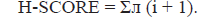Abstract
Background
This study aimed to establish an in vitro model of Alzheimer’s Disease (AD) to investigate the neuroprotective activities of allyl isothiocyanate (AITC) and sulforaphane (SFN).
Materials and Methods
Human neuroblastoma cell lines (SKNAS) were used for the in vitro model of AD after amyloid-β25−35 (Aβ25–35) treatment. Cytotoxicity analysis was performed using the (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) assay. Indirect immunocytochemical methods were used to assess the tau protein, alpha-synuclein (α-synuclein), and β-amyloid distribution in the in vitro model of AD and SKNAS cells.
Results
An in vitro AD model was induced by treatment of SKNAS cells with 1 µM of Aβ25–35 for 48 h. AITC and SFN were applied for 48 h, and the optimal concentrations were determined as 50 µM AITC and 15 µM SFN. Reduced tau immunoreactivity was shown after AITC and SFN administration in SKNAS cells and in vitro models, demonstrating that AITC and SFN prevented amyloid plaque production in the in vitro AD model control group by reducing the β-amyloid level, α-synuclein levels were similar in control and in vitro AD model cells. Reduced α-synuclein levels were observed after SFN treatment in the AD model cells and AITC treatment in the control cells.
Conclusion
It could be concluded that AITC and SFN are potential components as neuroprotective agents against AD.
Key Messages
For the in vitro Alzheimer’s model, 1 μM Aβ₂₅−₃₅ was applied to SKNAS cells for 48 h. It was found that 48 h of 50 μM AITC and 15 μM SFN administration to both control SKNAS cells and Alzheimer’s model cells were suitable for cellular cytotoxicity. It was observed that the tau immunoreactivities, α-synuclein, and β-amyloid concentrations of SFN and AITC applied groups on in vitro Alzheimer’s model cells were lower than the control group cells.
Introduction
Alzheimer’s Disease (AD) is considered one of the most well-known neurodegenerative diseases and the most common cause of dementia. By 2050, the number of patients suffering from AD is expected to be approximately 100 million (Korkmaz & Kaya, 2019). Thus, AD is defined as a public health problem as well as a socioeconomic burden. The pathogenesis of AD is characterized by amyloid beta plaque formation and neurofibrillary tangles (NFTs) that develop due to tau hyperphosphorylation (Jellinger, 2022). Recent studies have emphasized that the aggregation of alpha-synuclein (α-synuclein), a neuronal presynaptic protein, in the brain also plays a crucial role in neurodegeneration and AD pathogenesis (Diao et al., 2013). Senile plaques composed of amyloid beta peptides cause neuronal toxicity, elevated reactive oxygen species (ROS) formation, and oxidative stress cause neurodegeneration (Cheignon et al., 2018). Tau is a microtubule-associated protein that is abundant in neurons, where it binds and stabilizes the microtubules. The interaction of tau protein with the microtubules is impaired in the course of AD, and the accumulation of tau protein in the brain results in neurodegeneration, causing AD (Carolindah et al., 2013).
Inflammation, oxidative damage, insulin resistance, cholesterol, trace elements, metal exposure, and carrying the Apolipoprotein E4 (ApoE) gene are among the risk factors for AD development (Yıldırım & Özcan, 2020). The defense systems of the brain are triggered by toxins and injury, such as lipopolysaccharide (LPS), leading to neurodegeneration. Neurodegeneration is anticipated to be initiated as a result of neuroinflammation. LPS activates inflammation in microglia by an elevated formation and accumulation of inflammatory and cytotoxic mediators like nitric oxide (NO), eicosanoids, arachidonic acid, ROS, prostaglandin E2 (PGE-2) and so on. Moreover, LPS increases the levels of proinflammatory cytokines, all of which are related to neuronal death. Although there is no definitive cure for AD, there are treatments that can change the progression of the disease. A therapeutic approach for the treatment of neurodegenerative conditions and neuroinflammation can be applied by taking control of microglial activation. In addition, treatments are available to reduce oxidative stress, ROS formation, and inflammation (Subedi et al., 2017).
