Abstract
The influence, on arsenic (As) urinary metabolic profile, of the level of As exposure was evaluated on chronic-exposed inhabitants of several locations of the Chaco-Pampean Plains in Argentina. Urinary As (UAs) was quantified as a measure of the level of exposure. The metabolic profile of UAs (inorganic As, monomethylarsonic acid, and dimethylarsinic acid) was also evaluated. The presence of T860C polymorphism on the arsenite methyltransferase encoding gene was investigated by desquamation of buccal cells. UAs showed a wide range of levels (from 18 µg/g to 4103 µg/g) of creatinine. A clear influence of age, gender, level of As exposure, and the presence of T860C polymorphism was observed on As metabolic profile. The influence of the level of exposure showed to be different between individuals carrying the wild type (WT) and the heterozygous (H) genotypes. Metabolic profile of individuals carrying the WT genotype seemed to be influenced by the level of exposure, while individuals with the H genotype did not. It is concluded that the level of As exposure seemed to have a significant influence on urinary metabolic profile of individuals carrying the WT genotype. In contrast, individuals carrying the H genotype seemed not to be affected the same way by increasing the As exposure level.
Introduction
Arsenic (As) is a metalloid with widespread distribution on the Earth’s crust. It occurs naturally in soil and surface and groundwater. It is highly toxic for humans, 1–4 and it is a known human carcinogen. 5 Long-term exposure of humans to As through drinking water is an important health problem in many regions of the world, 6–11 including Argentina. 9,12,13 Chronic exposure through drinking water results in hydroarsenicism, a chronic disease with characteristic skin lesions. 4,14,15 Since 1993, the World Health Organization recommends 10 μg of As per liter of drinking water as a provisional guideline value. 16 This value was selected to minimize the risks of chronic exposure to As according to the information available to date.
With a few exceptions, mammals metabolize inorganic As through methylation to monomethylarsonic acid (MMA) and dimethylarsinic acid (DMA). 17 These methylation reactions are mainly catalyzed by the enzyme arsenite methyltransferase (As3MT). All metabolites, including inorganic As, are eliminated in urine in different proportions. Human As methylation capacity has wide variability among individuals, and this variability is still subject of many investigations. 18–20
In the last decades, studies on As metabolism have guided the research toward the investigation of the influence of multiple factors on As methylation capacity and urinary As (UAs) metabolic profile. 18,21–25 Tseng 19 conducted a comprehensive review and discussion on many issues related to As metabolism such as ethnicity, age, gender, pregnancy status, smoking status, presence of some gene polymorphisms, and level of exposure among others. Polymorphism in genes encoding enzymes involved in As metabolism has been related to interindividual variation of urinary metabolite profiles. 18,20,22–25 As3MT, the main As metabolizing enzyme, is one of the most studied enzymes in that aspect. Among the 26 single nucleotide polymorphisms identified on the As3MT encoding gene, only 1 (T860C, Met287Thr) is exonic and nonsynonymous, has a 10% allele frequency, and has demonstrated to produce a variation in enzyme activity. 26
In Argentina, there are many regions with high levels of As in water. 27 In particular, the Chaco-Pampean Plains, located in North Central Argentina, are known for their elevated levels of As of their groundwater resources. 28,29 Another characteristic of this region is its population, with many years of residence in the area. The importance of the present work lies in the study of this particular context.
The aim of the study was to evaluate the influence of the level of As exposure on UAs metabolic profile and, in particular, its association with the presence of T860C polymorphism in the
Materials and methods
Study population
On April 2010, a cross-sectional study was carried out on the population of urban and rural areas of North Central Argentina. Three districts were included in the study: Districts of Banda and Copo in Santiago del Estero Province and District of Almirante Brown in Chaco Province. The area of study is located in the Chaco-Pampean Plains in Argentina, and it is known because of the high levels of As in many sources of groundwater.
A total of 120 inhabitants from 16 different locations agreed to participate in the study.
The study was approved by the Ethical Committee of the Hospital de Clínicas “José de San Martín” (Buenos Aires, Argentina), and informed consent was obtained from each participant.
