Abstract
Background
Hair color retention and rejuvenation have recently emerged as a new research hot spot, with many studies focused on developing treatments to prevent premature greying.
Aim
This study is aimed to explore the hair-blacking effects and possible mechanisms of Cerasus serrulata flower extracts (CSE) using a C57BL/6 premature greying mouse model.
Materials and Methods
The variations in hair follicle number, hair follicle depth and cortical thickness were evaluated by hematoxylin and eosin staining. The quantity changes of melanocytes were also evaluated using Masson-Fontana Melanin staining. The relative abundance of vascular endothelial growth factor (VEGF), β-catenin and tyrosinase transcripts following transformed growth were further quantified using RT-qPCR and immunofluorescence. The changes in protein level expression of β-catenin, VEGF and tyrosinase were also evaluated by western blot.
Results
The β-catenin and VEGF expression were significantly up-regulated in skin tissues treated with C. serrulata extracts, promoting hair growth and blackening.
Conclusion
CSE can promote the efficient hair-blacking likely via the VEGF pathway, making it a potential therapeutic candidate for treating premature greying.
Introduction
Hair, an important component in body image is known to display several important physiological functions including mechanical protection, ultraviolet protection, body temperature regulation, drainage, touch, and sexual representation.1, 2 Premature greying has major psychosocial and socio-economic impacts, as it is deemed to be a sign of pre-senility or reduced health and often leads to loss of confidence. 3 A recent epidemiological survey indicates that the rate of premature greying among young people especially less than 40 years of age, has reached about 33%. Most of people experiencing this condition reveal additional reductions in hair strength and present this pathology even during middle or high school. 4 It is widely believed that the white hair is associated with genetic factors, 5 physical stress, 6 malnutrition, 7 and ageing. 8 However, the underlying mechanism for hair greying still remains largely unclear, but most scholars believe that the mechanism is likely to center around the ability of melanin to infiltrate the hair matrix cells. 9
As a result, new technologies designed to treat grey hair and promote hair growth are attracting attention. There also are some synthetic hair dyes used to enhance a user’s natural color, but many of these treatments have involved compounds with potential mutagenic or carcinogenic effects. 10 In contrast, natural herbal products are mild, biodegradable and low toxicity. 11 In fact, there are many effective natural ingredients showing the potential applications as beauty products, and the researches into Chinese herbal cosmetics have revealed a rapid development in this filed. Especially, some studies profile the effects of various plant extracts on hair blackening, owing to their reduced toxicity. However, most studies evaluating plant-based hair dyes remain limited and their potential for adverse effects is still largely unknown. 12
In recent years, there has been significant progress in the evaluation of hair agents, facilitated by hair follicle models in C57BL/6 mice. 13 These models are commonly used to assess the effects and mechanisms of hair growth with many new assessments including gene and protein expression. These systems enable researchers to understand hair ageing and identify that hair greying is a periodic process. In addition, several studies have also revealed that the changes associated with hair greying are subjected to strict regulation, facilitated through the complex interactions of various signaling pathways including Wnt/β-catenin, (TGF)-β, and transforming growth factor pathways among others. 14 The dermal papilla cell (DP), which acts as the major mesenchymal component of the hair, is located at the deepest point of the hair follicle and is thought to play an important role in inducing new follicles and maintaining hair growth. 13 The β-catenin is a core member of the typical Wnt signal pathway, promoting the transcription of downstream target genes and inhibiting melanocyte formation. 15 In addition, β-catenin also plays an important part in inducing HF morphogenesis, promoting hair growth and maintaining the hair cycle. 16 The VEGF and its receptors are only expressed in hair follicles during the growth period and gradually decrease in the degenerative stage. 17 These proteins are also supported by tyrosinase, which regulates pigment production. 18
C. serrulata is a flowering cherry germplasm resource with a long cultivation history in China, widely distributed across the temperate regions of the northern hemisphere.19, 20 C. serrulata is widely used as an ornamental plant and its fructus has also become a popular edible fruit. It also produces a variety of polyphenols that are beneficial to human health, 21 suggesting that some active ingredients in C. serrulata are conducive to the human body. 22 In addition, this plant is known to effectively eliminate hydroxyl free radicals from the body and has been shown to inhibit oxidative stress in dermal papillary cells reducing premature white hair loss. 23
Given this, this study will examine the utility of applying Cerasus serrulata flower extracts (CSE) as a hair growth stimulant when compared with the US-FDA approved 2% Minoxidil, and provide several insights to support the development of novel treatments for premature greying.
Materials and Methods
Plant Materials and Chemicals
The C. serrulata used in this study was obtained from Guangdong Heji Biotechnology Company (Guangzhou, China) and identified by Professor Nian Liu (Zhongkai University of Agricultural Engineering, Guangzhou, China). The details of these products were listed in Table 1. All chemicals were analytical grade and purchased from Aladdin Reagent Co., Ltd. (Shanghai, China).
