Abstract
Background
Selenium (Se) plays a key role in controlling several processes in plant development, such as root growth and regulation of enzyme activity. However, research on the effects of Se in Sophora tonkinensis, a medicinal plant, has not been reported. To elucidate the effect of Se on the accumulation of secondary metabolites in tissue-cultured seedlings of S. tonkinensis.
Materials and Methods
Tissue culture seedlings of S. tonkinensis was treated with different concentrations of Se in the rooting medium. Agronomic traits of roots and concentrations of hormones and some secondary metabolites were measured from seedlings.
Results
The results show that (1) root growth of tissue culture seedlings were stimulated by Se; (2) a specific amount of Se enhanced the activities of peroxidase (POD) and superoxide dismutase (SOD), as well as the production and storage of soluble proteins and chlorophyll; and (3) Se treatment lowered brassinolide (BR) content, and 15 mg/L Se minimized abscisic acid (ABA) content, while 60 mg/L Se enhanced ABA content. The gibberellin (GA3) content in seedlings increased significantly compared with the control under the treatment of 60 mg/L Se.
Conclusion
Se concentration of 30 mg/L facilitated matrine and oxymatrine production and accumulation in stem/leaf tissues.
Introduction
Selenium (Se) is an element essential to some bacteria, some green algae, and mammals; however, it is not an essential element for higher plants (El-Ramady et al., 2015). Ideal Se concentrations are beneficial; as the concentration increases, Se begins to inhibit growth or the formation of endogenous hormones, as well as induce stress on plant growth (Terry et al., 2000). Therefore, researchers have more recently focused on testing the exposure of plants to different concentrations of Se to determine plant responses; they report widely varying abilities of plants to absorb and accumulate Se (Brown & Shrift, 1982; Rosenfeld & Beath, 2013; Trelease & Beath, 1949). Additional reports have demonstrated that Se causes changes in secondary metabolites in plants (Ahmad et al., 2016; Handa et al., 2019).
There are reports that Se can alleviate drought stress on tobacco (Nicotiana tabacum) growth (Han et al., 2022). Chu proposed that Se treatment can increase the production of wheat (Triticum aestivum) (Chu et al., 2013). Se can alleviate the toxic effects of many heavy metals (Bernat et al., 2014; Kumar et al., 2012; Qing et al., 2015; Saidi et al., 2014; Thiruvengadam & Chung, 2015; Zembala et al., 2010). Studies have shown that moderate amounts of Se remove cellular reactive oxygen species (ROS) by direct or indirect regulation of antioxidant activity (Huang et al., 2018; Wang et al., 2014). Suitable amounts of Se can decrease superoxide anion content in the roots of Vicia spp. and enhance the activity of glutathione peroxidase (POD) to detoxicate plants of lead (Mroczek-Zdyrska & Wójcik, 2012).
S. tonkinensis is a medicinal plant commonly used in China that is mainly found in karstic-rocky hills in southern China. Considering the high content of Se in the soil in the growing area of S. tonkinensis, Se may have a certain impact on its growth, development, and secondary metabolism. However, research on S. tonkinensis mainly reports its chemical composition, properties, pharmacological and toxicological effects, active ingredients, and so on. There are no reports on Se and its effects on tissue culture seedlings. This experiment determined an ideal Se concentration for improving growth, physiological and biochemical measures, endogenous hormone production, and major secondary metabolite accumulation in S. tonkinensis seedlings and proposed a theoretical basis to improve the culture ofS. tonkinensis.
Materials and Methods
Materials
S. tonkinensis seedlings were provided by the Guangxi Key Laboratory of Medicinal Resources Protection and Genetic Improvement at the Guangxi Botanical Garden of Medicinal Plants. The materials were collected from the botanical garden in Nanning, China, and were introduced from wild materials found in Jingxi city of Guangxi to the Botanical Garden. The tissue culture seedling of S. tonkinensis was obtained from the in vitro library of the botanical garden. In the experiment, stem segments with buds were collected from tissue culture materials and put into the experimental medium as new explants.