Recently, research efforts have focused on the therapeutic potential of bioactive compounds and functional foods in AD. Studies showed a strong association between organosulfur compounds with anti-inflammatory, ROS-suppressing, and antioxidant properties and the prevention as well as the treatment of AD. Furthermore, studies also suggest that allyl isothiocyanate (AITC) and sulforaphanes (SFNs), which are isothiocyanates formed as a result of the hydrolysis of glucosinolates, have neuroprotective effects (Jaafaru et al., 2018). SFN and AITC are found in Brassica vegetables such as broccoli, zucchini, cabbage, kale, mustard, cauliflower, onions, garlic, horseradish, and turnips (Latronico et al., 2021; Subedi et al., 2017). Although the effects of SFN on AD are not clear, it has been reported that it has a protective role against the progression of AD through mechanisms like inhibition of oxidative stress and prevention of amyloid beta accumulation (Tarozzi et al., 2013). Previous studies also showed that SFN induces proteasome expression in neuroblastoma cells through carbonyl formation and hydrogen peroxide-induced cytotoxicity and reduces amyloid-beta peptide-mediated cytotoxicity, supporting its beneficial effects in AD. ROS has a potential role in neuronal cell death. SFN has been shown to prevent the development of oxidative stress by reducing ROS formation and the mitogen-activated protein kinase (MAPK) phosphorylation by inhibiting caspase-3 enzyme activation (Kala et al., 2018).
On the other hand, AITC has shown potential anti-inflammatory effects by decreasing some inflammatory cytokines (interleukin [IL]-1β, tumor necrosis factor-alpha [TNF-α], IL-6, c-Jun N-terminal kinases [JNK], and nuclear factor kappa light chain enhancer of activated B cells [NF-ĸb]) pathways and activating the nuclear factor erythroid 2-related factor 2 (NRF2) pathway and antioxidant enzymes. The neuroprotective effect of AITC occurs by suppressing neuroinflammation and protecting against oxidative damage via its antioxidant activity (Caglayan et al., 2019). It has been reported that a diet rich in cruciferous vegetables has potential benefits for the prevention of neurological diseases and may be used as a complementary treatment for AD (Latronico et al., 2021).
The presented study aimed to establish an in vitro AD model in order to observe and examine cell viability along with the neuroprotective effects of SFN and AITC on human neuroblastoma cell lines (SKNAS) cells.
Materials and Methods
Cell Culture
A neuroblastoma cell line (SKNAS, ATCC: CRL-2137) was used for the study. The SKNAS cells were cultured in low glucose medium (Capricorn Scientific, low glucose medium), containing 1% penicillin/streptomycin (Capricorn Scientific, PS-B) and 10% heat-inactivated fetal bovine serum (FBS) (Capricorn Scientific, FBS-HI-11B, USA). A humidified chamber of 5% carbon dioxide (CO2) and 95% air at 37°C was used to incubate cells. Developed cultured cells were subcultured using trypsin-ethylenediaminetetraacetic acid solution (0.25%, Capricorn Scientific, TRY-1B).
In vitro AD Modeling with Aβ25−35
Aβ25−35 was used for the AD model and was dissolved in distilled water (1 mM) and allowed to aggregate in an incubator at 37°C for 5 days. SKNAS cells (5 × 10³) per well were seeded into a black 96-well plate. Each well was added 100 µL of culture medium, and Aβ25−35 was applied at two different concentrations (1 and 5 µM) for 24 or 48 h to create the model. Thioflavin T (ThT) dye was used to control amyloid accumulation. ThT was dissolved in PBS, and at the end of 24 and 48 h, 200 µL was seeded in each well. Incubation of the cells took place at 60°C (30 min) and then measurements were obtained using a Microplate Reader (SpectraMax M5 Multi-Mode, USA) at 440–482 nm.
