Abstract
With the development of the times, electronic cigarettes (e-cigarettes) are being received by more and more people. We compared the different effects of e-cigarettes and tobacco cigarettes on human umbilical vein endothelial cells (HUVECs) treated with the typical e-cigarette aerosol extracts (ECA) and cigarette smoking extracts (CS) sourced from commercial retail stores. HUVECs were treated with different kinds of ECA or CS with different nicotinic concentrations (0.03125, 0.125, 0.5, 2, 8, or 32 μg/mL). Cell viability was examined by the MTT assay. The cell apoptosis was investigated by acridine orange (AO) and Hoechst 33258 staining. The RT-PCR and western blot assays were used to analyze the adhesion molecules and inflammation cytokines released by HUVECs. Furthermore, the intracellular reactive oxygen species (ROS) was observed by fluorescence microscopy. Our data showed that the CS (nicotine concentration at 0.125 μg/mL could decrease the viability of HUVECs by 71%, but not the four kinds of ECA. The apoptotic ratio was about 32.5% in the CS group. No matter the levels of adhesion molecules, inflammation cytokines or ROS, they were higher in CS groups than in ECA groups. Overall, the four kinds of e-cigarettes induced significantly less cytotoxicity than the commercially available tobacco cigarettes in HUVECs. The CS showed the most severe impact on HUVECs. ECA might provide a harm reduction measure, especially in cardiovascular risk, after people switch from tobacco cigarettes to e-cigarettes.
Keywords
Introduction
The use of electronic cigarettes (e-cigarettes), also known as electronic nicotine dispensing systems (ENDS), is steadily increasing in the market. It has been more than a decade since the first generation of e-cigarettes appeared on the market in the European Union in 2006 and in the United States of America in 2007. So far, it has been controversial whether e-cigarettes are less harmful to humans than tobacco cigarettes. 1 Unlike tobacco cigarettes, which need to be burned, e-cigarettes are electronic devices essentially consisting of a rechargeable battery, an e-liquid filled cartridge, a heating element (atomizer) to heat the e-liquid into aerosol, and a mouthpiece. 2 The e-liquids generally consist of glycerin, propylene glycol, nicotine in various concentrations, and different flavors. Much research reported that the aerosol vaporized by the atomizer had no harmful effects.3,7,9 However, some studies also reported that the process of heating atomization could be accompanied by the creation of some new substances, which might be hazardous.4,5 Thus, extensive studies are needed to evaluate the safety and toxicity of e-cigarettes.
At present, cardiovascular disease has become the leading cause of death worldwide. In 2016, an estimated 17.9 million people died from cardiovascular disease, accounting for 31% of all global deaths. 6 In America, it killed approximately 630,000 persons per year. 6 Although there are many factors that could lead to cardiovascular disease, traditional smoking is always the most preventable risk factor for cardiovascular disease. Since the generation of e-cigarettes, there has been a lot of research on the relationship between e-cigarettes and cardiovascular disease. Until now, many preclinical and clinical studies have proved that e-cigarettes have less risk than tobacco cigarette smoking.7,8 After people, particularly females, switched from tobacco cigarettes to e-cigarettes, their vascular health was significantly improved within 1 month. 9 Thus, many researchers consider that the adoption of e-cigarettes might result in an overall benefit for public health.3,7,9 However, a growing body of evidence suggests that e-cigarette use may increase the risk of cardiovascular disease.4,5,10 Actually, the effects of e-cigarettes on cardiovascular physiology are not fully known.
Vascular endothelial cells (VECs) compose the innermost layer of the vessels that are in direct contact with the blood. Due to close contact with various factors in the blood vessels, VECs could respond to changes in the blood such as nutrients, oxygen, and hazardous substances quickly. Through the alterations of adhesion molecules and permeability, VECs regulate the migration of cells and compounds out of the vascular space, which induces cardiovascular diseases, especially atherosclerosis. 11 And nowadays, VECs are recognized as novel immune cells. They play a very important role in both innate and adaptive immune responses. Once the physiological activity of VECs is abnormal, both local and systemic inflammatory responses will be induced, which could finally lead to cardiovascular diseases. 12 Thus, the dysfunction of VECs would be an important initiator of cardiovascular diseases. It has been reported that e-cigarette aerosol extracts (ECA) can induce DNA damage and VEC death. 13 Compared with e-cigarette products (Vype ePen and Vype eStick) and 3R4F cigarettes, e-cigarettes did not inhibit VEC migration. 14 The cytotoxicity of VECs was examined in 11 e-cigarettes vapors from the US and EU market. 15 However, there is still a lack of evidence for the cytotoxicity evaluation of VEC by e-cigarettes and tobacco cigarettes. In particular, different brands of e-cigarettes have different components that would lead to different risks to VECs. Thus, it is necessary to do more research about the cytotoxicity evaluation comparing e-cigarettes with tobacco cigarettes.
This study was aimed to investigate the cytotoxicity effects on VECs of the different typical commercially available e-cigarettes and tobacco cigarettes in China.
