated analytical method was successfully utilized to investigate the pharmacokinetics of IA1 in normal and antibiotic-treated rats. The bioavailability of IA1 and conversion from IA1 to IA were significantly inhibited by antibiotic-treated rats after oral administration of IA1. Fragment ions at m/z 483.3155, 455.3159, 439.3233, 421.3136, 395.3362, 152.9952, 113.0256, and 71.0531 were characteristic of the IA1 moiety. IA1 was metabolized in rat plasma by biotransformation routes involving deglycosylation, decarboxylation, isomerization, hydrogenation, dehydrogenation, and oxidation. Considering database analysis, IA and IA1 play synergistic role in common pharmacological pathways, such as hypertrophic cardiomyopathy and dilated cardiomyopathy.
Conclusion
The experiments illustrated that β-glucosidase activity inhibited by antibiotics suppressed the hydrolysis reaction of IA1 in the intestinal tract. IA1 and IA play a synergistic role in exerting effects.
Introduction
Triterpene saponins are widely distributed in natural medicinal plants and exhibit various bioactivities, such as anti-inflammatory, anti-obesity, anti-tumor, and anti-hypertensive activities (Biswas & Dwivedi, 2019). A significant characteristic of triterpene saponins is their restrictive bioavailability in the intestine, which results from their strong polarity. Ilexsaponin A1 (IA1) is an ursane-type and representative saponin in Traditional Chinese Medicine (TCM) with a long history, such as Ilex pubescens and Ilex hainanensis (Cao et al., 2018). Itwas reported that IA1 exhibited potential for preventing cardiovascular or vascular insufficiency-related diseases, reducing inflammation, and improving intestinal barrier function (Zhao et al., 2019). After oral administration, IA1 inevitably contacts the gut microbiota and could be biotransformed in the gastrointestinal tract to increase its fat solubility before entering the bloodstream (Gong et al., 2020). The metabolization of IA1 in intestinal microflora generates multiple metabolites with pertinent pharmacological effects. Studies have shown that IA1 can be metabolized into the dominant metabolite, ilexgenin A (IA) (Zhao et al., 2011). Notably, IA and IA1 are a pair of glycosides and aglycones generally found in natural medicinal plants (Zhao et al., 2018). The conversion process from IA1 to IA was performed by the β-glucosidase enzyme in intestinal microflora (Tannock, 2001). The transformation of IA1 to IA seems to be the pharmacodynamic basis for the effectiveness of IA1. To date, the conversion of IA1 to IA after administration of IA1 has never been calculated.
Antibiotics have been widely and globally applied to confront bacterial infections (Xu et al., 2023). Administration of antibiotics leads to a decrease in the abundance of beneficial symbiotic bacteria and an increase in the abundance of potentially harmful micro-organisms (Zimmermann & Curtis, 2019). Antibiotic administration triggers perturbations in the intestinal microbiota. The biotransformation of triterpene saponin processed by intestinal microflora could be strongly influenced by exposure to antibiotics, which are directly associated with their absorption and biological actions. The pharmacokinetic profile is the key means to monitor the dynamic processes of IA1 and its metabolite IA in vivo after IA1 administration. In early research, IA was quantitatively determined in rat plasma after oral administration or intravenous injection of IA (Li et al., 2012; Liu et al., 2010). However, few studies have demonstrated that abnormal intestinal microflora metabolism affects the pharmacokinetics of IA1 and its metabolite, IA.
Literature reported that the maximum plasma concentration of triterpene saponin was confined to the nanogram level regardless of monomer ingredient or complex administration (Yang et al., 2018). A more sensible strategy was needed to measure the lower absorption of triterpenoid glycosides and aglycones. Multiple studies have confirmed the superiority of ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry (UPLC-QTOF-MS) techniques, which exhibit a higher mass resolution, sensitivity, and accuracy for drug metabolite determination (Hu et al., 2021; Xu et al., 2019). In our paper, the metabolic profile and pharmacokinetic behavior of IA1 and its metabolite IA were comprehensively analyzed, as well as the striking speed, during continuous periods after intragastric administration of monomer IA1; this analysis was based on obtaining accurate masses, elemental compositions, and product ions. The collaborative pharmacological target pathways of IA1 and its metabolite, IA, were investigated using network pharmacology tools. Knowledge of the metabolism, transformation, and pharmacological action of IA1 should provide a reference for in vivo pharmacodynamic mechanisms.
