Abstract
Background
The present experimental work aimed to evaluate the chemical composition and in vitro and in vivo effects of Curcuma zedoaria essential oil (CZEO) compared with metronidazole (MTZ) for Giardia lamblia infection treatment. The constituents of CZEO were recognized by gas chromatography/mass spectrometry (GC/MS) examination.
Materials and Methods
To assess the in vitro anti-Giardia effects of CZEO, clinical isolates of G. lamblia (cysts and trophozoites) were treated with various concentrations of CZEO (25–100 µg/mL) for 10–360 min. Infected BALB/c mice orally received the CZEO at 200–600 µg/kg/day doses for 7 days. The mean number and viability of excretory cysts were assessed using vital 0.1% eosin staining. Giardia parasite antigen in the stool of the tested mice was also detected on days 5, 10, 15, and 20 post-infection through ELISA using the monoclonal antibodies.
Results
Based on GC/MS, the major components were curzerenone (28.6%), 1,8-cineole (21.3%), and delta-3-carene (10.6%), respectively. CZEO significantly (p < 0.0001) reduced the viability of G. lamblia cysts and trophozoites as a dose-dependent response. CZEO at the tested doses reduced the mean number and viability of the excreted G. lamblia cysts. The highest reduction of Giardia antigen was detected in mice cured with CZEO at the 600 µg/kg dose; on days 5, 10, 15, and 20, the rate of reduction was 85.97%, 77.86%, 97.09%, and 98.75%, respectively. Biochemical examinations confirmed no significant alteration between the liver and kidney enzymes of the mice cured with CZEO and the control group.
Conclusion
The C. zedoaria essential oil showed potent in vitro and in vivo anti-Giardia effects and can thus have Giardia infection therapeutic potentials. Still, more studies on the exact mechanisms of action of this plant and its toxicity are warranted.
Introduction
Giardia lamblia is a well-known and common parasite of the gastrointestinal tract that causes giardiasis, an acute or chronic infectious disease, in several animals as well as humans. 1 Giardia infection or giardiasis causes more than 300 million human diarrhea cases reported annually, principally in children younger than 6 years old.2, 3 Infection with this parasite occurs mainly through the consumption of water and food contaminated with G. lamblia cysts and/or direct fecal-oral interaction with such cysts. 4 Clinical symptoms of giardiasis range from asymptomatic to transient or acute stage, with severe watery diarrhea, steatorrhea, malabsorption, abdominal cramps, and weight loss, especially in children. 5 The primary treatment for giardiasis is the chemotherapy with nitroimidazoles, particularly metronidazole (MTZ), as well as furazolidone, tinidazole, and quinacrine. 6 The most common side effects of MTZ and other nitroimidazole agents are teratogenicity, nausea, vomiting, rough tongue, gastrointestinal disorders, skin rash, urticaria, headache, dizziness, ataxia, dark urine, itching, pain during intercourse, and concentrated white discharge.7–9 Additionally, some studies showed the parasite’s resistance to the above drugs and the drugs’ contraindications during pregnancy. 10
A significant population of people around the world rely on medicinal herbs and their products as an economical, reachable, and beneficial substitute medicine for treating several diseases. 11 Plant-produced chemical compounds, or phytochemicals, have also been used in a variety of applications ranging from commercial to medicinal and therapeutic. 12
White turmeric (Curcuma zedoaria), which belongs to the Zingiberaceae family, has diuretic and demulcent properties and is used in folk medicine to treat or alleviate conditions such as digestive problems, vomiting, dyspepsia, skin diseases, and some infections. 13 Several therapeutic properties, for example, antinociceptive, antitumor, anti-inflammatory, antibacterial, antiparasitic, and antifungal activity, have also been reported for this herb. 14 Previous studies on the phytochemical study of C. zedoaria essential oils showed that the main phytochemicals in essential oils are oxygenated monoterpenes and sesquiterpene hydrocarbons. 15 In gas chromatography-mass spectrometry (GC-MS), compositions have been reported differently depending on the extraction method, type of analysis device, plant harvest time, and place.16, 17 The present experimental work is intended to assess the chemical composition and in vitro and in vivo effects of C. zedoaria essential oil (CZEO) in comparison with MTZ for the treatment of G. lamblia infection.