Materials and methods
Cell culture
Human umbilical vein endothelial cells (HUVECs) were obtained as described. 16 Cells were cultured in DMEM-H medium (Genview) supplemented with 10% fetal bovine serum (Applied Biosystems) in a humidified incubator at 37°C with 5% CO2. Fresh medium and subculture were replaced according to cell growth and morphology. Cells were used in the 5th to 10th passages. For the HUVEC experiment, cells in logarithmic growth phase were seeded in well plate at a density of 1 × 105 cells/ml. The cell morphology was observed by Nikon phase contrast microscope.
Preparation of e-cigarette aerosol extracts and cigarette smoking extracts
The ECA and CS were prepared by bubbling the RELX e-cigarette aerosol of commercially available filter cigarettes or the commercially available tobacco cigarettes through prewarmed (37°C) cell culture medium without additives. All the ECA and CS were generated according to the Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) Recommended Method No. 81 vaping regime, which specifies a square-wave puff profile, 55 mL puff volume, 3 s duration, and a 30 s interval. The smoking machine was calibrated to the average puff duration for e-cigarette users at a frequency of 10 puffs/hour. 17 For each batch of aerosol, 160 puffs were collected into 20 mL of culture medium without serum. Nicotine analysis of the sample was analyzed using a 1,260 Infinity HPLC-ELSD, equipped with a Sepax AAA column (4.6150 mm, 5 μm). The column temperature was 35°C. Standard or sample solution (10 μL) was injected into the HPLC system, and the flow rate was kept at 1.0 mL/min for 20 min. The detection wavelength was 254 nm. The mobile phase was a mixture of methanol: water = 3:1, and the mobile phase was filtered and degassed by ultrasound prior to testing. The nicotine concentration in the sample was calculated using the standard curve of nicotine liquid phase results. HUVECs were treated with different kinds of ECA or CS with different nicotinic concentrations (0.03125, 0.125, 0.5, 2, 8, or 32 μg/mL).
Determination of cell viability
Cell viability was determined by MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltertrazolium bromide; M5655 Sigma-Aldrich) assay as previously described. 18 Briefly, the HUVECs in logarithmic growth phase were seeded in 96-well plate. Cells were treated with different kinds of ECA or CS with different nicotinic concentrations (0.03125, 0.125, 0.5, 2, 8, or 32) for 44 h and then cells were incubated with 0.5% MTT for 4 h. After removing the medium, 100 μL dimethyl sulfoxide (DMSO) solution was added. The absorbance was measured at 570 nm using a SpectraMAX ABS microplate spectrophotometer (Molecular Devices). The cell viability was calculated by the ratio of OD.
Cell viability (%) = (sample group OD-blank group OD)/(control group OD-blank group OD) × 100%.
Cell death assay
The HUVECs in logarithmic growth phase were seeded in 24-well plate. A lactate dehydrogenase (LDH) assay was performed on HUVECs treated with CS for 48 h using a LDH kit (Nanjing Jiancheng, China) according to the manufacturer’s protocol. Light absorption was measured at 440 nm using the SpectraMAX ABS microplate spectrophotometer (Molecular Devices, USA).
The HUVECs in logarithmic growth phase were seeded in laser confocal petri dishes. Cells were stained with 5 μg/mL acridine orange (AO, Fluka) for 5 min at room temperature. Nuclear condensation and fragmentation were observed by a fluorescent microscope (Nikon Ci-L, Japan).
The HUVECs in logarithmic growth phase were seeded in laser confocal petri dishes. Cells were stained with Hoechst 33258 (10 μg/mL) (Sigma, St Louis, MO, USA 94403) for 15 min at 37°C. Stained cells were washed three times with PBS, and then viewed by fluorescent microscope (Nikon Ci-L, Japan). Cells were scored as apoptotic if their nuclei were much brighter or showed condensed chromatin and nuclear fragmentation.
Enzyme-linked immunosorbent assays
The HUVECs in logarithmic growth phase were seeded in 24-well plate. Cell supernatants were collected after being treated with CS for 24 or 48 h to determine IL-6, IL-8, IL-1, and TNF- concentrations according to the manufacturers' instructions (1,110,602, 1,110,802, 579,409, and 1,117,203, Beijing Dakewei Biotechnology Co, Ltd).