Epidemiologic information
The population inhabiting these areas is rural and dispersed. It is characterized by a high percentage of children under 13 years and a low socioeconomic status, with a high proportion (more than 10%) of people with unsatisfied basic needs. 30 Their economic activities are limited, and they consist mainly of agriculture and logging. Information about age, gender, area of residence, and years of residence in the area was collected by performing a standard questionnaire. Most of the population was native and has been living in the area for life.
Sampling
All individuals were asked to provide a first morning urine sample. A sample of desquamated buccal cells was obtained by scraping the oral mucosa with a swab (at least two swabs were used per person). A water sample from each source of drinking water (public water distribution systems, collection systems, tanks, public and private wells and boreholes, and an artificial open aqueduct) was also obtained to assess the different levels of exposure of the population.
For locations with more than one source of drinking water, an average value was calculated, and that mean value was used to assess the level of exposure for the location. All samples were kept on ice until frozen at −20°C.
As and metabolites analyses
As in water and UAs levels were determined using a flow injection hydride generation atomic absorption spectrophotometry (FI-HG-AAS; Varian™ VGA77, AA475, Agilent Technologies, Santa Clara, California, USA) according to the method followed by Navoni et al. 31,32 UAs was expressed in micrograms per gram of creatinine. Creatinine analysis was performed using the Enzymatic Assay Kit Method (Wiener lab®, Argentina).
The urinary concentration of As(III), As(V), DMA(III + V), and MMA(III +
Previously filtered urine was injected (duplicate analysis) directly into the chromatograph. Chromatographic conditions were set according to the method developed by Vélez et al. 33 for methylated As compounds in seafood products. Chromatographic eluates reacted with sodium borohydride and hydrochloric acid to generate the corresponding arsines, and each arsine was measured using AAS.
Quality controls for water and UAs quantitative analysis were performed using certified reference materials EnviroMAT™ EPH-2 (SCP Science, Quebec, Canada) and Lyphochek™ level 2 (Bio-Rad, Hercules, California, USA), respectively. The reliability of As species quantification was evaluated by spiking urine blank samples with 100 μg/l of As(III), As(V), DMA(V), and MMA(V) and analyzing their recoveries.
Results of the urine samples speciation were expressed as percentage of inorganic As (%IAs) (
Polymorphism analysis
The presence of the single nucleotide polymorphism Met(287)Thr (T860C), in the
Statistical analysis
Statistical analysis was performed using InfoStat software (InfoStat Group, Cordoba, Argentina).
36
Shapiro–Wilks’ one sample test was performed to check normality distribution of variables and residuals. Spearman correlation coefficient (
Multiple linear regression analysis was performed to assess the influence of the level of exposure, the age and the gender, in association with the presence of T860C gene polymorphism on As metabolic profile. For statistical analysis, age was considered as a dichotomous variable: children (under 13 years) and adults (13 years and older).
Results
The study population consisted of 120 individuals, of which 61 (50.8%) were women and 59 (49.2%) were men. According to age, 85 participants (70.8%) were children under 13 years and 35 (29.2%) were 13 years and older. Participants were from 16 different locations (Table 1).
Level of As in drinking water, number, age (range and median) and gender of the participants, mean urinary As (UAs) concentration and mean percentages of IAs, MMA and DMA according to location.

As: arsenic; UAs: urinary arsenic; IAs: inorganic arsenic; MMA: monomethylarsonic acid; DMA: dimethylarsinic acid.
aAverage value
Information from the questionnaire showed that the population has been exposed to As through drinking water for life (or most of their lives). Levels of exposure varied by place of residence and were within a wide range of concentrations, from 13 to 1148 µg/l (Table 1).