C. serrulata Sample Information.
Ethanol Extract
C. serrulata petals were dried at 60°C for 8 h, ground through a 40–60 mesh, mixed with 50% ethanol with a ratio of 1:60 (g/mL) to produce an aqueous solution and finally stirred at 80°C for 2 h. This extract was filtered by an 800-mesh press cloth and then placed at 70°C to remove the remaining ethanol under reduced pressure (N-1100, EYELA, Japan). The extracts were monitored for weight changes, and the evaporation was stopped once the filtrate reached 67% of its original mass. These concentrates were then spray dried using the following conditions: flow rate of injection = 2, temperature = 130°C, wind speed = 0.663 m/min, and pressure = 9 × 10 kPa.
Full-Spectrum Metabolite Analysis
The LC–MS/MS was used to identify small and medium molecular compounds within each of our samples for qualitative and quantitative analysis. The separation and identification of these substances by ultra-high performance liquid chromatography–mass spectrometry, combined with the annotation and classification of mass spectrometry database information were completed using precise characterization Instrument ASTAT-DAP: LC–MS (Thermo, Ultimate 3000 LC, HF) fitted with a C18 column (Zorbax Eclipse C18 (1.8 µm × 2.1 × 100 mm)). The separation conditions were as follows: column temperature = 30°C; flow rate = 0.3 mL/min; mobile phase A = water + 0.1% formic acid, mobile phase B = pure acetonitrile; injection volume = 2 µ; and active autokinetic nozzle = 4°C.
Acute Dermal Toxicity Test
The acute dermal toxicology study was performed in accordance with OECD guideline 402. 24 Briefly, a total of nine Wistar female rats weighing 200–220 g were randomly selected and divided into three groups of three animals each. These animals were then distributed as one per box and placed under standard conditions for a week of acclimation. The CSE ointment using the surgical gel as a vehicle was prepared and applied to the animals as follows: the limited test animals received 2000 mg/kg of the CSE ointment, and the vehicle group received 500 mg/kg of the surgical gel alone. Our third group acted as non-treated control. The applications were completed on the dorsal area (10%) of each animal, which was shaved prior to testing. All doses were only applied locally once on the first day of the experiment and then covered with a cotton dressing (impregnated with 2 g of surgical gel to saturate its absorption) for 14 days. Animals were monitored and weighed every third day and changes in physical appearance, behavior patterns, injuries, pain, signs of illness, and death were recorded for later evaluations. On day 15, the animals were sacrificed by cervical dislocation.
Experimental Animals
The 4–5-week-old male C57BL/6 mice (17–21 g) were purchased from the Animal Center at Sun Yat-sen University (SCXK2011-0029, Guangzhou, China), and all animals were under an adaptive feeding with a 12/12 h light/dark cycle at 23 ± 2°C. The mice were acclimatized for three days before starting the experimental procedures. All experimental protocols were approved by the Ethics Committee for animals in experimentation at Guangdong University of Technology (GDUTXS2022007, approval data: March 9, 2022).
Grouping and Modeling
Mice were first depilated and were randomly divided into the control, hydrogen peroxide (H2O2) model, Minoxidil, CSE-1%, CSE-5%, and CSE-10% groups, with 15 animals per group. Different concentrations of CSE were obtained by dissolving CSE in 30% ethanol. The pathological white hair mouse model was established according to the previously described literature. In addition, our model group was only treated with 5% H2O2 solution for two weeks while our experimental animals received 5% H2O2 daily to the dorsum skin. The control group was never treated with H2O2. Treated animals were given their interventions after fasting for 6 h with each animal receiving a 0.2 mL dose of the appropriate treatment, while the control group received a 0.2 mL dose of 30% ethanol. Minoxidil was used as the positive control and administered in the same way. Animals were treated for 14 days, 25 and hair growth was evaluated as described in Table 2.
Hair Growth Evaluation.

Histology
On the 14th day of treatment, the newly born hair in the depilated areas was shaved, and their removal area was sterilized with 75% ethanol. Long strips of skin (20 mm × 8 mm) were then collected from each group and fully rinsed in normal saline before being spread out on filter paper, dried at 25°C and wrapped in tin foil paper. Dark skin tissues from each group were then fixed in 4% paraformaldehyde before embedding in paraffin. Thick sections (4 µm) were treated continuously for H&E staining after being dewaxed and rehydrated and then evaluated using a light microscope. 26
Masson-Fontana Melanin Staining
Masson-Fontana stain was prepared as previously described and then used to stain the skin sections described above. Briefly, skin sections were immersed in melanin working fluid and capped before being washed 3 times for 18 h in distilled water. Samples were then dyed with VG, sealed with a transparent seal and imaged on a Nikon confocal microscope. Melanocytes were then quantified using ImageJ™ software (NIH, Bethesda, MD, USA). 16
Histopathology Assays
After dewaxing, rehydration, antigen retrieval and blocking, the skin sections were also incubated overnight with primary antibodies against vascular endothelial growth factor (VEGF), β-catenin and tyrosinase at 4°C before being washed in PBS. These samples were then incubated with an Horseradish Peroxidase-labeled secondary antibody (1:200) at 25°C, and visualized by DAB (3,3’-diaminophenyl). After staining, the sections were observed under a fluorescence microscope (1:200, Bioss Biotechnology Co., Beijing, China). 27 The expression of revelent indicators was calculated using ImageJ™ (NIH, Bethesda, MD, USA). Histological images were obtained by using a microscope.
