Abstract
Background
Caffeic acid (CA) or 3,4-dihydroxycinnamic acid is a polyphenolic compound primarily found in coffee, herbs, berries, and other fruits. Its antioxidant, anti-inflammatory, and immunomodulatory effects on multiple health conditions have been evaluated and reported. CA’s anti-tumor effect has been reported in solid tumors, but evidence regarding liquid tumors such as multiple myeloma (MM) is limited. The increasing incidence of MM globally provides a justified rationale to explore the potential of CA on human MM cells through caspase-dependent induced apoptosis.
Objectives
The study explores CA’s therapeutic effect and mechanism on multiple myeloma.
Materials and Methods
We performed flow cytometry at different concentrations and time points after treating human MM cell lines (MM.1R, RPMI8226, and U266) with CA to identify apoptosis and changes in mitochondrial membrane potential. A Western blot was used to assess the expression of an apoptosis-related protein in MM cell lines.
Statistical Analysis Used
Student’s t-test was used to evaluate the mean difference between the experimental group and the control group.
Results
CA markedly induced the apoptosis of MM cells in a dose- and time-dependent manner. After co-incubation with CA, JC-1 (a cationic lipid fluorescent dye was used as an indicator of mitochondrial transmembrane potential), flow cytometry results showed that the mitochondrial membrane potential of MM cells significantly decreased, and the Western blot showed activation and cleavage of caspase-3, which is the classical marker of the mitochondrial apoptosis pathway. The experimental group was statistically significant compared with the control group (p < 0.01).
Conclusion
Our research demonstrated that CA induced MM cell apoptosis through disturbing mitochondrial membrane integrity, followed by caspase-3 splitting, and suggested that CA might have tremendous therapeutic potential for MM treatment.
Key Messages
This study showed that caffeic acid (CA) significantly induces human MM cell apoptosis. The mechanism of the action exerted by CA is to activate caspase-3 and decrease mitochondrial function in MM cells. These results suggested that CA could be a potential therapeutic agent against multiple myeloma.
Introduction
Multiple myeloma (MM) is a kind of plasma cell malignancy that is virtually incurable and represents more than 10% of all hematologic malignant tumors (Zhou et al., 2021). The pathogenesis of MM remains unclear, and possible risk factors may be related to radiation and industrial or chemical toxins (Marin et al., 2019; Rodriguez-Otero et al., 2021). Genetic factors, chromosomal abnormalities, tumor microenvironments, and gene mutations have been identified as some of the causes of MM, but no specific chromosomal abnormalities have been found (Rodriguez-Otero et al., 2021). The abnormal proliferation of monoclonal plasma cells characterizes MM. Abnormal plasma cell accumulation, accompanied by overproduction of monoclonal immunoglobulins or light chains, causes severe consequences, including bone destruction, anemia, hypercalcemia, Bence Jones proteinuria, and chronic renal disease (Kyle & Rajkumar, 2008). The median overall survival of patients with MM is nearly 5 years (Lonial et al., 2015). Recently, the survival rate was improved by applying new therapeutic agents, including proteasome inhibitors, immunomodulatory drugs, monoclonal antibodies, and hematopoietic stem cell transplantation (SCT). However, long-term treatment results in the emergence of drug-resistant MM clones, especially in patients with adverse FISH cytogenetics. Multiple myeloma is an incurable disease that eventually relapses after enough time in remission and causes death, making it necessary to research new therapeutic drugs (Fulciniti et al., 2017).
MM progression was associated with decreasing cleaved caspase-3, which suggests an inhibition of apoptosis (Trotter et al., 2016). Studies also showed that the downregulation of caspase-3 resulted in increased cell viability and mobility, accelerated cell cycle progression, and reduced the number of apoptotic cells (Marin et al., 2019; Zheng et al., 2021).
