Abstract
Background
Grape seed extract (GSE) has demonstrated various pharmacological actions. Urolithiasis is the occurrence of calculus in the renal system. The present study evaluated the anti-urolithic effect of GSE on ethylene glycol (EG) and ammonium chloride (AC)-induced experimental urolithiasis in rats.
Materials and Methods
Rats were assigned into six groups; Normal control and Normal + GSE, in which rats received standard drinking water and GSE orally daily, respectively; Urolithiatic animals received EG with AC in drinking water for 28 days; Urolithiatic animals + GSE, in which rats were administered EG with AC in drinking water and GSE 100 and 200 mg/kg orally; and Urolithiatic + cystone, where rats received EG with AC in drinking water and 750 g/kg of cystone as a standard drug orally.
Results
Urolithiatic animals showed a significant decrease in excreted magnesium and citrate and antioxidant enzymes, whereas they exhibited amplified oxalate crystal numbers, urinary excreted calcium, phosphate, oxalate ions, uric acid, intensified renal function parameters, lipid peroxidation, and inflammatory mediators. Management with GSE and cystone significantly augmented urolithiasis inhibitors (excreted magnesium and citrate) and amplified the antioxidant enzymes’ activities. GSE reduced oxalate crystal numbers and urolithiasis promoters, including excreted calcium, oxalate, phosphate, and uric acid excretion, lessened renal function parameters, and declined lipid peroxidation and the inflammatory mediators.
Conclusion
GSE could protect against EG-induced renal stones as evidenced by mitigated kidney dysfunction, histological alterations, and oxalate crystal formation. This action may be related to the antioxidant as well as anti-inflammatory activities of the extracts.
Introduction
Urolithiasis is the occurrence of calculus anywhere in the renal system, including the kidneys, bladder, and ureter. Most renal calculi are composed of calcium phosphate and oxalate crystals. 1 The renal stone architecture involves urine super-saturation, crystal nucleation, growth, accumulation, and relocation to renal epithelial cell surfaces. 2 The disproportion between the urolithiasis promoters such as low urine volume, high urinary calcium, oxalate, uric acid, phosphate, and urolithiasis inhibitors, including macromolecules, citrate, and magnesium, may represent a potential factor in the development of lithogenesis (i.e., the formation of calculi). 3 Renal crystals are accompanied by renal cell injury, cell loss, and inflammation, which is primarily limited to areas around crystal deposits and fibrosis. 4 The oxalate build-up may lead to several pathological disorders, for instance, hyperoxaluria, calcium oxalate renal stones, and renal failure. 5 Calcium oxalate renal stones cause toxicity of epithelial cells via free radical-mediated actions, resulting in lipid peroxidation with subsequent membrane and organelle destruction followed by renal cell necrosis and apoptosis. 6 Reactive oxygen species (ROS) are produced through the involvement of the mitochondria, 7 and the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. In the presence of calcium oxalate renal crystals, a primary origin of receptor-linked ROS construction is NADPH. 8
In addition to ROS, inflammation plays an essential role in urolithiasis-induced renal damage. NF-κB plays a crucial part in the inflammation in hyperoxaluria and obstructive nephropathy via controlling genes regulation of the encoded proteins intricate in both the immune as well as the inflammatory responses such as cytokines, chemokines, growth factor immune receptors, and adhesion molecules, among others. 9 Inducing calcium oxalate urolithiasis in rats is comparable to calcium oxalate stone disease in humans. 10 Using ethylene glycol (EG) alone to cause urolithiasis has been used commonly as an experimental model; however, it showed moderately inconsistent outcomes. Thus, a modification was introduced by Fan et al. 11 to add ammonium chloride (AC). AC causes metabolic acidosis, increasing the rate of calcium oxalate renal stones deposition, and currently, EG and AC-induced urolithiasis are used extensively.12–14 Therefore, in the current study, we used a combination of 0.75% of EG (V/V) and 1% AC (W/V) to induce urolithiasis.
Though various treatment approaches for urolithiasis developed over the years, variations occur regarding the clinical indications and the effectiveness of these management options. 15 Currently, urolithiasis medical approaches are based on minimizing stone secondary risks rather than etiologies. Thus, searching for a natural source product may avert the causes and progress, or even the nephrolithiasis reappearance has attracted the scientific community. 16 Numerous polyherbal preparations are available for nephrolithiasis, for instance, Lithocare 17 and Gokshuradi Yog, 18 among others.