UAs concentration was determined for the 120 participants, and it ranged from 18 to 4103 µg/g of creatinine. Correlation between UAs (micrograms per gram of creatinine) and As in drinking water (micrograms per liter) was assessed for 114 people. Six participants were excluded because their place of residence couldn’t be located. A positive correlation (Spearman’s correlation coefficient (
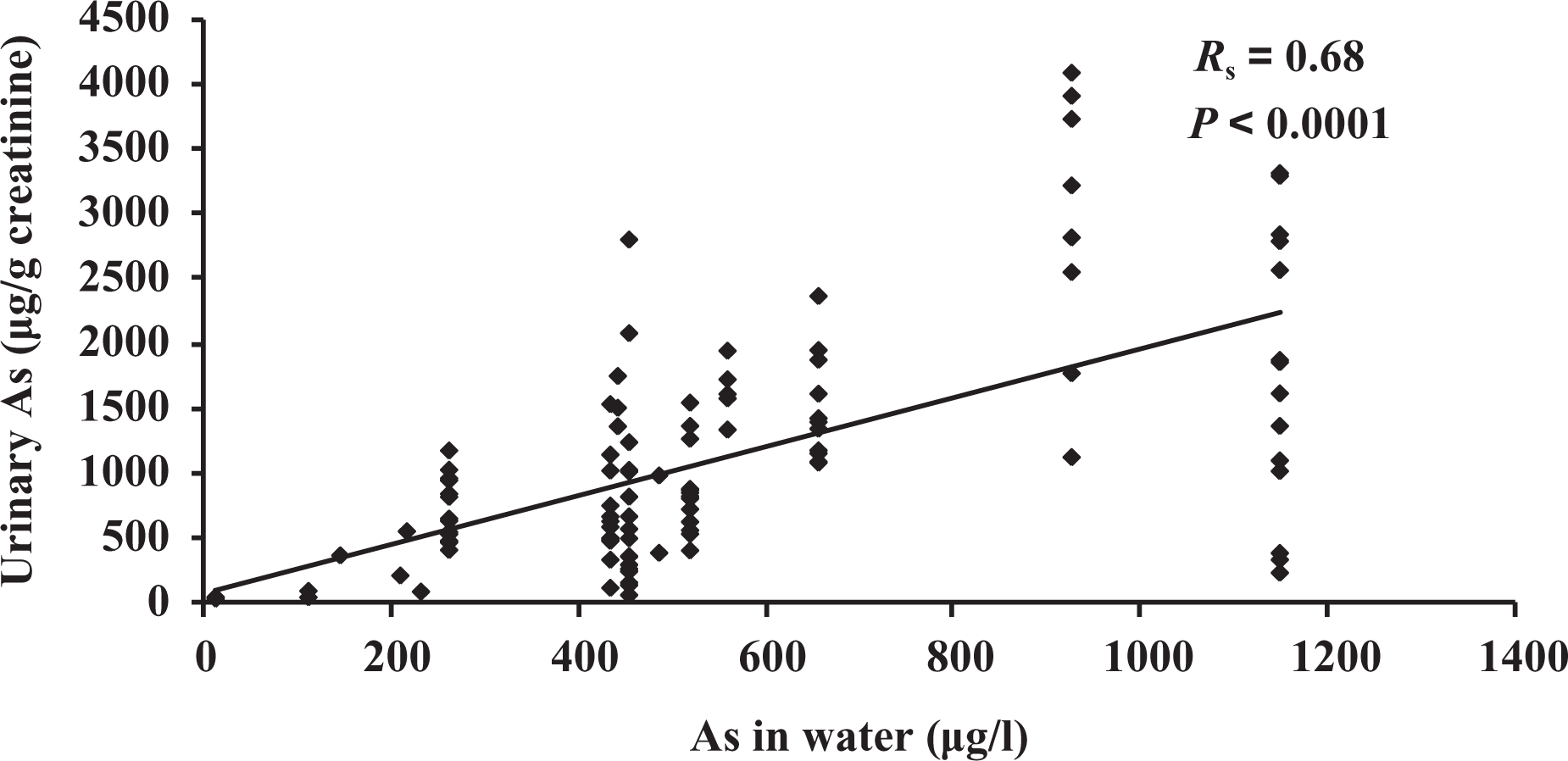
Correlation between UAs levels and drinking water concentration. UAs: urinary arsenic.