CA is found in a variety of plants, and it is a polyphenolic antioxidant compound. CA has antioxidant, anti-inflammatory, immunomodulatory, and anticancer effects (Tyszka-Czochara et al., 2017). The anticancer properties of CA are associated with its antioxidant and pro-oxidant capacity, attributed to its chemical structure that has free phenolic hydroxyls, the number and position of OH in the catechol group, and the double bond in the carbonic chain (Espíndola et al., 2019). Studies have demonstrated that CA has anti-tumor effects on human cervical cancer (Chang et al., 2010), colon cancer (Murad et al., 2015), breast cancer (Kabała-Dzik et al., 2018), tongue squamous cell carcinoma (Celińska-Janowicz et al., 2018), and non-small-cell lung cancer (Min et al., 2018). Apoptosis of colon cancer cells caused by CA might be caspase-dependent, as reported by Sim et al. (2019). Pelinson et al. (2019) found increased gene expression of caspases 1, 3, and 8 in solid tumor cells (melanoma cancer cells) after the treatment with CA, which indicated that CA-induced cell apoptosis might be caspase-dependent.
Numerous anticancer approaches are dependent on apoptosis induction in cancer cells to contract the tumor cell’s size. Executioner caspases play a crucial role in the apoptosis of cancer cell tumors, specifically caspase-3, which has proteolytic activity and causes cell death (Boudreau et al., 2019). However, minimal studies exist in the context of liquid tumors and caspase-3’s role in inducing apoptosis in liquid tumors. Based on the available study results, the current study explores the role of CA in inducing apoptosis in MM cells through a caspase-3-dependent pathway.
Materials and Methods
Cell Culture and Reagents
Three commonly used human myeloma cell lines, such as MM.1R, RPMI8226, and U266, were purchased from ATCC (Manassas, VA, USA). Cells were grown in RPMI-1640 medium (CF0001, Sparkjade Science Co., Ltd., China) with 10% fetal bovine serum (FBS, 04-001-1ACS, Biological Industries, Kibbutz Beit Haemek, Israel) and 100 U/ml of penicillin and 0.1 mg/ml of streptomycin (SV30010, HyClone, Logan, UT, USA). They were cultured in a humidified atmosphere containing 5% CO2 at 37℃. CA is a polyphenolic antioxidant compound purchased from Sigma-Aldrich (St. Louis, MO, USA).
Flow Cytometric Analysis of Apoptosis
MM.1R, RPMI8226, and U266 cells were co-incubated with different concentrations of CA (100, 200, 400, and 800 µM) and seeded into 6-well plates at a density of 1 × 106/well, respectively. The co-incubation with CA was kept for 48 h, and readings were taken at 12, 24, and 48 h. Following the instructions, we used flow cytometry (FCM) to detect the apoptotic rate through an Annexin V-PI apoptosis assay kit (KeyGEN BioTECH, Jiangsu, China). After the suspension cell was harvested, the cells were washed twice with 2 mL of 0.01M phosphate-buffered saline (PBS). The collected cells (approximately 3 × 105 cells) were resuspended in 500 µL of binding buffer. 5 µL of Annexin V/FITC solution and propidium iodide each was added, and cells were cultured at room temperature of 25°C and away from light for 15 min and then detected immediately. A BD FACSAria III flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA) was used to analyze the samples.
Mitochondrial Membrane Potential Analysis
JC-1 apoptosis detection kit (KeyGEN BioTECH, Jiangsu, China) was used to assess mitochondrial membrane potential (MMP) change. When the MMP level is high, JC-1 mainly exists in polymer aggregates and shows red fluorescence; however, when the MMP level decreases, it primarily exists as a monomer and produces green fluorescence. MM.1R, RPMI8226, and U266 cells were sown in 6-well plates (3 × 105 cells/well) at a time and co-incubated with 400 and 200 µM CA for 24 h separately. The suspension cells were aspirated, washed twice, and resuspended in 500 µL of ready-prepared JC-1 solution. Following a further incubation for 20 min, the cells were collected and washed twice with 1 mL of 1× diluent incubation buffer. Finally, we resuspended the solution in 500 µL of 1 × incubation buffer and evaluated it by FCM.
Western Blot Analysis
After treatment with different concentrations of CA (400 and 800 µM for RPMI8226 cells, 200 and 400 µM for U266 cells) for 24 h, the cellular protein was extracted following the BCA Protein Assay Kit (KeyGEN BioTECH, Jiangsu, China) protocol. Equal amounts of protein were loaded onto SDS-PAGE (Beyotime, Shanghai, China) and then transferred onto a PVDF membrane (Millipore, UK). The expression of caspase-3 at the protein level was detected by Western blot according to the antibody instructions. Briefly, the membrane was incubated with β-actin (rabbit polyclonal, 1:1000, Abcam) and caspase-3 (mouse polyclonal, 1:1000, CST) primary antibodies overnight at 4℃. After being blotted with secondary antibodies, signals were observed with enhanced chemiluminescence (ECL).