Grape seed extract (GSE) consists of numerous compounds, including catechin, epicatechin gallate, epicatechin gallate, and epigallocatechin. 19 GSE exhibited an extensive range of pharmacological as well as therapeutic aids. GSE demonstrated antioxidant, 20 anti-arthritic, 21 anti-inflammatory, 22 anti-obesity effect, 23 hepatoprotective, 24 neuroprotective, 25 and cardioprotective 26 actions. Furthermore, GSE alleviated diverse drug and chemical-induced toxicities such as arsenic-induced lung damage through NF-κB signaling, 27 titanium dioxide nanoparticle (TiO2-NPs)-induced hepatotoxicity through TLR-4/NF-κB, 28 thioacetamide-induced hepatic fibrosis, 29 and cisplatin-induced nephrotoxicity. 30 As for the clinical trial, a randomized, double-blind, placebo-controlled trial investigated the actions of GSE on blood pressure and vascular endothelial function in middle-aged Japanese adults with prehypertension. The study verified that GSE consumption might help maintain vascular elasticity and normal blood pressure. 31 The main target of the current study is to explore the anti-urolithic properties of GSE on EG and AC-induced urolithiasis in rats and to investigate the mechanism of action of GSE anti-urolithic actions.
Materials and Methods
Grape Seed Extraction
Grape (Vitis vinifera, family Vitaceae) seeds were obtained from a commercial source in Al-Taif, Western Province, Kingdom of Saudi Arabia, 32 in September 2021. The seeds were identified by experts in the college of agriculture and food sciences at King Faisal University to be from different local cultivars of grapes in Saudi Arabia. The seeds were washed, examined for foreign particles, and then dried at 25°C until constant weight (for one week). 300 gm of the dried grape seeds was grounded into a fine powder, then macerated in 70% ethanol (25% w/v, 3 days, room temperature) according to Badavi et al., 33 and then filtered. The filtrate was evaporated under reduced pressure to obtain GSE with a yield of 15% (w/w of dry seeds). GSE was dissolved in 0.1% DMSO to get the working solution, from which doses were given to the animals as mentioned before. 34
Animals and Ethical Statement
Wistar male rats (6–8 weeks; weight: 160–200 g) were obtained from Experimental Animal Research Centre, King Saud University, Riyadh, KSA. The animals were kept in standard laboratory food and water, ad libitum, in a ventilated cage system (12 h light/dark cycles, 20.3–23.1°C) throughout the experiment. The Institutional Animal Care and Use Committee of King Faisal University permitted the experimental protocol (KFU-REC-2022-APR-EA000590). All the experiments were implemented following the applicable processes and guidelines of the Ethical Conduct for the Use of Animals in Research at King Faisal University.
Induction of Urolithiasis
An EG- and AC-induced hyperoxaluria model was executed in the current study for induction of urolithiasis according to Azimi et al. 35 In brief, the animals were administered 0.75% v/v EG with 1% w/v AC in drinking water, ad libitum, for three days to accelerate lithiasis, followed by only 0.75% v/v EG was administrated for 28 days.13, 36 to induce nephrolithiasis. 37 AC (1%) was given only for the first 3 days, as administration of AC for more than 5 days can lead to extreme weight loss and ultimate animal death. 38
Experimental Design
Rats were assigned into six groups (n = 6). Group I: Normal control animals had free access to standard animal feed and tap water as drinking water; Groups II: Normal + GSE rats received tap water as drinking water, as well as GSE (200 mg/kg, dissolved in 0.1% DMSO) orally daily. Group III: Urolithiatic control animals in which rats received EG with AC in drinking water as mentioned before to induce urolithiasis and 0.1% DMSO (vehicle) orally; Groups IV–V: Urolithiatic animals + GSE, in which rats administered EG with AC in drinking water, as well as GSE (100 and 200 mg/kg, orally, dissolved in 0.1% DMSO), respectively. Group VI: Urolithiatic rats + cystone, received EG with AC in drinking water, as well as 750 mg/kg of cystone (orally, dissolved in 0.1% DMSO) as a positive control drug.