As metabolic profile was assessed for the 120 participants. Mean UAs and mean percentage of inorganic As (
Urinary %MMA was found to be directly associated with the level of UAs (
A multiple linear regression analysis was performed considering the age, gender, presence of T860C polymorphism, and UAs as predictors of As metabolic profile. The results showed a marked influence of age, UAs level, gender, and polymorphism as predictors of urinary %MMA. The results also showed a marked influence of gender, UAs level, and polymorphism as predictors of urinary %DMA, but the influence of age is just not significant (Table 2).
Results of the multiple linear regression analysis of the variables urinary %MMA and %DMA, considering age, gender, UAs, and presence of T860C polymorphism as predictors of As metabolic profile.
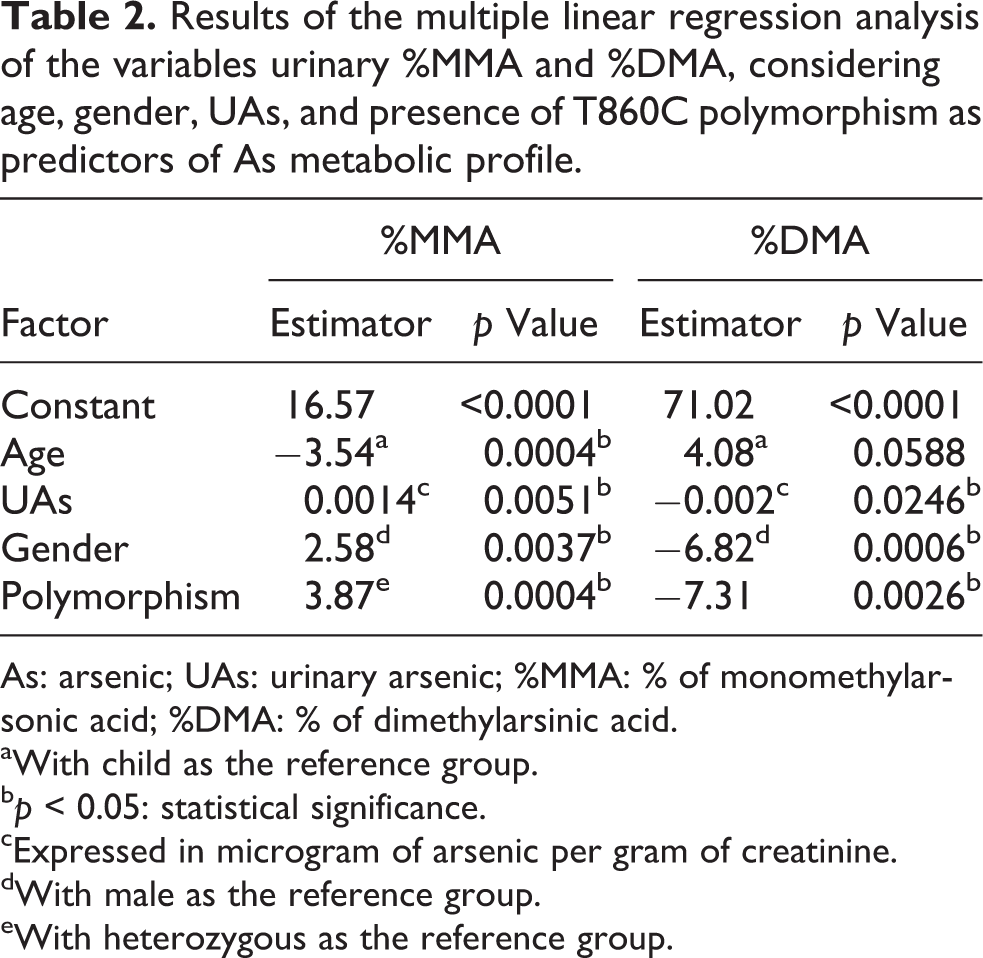
As: arsenic; UAs: urinary arsenic; %MMA: % of monomethylarsonic acid; %DMA: % of dimethylarsinic acid.
aWith child as the reference group.
b
cExpressed in microgram of arsenic per gram of creatinine.
dWith male as the reference group.
eWith heterozygous as the reference group.
The presence of T860C polymorphism was investigated in DNA extracted from desquamated buccal cell samples. The population showed to be in Hardy–Weinberg equilibrium for the selected polymorphism. Wild type (WT) genotype (TT) of
Observed and expected genotypes for the T860C polymorphism.