Se Treatment and Experimental Setup
Tissue culture of S. tonkinensis was carried out with different Se concentrations (0, 15, 30, 45, and 60 mg/L), and the agronomic traits of the roots, the physiological and biochemical indices, and the content of medicinally active ingredients were determined after the tissue culture seedlings had grown for 45 days. The concentrations of Se were set in the medium by adding different doses of plant nutrient solution containing Se amino acids (Se content: 1,500 mg/L, provided by the Guangxi Zhuang Autonomous Region Center for Analysis and Test Research). All tissue cultures were grown in bottles, and there were 4–6 seedlings in each bottle.
Identification of the Agronomic Traits of Roots
The root length and root number of each bottle of tissue culture seedlings were recorded, and the rooting rate was calculated.
Determination of Enzyme Activities
After harvesting, the seedlings were weighed, labeled, and sealed in transparent plastic bags to be vacuumed, rolled into cylinders, and placed in liquid nitrogen for 5 min before storage at −80°C. The seedlings for the determination of enzyme activities were removed and immediately frozen in liquid nitrogen. Then, the seedlings were ground and transferred to phosphate buffer (pH = 6.8) with polyvinyl pyrrolidone (PVP). The supernatant-containing enzyme was obtained after centrifugation at 14,000 rpm for 10 min. POD activity was measured by monitoring the increase in absorbance at 470 nm in phosphate buffer (pH = 6.8) with guaiacol. The protocol refers to the method proposed by Pütter (1974). Fifty microliters of supernatant were added to 3 ml of guaiacol solution. Immediately after stirring, absorbance measurement was carried out at a wavelength of 470 nm, and the 0 s, 30 s, and 60 s absorbance values were recorded.
Superoxide dismutase (SOD) activity was measured based on its performance in inhibiting the photochemical reduction of nitro blue tetrazolium (NBT). The protocol refers to the method proposed by Durak (1993). A 1.3-ml reaction system (0.3 ml of 0.13 mol/L methionine, 0.3 ml of 0.75 mmol/L NBT, 0.3 ml of 0.1 mmol/L ethylenediaminetetraacetic acid-Na2, 0.3 ml of 0.02 mmol/L riboflavin, and 0.1 ml supernatant) was used to react under light at 30°C for 30 min, and then the absorbance of the system was measured at 560 nm to evaluate the reaction.
Glutamic oxaloacetic transaminase (GOT) activity was measured on its performance in producing pyruvic acid in GOT substrate solution (0.0292 g α-ketoglutaric acid (KCTS), 2.66 g DL-aspartic acid, adjusting the pH = 7.0 with 10 mol/L NaOH, and fixing the volume to 100 ml with 0.1 mol/L phosphate buffer (pH = 6.8)). The protocol refers to the method proposed by Tsuchida et al. (1990). A 0.1-ml supernatant was added into the 0.5-ml substrate solution, and then the whole reaction system was bathed at 37°C for 30 min. A total of 0.5 ml of 1 mol/L 2,4-dinitrophenylhydrazine was added to the reaction system and incubated at 37°C for 20 min to measure the pyruvic acid content. The reaction system was terminated by adding 50 ml of 0.4 mol/L NaOH. The results of the reaction were obtained by measuring the absorption at 520 nm.
The glutamic-pyruvic transaminase (GPT) activity measurement method is similar to the GOT activity measurement. The only difference was that 2.66 g of DL-aspartic acid was replaced by 1.78 g of DL-alanine in the GPT substrate solution, and the remaining experimental method was consistent with the GOT activity measurement.
Measurement of Lipid Peroxidation
According to Wang and Huang’s method (Wang & Huang, 2015), the damage degree of the cell membrane was estimated by measuring the content of malondialdehyde (MDA). For this measurement, the control tube contained 2 ml of 0.1 mol/L phosphate buffer, 2 ml of 5% (w/v) trichloroacetic acid (TCA) solution, and 2 ml of 0.67% (w/v) thiobarbituric acid (TBA) solution. The sample tube contained 2 ml of enzyme solution, 2 ml of 5% (w/v) TCA solution, and 2 ml of 0.67% (w/v) TBA solution. The mixture was placed in 93°C water for 30 min. The reaction was stopped after the tubes were placed on ice. The samples were centrifuged, and the absorbance of the supernatant was measured at 450, 532, and 600 nm.