Real-Time PCR
The HUVECs in logarithmic growth phase were seeded in 6-well plate. Total RNA was extracted from the HUVECs using TRIzol (Thermo Fisher Scientific Co, Ltd, USA) at low temperatures. And cDNA was generated using a commercial RT-PCR kit (ABclonal Co, Ltd, Wuhan, China). Then, real-time PCR was conducted using the SYBR Green Quanti Tect RT-PCR kit (ABclonal Co, Ltd, Wuhan, China) and each of the samples was analyzed in triplicate. For each sample, Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal reference. The relative mRNA expression of the target genes was calculated by the 2−∆∆Ct method. The specific primers were for TNF-α, sense 5′-AGCTGGTGGTGCCATCAGAGG-3′, anti-sense 5′- TGGTAGGAGACGGCGATGCG-3′; IL-8 sense 5′-AACTGAGAGTGATTGAGAGTGG-3′, anti-sense 5′- ATGAATTCTCAGCCCTCTTCAA-3′; IL-6 sense 5′- CACTGGTCTTTTGGAGTTTGAG-3′, anti-sense 5′- GGACTTTTGTACTCATCTGCAC-3′; IL-1β sense 5′-GCAGAAGTACCTGAGCTCGCC-3′, anti-sense 5′-CCTTGCTGTAGTGGTGGTCGG-3′; ICAM-1sense 5′-TGCAAGAAGATAGCCAACCAAT-3′, anti-sense 5′-GTACACGGTGAGGAAGGTTTTA-3′; VCAM-1 sense 5′-CAGGCTGGAGATAGACTTACTG-3′, anti-sense 5′-CCTCAATGACAGGAGTAAAGGT-3′ GAPDH sense 5′-GAAGGTGAAGGTCGGAGTC-3′, anti-sense 5′-GAAGATGGTGATGGGATTTC-3′.
Western blot analysis
The HUVECs in logarithmic growth phase were seeded in 6-well plate. Western blot was performed as described previously. 16 Cells were lysed with protein lysate (Beyotime Biotechnology) for 10 min at RT. The protein concentration was determined by the BCA protein assay kit (Beyotime Biotechnology). The cellular proteins (20 μg) were applied to a 12% SDS-polyacrylamide gel and electroblotted onto a polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with 5% (w/v) nonfat dry milk in PBS-Tween 20 (PBST; 0.05%) for 1 h and incubated with anti-ICAM-1 (A5597, ABclonal), anti-VCAM-1 (A0279, Abclonal), or anti-actin (ACTB) antibody (Sigma) at 4°C overnight. After washing in PBST, the PVDF membrane was incubated with appropriate HRP-conjugated secondary antibodies (ABclonal) for 1 h at RT. The immunoreactive bands were chromogenously developed with the use of 3, 3′-diaminobenzidine. The relative quantity of proteins was analyzed by the use of Image J software.
Intracellular ROS assay
The HUVECs in logarithmic growth phase were seeded in laser confocal petri dishes. The levels of intracellular ROS were detected in cells by the use of a fluorescent probe, 2′, 7′-dichlorodihydrofluorescin (DCHF, Sigma, USA). 19 Within cells, the DCHF could be oxidized into fluorescent 2, 7-dichlorofluorescin (DCF) by intracellular ROS. Fluorescence was monitored by a fluorescent microscope (Nikon Ci-L, Japan). The amount of ROS was quantified as relative fluorescence intensity of DCHF per cell in the scanned area. The value of the figure represented the relative fluorescent intensity. The average fluorescence intensity was calculated by the Image-J software.
Statistical analysis
All experiments were repeated at least 3 times independently. The normal distribution was firstly analyzed by SPSS v11.5 (SPSS Inc. Chicago, IL). All the data were expressed as mean ± SEM and analyzed with one-way ANOVA with use of SPSS v11.5 to compare the treatment means when data was normally distributed. The Tukey-Kramer multiple comparison procedure was used for Post-Hoc comparisons. P < .05 were considered statistically significant. Images were processed by the use of GraphpadPrism 5 (GraphPad Software, La Jolla, CA, USA) and Adobe Photoshop CS6 (Adobe, San Jose, USA).
Results
Cell viability assessment
E-cigarette aerosol extracts and CS were collected by using the system in Figure 1(a). Nicotine in CS and ECS was quantitatively detected by HPLC (Supplemental Figure 1). Then, the nicotine in the samples was diluted to 0.03125, 0.125, 0.5, 2, 8, or 32 μg/mL. HUVECs were treated with different kinds of ECA and CS with different nicotine concentrations for 48 h. After treatment with CS with 32 or 8 μg/mL nicotine, respectively, the cell viability decreased to 57.7 or 55.6%, which presented significant differences compared with that of air group (p < .001). In e-cigarette groups with 32 μg/mL nicotine, the cell viability decreased to 64.6% in grape group (p < .01) and 67.4% in mung bean group (p < .05). In e-cigarette groups with eight or 2 μg/mL nicotine, the cell viability decreased to 66.3 or 69.9% in grape groups (p < .05) and there were no differences in cell viability between the mung bean, watermelon, cola groups, and the air group. In groups with the nicotine concentration of less than 0.5 μg/mL, there were no differences in cell viability between all four kinds of e-cigarette groups, and the air group. However, the cell viabilities were still decreased in CS groups, which were 70.6 or 71% in groups with a nicotine concentrations of 0.5 and 0.125 μg/mL (p < .05) (Figure 1(b)). In the groups with nicotine concentration of 0.03125 μg/mL, the cell viabilities were not changed in all treated groups. These results showed that the tobacco cigarette could decrease the viability of HUVECs when the cells were treated with the nicotine concentration higher than 0.03125 μg/mL. The e-cigarette of watermelon and cola with the nicotine concentration of up to 32 μg/mL did not decrease the cell viability. The e-cigarette of grape and mung bean with the nicotine concentration of up to 0.5 or 8 μg/mL did not decrease the cell viability. The IC50 of the tobacco group, grape group, and mung bean group were 71.18, 117.7, and 261.8 μg/mL, respectively. The nicotine concentration of 0.125 μg/mL was selected in the following studies to investigate the differences between e-cigarettes and tobacco cigarettes. The cell viability of HUVECs treated with different concentrations of CS. (a) Scheme of the CS generating machine. For ECA generation, the e-cigarette/t-cigarette ① was attached to the machine. The peristaltic pump ⑤ was activated and draw the cigarette smoke through a chemically inert tube ②, and then into the gas washing bottle ③, where the smoke bubbles through M199 culture medium ④. The culture medium ④ was the CS used for further experiments. (b) HUVECs were treated with different kinds of ECA or CS with different nicotinic concentrations (0.03125, 0.125, 0.5, 2, 8, or 32 μg/mL) for 48 h. The cell viability was detected by MTT assay. (Data are expressed as means ± SEM, #p > .05, *p < .05, **p < .01 and ***p < .001, n = 3).
