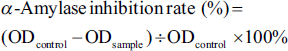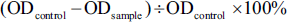Abstract
Background
Metabolic syndrome is an assortment of conditions that often happens together and upsurge your risk of diabetes, stroke, and heart disease. One strategy for the prophylaxis and treatment of diabetes and obesity is to inhibit the enzymes activities, which include α-glucosidase, α-amylase, and pancreatic lipase.
Objectives
To screen the effect of 16 Chinese medicinal herbs on inhibition of α-glucosidase, α-amylase, and pancreatic lipase.
Materials and Methods
The water extraction of 16 traditional Chinese herbal medicines was used to estimate activity in vitro from different families against α-glucosidase, α-amylase, and pancreatic lipase by using spectrophotometry with p-nitrophenyl-α-D-glucopyranoside (PNPG), starch derivatives, and 4-methylumbelliferone (4-MUO), respectively, as a substrate.
Results
The results showed that the water extraction yield of the 16 rhizomes ranged from 0.2% to 4.0%. Among the tested extracts, the extract from Gastrodia elata exhibited the strongest effect on α-amylase (The half-maximal inhibitory concentration (IC50)) = 4.10 mg/mL). The extracts from Pinellia ternate and Arisaema heterophyllum exhibited noteworthy effects on α-glucosidase (IC50 = 29.21 and 41.13 µg/mL, respectively), and Corydalis turtschaninovii possessed the highest effect on pancreatic lipase (IC50 = 4.11 µg/mL) and a stronger effect than orlistat (IC50 = 4.52 µg/mL).
Conclusion
This study provides a basis for screenings and in-depth studies of anti-obesity and anti-diabetes drugs. Further isolation, identification, and characterization of active compounds should be carried out in the future.
Introduction
Metabolic syndrome is a pathological state categorized by insulin resistance, central obesity, hyperlipidemia, and hypertension (Andrikakou et al., 2022). Due to the unhealthy eating habits and lifestyles of modern society, the number of patients with metabolic syndrome is each year (Di Marzo & Silvestri, 2019). The number of patients with metabolic syndrome globally will reach about 2.568 billion in 2040. Thus, it signifies a serious danger to human life and health, a global medical and health challenge (Saklayen, 2018). The pathogenesis of the metabolic syndrome is complex and not fully rich. However, obesity and insulin resistance are documented as important pathogenic factors (McCracken et al., 2018; Xu et al., 2018a).
Insulin resistance is the foundation for metabolic disorders such as diabetes, obesity, hypertension, and atherosclerosis. It is tangled in the formation of many diseases, and it results in tremendously serious harm to the body (Gluvic et al., 2017). Diabetes is a metabolic disease induced by inadequate insulin secretion or a lack of insulin sensitivity in peripheral tissue. It is considered tenacious hyperglycemia, and it may cause chronic injury, dysfunction, or tissue failure (e.g., blood vessels, eyes, heart, kidneys, and nerves) (Arneth et al., 2019). As per survey data from the International Diabetes Federation (IDF), the number of adult diabetic patients globally stretched to 425 million in 2017, with China serving as home to the major population (114 million patients) (IDF, 2018). Type 2 diabetes patients account for more than 90% of all patients (Fitipaldi et al., 2018).
Two major enzymes concerned with diabetes metabolism are α-glucosidase and α-amylase (Trinh et al., 2016). It has been stated that α-amylase inhibitors can effectively inhibit the activities of saliva and pancreatic amylase in the oral cavity and gastrointestinal tract as well as deter the hydrolysis of amylose to maltose in food (Takahama & Hirota, 2018). In addition, α-glucosidase inhibitors can inhibit the hydrolysis of oligosaccharides and disaccharides changing to monosaccharides, decrease glucose absorption and postprandial blood glucose, and advance glucose metabolism disorder in diabetic patients (Sohrabi et al., 2022). Presently, the representative α-glucosidase-inhibiting drugs for the clinical treatment of diabetes involve miglitol, acarbose, and voglibose. Prevailing hypoglycemic inhibitors have numerous side effects, including flatulence, bowel sounds, diarrhea, abdominal distension, and abdominal pain. Therefore, α-amylase and α-glucosidase inhibitors on hypoglycemia, blood lipids, and weight loss have established much courtesy.