Relative Body Weight Measurement
For each animal, the body weight was recorded weekly. The changes in animals’ body weights were expressed as relative body weight (RBW), which is obtained via dividing the initial body weight by the absolute body weight multiplied by 100, as mentioned before. 39
Assessment of Urine Samples
Twenty-four hours urine collection, at day zero, on the 14th day and the 28th day, was achieved via individually housing the animals in metabolic cages (one rat per cage). A drop of urine is spread on a glass slide and visualized using optical and polarized light using a Leica DM3000 light microscope. The crystals were counted in five fields, and the results represented the mean as mentioned before. 40 A portion of the 24 h urine samples was used to measure citrate levels, agreeing to the colorimetric kit instructions. Another portion of the urine samples was acidified by adding 5 M HCl to measure oxalate, calcium (Ca2+), 41 and magnesium (Mg2+) concentrations using commercially available kits, whereas inorganic phosphate excreted in urine was calculated using Daly and Ertingshausen’s process. 42 Diagnostic kits were used for the uric acid concentration determination. 43
Serum Analysis for Renal Function and Inflammatory Markers
After 28 days of experimental procedure, animals were euthanatized, and blood was collected from the retro-orbital sinus and centrifuged (10 min, 1200×g). Serum obtained was stored at −20°C for further analysis of biochemical parameters. Renal function was examined by measuring serum levels of creatinine (Cr, Cat. No; ab700460), uric acid (Cat. #; ab65344), blood urea nitrogen (BUN, Cat. No; ab83362), and urea (Cat. No; ab83362) using the indicated colorimetric or ELISA kit acquired from Abcam Inc. (MA, USA). The procedures were performed in harmony with the manufacturer’s directions.
Inflammation markers including NFκB (ab176648), TNF-α (ab46070), IL-1β (ab100768), and IL-6 (ab100772); ELISA Kits were obtained from Abcam Co., Eugene, OR, USA, and measured according to the manufacturer’s instructions using a microplate reader SpectraMax i3X (Molecular devices San Jose, CA, USA).
Renal Homogenate Analysis
At the end of the experiment and after sacrificing the animals, the left kidney from each animal was removed, washed in ice-cold normal saline, and weighed. The left kidneys were used for lipid peroxidation (malondialdehyde, MDA) determination and antioxidant enzyme assessment, for instance, catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPX) enzyme activities using the appropriate diagnostic kit. To detect the effect of autophagy on kidney injury due to calcium oxalate nephrolithiasis, we measured the release of urinary, renal tubular injury markers, including neutrophil gelatinase-associated lipocalin (NGAL, Cat. No; ab207925) and kidney injury molecule-1 (Kim-1, Cat. No; ab119597) 44 using ELISA kits acquired from Abcam Inc. (MA, USA).
Renal Histopathology
The right kidney was fixed in 10% neutral buffered formaldehyde, followed by dehydration, clearing, and paraffin embedding of renal tissues. Renal sections of 5 µm in thickness were prepared and stained with hematoxylin and eosin (H&E).
Statistical Analysis
Data were presented as mean ± SD. For multiple comparisons, one-way analysis of variance (ANOVA) followed by Tukey–Kramer as a post hoc test was performed. The .05 level of probability was used as the significance level. All statistical analyses were performed using Graph Pad software (version 8, San Diego, CA, USA).
Results
Effects of GSE on Body Weight
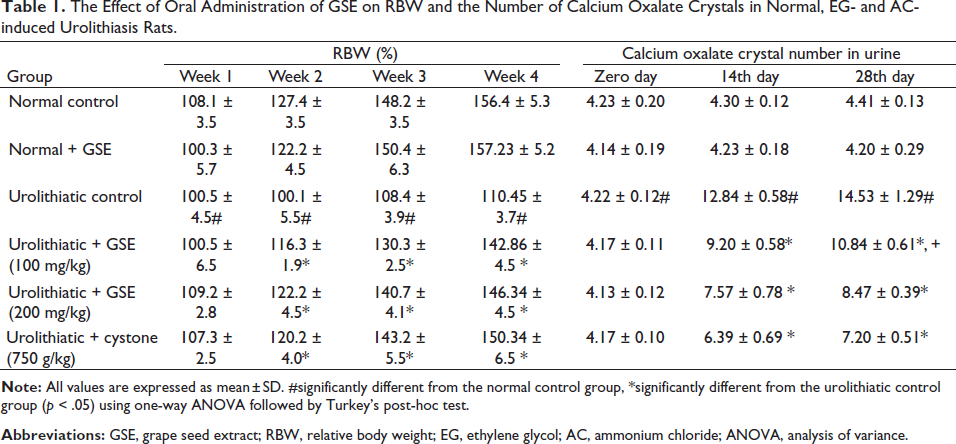
Normal control rats exerted a significant increase in RBW, whereas urolithiatic animals showed a significant decrease in RBW p < .05. Cystone and GSE (100 and 200 mg/kg/BW) significantly prevented this detrimental RBW reduction, as presented in Table 1.
