Abstract
Background:
Gestational diabetes mellitus (GDM) is a condition of glucose intolerance and insulin resistance only diagnosed during pregnancy. GDM has exhibited several adverse effects on both mother and offspring. The current research focuses on discovering visnagin’s beneficial properties against the streptozotocin (STZ)-induced GDM in rats via alleviating the inflammation and oxidative stress.
Materials and Methods:
GDM was caused in the pregnant rats by the administration of 25 mg/kg of STZ by the intraperitoneal route and then treated with 20 mg/kg of visnagin for 20 consecutive days. The rats’ body weight was measured, and fasting blood glucose (FBG) status was determined using a standard glucometer. The contents of total cholesterol (TCh), triglycerides (TG), low-density lipoprotein (LDL), and high-density lipoprotein (HDL) were assessed using kits. The MDA level, total antioxidant capacity (TAC) status, and activities of catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione S-transferase (GST) were determined using assay kits. Kits also assessed the contents of TNF-α and IL-1β. The contents of TNF-α and IL-1β effectively improved the body weight and decreased the FBG status in the GDM rats. The visnagin also decreased the TCh, TG, and LDL, and elevated the HDL content. The content of MDA was decreased and the visnagin treatment increased SOD, CAT, GST, and GPx, and the visnagin treatment increased SOD, CAT, GST, and GPx activities SOD, CAT, GST, and GPx activities. The visnagin effectively decreased the STZ-induced histopathological alterations in the pancreas.
Conclusion:
Altogether, our investigation results suggest a beneficial role visnagin against STZ-induced GDM in rats via inhibiting the inflammatory responses. Hence, it can be a talented therapeutic candidate for the successful management of GDM.
Introduction
Gestational diabetes mellitus (GDM) is a common metabolic complication characterized by glucose intolerance, inflammation, oxidative stress, and hyperinsulinemia, which arises only during pregnancy. 1 Several risk factors such as the age of gestation, obesity, and heredity may contribute to the initiation of GDM. 2 Hyperglycemia during pregnancy is connected with numerous harmful effects on both mother and offspring. A woman with a GDM history has an increased risk of developing insulin resistance (IR) associated with type-2 diabetes and cardiovascular complications. 3 The incidence rates of GDM have increased gradually worldwide each year due to lifestyle changes. 4 The exact pathophysiology of GDM has not been fully discovered yet; however, several research reports have highlighted that insulin-antagonistic hormones such as progesterone, estrogen, and prolactin produced by the placenta initially play a significant role in the development of GDM. 5
During the early phases of pregnancy, the increased inflammatory responses are tightly connected with higher risks of developing GDM and the progression of hyperglycemia. Additionally, oxidative stress was well-established in GDM, and antioxidant status was found deceased in the GDM women.6, 7 The chronic hyperglycemic condition has a straight connection to the inflammatory reactions, where the augmentation of cytokines can trigger the pancreatic β-cell destructions and result in dysfunction of the endocrine pancreas. 8 The inflammatory regulators such as TNF-α, IL-6, and IL-1β are critical factors in the initiation of GDM. Elevated β-cells apoptosis has been extensively reported as a crucial factor of GDM in animal models and human cases. 9 A TNF-α and its specific roles in insulin production and activity impairment were well elucidated. 10 The many inflammatory cytokines play a crucial role in the β-cell apoptosis, and other mechanisms were also reported in the dysfunction of β-cells.11, 12 Oxidative stress is connected with the stimulation of inflammatory cascades and is characterized by the irregular production of inflammatory mediators. 13 The fundamental molecular mechanisms of IR remain to be elucidated. However, several reports highlighted that local and systemic accumulation of TNF-α and IL-6 could be a critical factor in causing IR during GDM.14–16
The occurrence of GDM is increasing rapidly in women because of the elevated epidemiology of obesity; however, the successful treatment options for GDM remain controversial. 17 GDM is tightly connected with several neonatal complications such as increased obesity risks and cardiometabolic disorders, which contribute to diabetes and other metabolic syndromes. Several reports suggest that long-term GDM complications in women have an increased risks of cardiovascular complications.18, 19 During GDM treatment, insulin is generally used to decrease the blood glucose level however IR reduces its clinical usage in preventing neonatal and maternal complications. 20 Hence, there is a great demand to explore and discover new safer and more effective drugs, with the ability to decrease blood glucose and inhibit IR for the successful management of GDM.