Cell Viability
The (3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyl tetrazolium bromide) (MTT; M2128; 98%, Sigma-Aldrich) assay was used to measure cell viability by making use of the study executed by Hoca et al. (2020). AITC (377430-100G, Sigma-Aldrich, USA) and SFN (574215-25MG, Sigma-Aldrich, USA) solutions were prepared in dimethyl sulfoxide (DMSO) (D8418, Sigma-Aldrich) and diluted in culture medium at 1−50 µM concentrations. SKNAS cells were collected and suspended in a medium; 5 × 10³ cells per well were seeded in plates (96-well). Cells were incubated for 3 days with a culture medium, and 1 µM Aβ25−35 was added to the group that would form the AD model and incubated for 48 h. Only the culture medium was added to the negative control group. AD cells as a study group, SKNAS cells as a control group, and negative control group cells were incubated with various AITC and SFN concentrations for both hours (24 and 48 h) at 37°C in 5% CO2. Following the incubation stage, to each well was added 10 µL of MTT solution for 4 h at 37°C in 5% CO2. Then, DMSO (50 µL) was put in to help dissolve the formazan salts. A spectrophotometer was used to measure the absorbance, and the value was 570–630 nm.
Immunocytochemistry
The study described by Hoca et al. (2020) was used to perform the indirect immunocytochemical method. This method of analysis helps assess the distribution of anti α-syn (sc-53955), anti-β-amyloid (sc-28365), and tau protein (sc-21796) in both cultured SKNAS and AD cells. The staining intensity of proteins was evaluated using the semi-quantitative approach to assign H-SCORE. The calculation of the value has been done with the following equation:
Statistical Analyses
The results of the research were shown as a mean-standard deviation. To identify the main differences among the model groups statistically, a two-way analysis of variance (ANOVA) was applied. For statistically presented data, GraphPad Prism 9 was used, and for accurate post hoc analysis, comparison tests such as Tukey and Sidak were used as appropriate. The p value was accepted as significant when p < 0.05.
Results
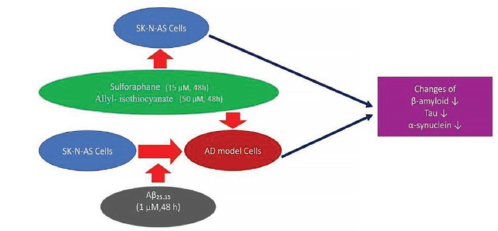
Assessment of the in vitro AD model SKNAS cells were applied at two different concentrations (1 and 5 µM) Aβ25−35 for 24 and 48 h (Figure 1). The ThT dye was used to control the AD model formation. It was observed that amyloid beta formation increased in a time-dependent manner. Therefore, 1 µM Aβ25−35 administered for 48 h was chosen to design this model.
Cell Viability and Evaluation of Growth Assay
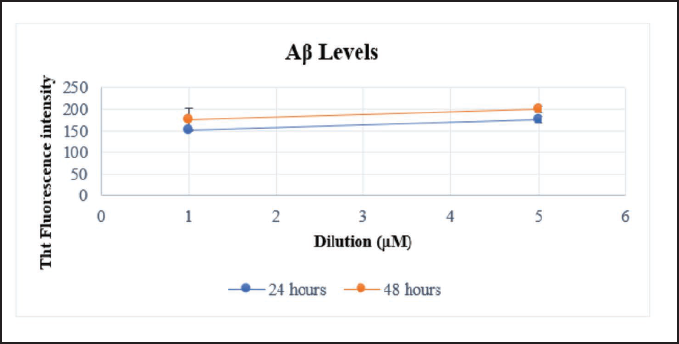
We divided our study groups into the SKNAS and AD cells. There was only culture medium in the negative control group. The SKNAS or Alzheimer’s model neuron cells served as the positive control group. In order to check cell viability, in vitro model of AD cells and SKNAS cell lines were treated with AITC and SFN at five concentrations (1−50 µM) for 24 or 48 h. As a result of the comparison of the in vitro model of AD cells and SKNAS cells, both AITC and SFN were found to produce non-toxic and neuroprotective effects relating to the incubation time and dose. AITC was more effective as the viability of the cells increased when kept in incubation for 48 h compared to the positive control group at 50 µM (Figure 2). Furthermore, cell viability was detected at a 15 µM concentration when treated for 48 h (Figure 2) in the SFN groups.