The Effect of Oral Administration of GSE on RBW and the Number of Calcium Oxalate Crystals in Normal, EG- and AC-induced Urolithiasis Rats.
Effects of GSE on Histopathological Examination
Figure 1a represents the histopathological examinations of renal samples obtained from different experimental groups. The study showed dilated proximal and distal tubules (black arrows) occupied with calcium oxalate crystals (blue arrows) and mild degenerative changes represented by hyper-eosinophilic cytoplasm of renal tubules and shrinkage of glomerular tufts. Cystone (750 mg/kg) and GSE (100 and 200 mg/kg) showed recovery of renal lesions characterized by individual tubular dilation, few deposits of calcium oxalate crystals (head arrow), and restoring renal degeneration.
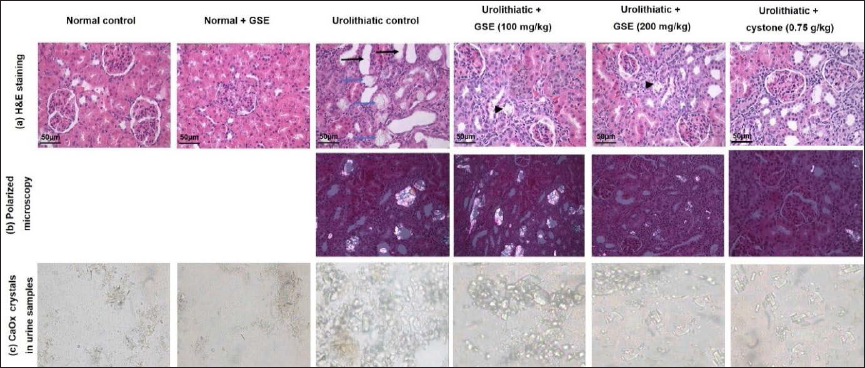
Effects of GSE on Urine Analysis
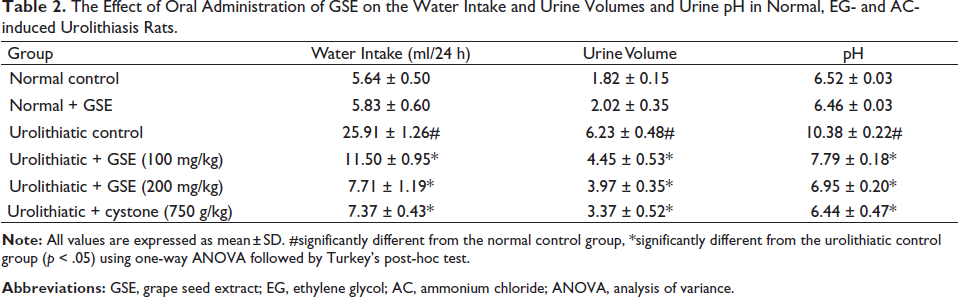
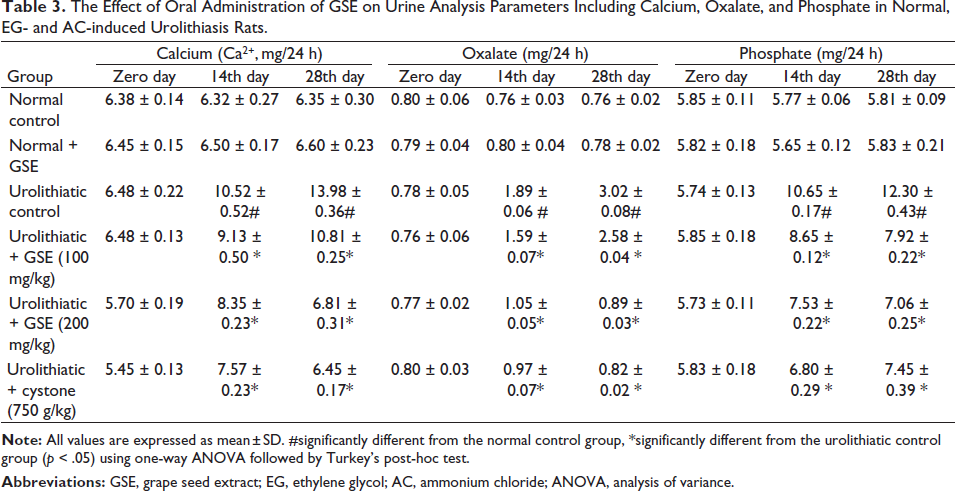
Urolithiatic animals exhibited a significant increase in water intake, urine volume, and pH. Treatment with GSE (100 or 200 mg/kg) and cystone (750 mg/kg) decreased these parameters. Oxalate crystals were undetectable in the urine samples obtained from the normal control or normal receiving GSE alone; however, urolithiatic rats’ urine displayed many oxalate crystals. The urine of urolithiatic rats receiving 100 mg/kg