Materials and Methods
Plant Materials and Essential Oil Preparation
C. zedoaria rhizomes were procured from a shop in Riyadh, Saudi Arabia, and recognized by a botanist at Shaqra University, Saudi Arabia. To separate the essential oil by hydro-distillation, powdered rhizomes (250 g) were used with Clevenger-type tools for 3 h. After dehydration, the EO was stored at 4°C until testing. 18
Gas Chromatography-Mass Spectrometry
CZEO compounds were detected using a Hewlett-Packard 6890 (Palo Alto, CA, USA) system armed with an HP-5MS column (30 m × 0.25 mm, film thickness: 0.25 mm) to accomplish the GC study based on the conditions and method described elsewhere. 18 The compounds were recognized by matching their retention indices (RI) and mass spectra (MS) from the literature, as well as by comparing them with the Wiley library database. 19
G. lamblia Cyst Collection and Isolation
To prepare G. lamblia cysts, positive samples of G. lamblia cysts were collected from fresh feces of 30 patients (aged 2–10 years) referred to Ad-Dawadimi Hospital in Riyadh Province, Saudi Arabia, from January to May 2021. All the specimens were placed in normal saline in sterile plastic containers and transferred to the Laboratory of Parasitology. To confirm G. lamblia infection, the stools were tested by direct methods and the formalin-ether test with a light microscope. 20 The cysts were then isolated by a highly concentrated solution of the cyst with a basic sucrose of 85% gradient technique. 20 Stool samples containing the cyst were diluted at 1:10 with normal saline, filtered, and then centrifuged at 4000 rpm for 10 min. Next, 5 mL of the stool solution was covered with 5 mL of 0.85 M sucrose and centrifuged at 700g for 8 min at 4°C. In the next step, the G. lamblia cysts were extracted using a Pasteur pipette and were gradually put on the upper part of a discontinuous density gradient, which contained 2- and 3-mL layers of 0.85 and 0.4 M sucrose. Finally, the concentrated cysts were kept at 4°C until testing. The standard method of vital cyst staining or 0.1% eosin was applied to estimate the viability of cysts, 21 and the dead and viable cysts appeared pink and colorless, respectively. The number of the cysts was counted with a hemocytometer and adjusted to approximately 10 5 cysts/mL in the distilled water (DW).
Excystation of G. lamblia Cysts to Gain Trophozoites
Excystation of G. lamblia cysts was performed based on the technique explained by Bingham et al. 21 Briefly, the cyst suspension was mixed with the induction solution, aqueous hydrochloric acid (pH 2.0), at a ratio of 1:9, and the combination was kept at 37°C for 1 h. After centrifuging the mixture at 700 rpm for 8 min and then removing the upper phase, the remainder was added to the excystation medium, which contained filter-sterilized TYI-S-33 culture medium supplemented with bovine bile and 20% heat-inactivated fetal calf serum. Culture tubes with 7 mL of the medium, improved with streptomycin (500 µg/mL) and penicillin (500 IU/mL), were kept warm at 37°C in a slant.
In Vitro Anti-Giardia Effects
To estimate the in vitro anti-Giardia effects of CZEO, G. lamblia cysts and trophozoites were exposed to CZEO (25–100 µg/mL) for 10–360 min. Briefly, 1 mL of each concentration was put in each test tube, and then 10,000 cysts and trophozoites were placed in the test tubes, which were kept warm at 37°C for 10–360 min. 22 After the incubation time, the supernatant phase of the solution was thrown away, and then 0.1% eosin stain (50 µL) was added to the remaining settled cysts and trophozoites. Smears were provided from the residue of cysts and trophozoites on a glass slide separately and considered by a light microscope at a 400× magnification.22, 23 The viability percentages of the treated cysts and trophozoites were calculated by counting 300 cysts, and the dead and viable parasites appeared pink and colorless, respectively. 21 The non-treated cysts and the cysts treated with MTZ were considered the negative and positive controls, in that order.
Ethical Statement
We state that processes containing human participants were in agreement with the ethical criteria of the institutional and national research committee and with the 1964 Helsinki Declaration. 24 An informed printed agreement was gotten from the outpatients. The process was approved by the Ethical Committee of the Faculty of Science and Humanities, Shaqra University, Ad-Dawadimi, Saudi Arabia (SH-27-2020).