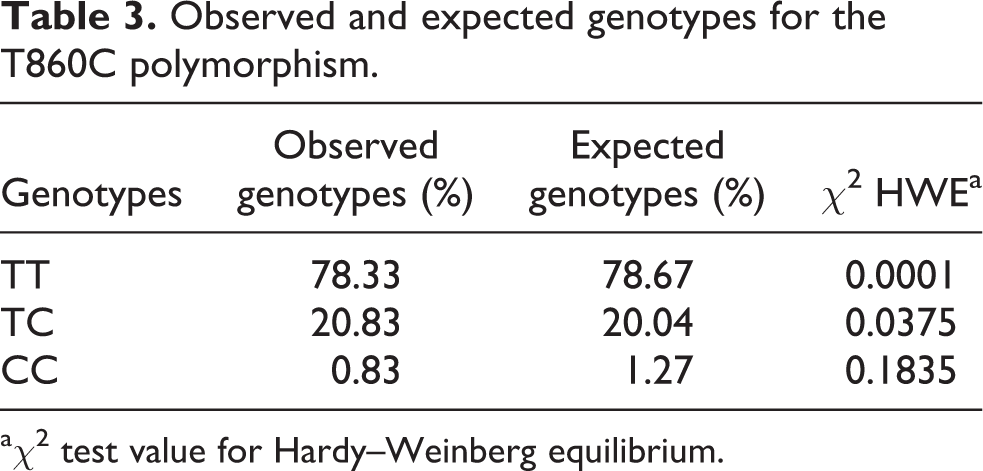
aχ 2 test value for Hardy–Weinberg equilibrium.
Multiple linear regression analysis, with adjustment for age, gender, and UAs, was performed separately for individuals carrying the WT and the H genotypes. Results showed a marked positive influence of UAs as a predictor of urinary %MMA for individuals carrying the WT genotype, and no statistically significant influence as the predictor of urinary %MMA for individuals carrying the H genotype (Table 4). In conjunction with this, a slight (negative) association of UAs as a predictor of urinary %DMA was observed for individuals carrying the WT genotype, and no statistically significant influence was observed for individuals carrying the H genotype (Table 4). Finally, unlike what was observed (Table 2) for the total sample population, for individuals carrying the H genotype, age showed to be a predictor not only for urinary %MMA but also for urinary %DMA and gender showed not to be a predictor for urinary %MMA (Table 4).
Results of the multiple linear regression analysis of the variables %MMA and %DMA, for individuals carrying the WT and the H genotype.

UAs: urinary arsenic; %MMA: percentage of monomethylarsonic acid; %DMA: percentage of dimethylarsinic acid; WT: wild type; H: heterozygous.
aWith child as the reference group.
b
cExpressed in micrograms of arsenic per gram of creatinine.
dWith male as the reference group.