Statistical Analysis
All FCM detections were performed in three independent experiments. GraphPad Prism 6 software (La Jolla, CA, USA) was used for analyzing all statistical data. A Student’s t-test analyzed the results. The data were presented as mean ± SEM. Statistical significance is set at P < 0.05.
Results
Caffeic Acid-induced Apoptosis in Human Myeloma Cell Line
To investigate whether CA could induce apoptosis in MM cells, we used the Annexin V-PI kit to detect apoptotic cells after treatment with CA. The apoptosis ratio treated with CA increased in a dose- and time-dependent manner against the standard control (Figure 1). The data in Figure 1A show that the percentage of apoptotic cells increased significantly after 24 and 48 h compared with 12 h treatment with CA in MM.1R cells. With the increase of concentration from 100 to 800 µM, the number of apoptotic cells also increased at 24 h (p < 0.01) compared to the control group. The apoptosis ratio in MM.1R cells after 12 h was increased by 13.2% ± 1.4%, 14.3% ± 0.4%, 21.4% ± 3.3%, and 41.6% ± 0.4% for 100, 200, 400, and 800 µM, respectively, while apoptosis ratio in RPMI8226 cells also increased with the increase of concentration by 20.4% ± 0.5%, 21.6% ± 2.5%, 31.9% ± 3.2%, and 90.3% ± 1.7% at 100, 200, 400, and 800 µM, respectively. With increased time, apoptotic cells increased at 12, 24, and 48 h in RPMI8226 cells. When the concentration of caffeic acid was 400 µM, there was statistical significance at 12, 24, and 48 h compared with the control group (p < 0.01). The IC50 was 440 ± 30 µM, 410 ±10 µM, and 170 µM, respectively (Figure 1B). The same trend was observed in U266 cells after 12 h of treatment, and the apoptosis ratio was 7.3% ± 0.4%, 18.1% ± 0.4%, 72.2% ± 3.5%, and 86.8% ± 0.6%, respectively (Figure 1C). CA had a more prominent effect, especially at 400 and 800 µM. CA-induced apoptosis in U266 cells at 200, 400, and 800 µM was statistically significant compared to the control group (p < 0.01) (Figure 1C). After exposure to different concentrations of CA for 12, 24, and 48 h, the number of apoptotic cells in U266 increased over time. The proportion of apoptotic cells added to the IC50 was 320 ± 10 µM, 260 µM, and 220 ± 10 µM at 12, 24, and 48 h.
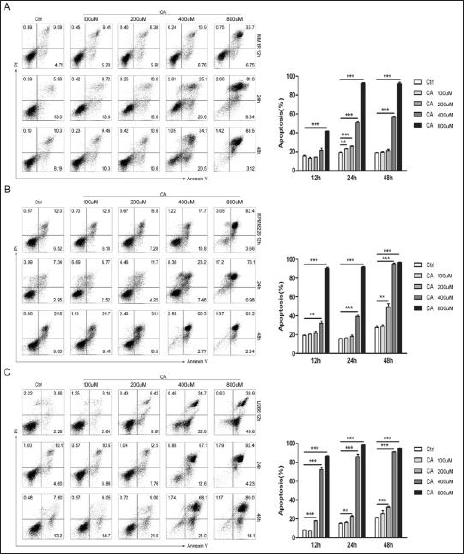
Caffeic Acid Changes the MMP of Apoptotic Cells
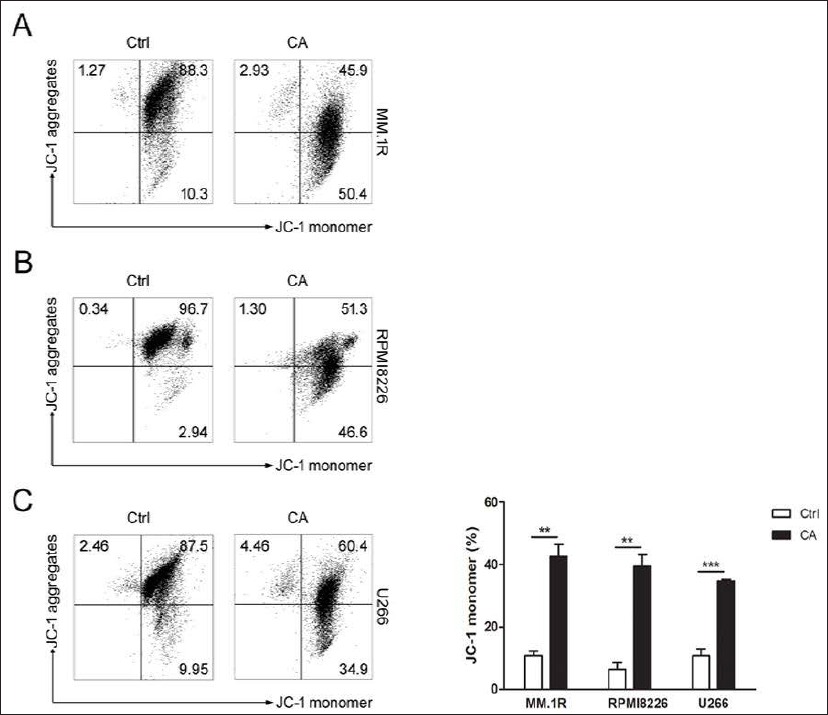