Cell Morphology
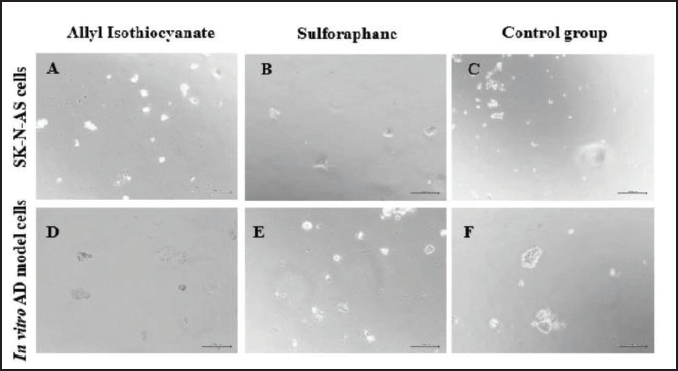
In vitro AD model cells or SKNAS cells exposed to either AITC or SFN were used as study groups. The control group of SKNAS cells and in vitro AD model cells did not receive any treatment. The morphology of SKNAS cells was similar after the administration of either AITC or SFN (Figure 3A and C). While the forms of in vitro model of AD cells incubated with AITC (Figure 3D) or SFN (Figure 3E) were generally indistinguishable from SKNAS cells (Figure 3A and B), retracting, swollen, and round cell morphology was indicated (Figure 3F).
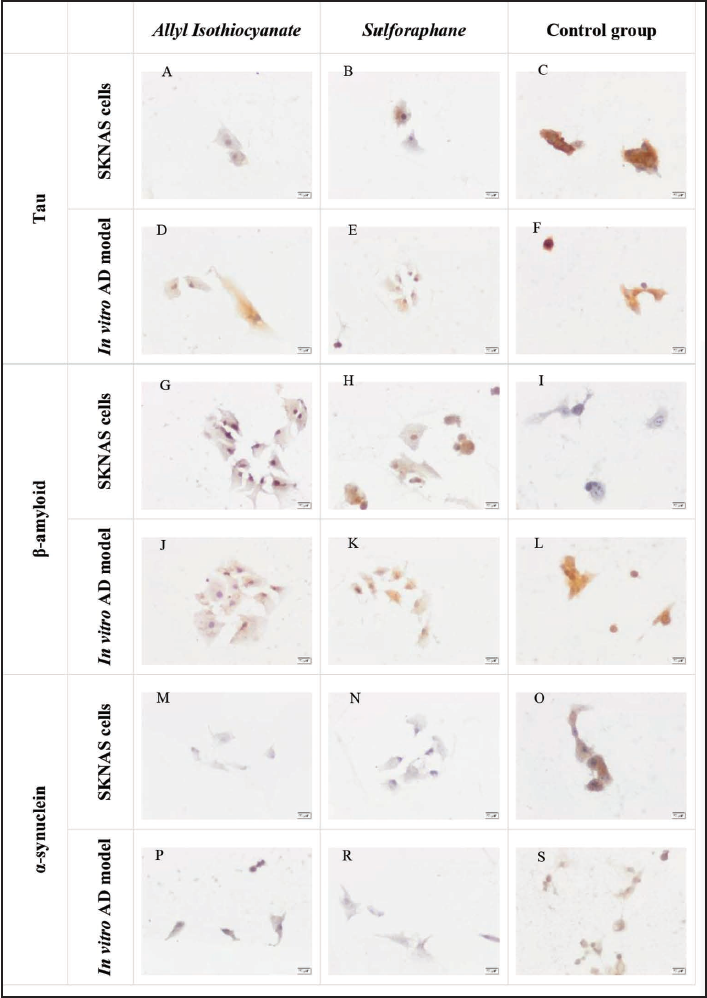
Immunohistochemical Assessment
In SKNAS cells, tau immunoreactivity was found to be weak, moderate, and strong in AITC-applied, SFN-applied, and control groups, consecutively (Figure 4A, B, and C). The dissimilarity among the three different groups was statistically significant (p ˂ 0.05). Therefore, the tau immunoreactivity of AITC-applied and SFN-applied SKNAS cells was significantly less than that of the control group of SKNAS cells (p ˂ 0.05; Table 1). In in vitro model of AD cells, the tau immunoreactivity of the AITC-applied group was weak (Figure 4D), whereas that of the SFN-applied group (Figure 4E) was moderate in comparison to the control group rates (p ˂ 0.05; Figure 4F and Table 1). Moreover, the H-SCORE for tau immunoreactivity showed significantly reduced tau appearance in AITC-applied (p ˂ 0.05) and SFN-applied (p ˂ 0.05) groups than in the control group (Figure 4, Table 1). Strong, moderate, and weak β-amyloid immunoreactivity was observed in AITC-applied (Figure 4G), SFN-applied (Figure 4H), and control groups in the SKNAS cells (Figure 4I). The H-SCORE values for β-amyloid immunoreactivity were 284.22 ± 40.37, 273.57 ± 40.74, and 