Since UAs seems to influence %MMA and %DMA only for individuals carrying the WT genotype, correlation between UAs and As urinary metabolites (MMA, DMA, and IAs) was assessed for the two populations (WT and H genotype individuals) separately. Figure 2(a) shows the variation of urinary %MMA, for WT and H groups, as a function of UAs level. A clear correlation (
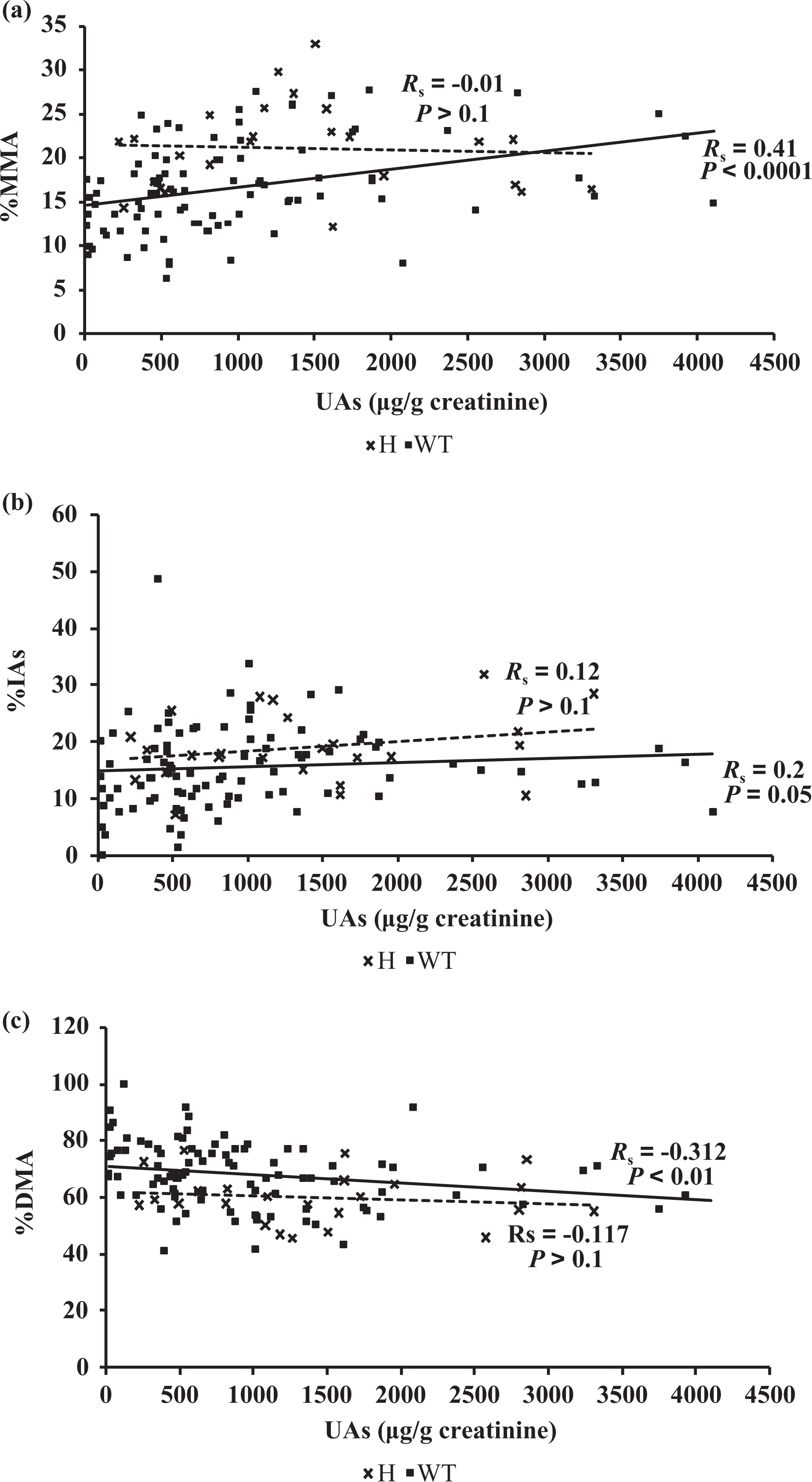
Urinary (a) %MMA, (b) %IAs and (c) %DMA according to UAs levels for individuals carrying the WT and the H genotype for the T860C polymorphism on the
Results showed a correlation between UAs levels and proportions of metabolites only for individuals carrying the WT genotype, and no correlation was observed for individuals carrying the H genotype.
Finally, a new multiple linear regression analysis (with adjustment for age, gender, presence of T860C polymorphism, and UAs) was performed only for individuals with UAs over 1500 µg/g of creatinine (
Results of the multiple linear regression analysis of the variables urinary %MMA and %DMA for individuals with UAs over 1500 µg/g of creatinine, considering age, gender, UAs, and presence of T860C polymorphism as predictors of As metabolic profile.

As: arsenic; UAs: urinary arsenic; %MMA: % of monomethylarsonic acid; %DMA: % of dimethylarsinic acid.
aWith child as the reference group.
b
cExpressed in micrograms of arsenic per gram of creatinine.
dWith male as the reference group.
eWith heterozygous as the reference group.