Determination of Soluble Protein Content
Coomassie brilliant blue staining was used to determine the total soluble protein content of each supernatant with a protein assay kit (Tiangen Biotech Co., Ltd., BJS China, TIANGEN®, catalog number PA102).
Determination of Soluble Sugar Content
According to Wang and Huang’s method (Wang & Huang, 2015), the content of soluble sugar was measured using the Anthrone method. After the seedling leaves were boiled, the supernatant was added to an Anthrone solution (0.2 g of Anthrone dissolved in 100 ml of sulfuric acid) and boiled for 10 min. The solution absorbance at 620 nm was measured (glucose solution was used as the standard to calculate the content).
Determination of Chlorophyll Content
We used 95% ethanol to extract chlorophyll from the sample, and the absorption values of the extract at 665, 649, and 470 nm were measured. The chlorophyll content was calculated using Wang and Huang’s method (Wang & Huang, 2015).
Quantification of Plant Hormones by Enzyme-Linked Immunosorbent Assay
The endogenous hormones abscisic acid (ABA), gibberellin (GA), Zeatin (ZA), brassinolide (BR), and Methyl Jasmonate (JA-ME) were measured. They were determined by an enzyme-linked immunosorbent assay (ELISA). The plant hormone ELISA kits were purchased from Shanghai Enzyme-Linked Biotechnology Co., Ltd. (mlbio®, catalog numbers ml077235, ml077232, ml077233, ml036308, and ml077337).
Determination of Matrine and Oxymatrine Contents
After the tissue culture seedlings had grown for 45 days, the roots and rhizomes were collected and further dried for the determination of matrine and oxymatrine contents. The levels of oxymatrine and matrine were measured according to the guidelines of the Chinese Pharmacopoeia (edition 2015) (Chinese Pharmacopoeia Commission, 2015).
Statistical Analysis
All data passed the homogeneity test of variance, so data were analyzed by one-way ANOVA and LSD (α = 0.05) for multiple comparison tests in SPSS 17.0 software.
Results
Effect of Different Se Concentrations on Roots
As shown in Table 1 and Figure 1, Se concentration plays a key role in root growth. The 15 mg/L-treated seedlings produced the longest roots. The 30 mg/L treatment also benefited root growth, while roots were the shortest in the absence of Se. The root germination rate increased with increasing concentrations of 0, 15, and 45 mg/L Se. We determined that the optimal Se concentration of 15 mg/L was ideal for root growth, and the differences were statistically significant. However, under the highest Se treatment (60 mg/L), root length, number, and rooting rate all fell below average, and dark roots were found in seedlings.
Phenotypes of Seedlings Under Various Se Treatments.
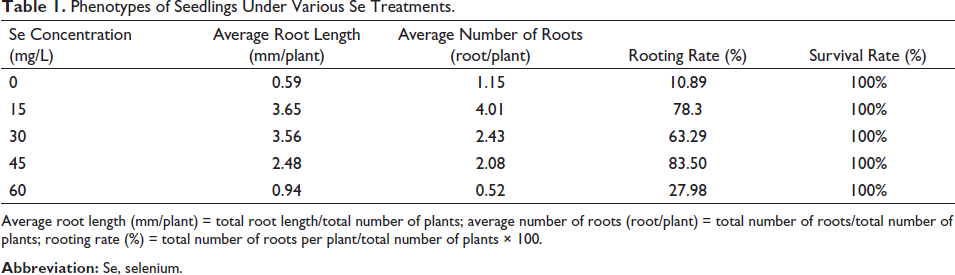
Average root length (mm/plant) = total root length/total number of plants; average number of roots (root/plant) = total number of roots/total number of plants; rooting rate (%) = total number of roots per plant/total number of plants × 100.

Effect of Se on Physiology and Biochemistry
The activity of POD was highest in the 15 mg/L group and then declined in the 30 mg/L group to the lowest level in the 60 mg/L group. The statistical analysis showed that the 15 mg/L and 60 mg/L treatments were significantly different from the control (0 mg/L) (Figure 2).