In vivo Anti-Giardia Effects
Animals
A total of 72 male BALB/c mice, aged 40–60 days and weighing 20–25 g, were accidentally divided into nine groups (eight mice per group). All the mice were kept under animal room conditions, with food and water available ad libitum all the time.
Induction of Animal Model Giardiasis
To establish the animal model of giardiasis, 200 µL of G. lamblia cysts (2×104/PBS) were orally administered by flexible plastic gavage needles into the mice in each tested group. To confirm the infection, mice were examined daily under a microscope from the first day of inoculation until G. lamblia cysts were identified in the mouse feces. The presence of G. lamblia cysts in the feces indicated that the mice were infected with the parasite. We adopted the formalin-ether technique to determine the presence of the G. lamblia cysts. 25
Treating the Infected Mice
On the 6th day post-infection, the mice were divided into five groups (eight mice per group), including infected mice treated with 100 µL CZEO at the dose of 200, 400, and 600 µg/kg/day orally for 7 days; whereas, the negative and positive control were treated with normal saline and MTZ (MTZ, Riyadh Pharma, Riyadh, Saudi Arabia) (15 mg/kg/day) orally for 7 days, respectively. 25
Parasitological Tests
After a 7-day treatment, mouse feces were tested by the formalin-ether method to confirm the existence of G. lamblia cysts and the percentage of reduction. The viability of excretory cysts was also assessed using vital 0.1% eosin staining. Cysts that took on the color of eosin were considered dead, and those that did not take on this color were regarded as viable. The body weight of the infected mice during the study period was also compared with that of positive and negative controls.25, 26
Infection Intensity Evaluation
Following proof of infection, the infection intensity was evaluated based on the method explained elsewhere. 27 Giardia parasite antigen in the stool of the tested mice was detected on days 5, 10, 15, and 20 post-infection via ELISA using the monoclonal antibodies (ProSpect Giardia Microplate Assay, Remel).
CZEO Toxicity on Liver and Kidney Function
To assess the toxicity of CZEO on liver and kidney function, 32 mice were divided into four groups (eight mice per group). Three groups of mice orally received 100 µL CZEO at the dose of 200, 400, and 600 µg/kg/day for 2 weeks. Control group mice received normal saline. One day after treatment, after collecting the blood specimens through the cardiac puncture, they were centrifuged at 4000 rpm for 10 min, and the separated serums were tested for evaluation of the serum level of liver and kidney enzymes. 22
Statistical Analysis
The data were analyzed using the SPSS 25.0 (SPSS, Inc.). The independent sample t-test, one-way analysis of variance (ANOVA), and Dunnett’s test were utilized to compare the findings. P < 0.05 was considered statistically significant.
Results
GC/MS Analysis
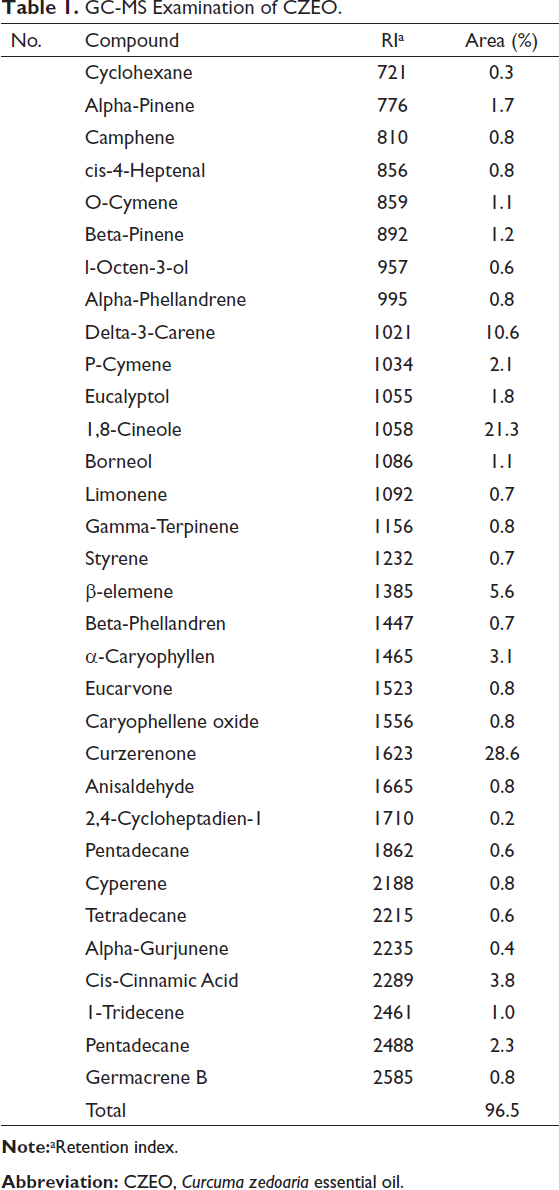
As shown in Table 1, 32 compounds were identified based on GC-MS, demonstrating 96.5% of the whole essential oil. The key constituents were curzerenone (28.6%), 1,8-cineole (21.3%), and delta-3-carene (10.6%).