Discussion
UAs level has been widely used as a biomarker to reflect the exposure to As through drinking water. 37,38 It has been described to correlate positively with As levels in drinking water as high as 620 µg/l37,39 and with estimated As daily intake. 38,39 However, there is scarce information about what happens at higher levels of exposure.
In this study, a positive correlation between UAs level and the level of exposure was observed. This correlation was defined for a wide range of concentrations (from 13 µg/l to 1148 µg/l). Although correlation seemed to decrease around the highest level of As in drinking water (1148 µg/l), it was still good enough to consider UAs as an indicator of the level of exposure.
Several factors have been identified to have influence on As metabolic profile in this study. Women showed to methylate As better than men, in concordance with the results of previous studies.
25,40
The action of estrogens on the synthesis of choline might explain the difference in the methylation profile between women and men.
19
Choline is involved in the remethylation of homocysteine to methionine, the latter is a precursor of
Age (expressed as child/adult difference) was also found to have influence on As metabolic urinary profile, although it showed to be a better predictor of urinary %MMA than of urinary %DMA. The marginal statistical significance for the influence of age on the urinary DMA may be due to the limited number of participants. The statistical significance of the later result would probably improve by increasing the number of individuals analyzed. Even so, these results do support the hypothesis that children have a more efficient second methylation step than adults. 25,41
It is a fact that in this study population there were more children than adults, so, it is possible, although we think unlikely, the existence of some bias due to the sample population.
A marked influence on the level of exposure (expressed as UAs concentration) was also observed on relative proportions of urinary MMA and DMA, in agreement with previously reported studies. 40–42 However, in the present study, this influence is described as of a higher degree and also for levels of exposure significantly higher.
Furthermore, the influence of the level of exposure on UAs metabolic profile showed to be different between individuals carrying the WT and the H genotypes. Metabolic profile for the individuals carrying the WT genotype seemed to be influenced by the level of exposure, while metabolic profile of those individuals with the H genotype did not. The reasons why metabolic profile of WT individuals changed at very high levels of exposure are not clear. The hypothesis of the existence of a methylation threshold was not supported by Hopenhayn-Rich et al. 43 However, in our study, the evaluated exposure levels covered a wider range (especially extended to higher levels), so it would be possible that a kind of saturation process might be involved.
Little is known about the activity of the enzyme As3MT at high concentrations of As. Studies in mice chronically exposed for 32 and 91 days to 25 and 2500 µg/l of As in drinking water
44
demonstrated that no induction in liver, testes, kidneys and lung As3MT activity occurred, after the exposure to high concentrations of As. In addition, and in agreement with, what we described as the influence of the level of exposure on WT individuals’ methylation capacity are the studies conducted by Song et al.
45
and Ding et al.
46
Song et al.
45
suggest the inhibition of the conversion of IAs to MMA and of MMA to DMA by increasing concentrations of IAs. According to the authors, the latter type of inhibition showed to be greater than the former,
45
suggesting that inorganic As could compete with MMA for enzymatic methylation and then decrease DMA production. Ding et al.
46
found that the catalytic capacities of both variants of human As3MT (WT and M287T) seemed to saturate at substrate (As (
To our knowledge, this is the first study analyzing the urinary metabolic profile of an Argentinean population exposed to a wide range of As concentrations through drinking water. It is also the first study to analyze the influence of the level of exposure on As metabolic profile, for individuals carrying two different genotypes (WT and H for the T860C polymorphism) of the
Elevated proportions of MMA and low DMA/MMA ratio are associated with high risks to develop skin lesions, 47 , 48 urothelial carcinoma, 49 bladder cancer, 50 and lung cancer. 51 In addition, the presence of T860C polymorphism has been related to increased cytogenetic damage 52 , 53 and bladder cancer, 54 in people exposed to As at low to moderate levels. However, no reports of what happened at high to very high levels of exposure are found in the literature. Considering the results of this study, we suggest that a very high level of exposure would act as an additional negative factor on the metabolic capacity of the individuals carrying the WT genotype, placing them in the same unfavorable risk situation of the individuals carrying the H genotype, in relation to the development of the As-related deleterious effects.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by the Universidad de Buenos Aires [UBACYT 20020100100394].
