Abstract
Background
Endothelial cell (EC) inflammation plays a crucial role in the development of several cardiovascular disorders (CD), including atherosclerosis and sepsis. Ligustrazine (Lig), a bioactive constituent derived from Traditional Chinese Medicine Ligusticum chuanxiong Hort, has exhibited in vivo anti-inflammatory properties. Despite the observed positive outcomes, the exact mechanisms underlying these beneficial effects remain unidentified.
Aim
The goal of this research is to investigate the influence and potential mechanism of Lig on lipopolysaccharide (LPS)-induced inflammatory responses in human umbilical vein endothelial cells (HUVECs).
Introduction
These experiments investigate the effectiveness of Lig in preventing LPS-induced damage in HUVECs, with the goal of elucidating the underlying processes at work.
Materials and Methods
To evaluate HUVECs’ viability, a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay was conducted. Enzyme-linked immunosorbent assay (ELISA) was employed to measure changes in ICAM-1, interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and monocyte chemoattractant protein (MCP-1) levels. Real-time polymerase chain reaction (RT-PCR) was used to determine the levels of ICAM-1, IL-6, TNF-α, MCP-1, and toll-like receptors (TLR4) mRNA. Additionally, we performed WB analysis to assess the levels of nuclear factor-κB (NF-κB) p65, TLR4, IκBα, and p-IκBα.
Results
The findings demonstrated significantly suppressed cell viability due to LPS treatment, while Lig treatment increased cell viability in a concentration-dependent manner. Lig also effectively reduced the mRNA levels of ICAM-1, TNF-α, IL-6, and MCP-1. Furthermore, Lig pretreatment led to downregulation of TLR4, p-IκBα, and NF-κB p65 in HUVECs.
Discussion
The findings indicate that Lig reduces LPS-induced inflammation in HUVECs, and that the TLR4/NF-κB pathway is critical in increasing cell survival and minimizing inflammatory damage. This provides possible anti-inflammatory techniques for treating CD.
Conclusion
In conclusion, our work demonstrates Lig’s anti-inflammatory actions on LPS-stimulated HUVECs. The data suggest that Lig lowers inflammation via regulating the TLR4/NF-B pathway, boosting cell survival, and decreasing inflammatory responses.
Introduction
Atherosclerosis (AS) is a persistent inflammatory condition triggered by various factors (Bosseboeuf &Raimondi, 2020). Endothelial dysfunction is regarded as the instigator of atherosclerotic disease (Favero et al., 2014; Rasmussen et al., 2020). Lipopolysaccharide (LPS) plays a pivotal role in AS progression, as it binds to endothelial cell (EC) surface TLR4, activating the JAK-STAT pathway, MAPK, and nuclear factor-κB (NF-κB). Consequently, these processes induce the expression of adhesion molecules and promote inflammation (Iannotta et al., 2021).
Toll-like receptors (TLRs), belonging to the transmembrane protein receptor family, were initially discovered in the study of drosophila embryo development (Devarapu & Anders, 2018). TLRs are broadly dispersed throughout the body and expressed on the exteriors of roughly 20 different cell types, with TLR4 being preferentially expressed on the surfaces of normal vascular EC (Kiyan et al., 2019). TLR4, found in various inflammatory cells, is responsible for recognizing bacterial LPS. Mutations in TLR4 have been observed in diverse ethnic groups, highlighting their prevalence and significance (Jang et al., 2017). Studies have shown that an important transcriptional activator, NF-κB, exists in the downstream signaling pathway of TLR4 (Wu et al., 2019). NF-κB remains inactive under normal conditions but can be induced by specific factors. When engaged, NF-κB initiates transcription of several essential inflammation-related genes resulting in the inflammatory response (Zhao et al., 2018).
Ligustrazine (Lig), derived from the root of the Ligusticum chuanxiong plant, has emerged as a prominent subject of research in the treatment of cardiovascular disorders (CD), tumors, and various ailments (Feng et al., 2023; Wu et al., 2020). It has recently gained significant attention for its potential role in mitigating inflammatory responses (Alkreathy et al., 2020). Nevertheless, the precise mechanisms responsible for the favorable effects of Lig remain enigmatic. To illuminate this matter, this study explores the effectiveness of Lig in countering LPS-induced damage in human umbilical vein endothelial cells (HUVECs), with the objective of unraveling the underlying processes at play.