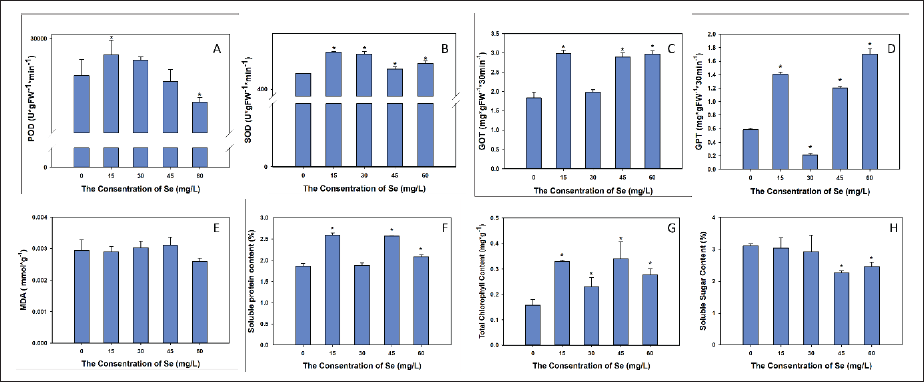
Se clearly enhanced seedling SOD activity. The activity was highest in the 15 mg/L group and lowest in the control group. We detected significant differences for all Se treatments from the control (p < 0.05) (Figure 2).
The activity of GOT increased with Se concentrations of 15, 45, and 60 mg/L. The ANOVA results showed that the 15, 45, and 60 mg/L treatments resulted in significantly higher GOT activity than the control (p < 0.05) (Figure 2).
Se plays a dual role in the regulation of GPT activity; that is, it inhibits GPT activity at a concentration of 30 mg/L but enhances GPT activity at other concentrations. The statistical analysis showed that the 15, 30, 45, and 60 mg/L treatments significantly differed from the control (Figure 2).
In this study, MDA content slightly increased with Se concentrations from 0 to 45 mg/L but dropped to the lowest content at 60 mg/L Se. However, the statistical analysis indicated that none of the Se treatments significantly differed from the control (Figure 2).
Seedlings that were treated with Se concentrations of 15, 45, and 60 mg/L produced significantly more soluble proteins than seedlings of the control (p < 0.05). The highest concentration of soluble proteins was observed in the 15 mg/L group (Figure 2).
Sugar content declined with Se at 45 mg/L and 60 mg/L. The statistical analysis showed significant differences between the 45 mg/L and 60 mg/L treatments and the control (p < 0.05) (Figure 2).
Chlorophyll contents fluctuated as Se concentration increased, peaking at 45 mg/L. All Se-treated seedlings contained significantly more chlorophyll than the control (p < 0.05) (Figure 2).
As Se treatments rose in concentration, ABA content initially dropped at 15 mg/L, and then more ABA content was found in seedlings with a 60 mb/L treatment. The statistical analysis of these data detected significant differences in the control between the 15 and 60 mg/L treatments but not between the 30 and 45 mg/L treatments (Figure 3).
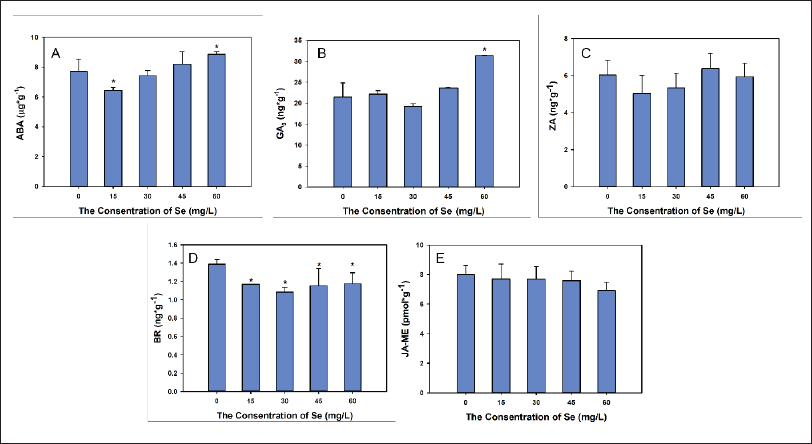
As Se concentrations increased, GA3 content initially declined to the lowest value in the 30 mg/L group and then increased to the highest value in the 60 mg/L group. However, only the GA3 content of the 60 mg/L treatment differed significantly from the GA content of the control (p < 0.05) (Figure 3).