of GSE showed a moderate number of oxalate crystals. The urine of urolithiatic rats that received GSE (200 mg/kg) or cystone showed fewer oxalate crystals, as illustrated in Figure 1c and Table 1. In addition, urolithiatic control exhibited a significant augmentation in urolithiasis promotors such as the urinary elimination of calcium, phosphate and oxalate ions, and uric acid. As for urolithiasis inhibitors, urolithiatic animals exhibited a substantial reduction in the excreted magnesium and citrate, linked to the normal control animals. Treatment with GSE or cystone significantly reduced urolithiasis promoters, including excreted calcium, oxalate, phosphate, and uric acid excretion, and augmented urolithiasis inhibitors such as excreted magnesium and citrate as compared with those of urolithiatic control rats (Tables 2−4).
The Effect of Oral Administration of GSE on the Water Intake and Urine Volumes and Urine pH in Normal, EG- and AC-induced Urolithiasis Rats.
The Effect of Oral Administration of GSE on Urine Analysis Parameters Including Calcium, Oxalate, and Phosphate in Normal, EG- and AC-induced Urolithiasis Rats.
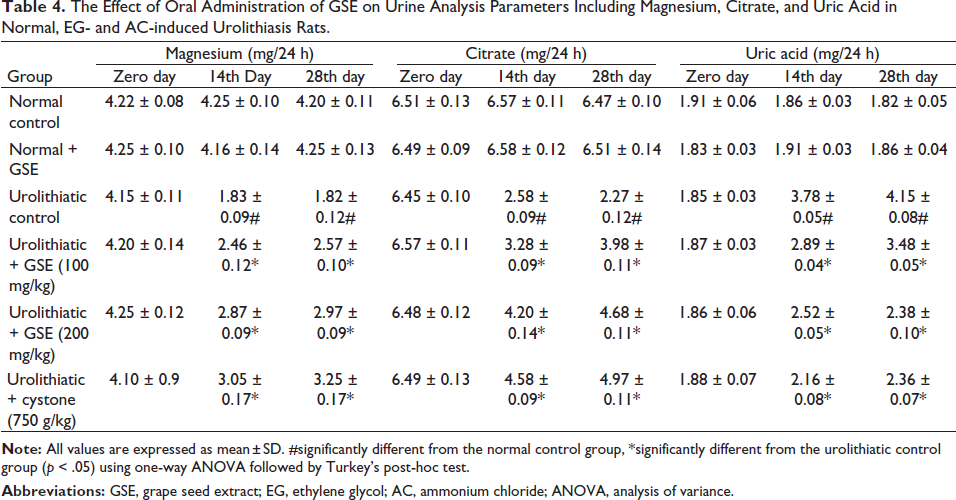
The Effect of Oral Administration of GSE on Urine Analysis Parameters Including Magnesium, Citrate, and Uric Acid in Normal, EG- and AC-induced Urolithiasis Rats.