Materials and Methods
Drugs and Reagents
The Lig hydrochloride utilized in this study was acquired from the Chinese National Institute for the Control of Pharmaceutical and Biological Products, while the LPS was obtained from Sigma-Aldrich. The enzyme-linked immunosorbent assay (ELISA) kit was procured from Elabscience Biotechnology Co., Ltd (Wuhan, China). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) was acquired from Invitrogen (California, USA), and Trizol was purchased from Ambion Co., Ltd (Texas, USA). Anti-NF-κB p65 antibody, anti-TLR4 antibody, and anti-p-IκBα antibody were obtained from Affinity Biosciences LTD (Beijing, China), while anti-IκBα antibody was sourced from Cell Signaling Technology. Anti-GAPDH antibody and bicinchoninic acid (BCA) were purchased from Xianzhi Biological Co., Ltd (Hangzhou, China). Pyrrolidine dithiocarbamate (PDTC) was procured from Sigma-Aldrich (Darmstadt, Germany). The 0.4% fetal bovine serum (FBS) was obtained from Elabscience Biotechnology Co., Ltd (Wuhan, China).
Cell Culture
The Shanghai Academy of Life Sciences provided the HUVECs. The cells were first seeded in a 75 cm2 culture flask and raised in M200 media with 2% low serum growth supplement (Cascade Biologics Inc.). The cells were sub-cultured at a 1:3 split ratio once they achieved about 80% confluence. The medium was changed after 24 h with M200 medium containing 0.4% FBS. It is worth noting that the cells used in this investigation were derived from the fourth-generation cultures.
Experimental Groups
The study comprised five distinct experimental groups, namely: (1) During the incubation phase, HUVECs were grown in drug-free media in the control group. Cells were treated to 1 mg/L LPS for 24 h in the LPS group. (2)The LPS and Lig (20 µg/mL) group, in which cells were marinated with Lig (20 µg/mL) for 2 h, followed by the addition of 1 mg/L LPS for 24 h. (3) The LPS and Lig (50 µg/mL) group, in which cells received prior treatment with Lig (50 µg/mL) for 2 h before being treated with 1 mg/L LPS for 24 h. (4)The PDTC and LPS group, in which cells were exposed to PDTC for 120 min and subsequently exposed to 1 mg/L LPS for 24 h.
MTT Assay
The MTT chemosensitivity assay was conducted using a microplate reader to evaluate the cell viability of HUVECs. The cells were planted in 96-well plates at 105 cells/mL. After 24 h of adherence and growth, the cells underwent pretreatment with Lig, followed by washing with phosphate-buffered saline (PBS). Subsequently, the cells were exposed to LPS. Each group was then treated with MTT solution (1 mg/mL). Formazan crystals were dispersed by adding 150 µL of dimethyl sulfoxide (DMSO). The absorbance value, which reflects cell viability, was taken with a microplate reader after shaking for 10 min. Cell vitality was determined as a proportion, with each experiment performed in quintuplicate using three separate cell cultures.
ELISA
The levels of the target genes were quantified using ELISA kits according to the manufacturer’s guidelines. Subsequent to treatment with various drugs and delivery methods, the cell supernatants were gathered and subjected to analysis following the provided protocols.
Quantitative Real-time Polymerase Chain Reaction (RT-PCR)
Trizol was utilized for total RNA extraction. Stringent precautions were taken to ensure RNase-free conditions, employing RNAse-free glassware and plastic ware. The isolated RNA samples underwent agarose gel electrophoresis for quality assessment, with the A260/A280 ratios falling within the range of 1.80 to 2.0, as measured on a spectrophotometer.
For cDNA synthesis and amplification, a TaKaRa RNA PCR Kit (AMV) Ver3.0 was utilized. PCR amplification was conducted for the target genes, utilizing primers designed specifically for each gene. A negative control (no RT) using RNA solution without reverse transcription was included to guarantee experimental correctness. GAPDH was also used as an internal control for normalization. Table 1 lists the primer sequences that were used.
Gene Expression Quantification Primer Sequences.