The ZA contents fluctuated insignificantly with increasing Se concentrations, reaching the lowest value at 30 mg/L Se and the highest value at 45 mg/L Se; however, the differences in all treatments from the control were not significant (Figure 3).
As Se increased, the BR content dropped sharply until its nadir at 30 mg/L Se, and then the content rose slightly at higher treatment concentrations. The statistical analysis showed that all Se treatments produced significantly lower BR contents than the control (p < 0.05) (Figure 3).
The Se treatments slightly produced lower JA-ME; however, the difference was not significantly different from all other Se treatments (p < 0.05) (Figure 3).
Matrine and Oxymatrine Contents in Seedlings
In the whole-plant samples of seedlings, as Se treatments increased in concentration, the contents of both metabolites increased and then decreased (Figure 4). Other Se-treated groups, except the 45 mg/L Se treatment, contained significantly more metabolites (matrine and oxymatrine) than the control contained in the whole plant, indicating that Se is helpful in the synthesis and accumulation of these products (Figure 4). However, in the stem/leaf samples, the oxymatrine content decreased after Se treatment. More oxymatrine accumulated on roots in Se-treated samples (excluding 45 mg/L Se-treated samples), indicating that Se plays a role in oxymatrine distribution in plants. Matrine in samples treated with 15, 45, and 60 mg/L Se also had less content in stems/leaves than in the control and more accumulation in roots (Figure 4), which also indicated that a specific concentration of Se treatment may change the distribution of matrine.
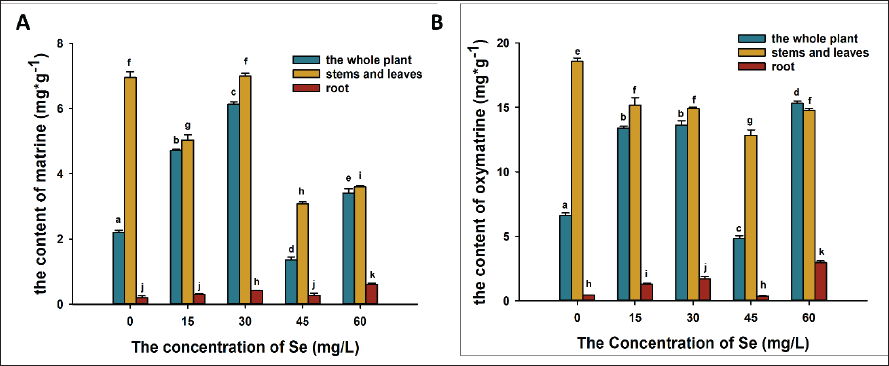
Discussion
S. tonkinensis Having a Strong Tolerance to Se
In this study, researchers have demonstrated that providing a moderate amount of Se can promote root growth, but Se concentrations that are too high will have negative effects on root growth. This result is consistent with previous studies, in which Se at low concentrations is beneficial for higher plants, but at high concentrations, it will cause toxicity. The root growth of S. tonkinensis was inhibited by 60 mg/L Se treatment, but the root growth was still better than that of the Se-free treatment. The soil Se content in Guangxi, which is the main distribution area of S. tonkinensis is relatively high (Ban & Ding, 1992), indicating that S. tonkinensis is likely to have a high tolerance for high concentrations of Se.
Effect of Se on the Physiology of S. tonkinensis and its Mechanism
The high soluble protein content and levels of GOT and GPT activities measured in the 15, 45, and 60 mg/L Se treatments indicate that a specific concentration of Se may promote nitrogen metabolism. In this study, Se is added to the culture medium in the form of organic matter (Se amino acid). There was a report showing that the uptake of organic forms of Se, such as selenomethionine (SeMet), in wheat seedlings is coupled to metabolism (Abrams et al., 1990), so the transformation of nitrogen metabolism in the study may be related to the control of organic Se absorption by roots. Se toxicity is also involved in reactive nitrogen species (RNS), such as nitric oxide (NO) and peroxynitrite (Chen et al., 2014; Lehotai et al., 2016a, 2016b), which may also be another reason for the change in nitrogen metabolism caused by Se.