At present, the existing therapies for GDM only focus on hyperglycemia and have a fewer effects on reducing inflammation and oxidative stress.21, 22 Therefore, a new therapeutic option to effectively target and inhibit the inflammation and oxidative stress to alleviate GDM is highly warranted. Visnagin, a furanocoumarin derivative found extensively in the fruits of the Ammi visnaga plant, has already been reported to show several biological properties. 23 A previous study highlighted that the visnagin effectively protected cerulein‐triggered pancreatitis in animals via increasing Nrf2 expression and decreasing NF‐κB. 24 Additionally, visnagin exhibited the anti-inflammatory property by decreasing the NF‐κB expressions in lipopolysaccharide-stimulated microglial cells. 25 Liu et al. and Asnaniet et al.26, 27 reported that visnagin demonstrated cardioprotective effects against doxorubicin-stimulated cardiomyopathy. The cardioprotective effects of visnagin-encapsulated nanoparticles were well reported. 28 A recent investigation exhibited that the visnagin protected the testes of lead acetate‐intoxicated animals. 29 Fu et al., 30 reported that visnagin ameliorated the myocardial ischemic reperfusion injury. However, the therapeutic roles of visnagin against GDM were not elucidated yet. Therefore, this research work focused to discover the visnagin’s beneficial properties against the streptozotocin (STZ)-induced GDM in rats by the alleviation of inflammation and oxidative stress.
Materials and Methods
Chemicals
Visnagin, STZ, and other chemicals were purchased from Sigma-Aldrich, USA. All the ELISA kits for the biochemical experiments were procured from Thermofisher and Biocompare, USA, respectively.
Experimental Rats
The Wistar albino rats from both sexes weighing above 210 ± 30 g were used in this study. The animals were confined in a polypropylene cage and sustained in laboratory circumstances with temperature (22–26°C), humidity (40–70%), and 12-hr light/dark series. All animals were fed with a regular pelleted diet with pure drinking water. The rats were acclimatized for a week before conducting the main experiments.
Establishment of GDM
The experimental rats were continuously fed with a high-fat diet for eight consecutive weeks, and the bodyweight gain of all experimental rats was checked regularly. After that, the rats were fasted overnight, and then vaginal smears were completed each day to identify the estrous cycle. Then, the animals with the estrous stage were mated with active and healthy male rats at a 2:1 ratio. After 24 hr, the presence of sperm was checked using a microscope to ensure the pregnancy of rats for 0 days. Then, the rats with pregnancy were marked and separated from the other animals. The non-pregnant rats, after a week of mating, were removed from further experiments. The pregnant rats were injected with 1% of STZ (25 mg/kg) prepared in 0.1 M citrate buffer (pH 4.4) to initiate the GDM, and the control rats were injected with the same amount of saline without STZ. On the fourth day of gestation, animals with glucose content >10 mmol/L were deliberated as diabetic and further used as a GDM model.
Treatment Procedure
A set of six healthy pregnant rats without GDM was used as a control group (Group I). The rats with GDM were distributed into two clusters with six rats in each GDM group (Group II) and GDM+visnagin treated group (Group III). Group III rats were treated with 20 mg/kg of visnagin orally for 2 weeks. The rats from the control and GDM groups were provided with the same saline concentration. The group of rats was sustained until the parturition to examine the influence of visnagin on the litter numbers. After the 20th day of treatment, rats were sacrificed, blood was gathered, serum was gathered, and serum was separated for biochemical assays. The liver, pancreas liver, and pancreas tissues were removed and used for the biochemical and histopathological assays.
Determination of Body Weight and Fasting Blood Glucose (FBG)
The changes in the maternal body weight and FBG level of both control and treated rats were determined on the 18th day of gestation. Using the sensitive weight balance, all the experimental rats’ body weight was measured and data were tabulated. The changes in the contents of FBG in the rats were determined with the help of a commercially procured glucometer (Roche, USA).
Measurement of Lipid Profiles
The blood samples from the control and treated rats were gathered on the 18th day of gestation, and serum was separated to assess the lipid profiles. The status of TCh, TG, HDL, and LDL was examined with the aid of commercially procured kits by using the protocols described by the manufacturer (MyBioSource, USA).