Western Blot (WB) Analysis
Confluent HUVECs cultivated in 6-well plates were subjected to Lig pretreatment and subsequent LPS treatment, as previously outlined. Subsequently, the cells were lysed, and the supernatant was obtained through centrifugation at 4°C and 14,000 rpm for 30 min. The protein concentration in the supernatant was determined using the BCA method. Equal amounts of protein samples were loaded onto a 10%sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel and separated through electrophoresis. Next, the PVDF membrane was blocked with Tris-buffered saline-Tween-20 (TBST) containing 5% skim milk at room temperature for 2 h. Subsequently, the PVDF membrane was incubated with the primary antibody at 4°C overnight. Afterward, the membrane was washed and incubated with horseradish peroxidase (HRP) for 1 h. Finally, the presence of the antigen was detected using the enhanced chemiluminescence (ECL) method.
Statistical Analysis
The data are presented as the mean±SD. Multiple group comparisons were performed using ANOVA with GraphPad Prism 6.0. To compare the means between each pair of groups, we employed least significant difference (LSD) analysis. Statistical significance was defined as p < 0.05. *p < 0.05 compared to control group. #p < 0.05 compared to LPS group.
Results
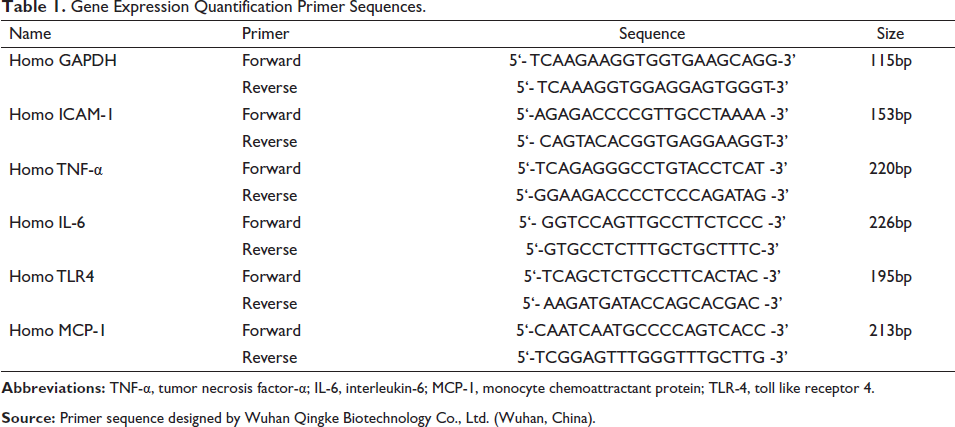
Impact of Lig on Cell Viability
Cell proliferation activity exhibited a concentration-dependent increase after 24 h of treatment with various concentrations of Lig. Figure 1 illustrates that LPS treatment significantly inhibited HUVEC proliferation. However, compared to the LPS group, the pretreatment of HUVECs with different concentrations of Lig resulted in a substantial enhancement of cell viability. The optimal concentration was found to be 50 µg/mL.
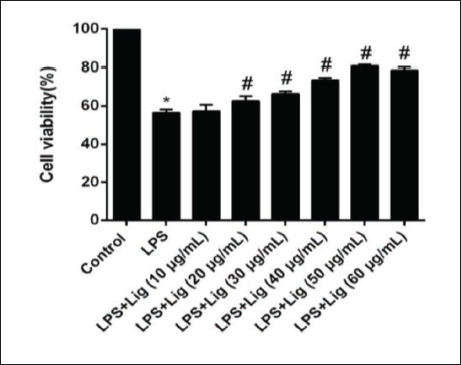
Modulatory Effects of Lig on ICAM-1, TNF-α, IL-6, and MCP-1 Levels in LPS-induced HUVEC Culture Medium
After exposing the cells to various concentrations of Lig for 24 h, a gradual reduction in the levels of the target genes was observed, indicating a dose-dependent effect. Figure 2 illustrates a significant increase in the production of the target genes in the culture medium upon LPS induction, signifying an intensified inflammatory response in the cells. Nonetheless, subsequent treatment with Lig led to a diminished production of the target genes in the culture medium, implying that Lig can downregulate the expression of these inflammatory markers, thereby exerting an anti-inflammatory and protective effect on the vascular endothelium.