To confirm the influence of CA on MMP, MM.1R and RPMI8226 cells were treated with 400 µM CA for 24 h, respectively, while U266 cells were co-incubated with 200 µM CA. JC-1 FCM analyzed the changes in MMP. As shown in Figure 2, CA caused a remarkable decrease in MMP in the experimental group compared to the control group (p < 0.01).
Caffeic Acid-induced Activation of Caspase-3
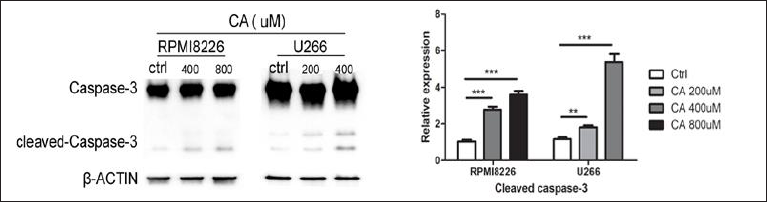
To research the mechanism of CA-induced apoptosis in MM cells, we used Western blotting to detect the expression of caspase-3. After co-incubation with 400 and 800 µM CA for 24 h, the expression of caspase-3 decreased, and the level of cleaved caspase-3 was sharply increased compared to the control group in RPMI8226 cells, as shown in Figure 3. The experimental group was statistically significant compared with the control group (p < 0.01). The same trend was shown in U266 cells after interactions with 200 and 400 µM CA for 24 h. These results suggested that CA-induced apoptosis of MM cells occurred through activating caspase-3.
Discussion
MM is categorized under plasma cell malignancy, characterized by proliferated monoclonal plasma cells in the bone marrow. As a result, there is an increase in monoclonal plasma proteins, bone displacement, and displacement of other hematopoietic cell lines. MM accounts for approximately 1% of all cancers (Kazandjian, 2016). The incidence of MM is related to many factors, such as chromosomal abnormalities, oncogene activation, tumor suppressor gene inactivation, and IL-6-dependent cytokine network disorders (Zhou et al., 2018). Modern drug therapy includes proteasome inhibitors such as ixazomib and bortezomib, immunomodulatory drugs such as lenalidomide and pomalidomide, and the anti-CD38 monoclonal antibody daratumumab (Yee & Raje, 2018). Despite the development of different chemotherapeutic agents, disease relapse and resistance warrant the exploration of new therapeutic agents. CA has been mostly explored for its anti-tumor properties for solid tumors, with limited studies on liquid tumors. The current study evaluated CA’s potential against human myeloma cells via apoptosis through a caspase-dependent pathway. The study found significant apoptosis of human myeloma cells in a caspase-dependent pathway. The study results implicate that CA could be an excellent therapeutic agent in treating liquid cancer; however, a more mechanistic study is warranted.