Obesity is an important cause of metabolic diseases, including hypertension, hyperlipidemia, arteriosclerosis, type II diabetes, and a variety of cancers (Engin, 2017). Pancreatic lipase is a triacylglycerol acyl hydrolase and is responsible for hydrolyzing dietary fat (50%–70%) in the gastrointestinal tract; it is a crucial target for the prevention and therapy of obesity (Liu et al., 2020). Pancreatic lipase inhibitors can decrease the decomposition and absorption of dietary fat in digestive organs by dipping the hydrolysis activity of pancreatic lipase, thus improving the symptoms of metabolic illnesses such as obesity and hyperlipidemia. Although it is available on the market, the pancreatic lipase inhibitor orlistat causes momentous adverse gastrointestinal reactions after long-term use, comprising diarrhea, watery or fatty stools, abdominal pain, nausea, and vomiting (Tak & Lee, 2021; Viner et al., 2010). Thus, there is a need to develop new pancreatic lipase inhibitors with stout inhibition capabilities and good safety performance. In recent years, investigators around the world have carried out a huge number of studies on Chinese herbal extracts, resulting in the chemical components of some that can regulate lipid metabolism and prevent obesity by inhibiting pancreatic lipase (Rajan et al., 2020; Seyedan et al., 2015). The fact that an assortment of Chinese medicinal herbs are employed as treatments for diabetes and lowering blood lipids provides imperative clues for the possibility that α-glucosidase, α-amylase, and lipase inhibitors are existing in traditional natural medicinal resources (Wang et al., 2017; Wu et al., 2019).
In this study, 16 rhizomes from Chinese medicinal herbs were designated as the research materials. Extracts were obtained by water extraction, destruction of enzymes, salting out, dialysis, and freeze-drying. The extracts inhibitory activities toward α-amylase, α-glucosidase, and lipase were examined, and their hypoglycemic, lipid-lowering, and anti-obesity effects were acknowledged. The rhizomes can be employed as raw materials to develop drugs or foods for the prevention of metabolic syndrome.
Materials and Methods
Materials
The rhizomes were obtained from a local market (Hanzhong, Shaanxi Province) in December 2018 and projected by Dr. Yong Wang from the Shaanxi University of Technology.
Chemicals and Equipment
Acarbose (purity > 95%) and orlistat (purity ≥ 97%) were acquired from Dalian Meilun Biotechnology (Dalian, Liaoning, China). At the same time, α-amylase and α-glucosidase were obtained from BIOTAI (Beijing, China) and Shanghai Guchen Biological (Shanghai, China), respectively.
A UV-6100s ultraviolet-visible spectrophotometer was found in Mapade (Shanghai, China). A microplate reader was acquired from Omega Instruments (Vermont, USA). Freeze dryers were procured from Martin Christ (Osterode am Harz, Germany).
Pre-treatment of Chinese Medicinal Herbs
Rhizomes of Chinese medicinal herbs without mechanical damage, disease, or insect pests were nominated as the test materials. They were gutted to confirm a lack of defects or broken skin. The 16 fresh rhizomes were located in a fume hood to dry at room temperature (25°C).