Materials and Methods
Chemicals
IA1 and IA were obtained from I. pubescens with over 98% purity in our phytochemistry laboratory. Digoxin was purchased from Weikeqi Biotechnology Co., Ltd., (Sichuan, China). Acetonitrile and methanol (LC-MS grade) were manufactured by Merck (Darmstadt, Germany). LC-MS grade formic acid was obtained from Fluka (Sigma, USA). Deionized water was prepared by the Millipore system from Shanghai Hitech Instruments Co., Ltd. (Shanghai, China). The antibiotics, including neomycin sulfate and streptomycin, were both from Biotopped Science & Technology Co., Ltd. (Beijing, China). p-Nitrophenol and p-nitrophenyl β-
Animal Experiments
Sprague Dawley rats (250–280 g) were purchased from the laboratory animal center at Guangzhou University of Chinese Medicine (Certificate No. SCXK 2013-0034). After the adaptation period, the animals were separately administered IA1 at low (IA1-L group, 9.96 mg/kg) and high (IA1-H group, 99.6 mg/kg) doses in normal and antibiotic-treated rats. During modeling, the IA1-L and IA1-H groups were used as normal control groups (administered 0.5% CMC-Na orally per day), and antibiotic-treated groups (IA1-L-Anti group and IA1-H-Anti group) were administered neomycin sulfate (100 mg/kg) and streptomycin (100 mg/kg) orally per day for 7 days. Fecal samples were collected, and the body weights of the rats were recorded every day. Food was removed for 12 h before blood collection, but water remained freely available. All groups were intragastrically administered IA1 on the seventh day. Blood samples (200 µL) were collected into heparinized tubes at 0, 0.25, 0.5, 0.75, 1, 1.5, 2, 4, 6, 8, and 12 h from the orbital sinus using heparinized glass capillary tubes. The supernatants were obtained after centrifugation at 3,000 rpm for 15 min. All samples were stored at −80°C until analysis.
UPLC-QTOF-MS Conditions
The experiments were performed on a UPLC system (LC-30AD, Shimadzu, Japan) and an AB SCIEX Triple TOFۛ 5600+ (Foster City, CA) mass spectrometer accompanied by a Duo Spray Ion Source. Common parameters of UPLC-QTOF-MS equipment were referred to previous research on triterpene saponin (Caoet al., 2018). The mobile phase was composed of 0.1% formic acid in water (A) and acetonitrile (B). Metabolic and pharmacokinetic investigations were conducted using an Acquity BEH C18 column (2.1 mm × 75 mm, 1.7 µm) and an Acquity BEH C18 column (2.1 mm × 100 mm, 1.7 µm), respectively. The gradient elution program was set as 65%–50% A (0–4 min), 50–20% A (4–7 min), and 20%–10% A (7–10 min) for the metabolic profiling. A gradient elution program was set as 35%–20% A (0–4.2 min) for the pharmacokinetic study. The precursor-to-product ion pairs, collision energy, and declustering potential values of IA and IA1 were m/z 501.32↓439.32 (−64 eV, −90 eV) and m/z 663.38↓501.32 (−37 eV, −100 eV), respectively. The target ion was monitored at m/z 779.43 for digoxin (internal standard, IS). The flow rate was 0.4 mL/min and the temperature was 40℃. The injection volume was 5 µL.
Determination of β-Glucosidase Activity
Fresh feces (0.2 g) collected from normal and antibiotic-treated rats were carefully mixed and suspended in 3.8 mL ice-cold phosphate buffer solution and then centrifuged at 200 rpm at 4°C for 5 min. The β-glucosidase activity of the supernatant was determined as previously described (Kang et al., 2016).