Cell apoptosis assessment
We then examined whether the decreased cell viability was due to the cell necrosis. Results from the LDH assay showed that there was no significant difference of the LDH release in HUVECs between the control and treatment groups with nicotine concentration of 0.125 μg/mL, which indicated that CS did not promote necrosis in HUVECs (Figure 2(a)). To determine whether the decreased cell viability was due to cell apoptosis, cells were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL. After a 48 h treatment, morphological changes were detected using acridine orange (AO) and Hoechst 33258 staining by fluorescent micrographs. We found that the ECA or CS induced HUVEC nuclear condensation and fragmentation, which are characteristics of apoptosis (Figure 2(b)). The cells showed condensed chromatin and nuclear fragmentation, which were much brighter in the Hoechst 33258 staining and were recognized as apoptotic cells. The proportion of apoptotic cells was about 32.5% in the tobacco group, 16.5% in the grape group, 10.25% in the watermelon group, 2.5% in the cola group, and 14.5% in the mung group (Figure 2(c)). The comparation of e-cigarette and CS on HUVEC apoptosis. HUVECs were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 48 h. (a) LDH release was detected by using the cell supernatant in each group. (Data are expressed as means ± SEM, #p > .05, n = 3) (b) After being treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 48 h, Acridine orange (AO) and Hoechst 33258 staining was did in HUVECs. Arrows indicated apoptotic cells and typical nuclear fragmentation. Scal bar: 10 μm (n = 3). (c) The apoptotic ratio was quantified by Hoechst 33258 staining at 48 h. The cells showed condensed chromatin and nuclear fragmentation which were much brighter in the Hoechst 33258 staining were recognized as apoptotic cells.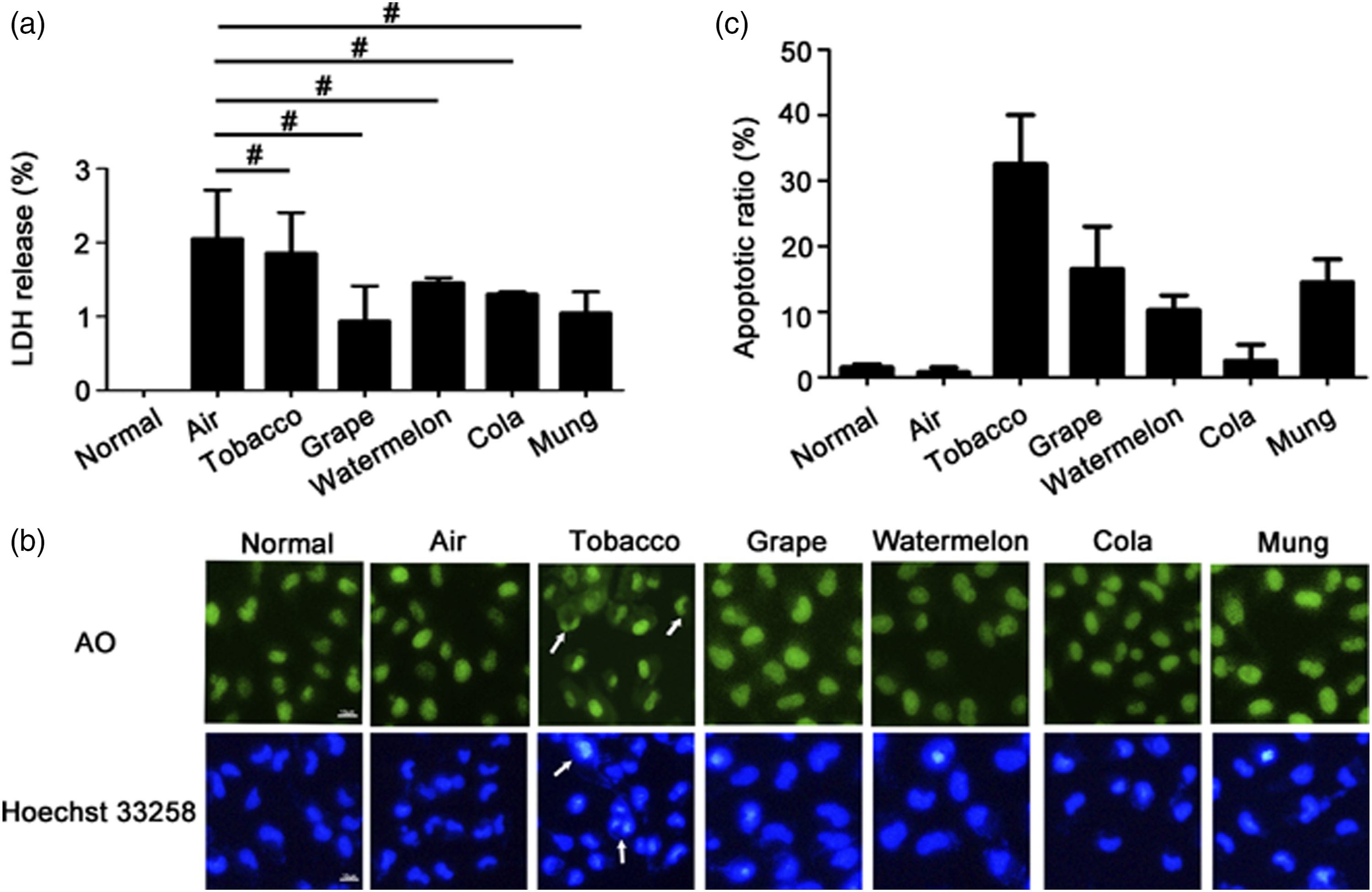