Immunofluorescence Assays
Sections were dried overnight and washed three times in PBS before being blocked with 5% BSA at 25°C for 1 h. They were then incubated overnight with the primary antibody (β-catenin rabbit polyclonal antibody, 1:800, GB11015) at 4°C before being rinsed and exposed to the appropriate secondary antibody (Cy3 conjugated goat anti-rabbit IgG (H+L), 1:300, GB21303) at 25°C for 1 h. Samples were then counterstained with DAPI (300 nM, G1012) at 25°C for 10 min, 27 and then imaged using a Nikon confocal microscope (200:1, Nikon, Japan).
RT-qPCR Analysis
Total RNA was extracted from mouse tissue by using the Trizol-chloroform method (Solarbio, China), and cDNA was synthesized from 2.5 µg of total RNA (cDNA synthesis kit, Takara). The subsequent RT-qPCR reactions were then performed using a Piko-real TM 96 real-time PCR system (Waltham, MA, USA, Thermo Scientific) and a Maxima SYBR Green/ROX qPCR off-line mixture. GAPDH was used as an internal control in RT-qPCR. The following primers were used: VEGF, 5’-CACTGGACCCTGGCTTTACTG-3’ (forward) and 5’-CTCAATCGGACGGCAGTAGC-3’ (reverse); tyrosinase, 5’-ACACACTGGAAGTATTTTTGAACA-3’ (forward) and 5’-TAGGTGCATTGGCTTCTGGG-3’ (reverse); β-catenin 5’-GGACCCCAAGCCTTAGTAAACA-3’ (forward) and 5’-TTATATCATCGGAACCCAGAAGC-3’ (reverse). 15
Western Blot
Samples from each of the treatment groups were then lysed and their protein content was quantified by Bradford assay. These samples were then resolved using 12% SDS–PAGE and transferred onto a nitrocellulose membrane. After blocking with 5% skim milk at 25°C for 2 h, the nitrocellulose membranes were immunoblotted with anti-VEGF (VEGF Rabbit monoclonal, 1:1000, GB11034) and anti-β-actin antibodies (β-actin mouse monoclonal s°C for 24 h, before being incubated with HRP-conjugated goat anti-rabbit IgG (H + L) (1:3000, GB23303) and HRP conjugated goat anti-mouse IgG (H + L) (1:3000, GB23301) (Servicebio Technology Co. Ltd., Wuhan, China) for 1 h at 25°C. Protein bands were visualized using an enhanced chemiluminescence kit (MultiSciences Biotech, Hangzhou, China), and quantitated using analysis software (Quantity One, Bio-Rad, Hercules, CA) and integral optical density (Wuhan Servicebio Technology Co., Ltd.).