Sample Preparation and Data Processing
Stock solutions of two analytes (IA and IA1) were prepared by dissolving two accurately weighed standards in methanol with final concentrations of 0.025 mg/mL for IA and IA1. Calibration standards were prepared by spiking the standard working solutions into the blank plasma to obtain the final concentrations for IA and IA1. Quality control samples were prepared at nominal concentrations of 1.14, 30.86, and 277.78 ng/mL. The detailed procedures for method validation are described in the supplementary material.
Frozen plasma samples were subjected to natural thawing at room temperature. The plasma samples from 0, 0.5, 1, 2, 4, 8, and 12 h were merged into equal 100 µL volumes, respectively. Plasma was precipitated with 300 µL methanol to determine the metabolites of IA1. Then, 150 µL methanol containing IS of 0.1 µg/mL was added to 50 µL of plasma sample in a 1.5 mL tube. After vortexing for 5 min, the samples were centrifuged at 12,000 rpm for 15 min. The residue was reconstituted with 100 µL methanol containing 0.1% formic acid and injected into the UPLC-MS system for pharmacokinetic studies.
A pharmacokinetic study was performed using noncompartmental analysis on DAS 2.1.1 software supplied by the Mathematical Pharmacology Professional Committee of China (Shanghai, China). Experiment data were drawn and statistically analyzed by Prism 6.0. All the data are expressed as the arithmetic mean ± standard deviation (SD).