Determination of Oxidative Stress and Antioxidant Biomarkers
The liver tissues were removed from control and experimental rats and cleansed with saline solution. Then liver tissues were homogenized using ice-cold buffered saline and then centrifuged at 15,000 rpm for 10 min. Then, the supernatant was collected and used for the biochemical examinations. The MDA status, TAC, and activities of CAT, SOD, GPx, and GST were investigated with the aid of commercially procured assay kits according to the manuals provided by the manufacturer (Thermofisher, USA).
Measurement of Inflammatory Cytokines Level
The contents of pro-inflammatory biomarkers such as TNF-α and IL-1β in the control and experimental rats were evaluated using commercially purchased kits according to the protocols given by the manufacturer (Biocompare, USA).
Histopathological Study
The pancreatic tissues were removed from the animals and managed with buffered saline. Then, pancreas tissues were embedded in the paraffin and then sliced into sections at 5 µm diameter. The pancreas sections were stained with hematoxylin and eosin, and finally, the changes in the histology of pancreatic tissues were identified under the optical microscope.
Statistical Analysis
All the data were statistically assessed using the GraphPad Prism software. The data were portrayed as mean ± SD of three separate measurements. All the data were measured by one-way ANOVA and Tukey’s post hoc assay. The p-value lower than 0.05 was deliberated as significant.
Results
Effect of Visnagin on the FBG and Bodyweight of the Experimental Rats
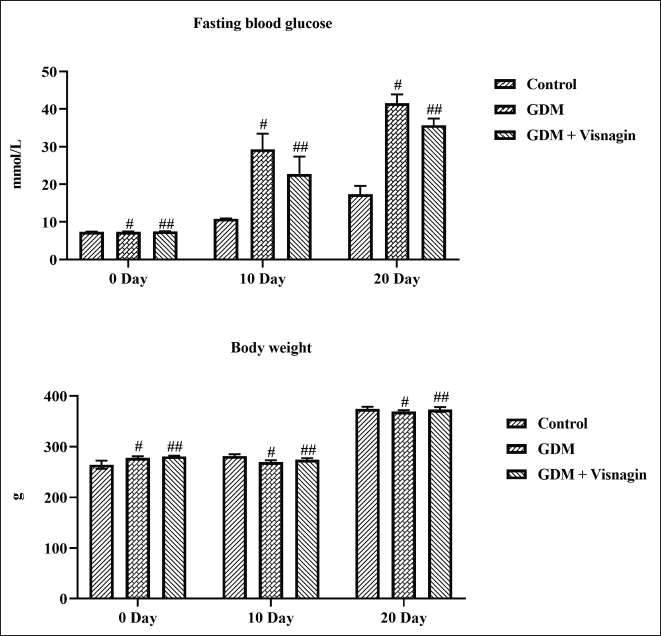
Figure 1 represents the effect of visnagin treatment on the FBG status and body weight of control and experimental rats. The GDM rats demonstrated the augmented FBG content on the 10th and 20th days. Interestingly, 20 mg/kg of visnagin treatment effectively decreased the FBG content in the GDM rats. On the other hand, reduced body weight was observed in the STZ-induced GDM rats. About 20 mg/kg of visnagin treatment also enhanced the bodyweight of STZ-stimulated GDM rats (Figure 1).
Effect of Visnagin on the Bodyweight, Live Pups, Survival Index, and Anogenital Distance of Pups
The effect of visnagin treatment on the number of live pups, body weight of pups, anogenital distance (AGD), and survival rate of pups was examined and outcomes are presented in Table 1. The reduced number of live pups and decreased survival index of pups were noted in the GDM rats. The GDM group also exhibited decreased body weight and the AGD of both pups sexes was stressed. However, the treatment with 20 mg/kg of visnagin was remarkably enhanced the number of live puppies and survival index. About 20 mg/kg of visnagin-treated AGD rats also demonstrated increased bodyweight of puppies and AGD of pups from both sexes (Table 1).
Effect of Visnagin on the Live Pups, Bodyweight, AGD, and Survival Index of Pups.