GC-MS Examination of CZEO.
In Vitro Anti-Giardia Effects of CZEO
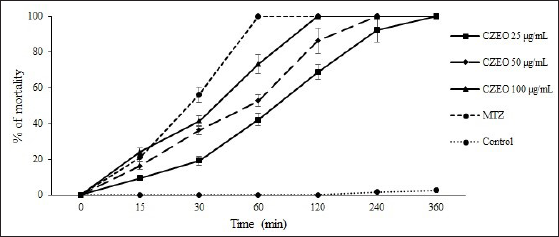
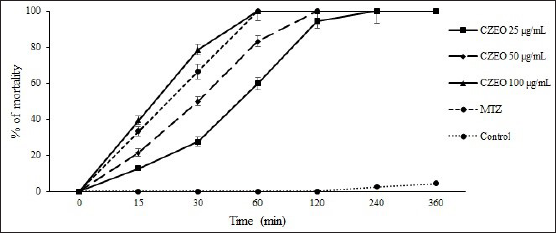
The in vitro anti-Giardia effects of various concentrations of CZEO on G. lamblia cysts at different times are shown in Figures 1 and 2. The results demonstrated that CZEO significantly (P < 0.0001) declined the viability of G. lamblia cysts as a dose-dependent response; at the concentrations of 50 and 100 µg/mL, it reduced the viability of G. lamblia cysts by 100% after 240 and 120 min, respectively. In the negative and positive controls, the viability of G. lamblia cysts was 2.6 and 100% after 360 and 60 min of incubation, respectively. Moreover, trophozoites were more susceptible to CZEO than cysts; the mortality rate was 100% after 240, 120, and 60 min of exposure of trophozoites to CZEO at concentrations of 20, 400, and 600 µg/mL, respectively.
In Vivo Anti-Giardia Effects of CZEO
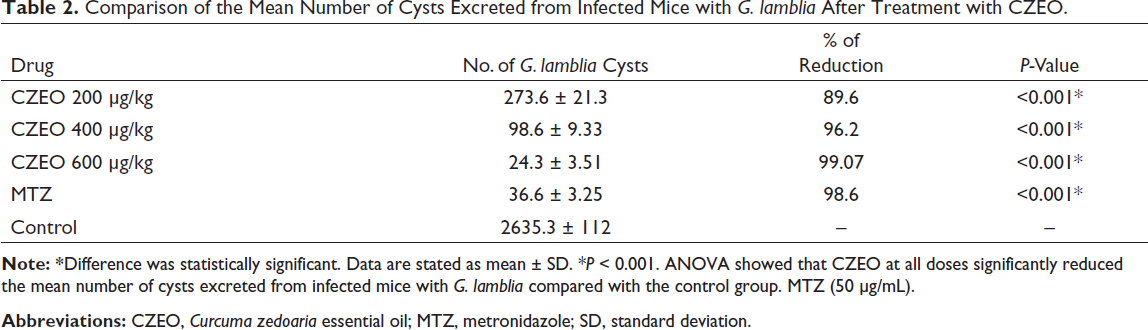
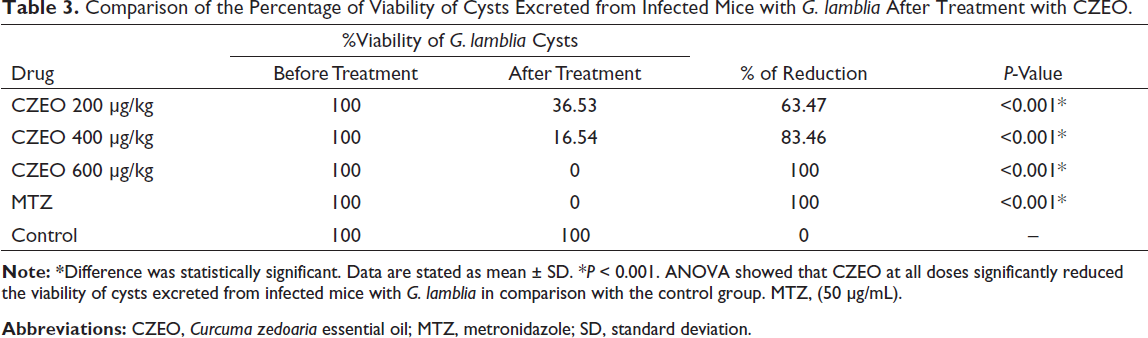
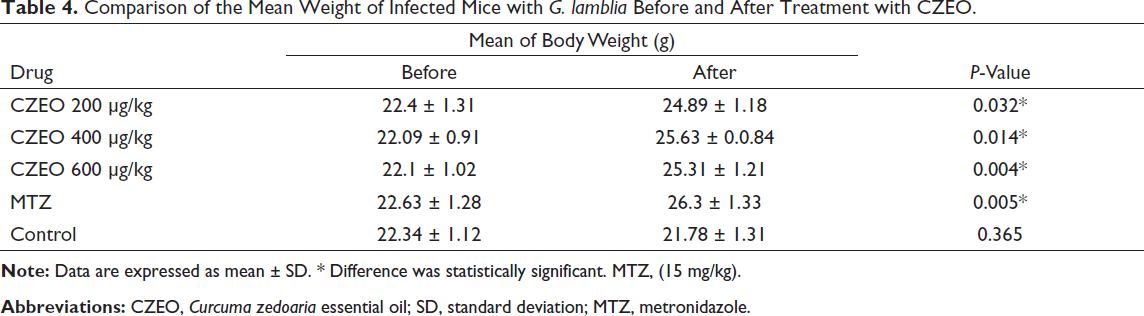
Following 7 days of curing infected mice with CZEO, mouse feces were tested by the formalin-ether method to confirm the existence of G. lamblia cysts and the percentage of reduction through the eosin staining. Table 2 presents the in vivo anti-Giardia effects of various concentrations of CZEO on the mean number of G. lamblia cysts excreted from mice infected with G. lamblia. After treatment