Network Pharmacology Study
IA1- and IA-related targets were collected using databases including Swiss Target Prediction (
Results and Discussion
Common Pharmacological Targets of IA and IA1
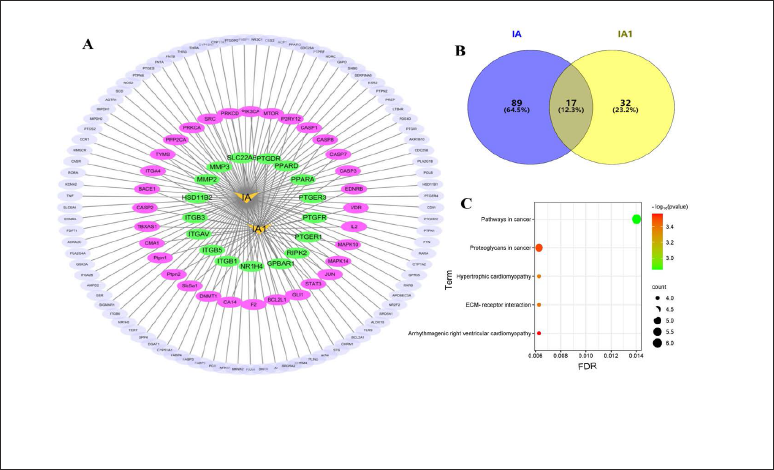
As shown in Figures 1A and B, 106 gene targets for IA were intersected with 49 gene targets for IA1, revealing 17 common targets. The KEGG pathways were retrieved based on screening significant enrichment (p < 0.05) and contribution value, which were mostly focused on pathways in cancer and proteoglycans in cancer (Figure 1C). Proteoglycans are the major macromolecules within the extracellular matrix. Dysregulation of extracellular matrix remodeling can contribute to tumorigenesis development (Wei et al., 2020). IA1 is among the most frequently studied proangiogenic phytochemicals, but increased rates of angiogenesis are tightly associated with cancer (Li et al., 2022). IA was reported to have anticarcinogenic activity and to obstruct the progression of cancer (Yun et al., 2021). The SwissADME online server (
Metabolite Profiling of IA1 in Rat Plasma
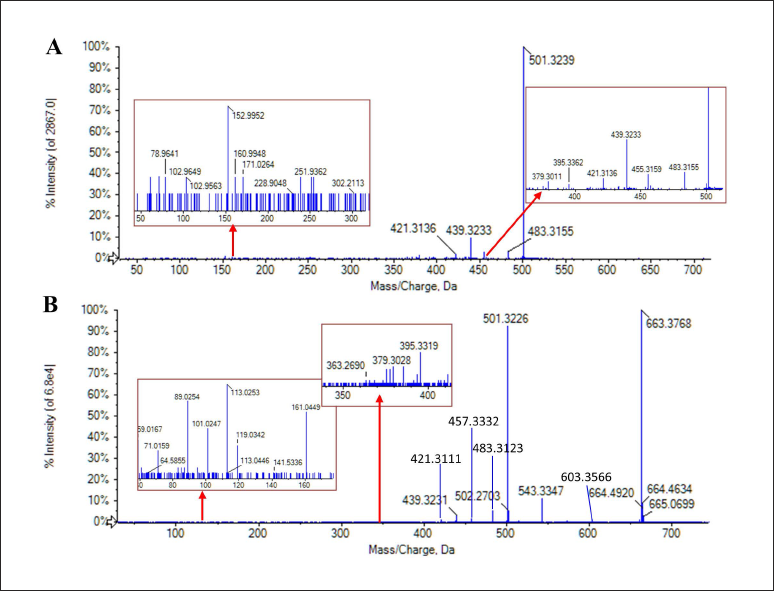
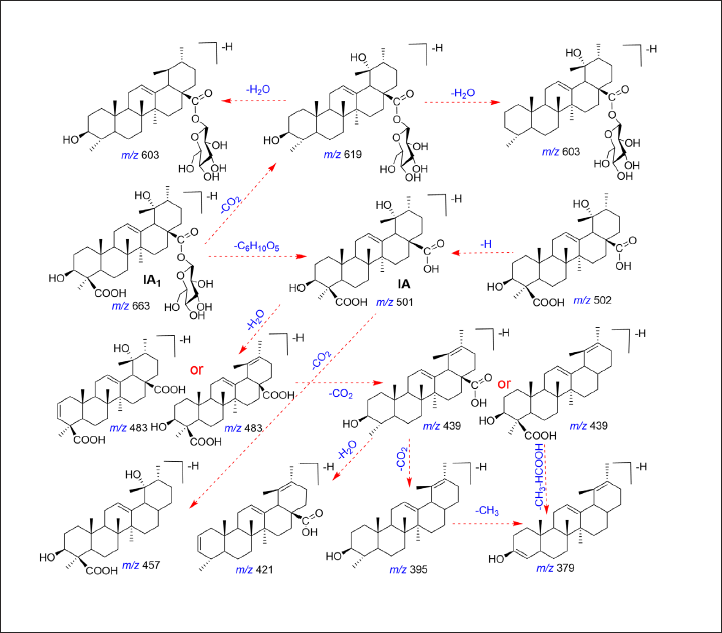
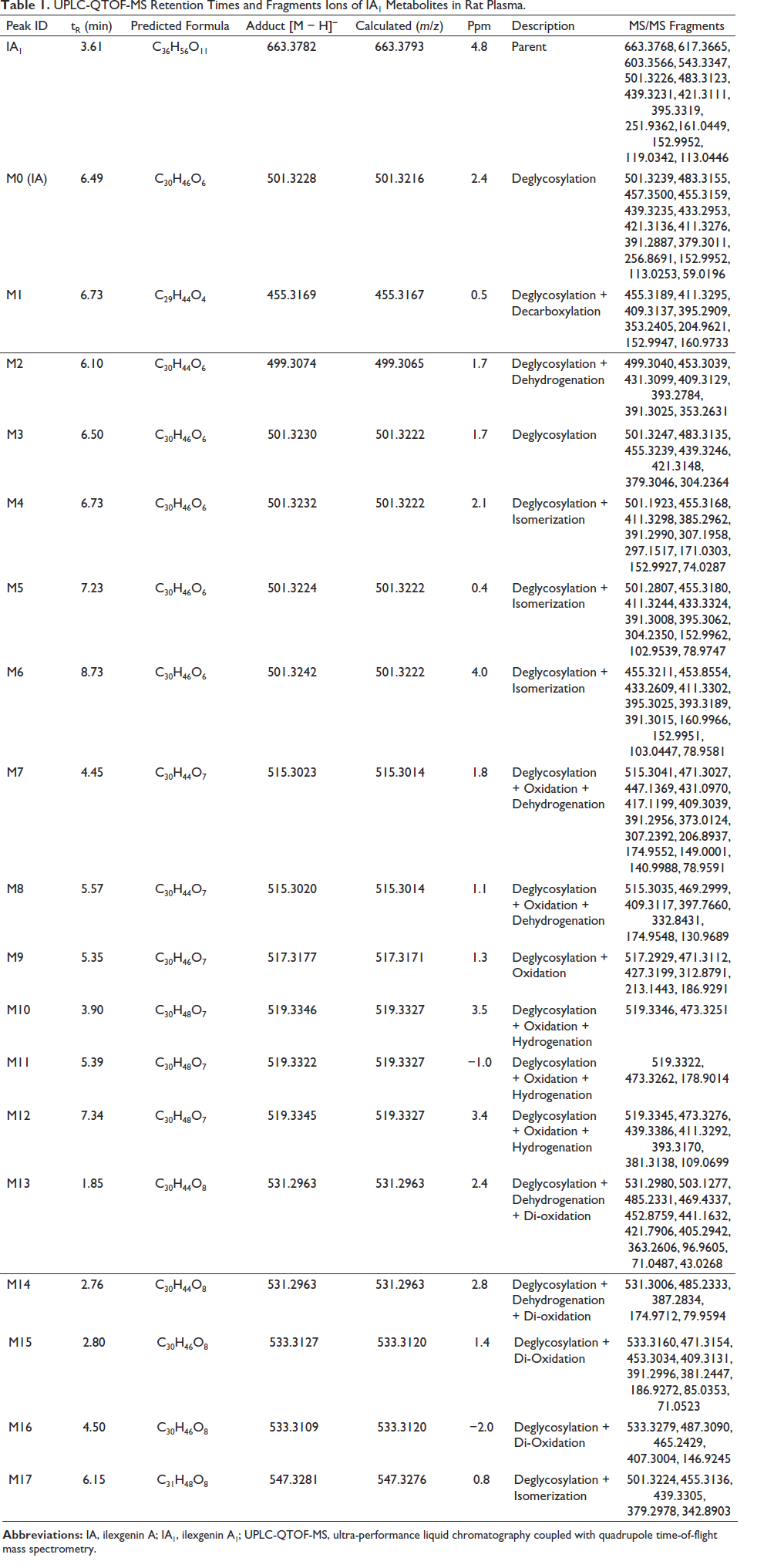