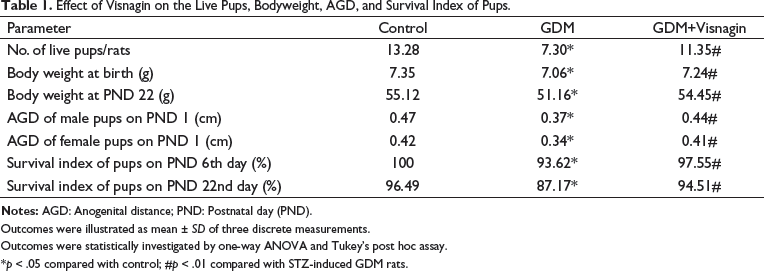
Outcomes were illustrated as mean ± SD of three discrete measurements.
Outcomes were statistically investigated by one-way ANOVA and Tukey’s post hoc assay.
*p < 0.05 compared with control; #p < 0.01 compared with STZ-induced GDM rats.
Effect of Visnagin on the Lipid Profiles of the Experimental Rats
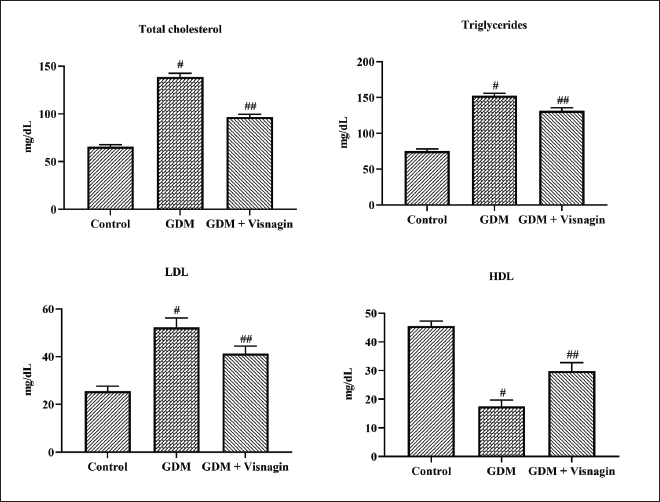
Figure 2 represents the effect of visnagin treatment on the contents of lipid profiles in the control and experimental rats. The augmented levels of TCh, TG, and LDL were observed in the serum samples of STZ-induced GDM rats. The GDM rats also demonstrated the depleted contents of HDL in the serum when compared with the control. However, 20 mg/kg of visnagin-treated GDM animals exhibited decreased levels of TCh, TG, and LDL and increased HDL (Figure 2).
Effect of Visnagin on the Oxidative Stress Biomarkers in the Liver Tissues
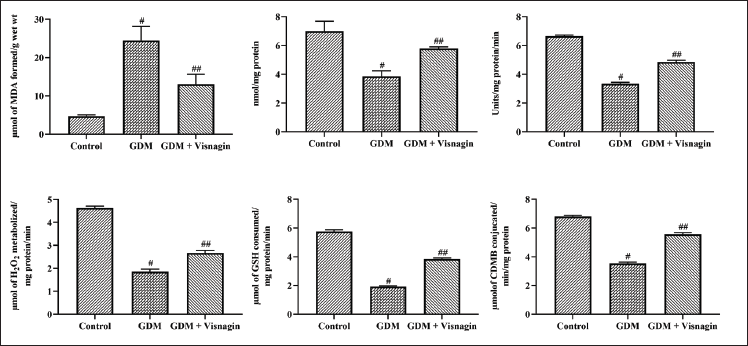
The MDA, TAC status, and SOD, CAT, and GST activities on the liver tissues were investigated, and the outcomes are furnished in Figure 3. The STZ-stimulated GDM animals exhibited enhanced MDA content and decreased TAC levels. The GDM rats also showed reduced SOD, CAT, GST, and GPx activities. As represented in Figure 3, 20 mg/kg of visnagin administration depleted the MDA status and augmented the TAC level in the GDM rats. About 20 mg/kg of visnagin treatment also exhibited improved CAT, SOD, GST, and GPx activities in the GDM rats (Figure 3).