Statistical Analysis
All data were expressed as the mean ± SD and statistical comparisons were performed using GraphPad Prism 8.0.2 (GraphPad Software, USA). Multiple comparisons were made using the Tukey’s post hoc test and statistical significance was set at P < 0.05.
Results
Quality Evaluation of CSE
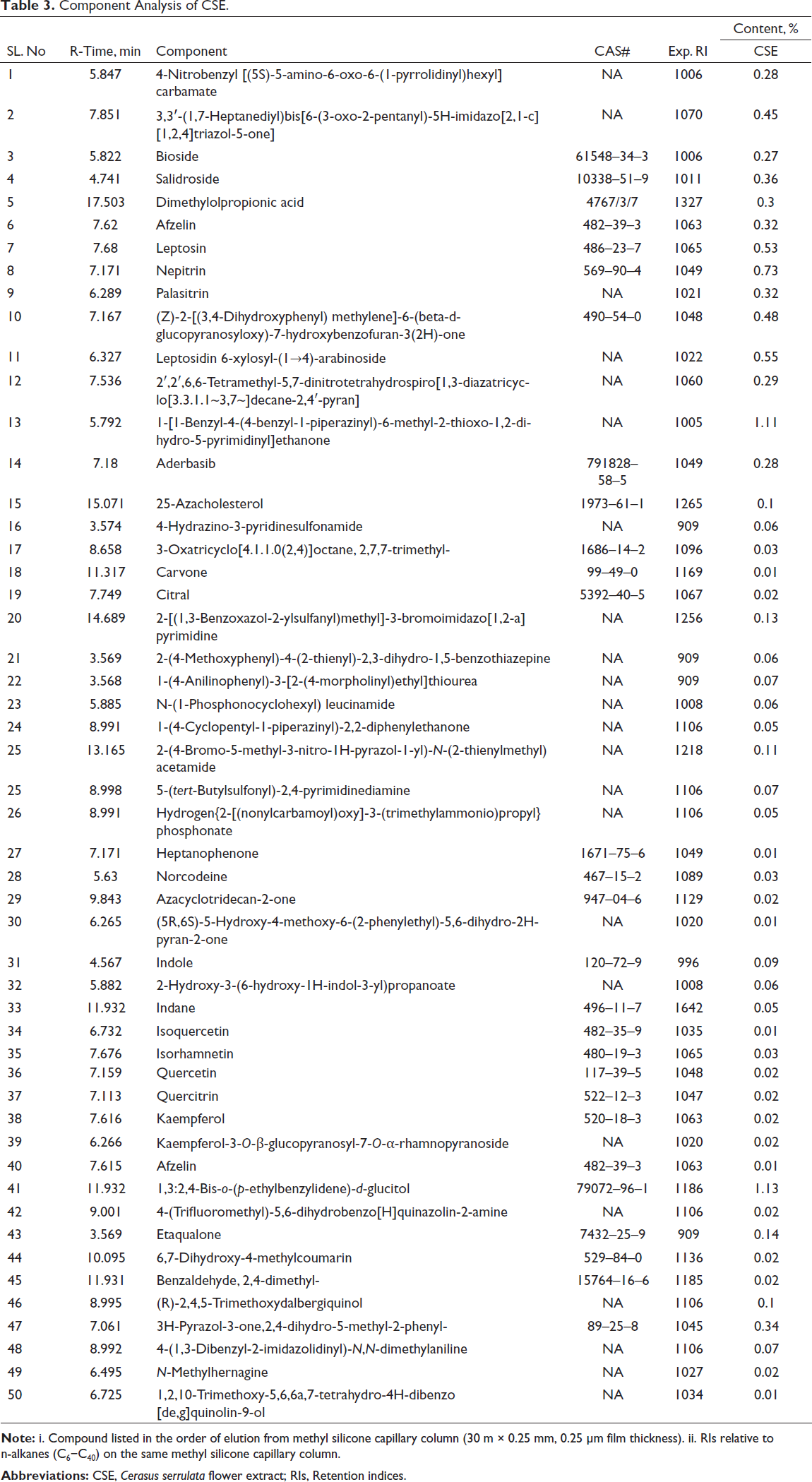
The quality of the CSE extracts was firstly confirmed using LC–MS/MS (Table 3). The evaluations demonstrated the presence of all of the critical CSE components, and revealed that 1,3:2,4-bis-o-(p-ethylbenzylidene)-
Component Analysis of CSE.
Acute Dermal Toxicity
Evaluations of acute dermal toxicity revealed that CSE is non-toxic up to a dose of 2000 mg/kg. These experiments revealed that there was no dermal toxicity in response to CSE treatment after 24 h of exposure with all treated animals presenting with normal skin, fur, eyes, and mucous membranes by normal respiratory, circulatory, autonomic and central nervous systems. Animals also retained their somatomotor activity, and there were no changes in behavior, tremors, convulsions, salivation, diarrhea, lethargy, sleep, or coma over the 14-day observation period (Table 4). Thus, it can confirm the safety of the extracts.
Observations of Changes in the Animal Welfare Parameters with 2000 mg/kg CSE.
Apparent Hair Follicle Recovery After CSE-Treated
Figure 1 shows that the number of hair follicle cells in the H2O2 model group was significantly reduced when compared to the control, while this was reversed in the Minoxidil group. In addition, the number of hair follicles between most of the Minodoxil and CSE groups was similar, although there was a significant increase in follicle cells in response to 5% CSE. This indicates that the active components in CSE can accelerate the growth of hair follicle cells and produce a similar result to a blue rinse. Additional evaluations of the cortical layer revealed significant increases in cortical thickness in the H2O2 group when compared to the control and a significant reduction in the Minodoxil group. Compared with the model group, the cortical thickness of the CSE-1% and CSE-10% groups were significantly decreased (p < 0.05).