Effects of GSE on Kidney Function Parameters
EG- and AC-induced urolithiasis animals displayed a significant intensification in several renal function parameters, comprising serum creatinine, BUN, urea, and uric acid, in relation to the normal animal group. In contrast, GSE administration, together with EG and AC, considerably lessened the serum concentrations of creatinine, urea, uric acid, and BUN compared to urolithiatic rats. These outcomes were comparable to those of the group receiving cystone as the standard drug, as presented in Figure 2a and b.
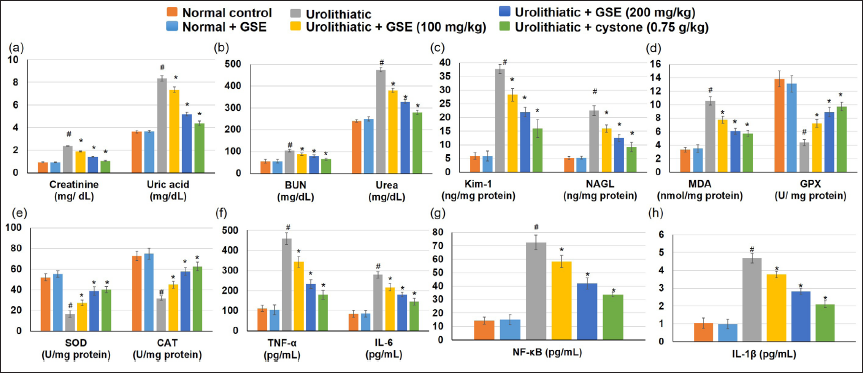
Effects of GSE on Renal Tubular Injury Markers
As shown in Figure 2c, urolithiatic rats displayed a significant intensification in renal tubular injury markers, including Kim-1 and NAGL. In contrast, GSE administration, together with EG and AC, considerably lowered both Kim-1 and NAGL.
Effects of GSE on Oxidative Stress Markers in Renal Tissues
The urolithiatic rats demonstrated a significantly higher MDA level in the kidney tissue compared with the control groups, whereas the administration of GSE and cystone declined the MDA level when linked to urolithiatic animals. As for the antioxidant enzymes, CAT, GPX, and SOD activities were significantly lower in urolithiatic animals than in control groups. Administration of GSE and cystone amplified the antioxidant enzyme activities more than in the urolithiatic rats. No significant differences were observed between normal control rats and those treated with GSE (Figure 2d and e).
Effects of GSE on Inflammatory Mediators
The urolithiatic rats demonstrated significantly higher inflammatory mediators, including TNF-α, NF-κB, IL-6, and IL-1β levels, in the kidney tissue when compared with the control groups, whereas the administration of GSE and cystone declined the TNF-α, NF-κB, IL-6, and IL-1β levels when linked to urolithiatic animals, as illustrated in Figure 2e, f, and g.
Discussion
Despite the progression in understanding the molecular mechanisms controlling the renal calculi formation and management, the pathological condition still occurs in clinical situations with a high prevalence and recurrence. In clinical practices, risk factors associated with urolithiasis include hyperoxaluria, hypocitraturia, abnormal urine pH, and low urine volume. 45 The current investigation was directed to evaluate the anti-urolithic action of GSE by inspecting the antioxidant and anti-inflammatory activity of the extract as a natural agent in comparison with cystone as a standard drug in EG-induced urolithiasis.