Preparation of Plant Extracts
The dried raw materials (100 g) were ground into powder. Then, purified water was added at a ratio of 1:4 (material to liquid, W/V), and the mixture was agitated for 12 h at 4°C. The supernatant was centrifuged at 8,000 rpm for 20 min. The residue was re-extracted by repeating the extraction procedure. Extracting solutions were then combined and quickly heated in a 70°C water bath for 30 min. After heating, the supernatant was salted out with 60% saturated ammonium sulfate at 4°C for 12 h, and it was centrifuged at 5,000 rpm for 15 min to obtain the precipitate. The precipitate was re-dissolved in phosphate buffered saline (PBS) solution (20 mM, pH 7.0) and dialyzed (regenerated cellulose, 3,500 Dalton cut off, Spectrum Labs Inc.) broadly with ultrapure water for 24 h at 4°C to eliminate the ammonium sulfate. The ultrapure water was substituted three times during dialysis. The dialysate was freeze-dried at –60°C in a freeze dryer for 72 h to form a dry powder, which was deposited at 4°C for additional analysis.
Measurement of α-Amylase Inhibitory Activity
3,5-Dinitrosalicylic acid (DNS) colorimetry was employed to assess the inhibitory activity of α-amylase (Ghosh et al., 2012; Tamboli et al., 2018). The determination method was as follows: First, draw 0.5 mL of the sample solution (10 mg/mL). Then, 0.5 mL (1 mg/mL) of α-amylase solution was added to the sample solution in a water bath (37°C) for 15 min. Next, 1 mL of 2% soluble starch was added and reacted for 5 min in a water bath. Finally, 3 mL of 3,5-DNS reagent was added, boiled for 5 min in a water bath, and rapidly put in an ice bath (0°C) for 5 min. The absorbance value was judged at a wavelength of 540 nm.
The control group used distilled water in the same procedure as labeled above, and the positive control group was acarbose. Three parallels were made for each sample. The inhibition rate formula for α-amylase inhibitor was as follows:
Measurement of α-Glucosidase Inhibitory Activity
α-Glucosidase inhibitory activity was measured by a micro-method. The sample solution (50 µL) was added to a 96-well plate. Then, 50 µL of PBS buffer solution (pH 6.5) and 10 µL α-glucosidase (4 U/mL) were added and preheated at 37°C for 15 min. Then, 50 µL of p-nitrophenyl α-D-glucosidase (PNPG) with a concentration of 250 mg/L was added, and the reaction was run for 15 min. After this, a 100 µL solution of Na2CO3 (1 mol/L) was located to immediately stop the reaction. Then, the absorbance value was analyzed at OD405 nm (ODsample). The blank control group (ODcontrol) and the positive control were ultrapure water and acarbose, respectively (Elya et al., 2012; Nguyen et al., 2016). Three autonomous parallels were made for each sample. The α-glucosidase inhibition rate (%) was dogged as the following formula:
Measurement of Pancreatic Lipase Inhibitory Activity
Copper soap colorimetry was employed to achieve pancreatic lipase inhibitory activity (Kwon et al., 2015). Briefly, 3 mL of phosphate buffer, 1 mL of bile salt, and 1 mL of olive oil were added to a 50-mL conical flask and shaken at 37°C in a constant temperature shaker (280 rpm) for 10 min. Then, 0.1 mL of lipase solution was injected, and the mixture was shaken (280 rpm) for 10 min. Immediately afterwards, 5 mL of toluene was placed and shaken (280 rpm) for 2 min. Then, the reaction was finished, and the fatty acids were extracted. The extraction solution was centrifuged at 4,000 rpm, and the organic and aqueous phases were clarified by layers. 1 mL of 5% copper acetate solution (pH 6.1) was added to 4 mL of the upper organic phase, and the resulting mixture was incubated for 3 min. The fatty acid and Cu2+ formed a green complex. Then, it was exposed to centrifugation for 8 min. The absorbance of a benzene solution containing copper fatty acid in the upper layer was evaluated by a spectrophotometer at 710 nm. The blank solution without lipase, which was prepared using the same method as the reference, was employed to get the concentration of fatty acid by comparing the standard curve of the absorbance of fatty acid.