During plant stress and aging, excess free radicals can accelerate the peroxidation of membrane lipids, resulting in damage to the cell membrane system. The enzymes POD and SOD can effectively remove free radicals, including ROS; thus, they are the main components of the enzymatic defense system. It is known that Se can exert its toxic effect by inducing oxidative stress (Van Hoewyk, 2013). Increased ROS production can be triggered by Se (Dimkovikj & van Hoewyk, 2014; Lehotai et al., 2012, 2016a, 2016b; Tamaoki et al., 2008). According to a previous study (Lehotai et al., 2016b), on a Se-enriching plant (Chinese brake fern, Pteris vittata), moderate amounts of Se (1–5 mg/L) can enhance the activity of POD and SOD. Our study supports their conclusion because the POD and SOD activities of the 15 mg/L Se-treated group were significantly higher than those of the control (p < 0.05), while the POD activity of the 60 mg/L Se-treated group was the lowest. On the contrary, the SOD activity of the 60 mg/L group was dramatically higher. This result is consistent with an existing study (Feng & Wei, 2012). SOD has higher activity than the control of zero treatment under the treatment of high concentrations of Se, while POD under the same treatment has little difference or is slightly decreased compared with the control. We hypothesize that the environmental stress of exposure to the high Se concentration stimulated a negative feedback mechanism of significant production of ROS to boost SOD activity.
No significant differences were observed in MDA contents among treatments with 0–60 mg/L. This may be explained by the mild growing temperature and sufficient water supplied to seedlings, as well as the fact that S. tonkinensis has a strong tolerance to Se. Plants did not need to adjust their MDA levels.
Moderate Se treatments can enhance photosynthesis in rice (Oryza sativa) (Zhang et al., 2014). Se also plays a role in maintaining chlorophyll content in potatoes under oxidative stress (Seppänen et al., 2003). The above reports show that Se can affect the photosynthetic system of plants. Previous studies have found that Se can increase the chlorophyll content of duckweed (Landoltia punctata) (Zhong & Cheng, 2016), which is consistent with our existing research results. This may be the response of plants to a certain concentration of Se stress.
Soluble sugar is one of the most active carbohydrates because of its role in regulating plant growth, development, and gene expression (Wang & Tang, 2014). Only under the treatment of high concentrations of Se (45 and 60 mg/L) did the soluble sugar content decrease significantly. This result suggests that plant accumulation of soluble sugars is difficult under extreme Se treatments. This result indicates that a high concentration of Se may weaken the ability of S. tonkinensis to cope with stress.
The Effect of Se on Root Hormones of S. tonkinensis is Related to the Root Phenotype
The accumulation of ABA is a plant response to abiotic stress (Vishwakarma et al., 2017). The ABA content was significantly lower in the 15 mg/L treatment than in the control (p < 0.05), indicating that this treatment did not stress the seedlings in this group. In contrast, as the Se concentration increased, the ABA content significantly increased. The data suggest that higher Se concentrations (60 mg/L) induced the ABA response, and thus ABA accumulated to a greater degree than in the control seedlings.
Seedlings treated with 60 mg/L Se accumulated significantly more GA3 than seedlings in the control and the other Se treatments. There are some reports that GA3 can inhibit adventitious root formation (Mauriat et al., 2014; Pizarro & Diaz-Sala, 2020; Zhang et al., 2021). The results showed that the decrease in rooting rate under 60 mg/L Se treatment may be related to the production of more GA3. Although the rooting rate of the 60 mg/L Se treatment was lower than that of the other concentrations of Se treatment, it was still higher than that of the Se-free control, which showed that the improvement in rooting rate by Se also involved other mechanisms, such as the phytohormone BR. The contents of BR in seedlings of all Se treatments were significantly below that of the control. This phenomenon can explain the effect of Se on roots; that is, a low concentration of BR promotes the growth of lateral roots (Mussig et al., 2003; Sathiyamoorthy & Nakamura, 1990).