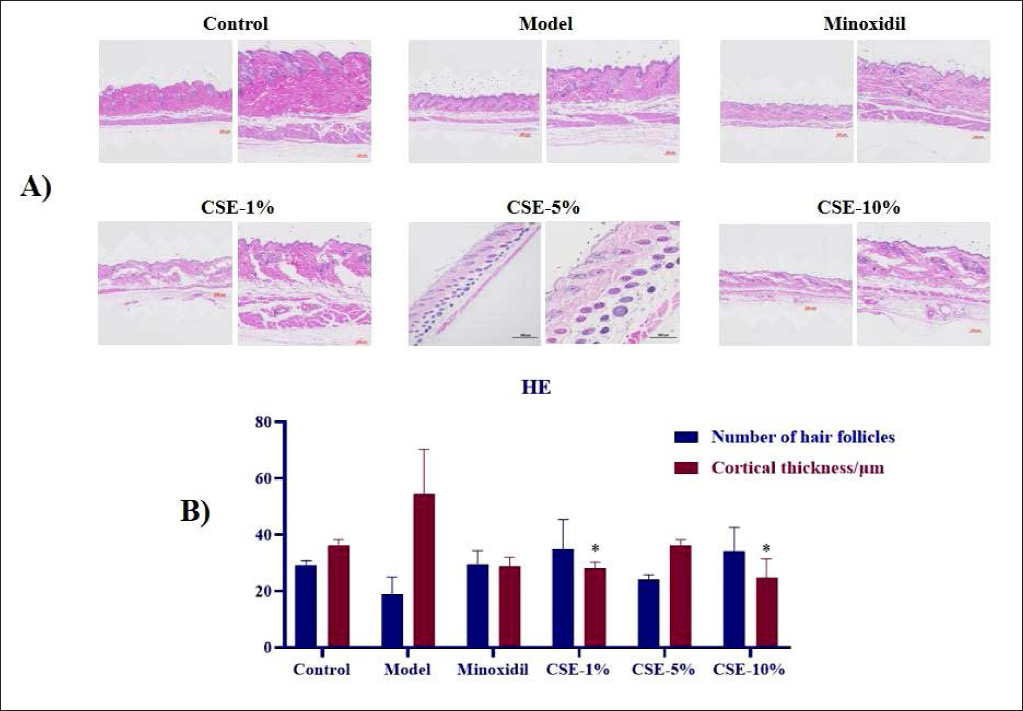
In addition, Table 5 summarizes the overall evaluations of hair growth in these samples, and reveals that Minoxidil, CSE-1%, CSE-5% and CSE-10% treatments induce significant increment in hair growth when compared to the peroxide model. The overall score from these groups when sorted from high to low is as follows: CSE-10% group > CSE-5% group > Minoxidil group > CSE-1% group > Control group > H2O2-5% model group, suggesting that higher doses of CSE had more beneficial effects on hair growth than both Minodoxil and low doses of CSE.
Evaluation of Hair Growth.

Histological Observation
Figure 2 reveals that the model group presented with clear reductions in melanocytes when compared to the control and that this was slightly increased in response to Minoxidil treatment (p < 0.05). In addition, the melanin content in the skin tissue of all groups of mice was increased to different degrees after treatment with different concentrations of CSE, among which, the CSE-10% group was the most significant (P < 0.05). Thus, it can infer that there are active ingredients in CSE which affect melanin production and that the application of an appropriate concentration of these compounds may help to restore melanin content in murine models of premature greying.
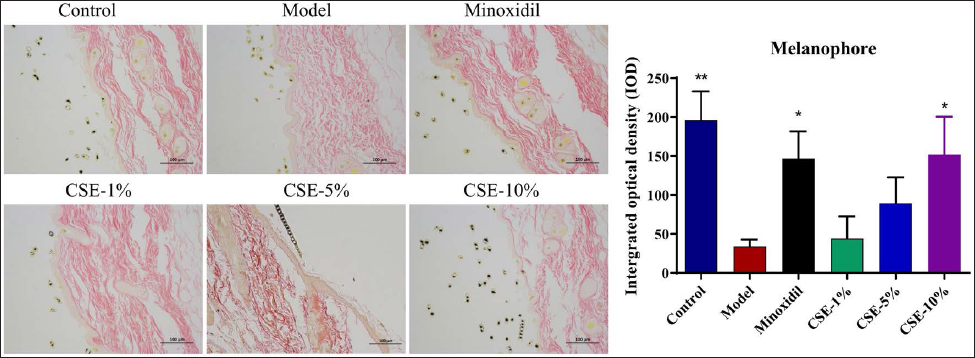
Hair-Blackening Effects Via Dynamic Changes in VEGF, β-Catenin and Tyrosinase Expression
VEGF is a vascular endothelial growth factor known to increase vascular permeability, promote angiogenesis, promote hair follicle regeneration and regulate hair follicle growth cycle and development. 17 Figure 3 shows significant reductions in the VEGF content of skin samples from the H2O2 group when compared with the control group (P < 0.05), and a significant increment in its expression for all three CSE treatment groups. A closer evaluation also revealed that this response was most significant in the CSE-5% group. In addition, the literature suggests that downregulation of tyrosinase inhibits the melanin biosynthesis pathway, 18 which is further confirmed by our observations that H2O2 treatment significantly reduces tyrosinase expression in these animals while both Minodoxil and CSE treatment upregulates its expression (P < 0.05).