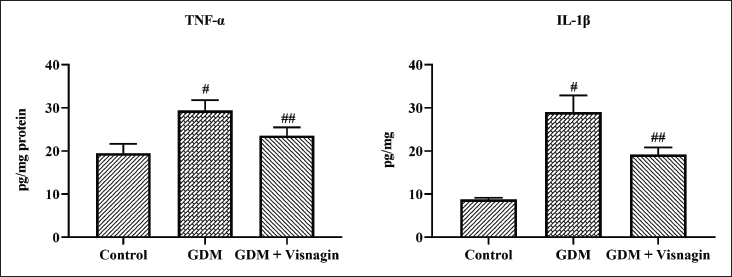
Effect of Visnagin on the Levels of Pro-inflammatory Markers in the Liver Tissues of GDM Rats
Figure 4 represents the effect of visnagin treatment on the contents of pro-inflammatory cytokines, that is, IL-1β and TNF-α, in the control and experimental rats. Figure 4 revealed the increased levels of IL-1β and TNF-α in the STZ-induced GDM rats. Interestingly, 20 mg/kg of visnagin administration appreciably decreased the IL-1β and TNF-α levels in the GDM rats (Figure 4).
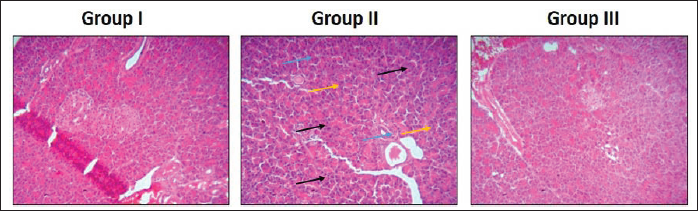
Effect of Visnagin on the Pancreas Histopathology of Experimental Rats
The effect of visnagin treatment on the histopathology of the pancreas in the control and experimental rats was assessed microscopically by the H&E staining technique and the findings are displayed in Figure 5. The normal cellular structures without the signs of inflammation were observed in the pancreatic tissues of normal pregnant rats. Contrastingly, when compared with the control, the pancreatic tissues from STZ-induced GDM rats demonstrated the signs of inflammation, inflammatory cell infiltrations, pancreatic islet cell contraction, and enlarged adipose size symptoms. Nonetheless, 20 mg/kg of visnagin administration appreciably decreased the pancreatic lesion, reduced adipose size, and decreased inflammation, which the STZ induces in the pancreatic tissues (Figure 5).
Effect of Visnagin on the Pancreas Histopathology of Experimental Rats. Group I: The Pancreas of Normal Pregnant Rats Demonstrated Normal Cellular Structures without the Inflammatory Signs. Group II: The Pancreatic Tissues of STZ-induced GDM Rats Exhibited the Inflammatory Signs (Blue Arrows), Inflammatory Cell Infiltrations (Black Arrows), and Pancreatic Islet Cell Contractions (Yellow Arrows). Group III: 20 mg/kg of Visnagin-treated GDM Rats Showed the Appreciable Reduction in the Histological Changes Induced by the STZ in the pancreatic Tissues.
Discussion
GDM is a special type of diabetes that appears only during women’s pregnancy.31, 32 GDM is developed due to poor glucose tolerance and IR during pregnancy. 33 GDM causes more risks of type-2 diabetes to both mother and fetus. 34 GDM may also lead to complications in fetal growth. It was reported that 15–45% of infants born to GDM women demonstrate macrosomia signs. 35 In pregnant women, normally endogenous glucose production was increased by more than 30% to fulfill the fasting energy requirements at the late gestational stage. However, a GDM woman’s dysfunction of β-cells is an indicator of IR and leads to hyperglycemia. 36 GDM has close connections with the overgrowth of fetuses and increased perinatal morbidity and mortality in both mothers and infants.37, 38
The exact pathophysiology of GDM development was not fully elucidated. However, IR is a major factor in GDM initiation and development. 39 Although the clear molecular mechanism is still unknown, many studies revealed that inflammation is a vital player in IR and pancreatic β-cells dysfunction.40, 41 During GDM, the inflammatory response plays a major role, which is defined by increased TNF-α, IL-1β, and IL-6 levels.42, 43
Several pieces of literature are highlighted that the increased maternal inflammation and oxidative stress can disturb the insulin signal transductions to initiate the IR, thus triggering the lipid and glucose metabolism disorders.44, 45 TNF‑α, IL‑6, and IL‑8 are the critical players of IR during GDM.46, 47 Furthermore, the elevated status of TNF-α, IL-6, and IL-8 was already been documented in pregnancy with GDM conditions, indicating that pro-inflammatory mediators could participate in the progression of IR during GDM. 48 Furthermore, the increased contents of these cytokines in blood stream can affect the biological activities of insulin that ultimately leading to IR.49, 50 The current findings revealed that the GDM rats showed increased levels of IL-1β and TNF-α; however, the visnagin treatment appreciably depleted the IL-1β and TNF-α levels.