The examination of adhesion molecule levels in HUVECs
Then, we investigated the effects of ECA and CS on inflammatory responses. The levels of adhesion molecules in HUVECs were evaluated. No matter the RT-PCR or the Western blot results showed that levels for the vascular cell adhesion molecule-1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1) were induced in the tobacco groups compared with those in the air groups (Figure 3). After being treated with ECA or CS for 12 h, the mRNA levels of VCAM-1 and ICAM-1 were not increased in HUVECs (p > .05). However, after being treated for 24 h, all four kinds of ECA elevated the mRNA level of VCAM-1 (Figure 3(a)). And the ECA of Grape and Mung elevated the mRNA level of ICAM-1 (Figure 3(a)). Although e-cigarettes increased the levels of VCAM-1 and ICAM-1 after being treated for 24 h, adhesion molecule levels were still much lower in e-cigarette treated groups than in tobacco cigarette groups (Figure 3(a)). The results of the protein levels of VCAM-1 and ICAM-1 were consistent with those of mRNA levels. After being treated with different ECA or CS for 24 h, tobacco cigarettes increased the levels of VCAM-1 and ICAM-1 (Figure 3(b) and (c)). The comparation of e-cigarette and CS on the levels of adhesion molecules in HUVECs. (a) HUVECs were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 12 or 24 h. The mRNA levels of VCAM-1 and ICAM-1 were detected by the RT-PCR assay. (b) HUVECs were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 24 or 48 h. The protein levels of VCAM-1 and ICAM-1 were detected by Western blot assay. (c) Quantification of VCAM-1 and ICAM-1 which were relative to that of ACTB. (Data are expressed as means ± SEM, #p > 0.05, *p < 0.05, **p < 0.01 and ***p < 0.001, n = 3).