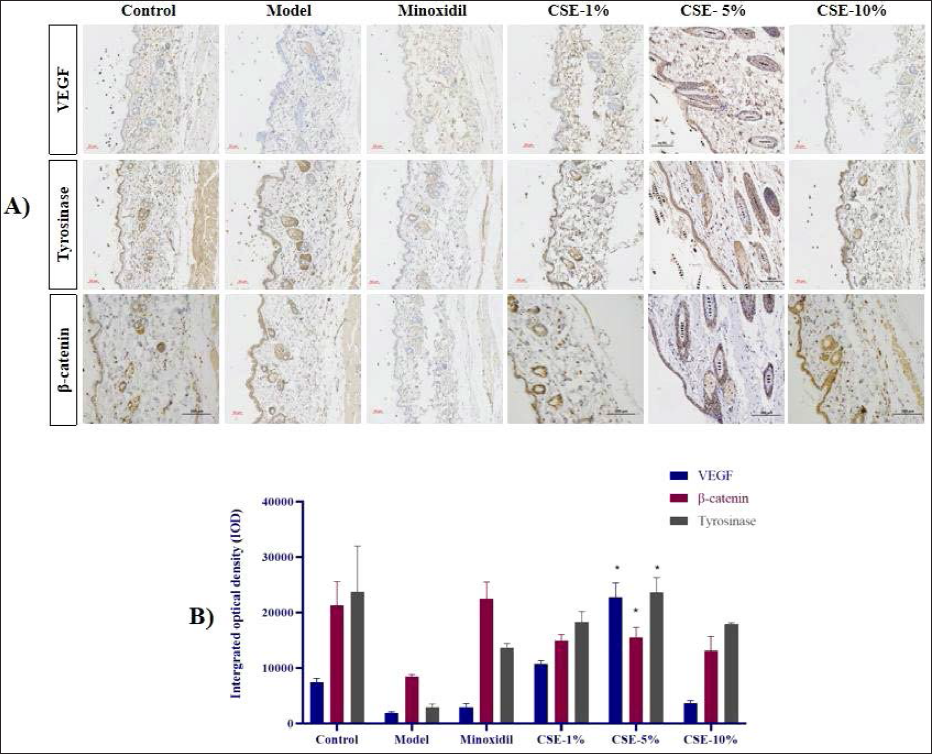
Finally, most studies suggest that β-catenin stimulates hair follicle development. 28 Given this, the β-catenin content of the CSE and Minodoxil groups were all significantly higher than that of the H2O2 samples (P < 0.05). Overall, these results suggest that the improvements in hair follicle health facilitated by CSE treatment are likely the results of increment in β-catenin, tyrosinase and VEGF.
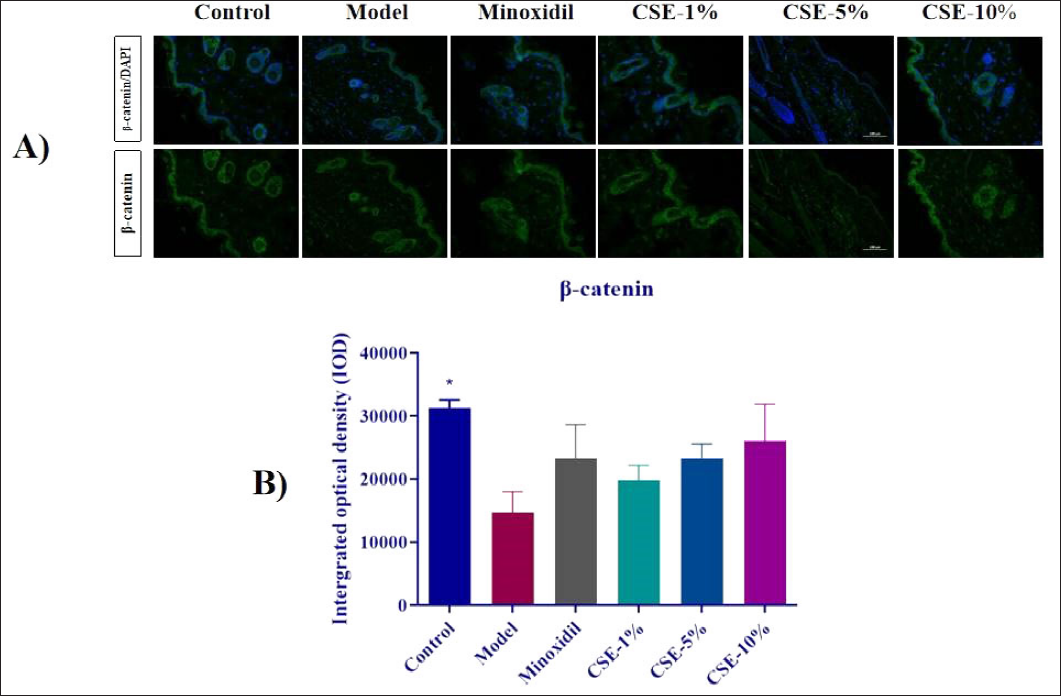
β-Catenin Expression in Treated Mice
Figure 4 revealed that all three CSE groups presented significant increment in β-catenin expression when compared to the control, and demonstrated that this effect was most prominent in the CSE-10% group, where β-catenin expression even exceeded the Minoxidil group.