Through MetabolitePilot™ and PeakView® 2.0 software, which are data processing and data mining techniques, a total of 17 metabolites of IA1 were filtered in rat plasma. The metabolic pathways of IA1 mainly refer to the phase I process. The corresponding MS2 spectra and fragmentation pathways of IA1 and IA are shown in Figures 2 and 3, res-pectively. Deglycosylation, isomerization, decarboxylation, hydrogenation, dehydrogenation, and oxidation occurred on the skeleton of IA1 could be quickly recognized depending on the mass shift related to characteristic ions, includingm/z 483.3155, 455.3159, 439.3233, 421.3136, 395.3362, 152.9952, 113.0256, and 71.0531. The formula, retention time (tR), ppm, and MS/MS fragment data for IA1 and M0−M17 are displayed in Table 1, and the extracted ion chromatograms of M0−M17 are illustrated in Figure S1. The proposed biotransformation pathways of IA1 are shown in Figure S2. Fragmentation analysis for metabolite (M0−M17) identification is depicted in the supplementary material. Metabolic types, such as deglycosylation, decarboxylation, hydrogenation, dehydrogenation, isomerization, and oxidation, are typically identified for triterpenoid saponins (Lü et al., 2019; Yan et al., 2012). Additionally, exiguous significant differences in metabolic types were observed between normal and antibiotic-treated rats.
UPLC-QTOF-MS Retention Times and Fragments Ions of IA1 Metabolites in Rat Plasma.