Several pieces of literature have demonstrated that hyperinsulinemia can increase the accumulation of free radicals and trigger oxidative stress damage to numerous organs.51, 52 The previous reports highlight that the accumulation of free radicals is increased, and the status of antioxidants was found depleted in women with GDM, which could be a major reason for congenital anomalies.53, 54 In GDM women, the excessive amount of glucose is auto‑oxidized, which results in the increased production of free radicals, while the antioxidant defense was also found to decrease in GDM women. 55 The CAT, SOD, and GPx are the crucial antioxidants, which detoxify the toxicity and adverse effects of free radicals. 56 GPx is a selenium-containing antioxidant enzyme that occurs in considerable amounts in the body, which detoxifies and converts hydrogen peroxide into water. During the diabetic condition, a decreased GPx activity was reported. 57 The over-accumulation of hydroxyl radicals and depleted antioxidants are the major factors of oxidative stress. 58 Here, our present findings represented that the GDM animals displayed increased MDA and decreased SOD, CAT, GST, and GPx activities. Interestingly, the visnagin administration appreciably decreased the MDA level and improved the SOD, CAT, GST, and GPx activities in the GDM rats.
Diabetic dyslipidemia is characterized by a depleted amount of HDL, augmented status of TCh, and LDL, and TG is tightly connected with the atherosclerotic damages. 59 However, the precise underlying molecular mechanisms of diabetic dyslipidemia are not fully elucidated yet, while obesity and IR are often reported to impair the lipid metabolism. 60 Followed by the increased IR, the circulating contents of lipids found increased in the blood. 61 However, maternal lipid metabolism was normally impaired in GDM women. 62 Therefore, the augmentation of lipid profiles, for example, TCh, TG, and LDL and depletion of HDL content are generally reported in GDM women. 63 Chen et al. 64 reported that the GDM mice exhibited the augmented status of TCh, TG, and LDL and a depleted amount of HDL in serum, which indicates that GDM might be associated with a disorder of blood lipid metabolism. Similarly, our results also witnessed that the GDM rats demonstrated increased contents of lipids and decreased HDL levels in the serum. However, the treatment with the visnagin effectively depleted the LDL, TCh, and TG and enhanced the HDL in the serum of GDM animals.
Conclusion
In conclusion, our findings from this study revealed a therapeutic property of visnagin against STZ-induced GDM in rats. The visganin treatment appreciably decreased the inflammatory markers, reduced oxidative stress, and improved the activities of antioxidant enzymes. The visnagin administration also modulated the status of lipid profiles in the GDM rats. Altogether, the current results witnessed the beneficial roles of visnagin against GDM. Furthermore, additional studies in the future can help to understand the underlying mechanisms of therapeutic roles of the visnagin and can lead to the development of visnagin as a new therapeutic candidate to treat GDM.
Abbreviations
GDM: Gestational diabetes mellitus; STZ: Streptozotocin; FBG: Fasting blood glucose; TCh: Total cholesterol; TG: Triglycerides; LDL: Low-density lipoprotein; HDL: High-density lipoprotein; CAT: Catalase; SOD: Superoxide dismutase; GPx: Glutathione peroxidase;
Footnotes
Acknowledgment
This work was supported by Jiyang District People’s Hospital, No.19, Xinyuan Road, Ji Nan City, 251400, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Research experiments were conducted in this article with animals were approved by the Ethical Committee and responsible authorities of Jiyang District People’s Hospital (No. JDPH -2022-016), following all guidelines, regulations, legal, and ethical standards as required for animals.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Summary
GDM is a condition of glucose intolerance and insulin resistance only diagnosed during pregnancy.
The visnagin also decreased the TCh, TG, and LDL, and elevated the HDL content.
The visganin treatment appreciably decreased the inflammatory markers, reduced oxidative stress, and improved the activities of antioxidant enzymes.