VEGF, β-catenin and Tyrosinase Expression in CSE Mice
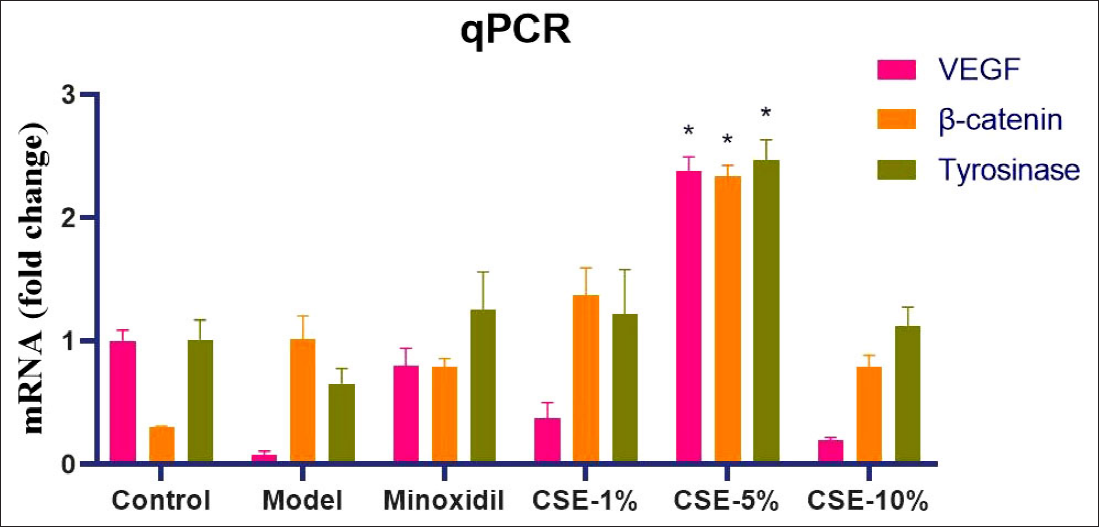
RT-qPCR revealed that Minoxidil and CSE treatments induced significant changes in VEGF, β-catenin and tyrosinase expression when compared to the H2O2-5% group (Figure 5) (P < 0.05). These differences were most significant in the CSE-5% group, confirming our earlier observations. Taken together, this data confirms that CSE mediates its therapeutic effects via changes in VEGF, β-catenin and tyrosinase signaling.
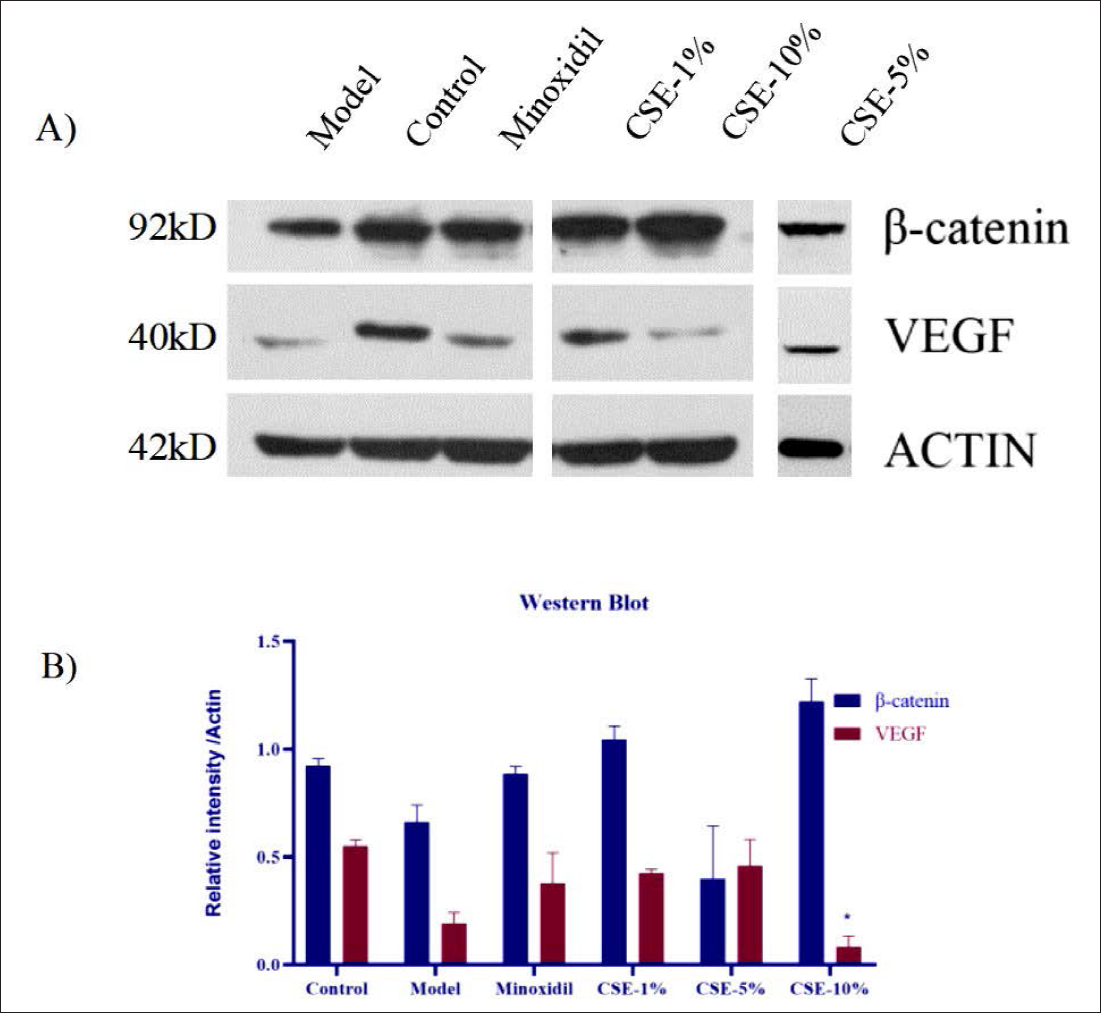
The Dorsal Skin of Treated Mice Facilitates its Hair-Blackening Effects
Figure 6 shows that β-catenin protein expression is significantly reduced in H2O2-treated animals when compared to the control and that the application of the CSE induces recovery in these expression levels, with CSE-10% producing the strongest response. This figure also confirms the upregulation of VEGF expression in the CSE-treated animals when compared to the model samples, with CSE-5% demonstrating the most significant effect. Taken together, this data supports the hypothesis that CSE induces hair follicle rejuvenation via changes in the VEGF, β-catenin and tyrosinase signaling pathway.