of mice infected with G. lamblia with different concentrations of CZEO for 7 days, the mean number of cysts significantly (P < 0.001) decreased; CZEO at the doses of 200, 400, and 600 µg/kg reduced the mean number of G. lamblia cysts by 89.6%, 96.2%, and 99.07%, respectively. Table 3 also indicates the in vivo anti-Giardia effects of various concentrations of CZEO on the viability of G. lamblia cysts excreted from mice infected with G. lamblia. Following treatment with different concentrations of CZEO for 7 days, the viability of cysts significantly (P < 0.001) declined; CZEO at the doses of 200, 400, and 600 µg/kg reduced the viability of G. lamblia cysts by 76.6, 83.46%, and 100%, respectively. The mean weight of mice before and after treatment of infected mice with different concentrations of CZEO is shown in Table 4. The results showed that after treatment, weight changes in the negative control group were not significant, and the mean of the changes was weight loss of 0.56 g, while in the infected mice treated with different concentrations of CZEO, significant weight gain was observed (P < 0.05).
Comparison of the Mean Number of Cysts Excreted from Infected Mice with G. lamblia After Treatment with CZEO.
Comparison of the Percentage of Viability of Cysts Excreted from Infected Mice with G. lamblia After Treatment with CZEO.
Comparison of the Mean Weight of Infected Mice with G. lamblia Before and After Treatment with CZEO.