120.83 ± 25.00 for AITC-applied, SFN-applied, and control groups, respectively (Table 1). The dissimilarity among the different groups was significant statistically (p ˂ 0.05). According to this difference, β-amyloid immunoreactivity was determined to be significantly greater in AITC-applied and SFN-applied SKNAS cells than in the control group of SKNAS cells (p ˂ 0.05; Table 1). Moderate, weak, and strong β-amyloid immunoreactivity was observed in AITC-applied, SFN-applied, and control groups in the in vitro model of AD consecutively (Figure 4J, K, and L). No significant difference was found (Table 1).
Tau, β-Amyloid and α-Synuclein Immunoreactivities in SKNAS and in vitro AD Model Cells (H-SCORE rates) Applied with AITC at 50 μM (48 h), SFN at 15 μM (48 h) and Control Group.
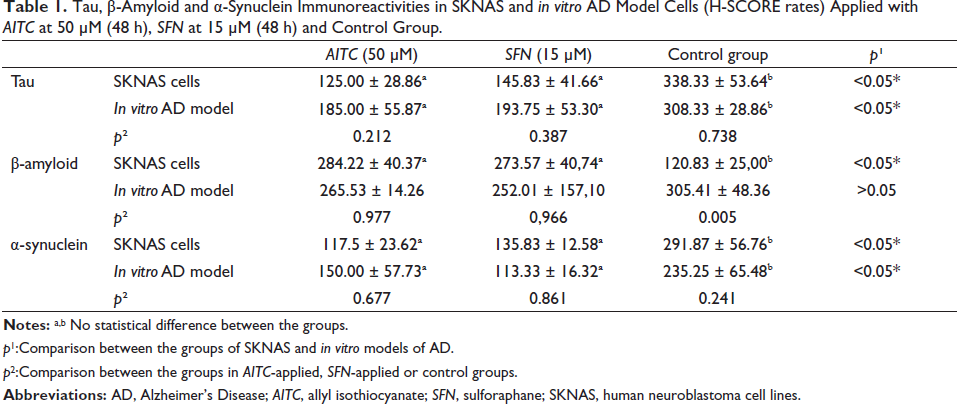
p1:Comparison between the groups of SKNAS and in vitro models of AD.
p2:Comparison between the groups in AITC-applied, SFN-applied or control groups.
In SKNAS cells, α-syn immunoreactivity of the AITC-applied group was found to be weak (Figure 4M), while that of the SFN-applied group (Figure 4N) was found to be moderate when compared to the control group, and the difference was rather significant (p ˂ 0.05). Therefore, α-syn immunoreactivity was significantly reduced in AITC-applied and SFN-applied SKNAS cells than in the SKNAS cells in the control group (p ˂ 0.05; Table 1). In addition, the H-SCORE for SKNAS control cells (Figure 4O) showed higher immunoreactivity for α-syn compared to the in vitro model of AD control cells (Figure 4S). The H-SCORE results for α-syn were 291.87 ± 56.76 and 235.25 ± 65.48 for SKNAS and in vitro AD model control cells, respectively, but the difference between the two groups was considered non-significant (p ˃ 0.05; Table 1).