One enzyme unit was measured as the number of enzymes desirable to produce 1 µmol/L fatty acid per minute under given conditions. Lipase activity was assessed using the formula: X = CV/Mt, where X is lipase activity (U/mg), C is the fatty acid concentration (µmol/mL), V is the solution volume of fatty acids (mL), M is the dosage of enzyme solution (mg), and t is the action time (min).
The lipase inhibitory activity was further designed. The ratio of the difference in enzyme activity before and after adding the lipase inhibitor is demarcated as the inhibited enzyme activity. In line with the lipase activity determination method described above, a quantitative lipase inhibitor was added before pre-heating, and the residual enzyme activity was restrained to calculate the enzyme inhibitory activity. The positive control was orlistat.
IC50 Determination
The half-maximal inhibitory concentration (IC50) value means the concentration of an inhibitor that is necessary to reduce half of the enzyme activity. Suitable extract concentrations were evaluated for the IC50 value. The Y axis (the percentage inhibition) and the X axis (the extract concentration) were designed, and a logarithmic regression analysis was achieved based on the mean inhibitory values.
Statistical Analysis
The data were analyzed using the SPSS 17.0 statistical software, and an ANOVA method was pragmatic for significance analysis, and p < 0.05 means significant differences.
Results
Extraction of 16 Chinese Medicinal Herbs
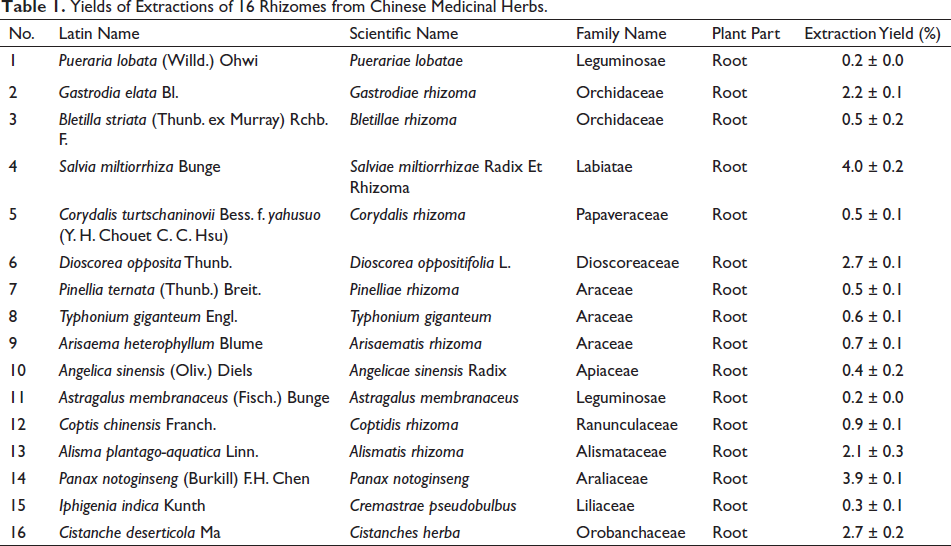
We employed water extraction, heat inactivation, salting out, dialysis, and freeze-drying to obtain 16 rhizome extracts. The consequences are revealed in Table 1. The extraction rate ranges from 0.2% to 4.0%. Astragalus membranaceus had the lowest extraction rate, and Salvia miltiorrizia had the highest extraction rate.
Yields of Extractions of 16 Rhizomes from Chinese Medicinal Herbs.