Discussion
In this study, we investigated the biochemical activity and underlying molecular mechanisms of CSE, a common natural ingredient in shampoo and hair care products used to combat premature greying. Our results suggest that CSE promotes macroscopic hair regrowth when evaluated in a mouse model. It also revealed that these effects were largely dose-dependent and that animals treated with 5% CSE displayed the best outcomes. In addition, our results also revealed that CSE includes various active substances that increase the numbers of melanocytes in the skin tissue of these mice which in turn promotes hair growth. We also noted that CSE treatment increases VEGF expression, limiting skin thickening and promoting the expression of both tyrosinase and β-catenin, which are known to exert positive effects on hair growth. Contrary to the predicted results, higher concentrations of CSE (10%) inhibited the expression of VEGF in the process of inhibiting skin thickening compared with lower concentrations (1% and 5%), suggesting that further evaluation and research should be conducted to support the molecular mechanism of CSE-mediated hair-blackening. Finally, while encouraging, our data highlights the complexity of these mechanisms and the need for further evaluation before CSE is widely applied in this setting.
Conclusion
C. serrulata is widely recognized for its outstanding anti-oxidant and anti-cancer activities, making it a favorite candidate in cosmetic, therapeutic, and food engineering applications worldwide. 29 These applications even extend to the pits, which are known to have significant nutritional benefits flowing from their high fatty acid El and triacylglycerol content. 30 Recent studies have shown that the seeds of this plant may also have the potential as a therapeutic adjuvant in the management of cardiovascular disorders. 31 Thus, when we combine these observations with our results, we clearly provide several important insights into the application of C. serrulata in health products. 32
Even though our study was primarily designed to evaluate the relationship between CSE-mediated signal activation and hair-blacking, our results may have broader implications including whether other Traditional Chinese Medicines (TCM) recorded as Qi and blood-promoting herbs may have potential applications in the treatment of premature greying. In addition, these results support the molecular evaluation of other Qi and blood-promoting TCMs in order to uncover their underlying mechanisms of action and support their application in therapeutic settings. 33 This evaluation revealed that C. serrulata, a popular TCM, activates and inhibits specific genes and signaling pathways to promote hair growth and reduce premature greying. 10 Given this, we propose that future studies should evaluate similar functions in other Qi and blood-promoting TCMs. Finally, we believe that our results support the hypothesis that further evaluations are likely to uncover other natural functional cosmetics and aid in the fulfilment of increasingly specific consumer demands.
Summary
This study was designed to evaluate CSE as a hair-blacking agent and revealed that: (1) CSE is an effective treatment for reversing premature ageing; (2) CSE-mediated changes in the hair follicle are evaluated by both histological and morphological analyses; and (3) CSE mediates these changes by inducing β-catenin, VEGF and tyrosinase expression.
The efficacy of CSE as a hair growth promoter is estimated. The effect of CSE on hair growth is determined by histological and morphological analysis. CSE can increase the VEGF of skin thickening and the expression of complex enzymes, and promote the expression of tyrosinase and β-catenin, causing hair blackening.
Footnotes
Authors Contribution
Jianfang Luo constructed the firing model, Wenyun Zhang, Jian He and Xian Xu performed the experiments. Xiaomin Zheng, Siyang Zhu and Tinggang He analyzed the data. Bing Zhang, Lanyue Zhang and Yong Ai wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experimental protocols were approved by the Ethics Committee for animals in experimentation at Guangdong University of Technology (GDUTXS2022007, Approval data: March 9, 2022).
Funding
The authors are grateful to the Innovation and Entrepreneurship Leading Team Project of Panyu District (Grant No. 2019-R01-6) and the Guangdong Provincial Key Laboratory of Plant Resources Biorefinery (No. 2021GDKLPRB02) for providing financial support.