Moderate, weak, and strong α-syn immunoreactivity were determined in the model cells of the AITC-applied, SFN-applied, and control groups (Figure 4P, R, and S; Table 1). α-syn H-SCORE values of the AITC-applied and SFN-applied groups were not higher than the control group; however, the difference was rather significant (p ˂ 0.05; Table 1). α-syn immunoreactivity between AITC-applied and SFN-applied in vitro AD model cells was less than in the control group of AD model cells, and the difference was statistically significant (p ˂ 0.05; Table 1).
Discussion
Alzheimer’s is one of the slowly progressive neurodegenerative conditions. Presently, there is no effective therapy or cure. The most noticeable pathology that AD can be identified with is the extracellular plaque formation produced through the Aβ accumulation (DeTure & Dickson, 2019). The production of intraneuronal NFTs composed of truncated and hyperphosphorylated cortical neurons’ tau proteins also plays a great part in the development of AD (Palmqvist et al., 2019). On the contrary, it is known that α-syn, which has similar properties with Aβ and tau proteins, causes aggregation and fibril formation, leading to neuronal death. This effect causes oxidative and inflammatory damage to the brain (Tuncel & Arslan, 2018). It is known that there is no effective treatment available to prevent the progression of this disease. Scientific research has shown that natural products have potentially beneficial effects on human health, such as anti-inflammatory, antioxidant, and cytoprotective effects in glial and neuronal cells (Chen et al., 2021). Among natural products, isothiocyanate SFN, derived from the hydrolysis of the glucosinolate glucoraphanin, which can be found in Brassica vegetables, has shown neuroprotective impacts in many in vivo and in vitro studies. Evidence demonstrates that the useful impacts of AITC and SFN could be primarily attributed to their outstanding antioxidant capacity and anti-inflammatory effects. Therefore, SFN and AITC appear to be promising compounds with neuroprotective properties that may play an important role in preventing neurodegeneration (Subedi et al., 2017; Tarozzi et al., 2013). In light of these data, we analyzed the neuroprotective activities of AITC and SFN on SKNAS and AD model cells in our study.
β-amyloid can be used to produce an AD model. Aβ has different isoforms: Aβ (140, 142, 143, 2535). Aβ1−40 and Aβ25−35 have been used to induce oxidative stress in different cell lines and examine the neuroprotective effects of the natural products (Carolindah et al., 2013). For the present study, we used Aβ25−35 to induce the in vitro model of AD, and 1 µM was determined as the optimal dose for 48 h. In another study, Aβ25−35 was used to create an AD model in neuroblastoma cell lines, as in our study, and the effective dosage was decided to be 2 µM administered for 24 h (Xu et al., 2018). It is known that amyloid β accumulation in loose plaques has an integral role in the evolution of AD by causing a chronic state of oxidative stress and inflammation as well as the formation of free radicals and inflammation (Markesbery et al., 2005). Our study showed reduced beta-amyloid levels in in vitro model of AD cells treated with AITC and SFN compared to in vitro AD model control cells. Consistently, in a study led by Bahn et al. (2019) on laboratory mice, SFN administration was shown to reduce the intensity of Aβ plaques.
Tau aggregation and hyperphosphorylated tau are important mediators of AD pathologies. Tau aggregation causes synaptic function loss, activation of microglia, mitochondrial damage, and ultimately neuronal death. Contrarily, it is evident that neuroinflammation mediated by primed microglia cells also contributes to AD pathogenesis. On the other hand, the presence of inflammation, especially the release of proinflammatory cytokines, leads to neuronal loss and tau hyperphosphorylation (Wang et al., 2015). In a study conducted by Bahn et al., phosphorylated tau (pTau) levels were analyzed in SFN and vehicle-applied 3× Tg-AD mice. The authors found an important reduction in pTau levels in the brain tissues following SFN treatment (Bahn et al., 2019). In our study, less tau immunoreactivity was detected after both AITC and SFN treatment in the SKNAS and AD models. Therefore, both agents may have reduced tau accumulation in an in vitro model of AD cells.