α-Amylase Inhibition Analysis
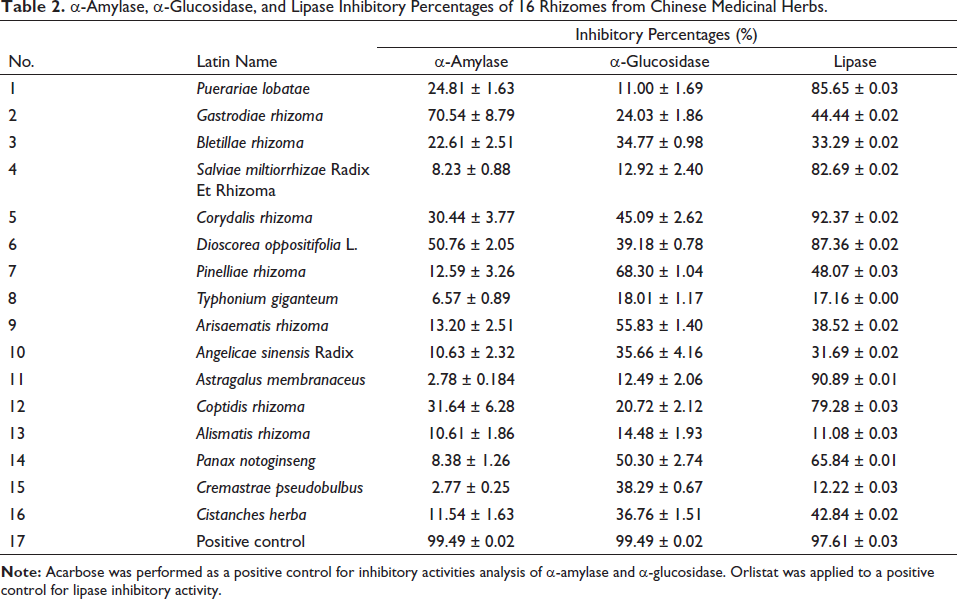
The inhibitory activities of the extracts varied from 2.78% to 70.54%. The inhibitory activity of 14 kinds of Chinese medicinal herb extracts concerning α-amylase was feeble and did not reach at least 50% of the enzyme inhibition level. In contrast, the inhibitory activity of Gastrodia elata and Dioscorea opposita regarding α-amylase was sturdy and attained more than 50% of the enzyme inhibition level (Table 2).
α-Amylase, α-Glucosidase, and Lipase Inhibitory Percentages of 16 Rhizomes from Chinese Medicinal Herbs.
The IC50 values of G. elata and D. opposite (inhibition rate greater than 50% against α-amylase) were 4190.26 µg/mL and 11850.12 µg/mL, respectively (Figure 1).
IC50 Values Against α-Amylase.

α-Glucosidase Inhibition Analysis
The inhibition (percentage) of the 16 rhizomes gotten from Chinese medicinal herbs against α-glucosidases. The inhibition rate of α-glucosidases ranged from 11.00% to 68.30% at a concentration of 0.2 mg/mL. The extract of Pinellia ternata disclosed the highest inhibitory activity (68.30%) (Table 2).
The IC50 values of three extracts (inhibition rate greater than 50% against α-glucosidase) vacillated from 29.21 to 198.29 µg/mL (Figure 2). The most active extracts against α-glucosidases were obtained from P. ternate (IC50 = 29.21 µg/mL) and Arisaema heterophyllum (IC50 = 41.13 µg/mL), but all extracts were weaker than acarbose (IC50 = 1.38 µg/mL).
IC50 Values Against α-Glucosidase.

Lipase Inhibition Assay
The extracts of the 16 rhizomes showed better inhibition of lipase activity. The inhibition rate fluctuated from 11.08% to 92.37% in concentration of 0.5 mg/mL. Among the 16 extracts, Panax notoginseng and Corydalis turtschaninovii were the most and least effective lipase inhibitors, respectively (Table 2). The IC50 values of seven extracts (inhibition rate greater than 50% against lipase) ranged from 4.11 to 80.20 µg/mL (Figure 3). The extract fromC. turtschaninovii exposed the lowest IC50 value (4.11 µg/mL) for lipase among all the extracts, and it displayed better performance than orlistat (IC50 = 4.52 µg/mL).
IC50 Values Against Lipase.