The examination of IL-6, IL-8, TNF-α, and IL-1β levels in HUVECs
First, the mRNA levels of IL-6, IL-1β, TNF-α, and IL-8 were examined by RT-PCR. After treated the cells with different ECA or CS for 12 or 24 h, the levels of IL-6, IL-1β, and TNF-α were not changed in all the ECA treatment groups. However, in the CS treatment groups, the levels of IL-6, IL-1β, and TNF-α were significantly increased compared those in air groups (Figure 4(a)–(c)). The levels of IL-8 were increased after cells were treated with tobacco cigarette (p < .01), watermelon ECA (p < .05) and cola e-cigarette (p < .05). However, the IL-8 levels in watermelon and cola e-cigarette groups were much lower than those in the tobacco cigarette group (p < .05) (Figure 4(d)). Thus, compared with the e-cigarette, the mRNA levels of inflammatory factors in HUVECs were induced higher by tobacco cigarettes. The comparation of e-cigarette and CS on the mRNA levels of pro-inflammation cytokines in HUVECs. HUVECs were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 12 or 24 h. The mRNA levels of (a) IL-6, (b) IL-1β, (c) TNF-α and (d) IL-8 in HUVECs were detected by RT-PCR assay. (Data are expressed as means ± SEM, #p > 0.05, *p < 0.05 and **p < 0.01, n = 3).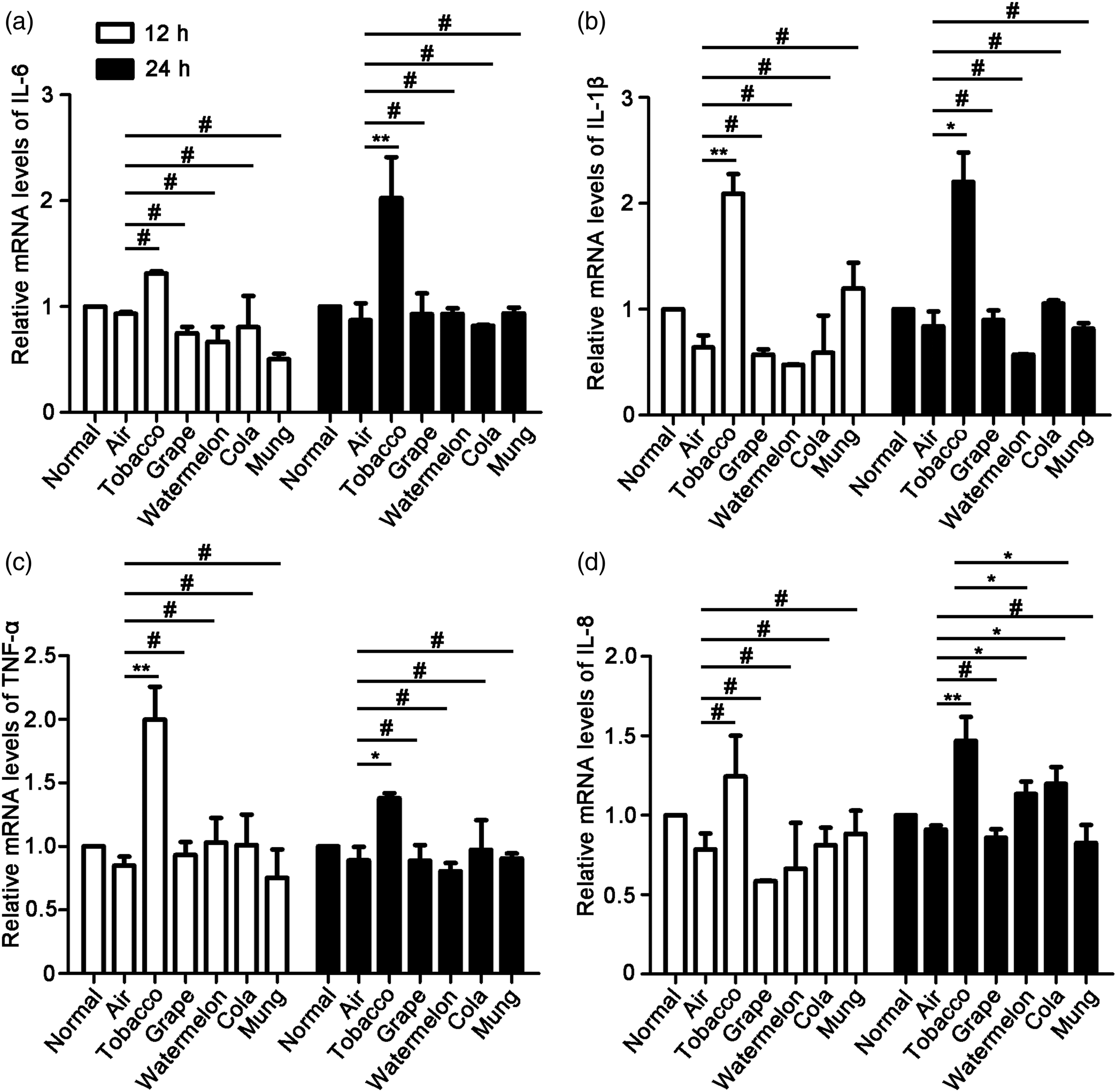
Then, we investigated the protein levels of IL-6, IL-1β, TNF-α, and IL-8 by ELISA assay (Figure 5). After treating the cells with different ECA or CS for 24 h, the levels of IL-1β and IL-8 were significantly increased in the tobacco cigarette treatment groups compared to those in the air groups (p < .05) (Figure 5(b) and (d)). After treating the cells with different ECA or CS for 48 h, the levels of IL-6, TNF-α, and IL-8 were significantly increased in the tobacco cigarette treatment groups compared those in air groups (p < .05, p < .01) (Figure 5(a)–(d)). However, all the four e-cigarette did not increase the levels of IL-6, IL-1β, TNF-α, and IL-8 after treated for 24 or 48 h (p > .05). The comparation of e-cigarette and CS on the protein levels of pro-inflammation cytokines in HUVECs. HUVECs were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 24 or 48 h. The protein levels of (a) IL-6, (b) IL-1β, (c) TNF-α, and (d) IL-8 in HUVECs were detected by ELISA assay. (Data are expressed as means ± SEM, #p > 0.05, *p < 0.05, and **p < 0.01, n = 3).