β-Glucosidase Activity and Rat Body Weight Trend
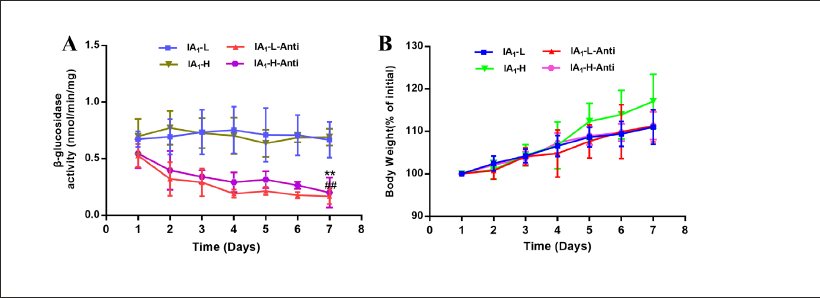
A comparison of β-glucosidase activity between the normal and antibiotic-treated groups was obtained by performing a t-test (Figure 4A). More days of modeling resulted in an increasing difference between the normal and antibiotic-treated groups. On the 7th day, the β-glucosidase activity of the normal group was approximately three times higher than that of the model group given antibiotics (p < 0.01). The β-glucosidase activity was regarded as an agreeable indicator of gut status (Lee et al., 2002). It was suggested that abnormal modifications of the intestinal flora were successfully triggered, which in turn led to a significant decrease in β-glucosidase activity. The type, quantity, proportion, location, and biological characteristics of the intestinal microbiota were altered once common intestinal flora disorders occurred (Patel et al., 2016). Additionally, the growth rate of rat weight gain recorded before administration every day was calculated. The weights of the rats in the normal and antibiotic-treated groups increased steadily, which indicated no significant differences among them (Figure 4B). Therefore, antibiotic treatment did not induce organ toxicities or physiological changes.
Method Validation for Pharmacokinetic Studies
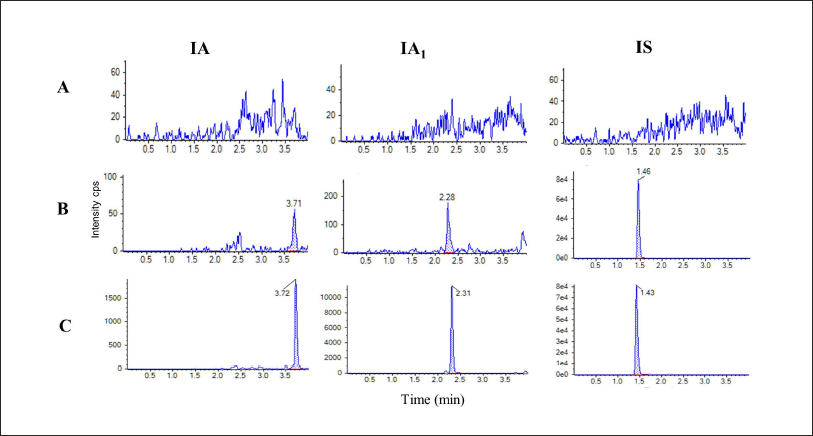
Under optimized conditions, the tR of IA, IA1, and IS were 3.71, 2.28, and 1.46 min, respectively. No interference at the tR of IA, IA1, or IS was discovered (Figure 5). The regression equations were y = 4.87995e−5x + 2.16161e−4 for IA and y = 2.37018e−4x + 0.00115 for IA1. The quantitative ranges of IA and IA1 were 0.38–1040 ng/mL and 0.13–1060 ng/mL, respectively, with correlation coefficients of 0.9954 for IA and 0.9927 for IA1. The detailed results of method validation are described in the supplementary material (Tables S1–S3).