Discussion
Central obesity and insulin resistance are the chief pathogenic factors for the metabolic syndrome and thus serve as good preliminary points for improving the syndrome. It is well known that one of the causes of postprandial hyperglycemia with type 2 diabetes mellitus is that, after eating, a large amount of starch is slashed into α-1,4 glucosidic bonds and hydrolyzed into dextrin and oligosaccharides. After the action of α-glucosidase, it is transformed into glucose for human absorption. α-Glucosidase and α-amylase are embattled in the prophylaxis and treatment of non-insulin-dependent diabetes mellitus. In the past, researchers from many countries have described that some plants have an anti-diabetes effect, and many of these studies used leaves, seeds, and bark (Ríos et al., 2015). Few investigations on the inhibitory activities of α-amylase and α-glucosidase have been carried out with rhizomes.
The root of Pueraria lobata has a long history as an edible medicinal herb for the treatment of diabetes in China. In preceding studies, a pure product, named pl70-1-1, was obtained from P. lobata by two chromatographic steps. It disclosed a better α-amylase inhibitory activity (IC50 = 3.945 µM) in vitro, which was 417 times higher than acarbose (positive control) (Xu et al., 2018b). This research designated that isoforms I, II, IX, and XII have inhibitory effects for human carbohydrate anhydrase and was reliable with the earlier studies (Mocan et al., 2018). Puerarin is an active component found in the rhizome of P. lobata. The results of this study specified that puerarin could have moderately prevented catalase formation and progress by the signaling pathway of Nrf2/mHO-1 in streptozotocin-induced diabetic mice (Zhang & Li, 2019). The three studies stated above check the anti-diabetic activity of the monomer components of P. lobata. In our study, we established that the crude extract of P. lobata had an inhibitory effect on α-amylase, but its activity was weaker than that of pure glucan. This may be because the extract is a mixture.
D. opposite (Dioscoreaceae) is another edible medicinal plant grown in China. Its rhizome nurtures the lungs, tonifies the spleen, strengthens the kidney, aids the meridians, and helps digestion. An afore-mentioned study presented that the ethyl acetate extract of Dioscorea bulbifera (Dioscoreaceae) exhibited high elevation (up to 72.06 ± 0.51% and 82.64 ± 2.32% resist to α-amylase and α-glucosidase in vitro, respectively) at a concentration of 0.1 mg/mL (Ghosh et al., 2014). In addition, polysaccharides from D. opposite can decrease blood glucose and blood lipids by regulating the activities of enzymes related to glucose and lipid metabolism, persuading the synergistic anti-oxidant effect, and improving insulin sensitivity. Li et al. (2017) found that D. opposite polysaccharides can efficiently lower fasting glucose tolerance and blood glucose (Li et al., 2017).
Lipid metabolism plays a key role in the balancing of body energy. An imbalance in the lipid metabolism will lead to obesity and hyperlipidemia, which in turn will cause a series of stern health-threatening illnesses, including atherosclerosis, hypertension, and diabetes. Studies have exposed that the great majority of enzymes are drawn into the lipid metabolism pathway, and their structures and functions are flattering and progressively clear (Liu et al., 2020). Thus, they may be a new candidate substance to cure obesity and related metabolic ailments. For example, lipases play an important role in the digestion of triglycerides and phosphates. The inhibition of lipase is the most broad target in efforts to develop and screen natural anti-obesity drugs. At present, orlistat is an isolated, clinically implemented drug that can be used in the treatment of obesity, and its mechanism of action includes inhibiting the activity of pancreatic lipase.
P. lobata also presented good anti-obesity effects, as stated by research on P. lobata extracts in animal bodies and adipocyte research. Buhlmann et al. (2019) confirmed the positive functions of P. lobata water extract on a diet-induced obesity mouse model and supplied evidence regarding the mode of action of this extract at the molecular level (Buhlmann et al., 2019). Another study of cells displayed that P. lobata root ethanol extract augmented adipogenesis but reduced lipogenesis, leading to a reduction in lipid accumulation in 3T3-L1 cells (Lee et al., 2015).