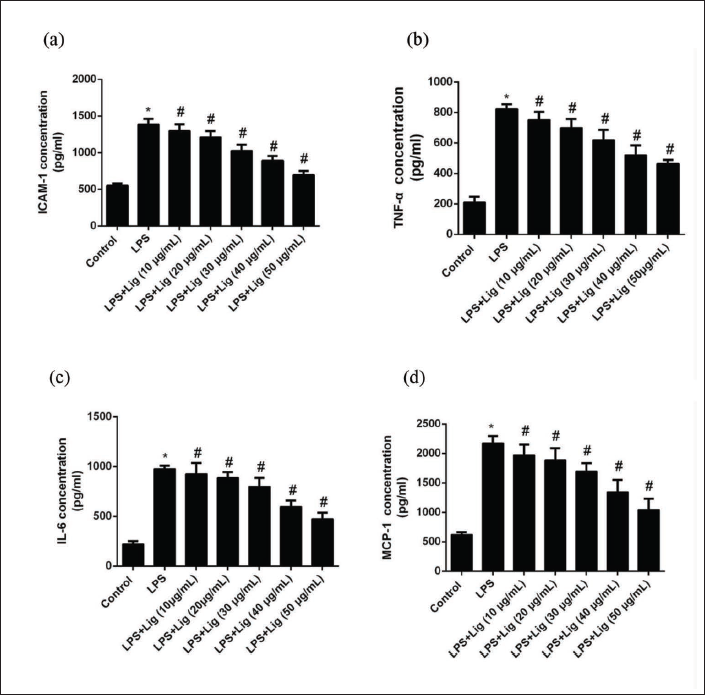
Impact of Lig on mRNA Expression and Levels of ICAM-1, IL-6, TNF-α, and TLR4 in LPS-induced EC
As illustrated in Figure 3, the mRNA levels of the target genes in the control group were remarkably low. However, after 24 h of LPS treatment, a substantial increase in the mRNA expressions of these inflammatory factors was observed. Importantly, pre-treatment with varying doses of Lig and PDTC substantially inhibited the effects of LPS.
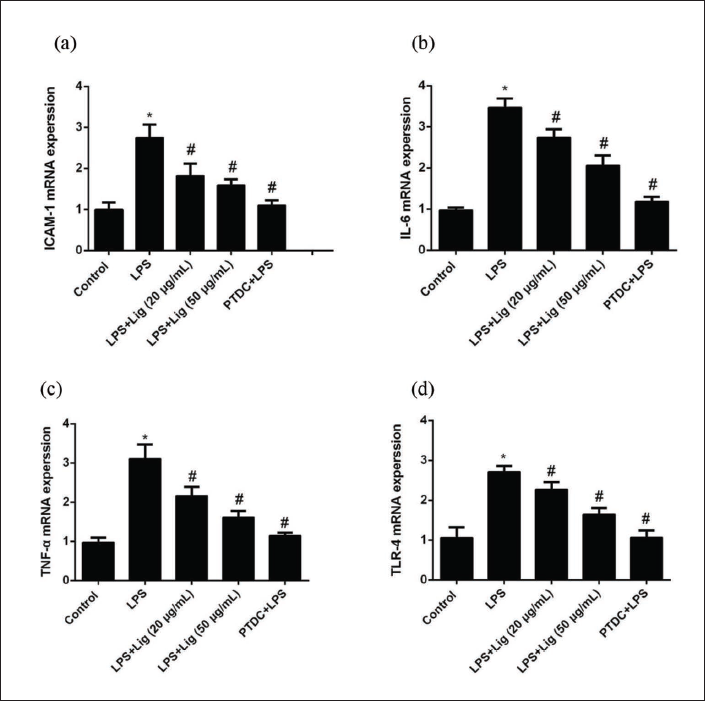
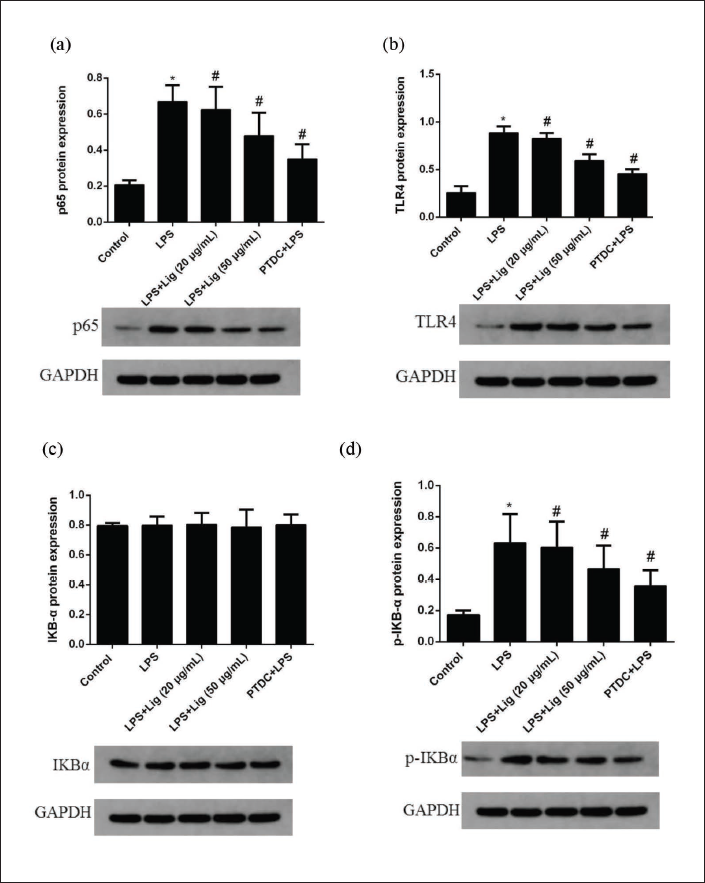
Modulation of NF-κB p65, TLR4, IKBα, and p-IKBα Protein Activities by Lig in LPS-treated EC
In Figure 4, it is evident that the activities of the target proteins in EC significantly increased following LPS treatment. However, pre-treatment with different concentrations of Lig effectively suppressed the activities of the target proteins induced by LPS. Similarly, pre-incubation with PDTC also demonstrated significant inhibition of the activities of the target proteins.
Discussion
AS is a multidimensional inflammatory disease characterized by complex molecular and cellular changes inside the artery wall (Blagov et al., 2023; Rubin et al., 2012; Sazonova et al., 2021). Inflammation is crucial in the onset, development, and pathophysiology of AS (Gusev &Sarapultsev, 2023; Wolf &Ley, 2019). As a result, HUVECs were used in this study due to their importance in vascular endothelial function. Traditional Chinese botanical treatments have received a lot of attention for their ability to treat AS, protect vascular EC, and promote tissue regeneration. This study focused on L. chuanxiong, a Chinese medicinal herb known for its ability to improve blood circulation and reduce stasis. Figure 1 depicts the concentration-dependent increase in cell viability caused by Lig therapy, in contrast to the suppressive impact seen with LPS treatment.