Application in Pharmacokinetic Studies
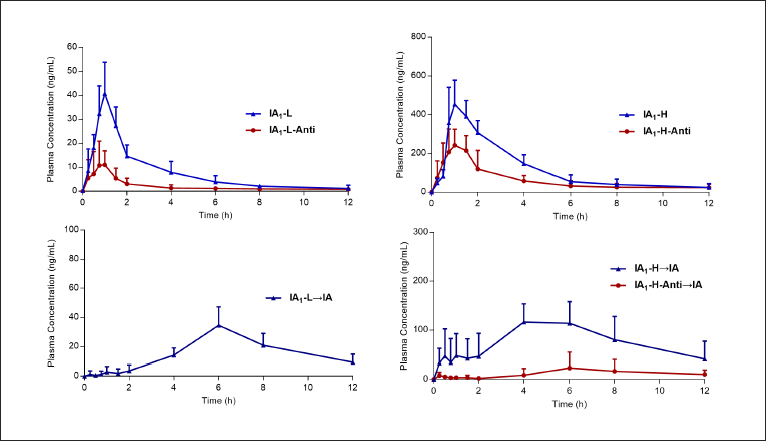
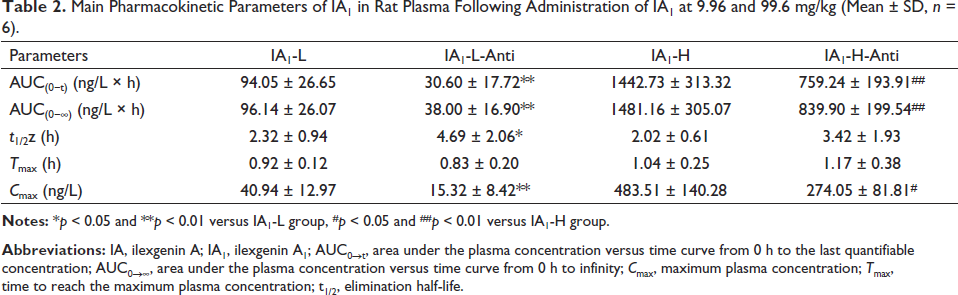
The validated analytical method was successfully applied to investigate the pharmacokinetics of IA1 and its metabolite IA after oral administration of IA1. The mean plasma concentration–time profiles (n = 6) of IA1 and its metabolite IA are shown in Figure 6, and the corresponding pharmacokinetic parameters of the tested compounds are listed in Tables 2 and 3. At the same dose, the pharmacokinetic parameters Cmax, area under the plasma concentration versus time curve from 0 h to the last quantifiable concentration (AUC(0−t)), and area under the plasma concentration versus time curve from 0 h to infinity (AUC(0−∞)) of IA1 in the IA1-L dose group were considerably increased compared with those of the IA1-L-anti group (p < 0.01). The differences in Cmax, AUC(0−t), and AUC(0−∞) between the IA1-H and IA1-H-anti groups were dramatically significant (p < 0.01, p < 0.05) in Table 2. The amount of IA1 taken up in the blood was relatively low, which is in accordance with earlier reports. The absorption of triterpenoid prototypes was severely limited by several properties, including low solubility, selectivity, and poor bioavailability (Zhou et al., 2017). The plasma concentration of IA1 at each time point in the intestinal flora disorder groups was lower than that in the normal group after intragastric administration, suggesting that the intestinal flora impeded the absorption of glycosides under the influence of antibiotics by monomer administration. However, herbal extracts can enhance the bioavailability of active compounds via ingredient–ingredient interactions, influence the activity of gut enzymes, and decrease antibiotic resistance, according to the overview (Su et al., 2020).
Main Pharmacokinetic Parameters of IA1 in Rat Plasma Following Administration of IA1 at 9.96 and 99.6 mg/kg (Mean ± SD, n = 6).
Main Pharmacokinetic Parameters of IA Converted by IA1 in Rat Plasma Following Administration of IA1 at 9.96 and 99.6 mg/kg (Mean ± SD, n = 6).