It is thought that reducing α-syn production in AD may be a good treatment approach. Thus, reducing the level of α-syn can also diminish the risk of proteins forming oligomers and prevent aggregate formation, which can preserve the vitality and functions of neurons (Çınar et al., 2019). Oxidative stress, excitotoxicity, inflammation, misfolded proteins, and neuronal loss are common characteristics of a wide range of chronic neurodegenerative diseases, such as AD. In a study conducted with a type of isoflavone, it was determined that isoflavone reduces α-syn induced neurotoxicity due to its antioxidant properties and thus has a neuroprotective effect (Pan et al., 2021). AITC, the activated form of glucosinolates present in Brassica vegetables, has shown neuroprotective activity in several experimental studies due to their peculiar ability to activate the Nrf2 /antioxidant response element (ARE) pathway, playing a role in boosting the neuronal natural phase 2 enzyme antioxidant defense system. In a study by Xiang et al. on rats using different doses of AITC and SFN, the protective effects of SFN and AITC were associated with Nrf2 nuclear translocation and upregulated expression of antioxidative systems regulated by Nrf2, including heme oxygenase-1, quinone oxidoreductase (NAD(P)H), and cysteine/glutamate exchange transporter. Thus, both AITC and SFN may be used to protect against neurological disorders (Xiang et al., 2012). In another study using the SH-SY5Y cell line, it was stated that SFN administration increased cell viability, decreased glutathione levels, and also increased antioxidant capacity, resulting in a greater effect than the control group (Marrazzo et al., 2019). In line with the aforementioned studies, the bioactive compounds used in the present study showed a neuroprotective effect by reducing α-syn due to their antioxidant properties. Similar α-syn values were observed in both the control group and the in vitro model in our study. Also, a reduction was found in α-syn levels in both AD model cells and control group cells following treatment with AITC and SFN.
Conclusion
The neuroprotective impacts and cell viability of AITC and SFN were examined in both SKNAS and AD model cells by the application of different concentrations of both compounds. Neuroprotective effects on AD model cells were detected after being applied with AITC and SFN. The data from this study may be useful for future studies where the neuroprotective effects of AITC and SFN can be investigated using various neuroblastoma cells in vitro.
Footnotes
Abbreviations
AD: Alzheimer’s Disease; AITC: Allyl isothiocyanate; SFN: Sulforaphane; SKNAS: Human neuroblastoma cell lines; ThT: Thioflavin T; NFTs: Neurofibrillary tangles; ROS: Reactive oxygen species; ApoE: Apolipoprotein E4; LPS: Lipopolysaccharide; NO: Nitric oxide; PGE-2: Prostaglandin E2; NRF2: Nuclear factor erythroid 2-related factor 2; α-synuclein: Alpha synuclein; Aβ: Amyloid-β; GSH: Glutathione; pTau: Phosphorylated tau; NAD(P)H: quinone oxidoreductase; FBS: Fetal bovine serum; DMSO: Dimethyl sulfoxide.
Acknowledgments
The authors of the article thank Near East University (NEU), Research Center of Experimental Health Sciences, Cell Culture Laboratory.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Scientific Research Project (BAP) unit of Near East University; [SAG-2-024].
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
AD is a progressive neurodegenerative disease that is indicated as one of the most important causes of dementia worldwide. In this study, the neuroprotective effects of SFN and AITC were investigated using different concentrations on both SKNAS cells and in vitro Alzheimer’s model cells. For the in vitro Alzheimer’s model, ThT staining with 1 µM Aβ25–35 was applied to SKNAS cells for 48 h. It has been determined that 48 h of administration of 15 µM SFN and 50 µM AITC to SKNAS cells and in vitro Alzheimer’s model cells are suitable for cellular viability. It was determined that the tau immunoreactivities of the groups treated with SFN and AITC on in vitro Alzheimer’s model cells were lower than those of the control group cells. Although β-amyloid immunoreactivity was found to be lower in in vitro Alzheimer’s model cells treated with SFN and AITC compared to the control group, this difference was not statistically significant. It can be emphasized that α-synuclein immunoreactivities were lower in the groups treated with SFN and AITC on in vitro Alzheimer’s model cells compared to the control group cells. In conclusion, SFN and AITC are sulfur compounds, and it is thought that they may have positive potential as neuroprotective agents in Alzheimer’s model cells in vitro.