The comparation of ECA and CS on reactive oxygen species levels in HUVECs
At the physiological level, ROS plays a very important role in the regulation of endothelium function, such as vascular tone maintenance, angiogenesis facilitation, and acute inflammatory responses to pathogen invasion. However, excessive ROS generation in VECs of pathological states would increase endothelial permeability and exacerbate endothelial inflammation. After the cells were treated with different ECA or CS, the intracellular ROS level was detected. We observed the fluorescence of each group with a fluorescence microscope. In the Tobacco group, the fluorescence was much higher than in the air group (Figure 6(a)). The average fluorescence intensity was calculated by the Image-J software. The relative ROS values of Normal group, Air group, Tobacco group, Grape group, Watermelon group, Cola group, and Mung group were 1.99, 2.50, 3.62, 2.85, 2.52, 2.36, and 2.44, respectively. The results showed that only CS incubation of HUVECs induced the ROS level. The comparation of e-cigarette and CS on the ROS level in HUVECs. Cells were treated with all kinds of ECA or CS with nicotine at 0.125 μg/mL for 48 h. (a) The relative intensity of ROS in HUVECs was detected with fluorescence microscope. Scal bar: 10 μm. (b) The quantity of ROS level in groups mentioned above. The value of the figure represented the relative fluorescent intensity per cell determined by laser scanning confocal microcopy. The average fluorescence intensity was calculated by the Image-J software. (Data are expressed as means ± SEM, #p > 0.05 and *p < 0.05, n = 3).
Discussion
The habit of smoking could date back thousands of years. Unlike in the past, smoking was considered a symbol of status and wealth. It is now clearly recognized that smoking is a major cause of many diseases, including tracheitis, lung cancer, hypertension, especially cardiovascular diseases. 20 Nowadays, the prevalence of smoking is very high all over the world. For governments, the economic burden of smoking and its associated diseases is high. 11.5% of deaths worldwide are linked to tobacco use. 21 For all the reasons mentioned above, e-cigarettes are accepted as an alternative to tobacco cigarettes, even as a tool to help tobacco smoking cessation. 22 Since e-cigarettes came onto the market about 20 years ago, their safety has always been studied. It has been revealed that the glucose and lipid metabolism could be disrupted by e-cigarette even without nicotine. 23 The e-cigarette could induce cytotoxicity and pro-inflammatory cytokine release in many kinds of cells, including bronchial epithelial cells, most lung and oral cavity derived cell lines.24–26 Different e-cigarette liquids have different cytotoxic effects because of the different components. Thus, the specific cytotoxicity of e-cigarette vapor is actually not well understood. Extensive studies are still needed to prove the safety of e-cigarettes.
In this manuscript, ECA and CS were used to treat HUVECs, and the concentration of nicotine was taken as the standard. This system consisted of an electronic smoking device to simulate human smoking. After smoking, the cigarette smoke constituents were dissolved in the culture medium to become the aqueous phase. 27 This system could mimic the habits of average smokers, determine the nicotine concentrations, and keep the comparatively constant chemical compositions. Although the system could not be exactly the same as the in vivo situation, especially the lung/blood barrier, the composition, concentration, and distribution of cigarette smoke are very similar to those in the in vivo situation. Thus, ECA and CS taken from this system were recognized and available to investigate the effects on molecules, cells, and tissues.17,28,29 It has been reported that the average nicotine concentrations of smokers in their plasma is 43.7 ng/mL +/− 38. 30 Thus, in this study, the dilutions used for the treatment of cells were upper than 0.03125 μg nicotine/ml. The results showed that at the concentrations of 0.5 and 0.125 μg/mL, the survival rate of CS group decreased to 70.6 and 71%, respectively. At the concentration of 0.03125 μg/mL, there was no significant change in cell viability. Our data suggested that no matter the CS or the ECA at the concentration of 0.03125 μg nicotine/ml did not decrease the cell viability after treated for 48 h. After treating the cells with cigarettes containing more than 0.125 μg nicotine/mL, CS decreased the cell viability significantly, but not ECA. The average nicotine concentration in the plasma was measured in the bodies of long-term smokers. Because the treatment time in this study is much shorter and it was not possible to simulate the status of long-term smoking, a slightly higher concentration than average (0.125 μg nicotine/ml) was chosen. LDH release, AO and Hoechst 33258 staining showed higher apoptosis rates in the CS group compared to ECA groups.