In the current study, urolithiatic animals showed a significant decrease in RBW, which presented as a preliminary indicator of renal toxicity, in accordance with earlier studies.16, 40 The management with cystone and GSE significantly prevented this detrimental RBW reduction. This prevention in RBW reduction may be due to the capability to antagonize the toxicity effects of EG and AC. EG’s final metabolite is oxalic acid, which acts mainly in the kidney, causing acute poisoning symptoms. 46 Due to renal stone induction, urolithiasis animals stopped eating; thus, their body weight declined. Furthermore, the urine of urolithiatic rats shows many oxalate crystals, as mentioned before in several studies. 17
Urolithiatic control animals exhibited a significant increase in the urine volume and pH and an upsurge in urinary calcium oxalate crystals numbers. Moreover, urolithiatic control exhibited a significant augmentation in the urinary elimination of calcium, phosphate, and oxalate ions, as well as a substantial reduction in the excreted magnesium and citrate as related to the normal control animals. Administration of EG led to hyperoxaluria and nephrotoxicity, which was evidenced by histopathological investigations and was in concordance with other studies that reported tubular hypertrophy and tubulointerstitial damage and widespread calcium oxalate crystal deposition in the kidney tubules in different urolithiatic rat models.16, 47 EG causes hyperphosphaturia, hypercalciuria, and hyperoxaluria, leading to urolithiasis.2, 11, 46 The formation of stones by EG is caused mainly by hyperoxaluria, which leads to elevation of both kidney excretion and retention of oxalate. 11 Due to EG, there is an imbalance between the lithogenesis promoters such as phosphate, oxalate, calcium, uric acid, and low urine volume on the one hand and inhibitors, including magnesium, and citrate, on the other hand. 10 Thus, the elevation of oxalate, calcium, and uric acid, as observed in this study, triggered the crystallization development and the crystal precipitation as calcium oxalate in nephrons, leading to renal epithelial cell damage. 48 The histological outcomes of the present study supported such signs by the existence of oxalate crystals in the lumen of the renal tubules, leading to inflammation and necrosis. As for hypercalciuria, the high level of urine calcium in the current experiment could be due to renal injuries, which cause calcium loss in the urine. This hypothesis was supported by Ibrahim and El-Khateeb, 49 who interpreted that membrane damage in hyperoxaluria results in renal leakage of calcium, which further leads to nucleation and precipitation of calcium oxalate or phosphate in the urine and subsequently crystal growth. Concerning hyperphosphaturia, increased excretion of urinary phosphate along with oxalate stress provides a condition suitable for stone formation by forming calcium phosphate crystals, which induces the deposition of calcium oxalate. 47 Management with GSE and cystone decreased urine volume and pH, diminished urinary calcium oxalate crystals, reduced the excreted calcium, oxalate, and phosphate excretion, and augmented urinary elimination of magnesium and citrate.
In the current study, urolithiatic animals displayed a significantly higher serum creatinine, BUN, urea, and uric acid than normal controls, indicating a severely deteriorated kidney function. All these effects could be attributed to lowered clearance from blood to urine due to a reduction in the glomerular filtration rate and the renal tubular cells’ damage. Urolithiatic animals exhibited a diminished glomerular filtration rate due to the obstruction to urine flow by stones in the urinary system. 17 In addition, proximal tubule cell damage inhibited the clearance of waste products, primarily nitrogenous substances such as blood creatinine, urea, and uric acid, resulting in augmented levels of these waste products in the blood,50, 51 and thus, renal damage and deteriorated kidney function. High urine uric acid levels increased the risk of stone formation as being one of the crystallization promoters. 52 The reason could be illustrated in uric acid-binding proteins’ ability to bind calcium oxalate and modify its crystallization, reflecting its essential role in the formation of stones. 1 In contrast, treatment with GSE, or cystone together with EG and AC, lowered the serum concentrations of creatinine, urea, uric acid, and BUN compared to urolithiatic rats. These changes may be attributed to increased blood creatinine, urea, and uric acid clearance into the urine. These outcomes are consistent with GSE actions in different models of kidney damage, including renal ischemia-reperfusion, 53 cadmium, 54 arsenic, 55 cisplatin, 30 and induced renal injuries. Kidney injury molecule-1 (Kim-1) is a type 1 transmembrane glycoprotein, and its expression is significantly increased in proximal tubule epithelial cells following renal injury, 56 whereas NGAL is widely used to reveal the early renal tubular injury and dysfunction in kidney disease. 57 In the existing investigation, urolithiastic animals demonstrated a significant intensification in renal tubular injury markers, including Kim-1 and NAGL, whereas GSE and cystone lowered both Kim-1 and NAGL, indicating deterred renal tubular injury.