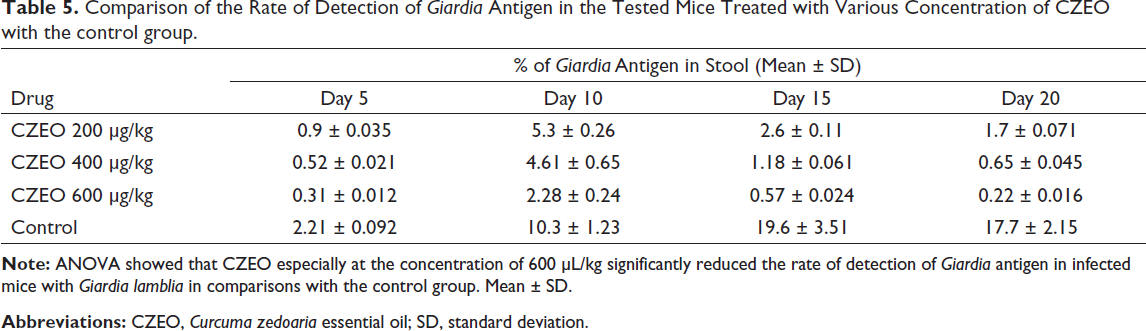
Based on the intensity of infection, the results of ELISA demonstrated a significant (P < 0.001) decrease in Giardia antigen in the stool of the mice cured with CZEO compared to the control groups. The highest reduction of Giardia antigen was detected in mice cured with CZEO at a dose of 600 µg/kg; on days 5, 10, 15, and 20 post-infection, the rate of reduction was 85.97%, 77.86%, 97.09%, and 98.75%, respectively (Table 5).
Comparison of the Rate of Detection of Giardia Antigen in the Tested Mice Treated with Various Concentration of CZEO with the control group.
Toxicity Effects of CZEO on Liver and Kidney Function
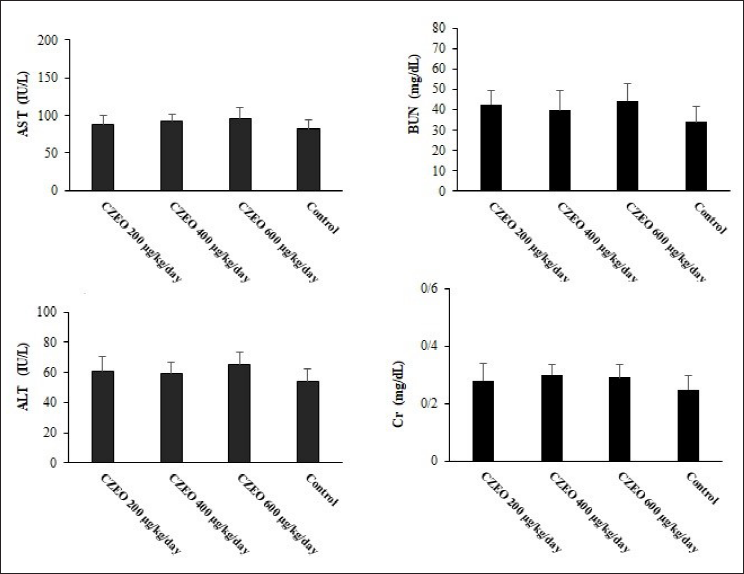
After pre-treatment of mice with CZEO, no mortality among the tested mice was reported, whereas significant weight gain was observed. Moreover, the findings of biochemical examinations revealed no remarkable difference between liver (ALT and AST) and kidney (Cr and BUN) enzymes in the mice cured with CZEO and the control group (P > 0.05) (Figure 3).
Discussion
MTZ is considered the most common synthetic drug used to treat giardiasis, but recent studies report that it has some disadvantages and side effects. 28 Today, the anti-Giardia effects of a number of plants (e.g., Carum copticum, Allium spp., Artemisia annua, and Zataria multiflora) have been reported in several studies; nevertheless, the comprehensive adoption of herbal medicines for Giardia infection is now suspended by the ambiguous results of the examination, which are not always satisfactorily powered. 29 White turmeric, due to some of its medicinal advantages in traditional (e.g., vomiting, stomachic disorders, diarrhea, and dysentery) and modern (e.g., antimutagenic, anti-diarrheal, anti-protozoal/antiamoebic, antimicrobial, anti-immunomodulatory, anti-platelets, anti-ulcerogenic, hepatoprotective activity, wound healing activity, antipyretic, anti-inflammatory, and antioxidant) medicine, can be a promising alternative agent for giardiasis treatment. 30 Therefore, we decided to assess the anti-giardial effects of CZEO in comparison with MTZ for the treatment of G. lamblia infection.