Current investigations suggest that inflammatory cytokines, namely ICAM-1 (Buckley et al., 2022), interleukin-6 (IL-6) (Rose-John et al., 2023), and MCP-1 (Bittar et al., 2020), synthesized during the inflammatory response, can precipitate EC injury and dysfunction. ICAM-1, an indispensable adhesion molecule belonging to the immunoglobulin superfamily, typically exhibits diminished expression levels in a quiescent state but can be upregulated by various factors, thereby contributing to the adhesion of diverse leukocytes (Kucukal et al., 2020). The adhesion and migration of monocytes (Mon), lymphocytes, and other leukocytes to vascular EC represent pivotal initial stages in the early development of AS (Jebari-Benslaiman et al., 2022). IL-6, a member of the chemotactic cytokine superfamily, is renowned as the most potent chemoattractant and activator of neutrophils to have been discovered thus far (Hansen, 2020). It instigates neutrophil activation and stimulates the release of granule contents, leading to endothelial injury. Furthermore, IL-6 can impair vital components of the endothelial matrix, such as collagen and elastin, through various processes, culminating in augmented endothelial permeability (Verboogen et al., 2019). MCP-1 has been identified as a significant pro-inflammatory cytokine that can be secreted by vascular EC, Mon, smooth muscle cells, and others (Choi et al., 2021; Singh et al., 2021). During inflammation, MCP-1 exhibits specific chemotactic and activating effects on Mon (Rodríguez-Frade et al., 1999). In this study, the levels of ICAM-1, TNFα, IL-6, and MCP-1 were assessed using ELISA kits. Following treatment with various concentrations of Lig for 24 h, a noteworthy reduction was observed in the contents of ICAM-1, tumor necrosis factor-α (TNF-α), IL-6, and MCP-1, clearly demonstrating a concentration-dependent effect. Based on these findings, it can be inferred that Lig exerts an anti-inflammatory and protective role in the vascular endothelium.