There have been numerous studies on the anti-obesity properties of D. opposite. The 15 phenolic compounds of D. opposite are operative for inhibiting pancreatic lipase, with 3,3′,5-trihydroxy-2′-methoxybibenzyl viewing the supreme inhibition function (IC50 = 8.8 µM; orlistat, 1.24 µM) (Kwon et al., 2003). Another study examined the anti-obesity effects of D. opposite n-BuOH extract in obese rats induced by a high-fat diet (Jeong et al., 2016). This exploration showed that the extract efficiently lessened fat accumulation in the mice and that D. opposite water extract exposed strong inhibitory activity (87.36 ± 0.02%) on lipase at the concentration of 0.5 mg/mL.
Coptidis chinensis is a frequently used antipyretic and dampening drug in clinical settings, and it has been recorded as one of the highest-grade herbs related to Traditional Chinese medicine. Modern research displays that berberine is one of the most imperative active ingredients of C. chinensis. It has antibacterial, antiviral, and anti-gastric ulcer pharmacological effects and plays a key role in hypoglycemic, hypolipidemic, anti-tumor, anti-cardiovascular, and cerebrovascular diseases. Previous studies have shown that berberine augmented the expression of adipose triglyceride lipase and caused adipocytes to undergo lipolysis through a pathway that induced AMPK activation but did not result in insulin resistance (Jiang et al., 2016; Yang et al., 2020). In the existing study, we found that C, chinensis water extract disclosed strong inhibitory activity (79.28 ± 0.03%) against lipase in vitro, with the IC50 value equaling 15 µg/mL (orlistat, 4.5 µg/mL).
Conclusion
This study assessed the inhibitory activities of α-amylase, α-glucosidase, and pancreatic lipase in 16 rhizomes extracted using water from Chinese medicinal herbs. The fallouts display that the extract of G. elata exhibited the most solid effect on α-amylase, the extracts of P. ternate and A. heterophyllum exhibited the finest effect on α-glucosidase, and the extract of C. turtschaninovii exhibited the stoutest effect on pancreatic lipase. Overall, this study offers a basis for further screenings and in-depth studies of anti-obesity and anti-diabetes drugs. Furthermore, in vivo experiments are desirable to confirm the treatment capacity of these extracts for diabetes mellitus and obesity.
Footnotes
Summary
A lot of Chinese medicinal herbs are employed as treatments for diabetes, and lowering blood lipids delivers imperative signs for the possibility that α-amylase, α-glucosidase, and lipase inhibitors are extant in traditional natural medicinal resources.
The purpose of this study was to explore the inhibitory activity of water extracts from 16 Chinese herbal medicines on α-amylase, α-glucosidase, and pancreatic lipase.
This study presented that the extract of G. elata exhibited the hardest effect on α-amylase, the extracts of P. ternate and A. heterophyllum exhibited the superlative effect on α-glucosidase, and the extract of C. turtschaninovii exhibited the sturdiest effect on pancreatic lipase. Our study may add a new pharmacological basis for the prophylaxis and treatment of metabolic diseases using Chinese medicinal herbs.
Abbreviations
IDF: International Diabetes Federation; IC50: The half-maximal inhibitory concentration; PNPG: p-nitrophenyl-α-D-glucopyranoside; 4-MUO: 4-methylumbelliferone; DNS: 3,5-Dinitrosalicylic acid; PBS: Phosphate buffered saline.
Acknowledgment
We are thankful to Ms. Yi Zhu for her technical work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Development Program of Shaanxi (2020LSFP2-38) and the Natural Science Basic Research Program of Shaanxi Province (2022JM-120).
Statement of Informed Consent and Ethical Approval
Before beginning the study, all participants provided the necessary ethical clearances and informed consent.