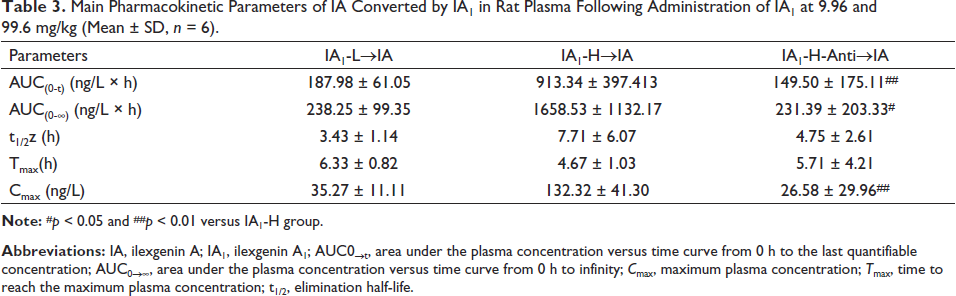
The pharmacokinetic behavior of IA transformed from IA1 was simultaneously investigated after oral administration of IA1 (Figure 6). The value Tmax of IA was observed at approximately 6.33 h in the IA1-L group, while the amount of IA could not be measured because the content was less than the lower limit of the quantification standard in the IA1-L-anti group in Table 3. The values of Cmax, AUC(0−t), and AUC(0−∞) in antibiotic-treated rats were significantly lower than those in normal rats (p < 0.01, p < 0.05) at the same dose of 99.6 mg/kg. Biotransformation of triterpene saponins mediated by intestinal microflora occurred through deglycosylation, glycosylation, oxidation, and dehydrogenation reactions (Fuet al., 2019). It could be interpreted that β-glucosidase activity inhibited by antibiotics suppressed the hydrolysis reaction of IA1 in the intestinal tract. Gut microbiota that expressed sundry enzymes could transform TCM compounds, which could generate active prodrugs with new bioactivity (Feng et al., 2019). Subsequently, the therapeutic effect could be altered as the enzymatic activities and diversity of the gut microbiota are affected by antibiotics (Li et al., 2019). The conversion product (IA) of IA1 could be an important pharmacodynamic basis for exerting antithrombosis, reducing inflammation, and promoting angiogenesis (Yang et al., 2017). From another perspective, utilizing gut microbiota to enhance the efficacy of IA1 should be a serious consideration.
Conclusion
The pharmacokinetics and metabolite profiles of IA1 were investigated based on the UPLC–MS/MS technique. Atotal of 18 metabolites of IA1 were identified in ratplasma by MetabolitePilot™ۛ 1.5 and PeakView® 2.0 software. Isomerization, decarboxylation, hydrogenation, dehydrogenation, and oxidation occurred on the skeleton of IA, which was transformed from IA1 through deglycosylation. The pharmacokinetic data demonstrated that the absorption of IA1 and the conversion efficiency of IA1 to IA were significantly impaired by disordered intestinal bacteria after monomer administration. Considering the database analysis, 1A and IA1 could play a synergistic role in common pharmacological pathway, such as hypertrophic cardiomyopathy and dilated cardiomyopathy. The converted product, IA, could be an important pharmacodynamic basis for IA1 exerting an effect. However, the effects of antibiotics on the efficacy of IA1 remain unclear. In addition, unsatisfactory drug likeliness criteria of IA1 and IA are adverse to clinical application. Therefore, these factors should be further considered in subsequent research.
Abbreviations
IA1: Ilexsaponin A1; IA: Ilexgenin A; TCM: Traditional Chinese Medicine; UPLC-QTOF-MS: Ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry; IS: Internal standard; PNPG: p-nitrophenol and p-nitrophenyl β-
Authors’ Contributions
All authors made contributions to the completion of the article. Di Cao performed the material preparation, experiments, and writing original draft. Xiuting Shen, Zhengjiao Wang, and Xiaojun Song handled the data analysis and manuscript review. Zhongxiang Zhao executed the supervision and revision of manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
The studies were approved by the ethics committee on the Care and Use of Laboratory Animals of Guangzhou University of Chinese Medicine.
Funding
This work was funded by grants from the National Natural Science Foundation of China (No. 81673565), the National Innovation and Entrepreneurship Training Program for College Students (No. 202110368059), and the Wuhu Science and Technology Program (No. 2022jc33).
Summary
The amounts of IA1 and its metabolite IA were simultaneously quantified in the plasma of normal and antibiotic-treated rats. β-Glucosidase activity inhibited by antibiotics suppressed the hydrolysis reaction of IA1 in the intestinal tract. IA1 was metabolized in rat plasma by biotransformation routes of deglycosylation, decarboxylation, isomerization, hydrogenation, dehydrogenation, and oxidation.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