The various Se concentrations had little influence on ZA and JA-ME contents, or perhaps more time (>45 days) was needed to effect any changes.
Treatment with 30 mg/L Se was Beneficial to the Accumulation of Matrine and Oxymatrine in Seedlings of S. tonkinensis
Se aided the production and accumulation of matrine and oxymatrine in seedlings, and the ideal concentration was 30 mg/L. At this concentration, some indices were significantly different from other concentrations of Se treatment. These indices suggest that the changes in nitrogen metabolism are closely related to the contents of matrine and oxymatrine. To the best of the authors’ knowledge, this is the first study that reports the secondary metabolites matrine and oxymatrine concentrated in the stem/leaf tissues of S. tonkinensis seedlings with Se treatments.
This study investigated the effects of various concentrations of Se on seedlings of S. tonkinensis. Generally, a small amount of Se is favorable for the growth and health of the plant, while excessive amounts produce negative effects. Under 30 mg/L Se treatment, the greatest accumulation of matrine and oxymatrine was observed. This research provides a foundation for future explorations in the application of S. tonkinensis. The effect of Se on plants involves multiple signaling pathways. Future research needs to prolong the processing time and carry out molecular biology research to indicate the change in the expression of related genes. In this study, the performance of indicators at the 30 mg/L Se concentration was specific to other Se treatment concentrations, so it is necessary to further strengthen the study of S. tonkinensis under this concentration treatment.
Conclusion
S. tonkinensis is likely to have a high tolerance to high concentrations of Se. A specific concentration of Se may promote nitrogen metabolism and increase the activity of SOD, the content of soluble protein, and chlorophyll in S. tonkinensis. Under the treatment of high concentrations of Se (45 and 60 mg/L), the soluble sugar content decreased significantly in S. tonkinensis. Higher Se concentrations (60 mg/L) induced the ABA response in S. tonkinensis. The contents of BR in seedlings of all Se treatments were significantly below that of the control. The treatment of 60 mg/L Se made the seedling accumulate significantly more GA3. Treatment with 30 mg/L Se was beneficial to the accumulation of matrine and oxymatrine in seedlings ofS. tonkinensis.
This reaction of S. tonkinensis to Se correlates with the formation of its medicinal efficacy; the high content of Se in a specific environment leads to the accumulation of effective ingredients. In addition, this reaction is also a manifestation of the plant’s adaptability to the environment. However, this study has not demonstrated the molecular mechanism underlying the response to Se, and the next step needs to further analyze which genes in plants participate in the response to Se.
Footnotes
Summary
S. tonkinensis is likely to have a high tolerance to high concentrations of Se. Treatment with 30 mg/L Se was beneficial to the accumulation of matrine and oxymatrine in seedlings of S. tonkinensis.
Abbreviations
Se: Selenium; POD: Peroxidase; SOD: Superoxide dismutase; GOT: Glutamic oxaloacetic transaminase; GPT: Glutamic-pyruvic transaminase; ROS: Reactive oxygen species; PVP: Polyvinyl pyrrolidone; MDA: Malondialdehyde; ABA: Abscisic acid; BR: Brassinolide; GA: Gibberellin; ZA: Zeatin; JA-ME: Methyl Jasmonate; RNS: Reactive nitrogen species; NO: Nitric oxide; TBA: Thiobarbituric acid; TCA: Trichloroacetic acid; NBT: Nitro blue tetrazolium; KCTS: α-Ketoglutaric acid; ELISA: Enzyme-linked immunosorbent assay; SeMet: selenomethionine.
Acknowledgments
We would like to thank Namuhan Chen from Baotou Medical College for his assistance in the laboratory.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangxi Science and Technology Project (GuiKeZY 20198018, GuiKeAD22080012) and National Natural Science Foundation of China (81460582, 81473309).
Statement of Informed Consent and Ethical Approval
This study focuses on plants rather than animals and humans. The studied plants are those that have been introduced and cultivated by the botanical garden, rather than protected wild populations. Therefore, obtaining informed consent and ethical statements is not necessary for this study.