TLR4 stands as a pattern recognition receptor, assuming a vital role in discerning conserved structures, activating signal transduction pathways, and instigating inflammatory responses throughout microbial evolution (Xia et al., 2021). Previous investigations have identified TLR4 as the primary receptor responsible for transmitting signals to cells in response to LPS, a pivotal component of endotoxin (Wu et al., 2019). Furthermore, downstream in the signaling cascade of TLR4, an indispensable transcriptional activator, known as NF-κB, has been unveiled (Wu et al., 2019). NF-κB stands as a pivotal regulator of gene transcription, exerting control over the expression of numerous genes, particularly those involved in the inflammatory response (Zhao et al., 2018). Activated NF-κB binds to corresponding sites in the promoter regions of genes, including cytokines and adhesion molecules, facilitating the transfer of information from the cytoplasm to specific regions of nuclear DNA (Jambusaria et al., 2020). This activation precipitates the initiation of transcription and translation processes in the respective genes. The observed content of inflammatory cytokines in EC caused by LPS in this study implies a potential association with the activation of the TLR4/NF-κB pathway. In the control group of this study, the mRNA expressions of ICAM-1, IL-6, TNF-α, and TLR4 were observed to be at very low levels. However, after 24 h of LPS treatment, there was a significant upregulation of the mRNA expressions of these inflammatory factors (Figure 3). Moreover, the effects of LPS were mitigated by different concentrations of Lig and PDTC in advance (Figure 3). To investigate the underlying process, the expression levels of NF-κB p65, TLR4, IKBα, and p-IKBα were assessed using WB analysis. Notably, Lig treatment led to the downregulation of NF-κB p65, TLR4, and p-IKBα expression. However, these effects of Lig were nullified by PDTC treatment (Figure 4). Consequently, this study exemplified that Lig treatment mitigates LPS-induced injury in HUVECs by reducing inflammation through the activated TLR4/NF-κB pathway.
Conclusion
The study’s findings reveal that Lig bestows a protective effect by augmenting cell viability while concurrently reducing the levels of ICAM-1, TNFα, IL-6, and MCP-1. Lig also exhibits anti-inflammatory properties, as evidenced by the reduction in mRNA levels of ICAM-1, TNFα, IL-6, MCP-1, and TLR4 in HUVECs. Moreover, Lig pretreatment induces a noteworthy downregulation of TLR4, p-IκBα, and NF-κB p65 expression in HUVECs. Taken together, these findings strongly imply that Lig protects HUVECs against LPS-induced inflammatory damage, which may be mediated through the modulation of the TLR4/NF-B pathway.
Footnotes
Abbreviations
EC: endothelial cell; CD: cardiovascular disorders; Lig: ligustrazine; LPS: lipopolysaccharide; HUVECs: human umbilical vein endothelial cells; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide; ICAM-1: intercellular cell adhesion molecule-1; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; MCP-1: monocyte chemoattractant protein-1; ELISA: enzyme-linked immunosorbent assay; RT-PCR: real-time polymerase chain reaction; TLR4: toll like receptor 4; NF-κB: nuclear factor-κB; p-IκB-α: phosphorylation of inhibitor of NF-κB alpha; AS: atherosclerosis; TLRs: toll-like receptors; PDTC: pyrrolidine dithiocarbamate; FBS: fetal bovine serum; PBS: phosphate-buffered saline; BCA: bicinchoninic acid.
Acknowledgments
This work was supported by the Hubei Science and Technology Planning Project [grant number2015CFC814];2021 Scientific research Plan guiding project of Hubei Education Department [grant numberB2021034] and 2021 Yichang Medical and health Project [grant numberA21-2-035].
The authors express their profound gratitude to all the personnel of the Second People’s Hospital of Yichang City for their invaluable technical support in conducting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Medical and Health Research Project of Yichang (A21-2-034, A21-2-035) and the Science and Technology Research Project of Hubei Department of Education (B2021034).