Vascular endothelial cells (VECs) line the innermost layer of vessels in the human body and are in direct contact with the blood. Thus, many diseases, including hematological diseases, metastasis of cancer, immune related diseases, and especially cardiovascular disease, are associated with endothelial dysfunction. 31 Many of the factors that cause atherosclerosis and cardiovascular disease, such as dyslipidemia, pathoglycemia, and smoking could induce VEC apoptosis. 32 At the beginning of atherosclerosis, the expression of adhesion molecules VCAM-1 and ICAM-1 in VECs are increased by risk factors, and the VECs are activated. The secretions of pro-inflammatory cytokines including TNF-α, IL-1β, IL-6, and IL-8 were induced in the activated VECs. 33 Then the high levels of adhesion molecules and pro-inflammatory cytokines induce the expression of cell adhesion molecules (CAMs) and chemokines, which recruit the leukocytes to attach to VECs. The adhesive leukocytes penetrate the endothelium and infiltrate into the intima to become foam cells. 34 Actually, the adhesion of leukocytes is also related with the process of inflammation, infection, and cardiovascular disease. 35 RT-PCR and ELISA data showed that after treated for 24 h, levels of IL-6, IL-1β, TNF-α, and IL-8 were significantly lower in the e-cigarette groups than those in the conventional cigarette group. RT-PCR and Western blot data showed that the levels of adhesion molecules VCAM-1 and ICAM-1 in the e-cigarette groups were much lower than those in the traditional cigarette group. Thus, compared to e-cigarette smokers, tobacco cigarette smokers might be more likely to develop atherosclerosis, inflammation, infection, or cardiovascular disease.
ROS are oxygen-related species of high chemical reactivity, which include oxygen radicals and peroxides. 36 At the physiological level, ROS plays a very important role in the regulation of endothelium function, such as vascular tone maintenance, angiogenesis facilitation, and acute inflammatory responses to pathogen invasion. 37 However, excessive ROS generation in VECs of pathological states would increase endothelial permeability and exacerbate endothelial inflammation. And increased endothelial permeability and inflammation are two major hallmarks of the life-threatening conditions that induce diseases such as acute lung injury, acute respiratory distress syndrome, and sepsis. 38 Additionally, excessive accumulation of ROS could also induce cell senescence, which is a risk factor for the development of hypertension and atherosclerosis. 39 Our data showed that the levels of ROS were much higher in tobacco cigarettes treated group. Thus, based on our data, compared to e-cigarette smokers, tobacco smokers might be more likely to develop diseases induced by excessive ROS production.
Until now, there have been some reports about the effects of e-cigarettes on HUVECs. It has been reported that ECA could increase the ROS level and induce HUVEC apoptosis.13,15 The comparative effects of e-cigarette and tobacco cigarette on endothelial migration were also investigated. 14 However, there is no report about the comparative effects of e-cigarette and tobacco cigarette on endothelial inflammation responses. Additionally, the e-cigarettes from different brands have different ingredients in the liquid, which would induce different effects on HUVECs. The typical e-cigarette brand in China was chosen for this investigation. Our study proved that tobacco cigarette showed the much more severe effects on endothelial cells than the four kinds of e-cigarettes (mung bean, watermelon, cola, and grape). Based on our data, the tested e-cigarettes seem to be safer than the selected commercial tobacco cigarettes. However, the long-time treatment experiments were needed to further clarify the safety evaluation in our next step.
Conclusions
In summary, in this study, we compared the effects of tobacco cigarettes and e-cigarettes on HUVECs. Although e-cigarettes also induced the levels of adhesion molecules and pro-inflammatory cytokines in HUVECs, tobacco cigarettes showed much more severe toxicological data, including lower cell viability, higher apoptotic rate, and higher levels of inflammation and ROS in HUVECs. We examined the effects of conventional cigarettes and e-cigarettes on endothelial cell survival, inflammatory response, adhesion molecule expression, and ROS level. Based on these data, we concluded that in these aspects, e-cigarettes appeared to be less toxic than smoking cigarettes. And e-cigarettes might be a good alternative to tobacco cigarettes. However, this does not mean that e-cigarettes are not harmful at all. In fact, studies on the long-term effects on human health are necessarily required.
Supplemental Material
Supplemental Material - A comparative assessment of e-cigarette aerosol extracts and tobacco cigarette smoke extracts on in vitro endothelial cell inflammation response
Supplemental Material for A comparative assessment of e-cigarette aerosol extracts and tobacco cigarette smoke extracts on in vitro endothelial cell inflammation response by Le Su1, Minghan Zhao, Feifan Ma, Zaiyong An, Qiulin Yue, Chen Zhao, Xin Sun, Song Zhang, Jing Xu, Xingtao Jiang, Kunlun Li and Lin Zhao in Human & Experimental Toxicology.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Taishan leading talent project (Grant number LJNY202015 and tscy20180507); Spring Industry Leader Talent Support Plan (Grant number 2017035 and 2019042); Key R&D Program of Shandong Province (Grant number 2019YYSP019 and 2019QYTPY024); Yantai Development Zone Science and Technology Leading Talents Project (Grant number 2020CXRC4); Science, education and industry integration and innovation pilot project of Qilu University of Technology (Shandong Academy of Sciences) (grant number 2020KJC-YJ01 and 2020KJC-GH10) and National key plan “science and Technology to help the economy” special project and University, government, industry, research Collaborative innovation Fund project (2020-CXY45).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