Apoptosis is a mechanism for maintaining homeostasis in body tissue and a process of programmed cell death that occurs during development and senescence (Saiki et al., 2021). It is an irreversible and energy-dependent cascade reaction. So far, studies have shown that there are two major apoptotic signaling pathways, namely, the intrinsic pathway and the extrinsic pathway. The extrinsic signaling pathway is activated by transmembrane death receptors (members of the TNF receptor family) receiving external stimuli. The mitochondrion initiates the intrinsic signaling pathway. Signals are produced in the cell and act directly on the targets, leading to a change in mitochondrial membrane permeability, contributing to the loss of MMP and the release of cytochrome C, a pro-apoptotic protein, into the cytoplasm (Elmore, 2007). Mitochondrial membrane permeability is associated with apoptosis induction. Studies have shown that a decline in MMP is an early manifestation of apoptosis, which occurs before changes in cell morphology (You et al., 2018). Studies have indicated that CA decreases the MMP and has a high level of cytotoxicity against HL-60 cells (Skała et al., 2018). Our results showed a remarkable decrease of MMP in 400 µM CA-treated MM.1R, RPMI8226 cells, and 200 µM CA-treated U266 cells after incubation for 24 h, indicating that CA damages MM cell mitochondrial membrane integrity and that cell apoptosis induced by CA is at least in a part mitochondrial pathway dependent. Increasing reactive oxygen species (ROS) leads to the loss of MMP. ROS restrains Bcl-2 expression and increases Bax expression, which induces cytochrome C release. An apoptosome is composed of cytochrome C bound to apoptosis protease activating factor-1 (Apaf-1) and procaspase-9. Activated caspase-9 can activate other effector caspases, including caspases 3 and 7, which cause cell death (Pistritto et al., 2016). Both the extrinsic signaling pathway and the intrinsic signaling pathway ultimately converge to activate and cleave the effector caspase-3, resulting in cellular biochemical features and morphological changes of apoptosis (Ding et al., 2021; Elmore, 2007; Rajendra Prasad et al., 2011). Fibrosarcoma (HT-1080) cells treated with CA show an increase in intracellular ROS, which may induce DNA fragmentation and apoptosis of tumor cells (Rajendra Prasad et al., 2011). It has been demonstrated that gene expression of caspase-1, caspase-3, and caspase-8 is increased in melanoma cancer cells with CA treatment (Pelinson et al., 2019). The increased caspase-9, caspase-8, and caspase-3 proteins and cleavage of PARP in CA-treated HL-60 cells imply that it may induce apoptosis through the activation of caspases (Skała et al., 2018; Yen et al., 2018). Our Western blot analysis results indicate that the CA-induced activation of caspase-3 suggests activation of the apoptosis effector.
Conclusion
Our study suggests that CA induces apoptosis in MM cells in a dose- and time-dependent manner. It also changes the MMP in MM cells. This is the first time we have studied the role of caspase-3 in inducing apoptosis in liquid tumors. We find that CA activates caspase-3 and increases the level of cleaved caspase-3 in MM cells. Based on the available study results, we speculate that CA induces apoptosis of MM cells through the caspase-dependent pathway. However, CA’s molecular mechanisms, whether other pathways were involved in cell apoptosis, and its effect on MM in vivo require further study. In short, CA provides a new idea for the clinical treatment of multiple myeloma.
Footnotes
Summary
Caffeic acid found potential anti-cancer activity against human multiple myeloma cells.
Abbreviations
CA: caffeic acid; ECL: enhanced chemiluminescence; FCM: flow cytometry; MM: multiple myeloma; MMP: mitochondrial membrane potential; ROS: reactive oxygen species; SCT: stem cell transplantation.
Acknowledgments
The authors would like to thank Editage (
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Funding
This study was supported by the Rongxiang Regenerative Medicine Foundation of Shandong University (Grant no. 2019SDRX-05), the Shandong Science and Technology Development Plan on Medicine and Hygiene in China (2017WS656), and the Key Research and Development Program of Jining City (2018SMNS008).