We found 32 compounds in CZEO based on GC-MS; whereas, the major components were curzerenone (28.6%), 1,8-cineole (21.3%), and delta-3-carene (10.6%), respectively. 1,8-cineole, α-copaene, curzerenone, camphor, elemol, β-caryophyllene, and elemol are reported to be constituents of C. zedoaria rhizome.31, 32 However, studies have shown that the structure of essential oils is different depending on some criteria, such as the extraction method, type of analysis device, plant harvest time, and place.16, 17
Our results showed that the CZEO reduced the viability of G. lamblia cysts and trophozoites as a dose-dependent response, with trophozoites being more susceptible to CZEO than cysts. In vivo assay also exhibits that after treatment of infected mice with different concentrations of CZEO (especially at the dose 600 µg/kg) for 7 days, the mean number of cysts decreased. In addition, we found a significant decrease in Giardia antigen in the stool of the tested mice treated with various concentrations of CZEO compared with the control groups.
As for the antiparasitic effects of C. zedoaria, Senathilake et al. (2016) have demonstrated that hexane and chloroform extracts of the rhizome of C. zedoaria considerably decreased the motility and viability of microfilariae and adult worms of Setaria digitata in vitro. 33 Mahmoudvand et al. (2020) exhibited that CZEO, especially at concentrations of 150 and 300 µL/mL, had remarkable protoscolicidal effects in vitro and ex vivo against the protoscoleces of Echinococcus granulosus. 18 Based on reviews, the antimicrobial effect of various essential oils is directly related to the presence of some compounds, including monoterpenes, sesquiterpenes, and hydrocarbons. 34 Our results showed that the main components of CZEO are monoterpenoids such as curzerenone, 1,8-cineole, and delta-3-carene. Regarding the antimicrobial mechanisms of monoterpenoid compounds, recent studies have confirmed that these compounds exert their antimicrobial activities by affecting cell permeability and subsequent cell wall destruction, penetrating the microbes, interacting with crucial intracellular sites, inhibiting oxygen uptake and virulence factors, and so on.35–37 Hence, it is suggested that the anti-Giardia effects of C. zadoaria are linked to the existence of these components in its essential oil; however, more surveys are essential to clarify these mechanisms.
In terms of CZEO toxicity, our results confirmed that after pre-treatment of mice with CZEO, no mortality among the tested mice was reported, but there was significant weight gain. Biochemical examinations also revealed no remarkable difference between liver and kidney enzymes in the mice cured with CZEO and the control group. Likewise, Mahmoudvand et al. have revealed that, in BALB/c mice, the intraperitoneal administration of CZEO at the doses of 150, 300, and 600 µg/kg for 14 days resulted in no mortality or toxicity on the hematologic and chemical parameters (liver and kidney enzymes) compared with the group receiving normal saline. 33
Conclusion
The results indicated that C. zedoaria essential oil showed the relevant effects on growth rate of trophozites and cysts of G. lamblia clinical isolates; the results of the in vivo assay also revealed that CZEO treatment of infected mice declined the mean number and viability of the excreted G. lamblia cysts and intensity of infection with no significant toxicity on the liver and kidney enzymes. However, further studies on the exact mechanisms of action of this plant and its toxicity, especially in human volunteers, are required.
Footnotes
Abbreviations
Acknowledgment
We thank the staff of the Biological Science Department, Faculty of Science and Humanities, Shaqra University, and the staff of the Department of Biology, University of Tabuk, Saudi Arabia, for their technical support.
Summary
We assessed the in vitro and in vivo effects of CZEO against G. lamblia.
CZEO considerably declined the viability of G. lamblia cysts and trophozoites as a in vitro dose-dependent response.
CZEO at the tested doses reduced the mean number and viability of the excreted G. lamblia cysts in vivo.
There was no significant difference between the liver and kidney enzymes of the mice treated with CZEO and the control group.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study received the approval of the Ethics Committee at Almaarefa University, Saudi Arabia (IRB23-030).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