Exposure of the renal epithelial cells to high oxalate levels and calcium oxalate/ phosphate crystals generates excess ROS, causing injury and inflammation. 8 Administration of EG and AC resulted in elevated MDA levels and lowered CAT, GPX, and SOD activities. At the same time, the administration of GSE and cystone deteriorated MDA levels and amplified the antioxidant enzyme activities. The increase in lipid peroxidation and ROS resulted from calcium oxalate crystal formations in the tubular cells. These data go in parallel with many publications, which demonstrated oxidative stress and lipid peroxidation in the stone formation status.6, 13, 58 Augmented calcium oxalate crystals caused the abnormal formation of ROS and oxidative stress, which resulted in injuries in the renal tubular cells. 6 Oxalate caused increased ROS generation and reduced aminophospholipid translocase activity, which strongly relates to hyperoxaluria, promoting calcium oxalate urolithiasis, and facilitating the redistribution of phosphatidylserine in epithelial cells of the kidney. 59 Afzal et al. 13 reported that oxalate concentration amplified in urine due to inducing lipid peroxidation and caused glomerular and tubular damage. Furthermore, Huang et al. 58 stated that elevated oxalate concentration in urine induced lipid peroxidation and renal damage by reacting with polyunsaturated fatty acids in the cell membrane. The hyperoxaluric rats exhibited a deficiency in GSH content, which was related to the reduction in GSH-synthesizing enzyme activities, glutathione reductase, and glucose 6-phosphate dehydrogenase.18, 60 On the other hand, GSE exhibited remarkable antioxidant activity in different non-renal models, for instance, type-1 diabetes, 61 titanium dioxide nanoparticle-induced hepatotoxicity, 28 and D-galactose-induced ageing mouse model, 62 and in diverse renal models including renal ischemia-reperfusion, 53 carboplatin and thalidomide, 34 cisplatin, 30 and induced renal damage.
The inflammation status observed in the current study was detected in the histopathological observations in urolithiastic animals and was confirmed by the significant increase in the levels of the inflammatory mediators, including TNF-α, NF-κB, IL-6, and IL-1β. However, the administration of GSE and cystone decreased the inflammatory mediators’ levels and improved the inflammation. In an inactivated state, cytosolic NF-κB is complexed with the inhibitory protein IκBα. Extracellular signals such as TNF-α and IL-1β can activate IκB kinase, which dissociates NF-κB from IκBα, causing proteasomal degradation of IκBα. This causes the release of the p65/p50 heterodimers and their translocation into the nucleus, where they promote NF-κB target genes involved in inflammatory, immune, and acute phase responses.63, 64 GSE exhibited anti-inflammatory actions in different inflammatory conditions, such as in endotoxin-stimulated RAW 264.7 macrophages by inhibiting the NF-κB signaling pathway 65 and in titanium dioxide-induced hepatotoxicity via deterring the TLR4/ NF-κB signaling pathway. 28 Furthermore, GSE showed an anti-inflammatory effect in many other models, including the monocrotaline-induced pulmonary arterial hypertension 66 and D-galactose-induced ageing. 62 As for renal action, GSE demonstrated reno-protective activity via attenuating inflammation in diverse renal models, including renal ischemia/reperfusion, 53 arsenic, 55 carboplatin, and thalidomide-induced renal injury 34 and renal fibrosis. 67 Based on the present results and previous literature, it could be suggested that GSE produces anti-inflammatory actions by affecting the canonical pathway of NF-κB activation.
Conclusion
The current study addressed the recent approaches in urolithiasis management based on diminishing secondary stone risks rather than etiologies. The present study verified that GSE could protect against EG-induced rat kidney dysfunction. GSE could improve the histopathological alterations, the oxalate crystal formation, and the oxidative stress and inflammation status accompanying urolithiasis. Despite these potential effects, further clinical trials are needed to evaluate the safety and effectiveness of GSE in human beings.
Footnotes
Summary
GSE protected against EG-induced renal stones, as demonstrated by improved kidney function, depressed histological alterations, and oxalate crystal formation. The GSE effect could be because of its antioxidant and anti-inflammatory effects.
Abbreviations
Acknowledgment
The authors are grateful to the college of clinical Pharmacy and Deanship of Scientific Research at King Faisal University for financially supporting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [GRANT745].
Statement of Informed Consent and Ethical Approval
The Institutional Animal Care and Use Committee of King Faisal University permitted the experimental protocol (KFU-REC-2022- APR-EA000590). All the experiments were implemented following the applicable processes and guidelines of the Ethical Conduct for the Use of Animals in Research at King Faisal University (found in the methodology section).
