Abstract
Background
An experimental design to obtain bioactive ingredients from Oroxylum indicum (L.) Kurtz was performed applying microwave-assisted extraction (MAE). Recently, tremendous research interest and benefits have been observed for extracting active constituents from plant materials using the microwave.
Objectives
This work aimed to develop an optimized, green, efficient, alternative procedure for extracting flavonoid and phenolic (total flavonoid content [TFC] and total phenolic content [TPC]) from leaves of Oroxylum indicum (L.) Kurtz.
Materials and Methods
A total of 1 g of the powdered sample was extracted using classical Soxhlet apparatus for 48 hr to ensure exhaustive extraction; simultaneously, maceration and ultrasound-assisted extraction were also carried out using 1 g of the plant sample for 12 hr and 60 min at 25˚C, respectively. The optimization of MAE was done using Taguchi L9 orthogonal design. The optimum operating conditions were microwave power (160 W), soaking time (4 min), irradiation time (4 min), and temperature (57˚C).
Results
The microwave method has shown 70% higher yield than other conventional techniques in a shorter time, along with increased TFC and TPC.
Conclusion
The TPC, TFC, and total antioxidant capacity (TAC) values are higher in the extract obtained by MAE extracts in comparison to other techniques. The extraction technique will be an ideal tool to safeguard the thermolabile plant constituents that will be more environmentally friendly.
Keywords
Introduction
In recent decades, the role of herbal medicines has become exceptionally successful in disease prevention and management. Different bioactive compounds are present in plants naturally, such as phenolic compounds, which have numerous health benefits for humans. However, we must consume a lot of plant materials to achieve the effective dose of these pharmacologically active compounds, as they are available in trace quantities naturally in plants which may be impossible practically. Hence, extraction of bioactive compounds from the plant material is the best way to achieve health-promoting properties. As extraction is one of the best methods, it has been practiced for centuries to concentrate the active pharmaceutical ingredients and add them to food products as functional ingredients.
The plant Oroxylum indicum belonging to the family Bignoniaceae is an evergreen or partly deciduous, small to medium-sized tree with light greyish brown, soft, and spongy bark. 1 It is found in many parts of the world, particularly in South Asia, Southeast Asia, and China. 2 This plant is reported to possess various activities such as gastroprotective, anticancer, anti-inflammatory, antioxidant, and immune stimulant. 3 Researchers have focused on more efficient extraction techniques for recovering phytochemicals from plant materials. Those techniques allow a selective extraction and isolation of phytochemicals with significant yield value, less solvent consumption, energy utilization, and ultimately less environmental impact. Nowadays, the potential genuine benefit seems to be overshadowed by the safety and quality-related problems, such as reliable extraction, lacking high performance, analytical methodologies, and techniques, used for establishing a standard therapeutic functionality related to herbal medicine. 4 The traditional extraction techniques, which have been in use for many years, take a long time and use a lot of solvents. Microwave extraction can boost yield while requiring less solvent and less time at the same temperature. 5 According to a survey, the surface layer of fruits and vegetables contains the majority of the flavonoid content. Due to enzymatic and thermal degradation, the flavonoid concentration is further reduced during additional extraction processing steps such as heat treatment, maceration, and juice extraction. 6 The limits of existing procedures for the extraction of these substances are continually being addressed by the development of new techniques and tactics like microwave-assisted extraction (MAE). 7
Particularly, in the last few decades, researchers have studied green extraction methods; the majority of these methods are quick and solvent-intensive but effective. The cutting-edge method of MAE produces high-quality natural plant extracts.
As per the previous literature, the extraction was conducted in usual methods such as maceration, percolation, and soxhlation, by taking seeds and barks, but the percentage of yield of the extract is very less. Some methods similar to Jagetia et al. had taken the stem barks for extraction using a Soxhlet apparatus. 8 It was also reported that the extraction by sonication with methanol, but the percentage of yield was not so encouraging. 9 The air-dried and powdered plant materials were weighed (15 g) and macerated exhaustively at room temperature with solvent (80% ethanol v/v, 2 × 150 mL). 10 Moreover, in all these cases, the percentage yield was a less and more time-consuming process.
This study was purposed to develop a working method with the help of MAE method to obtain pharmacologically active ingredients from O. indicum. For the same, the steps followed are (1) recovery of bioactive compounds (as TPC and TFC) under different MAE conditions, (2) finalizing the factors influencing MAE in the composition of extracts, and (3) obtaining high-quality extracts of specific phytoconstituents from O. Indicum leaves by adopting Taguchi L9 orthogonal design. The independent variables were matrix characteristics, extraction time, percentage of solvent (ethanol, methanol, or water), matrix characteristics, and temperature. The responses are obtained in terms of percentage of yield and phytochemical composition (phenolic compounds) determined as TPC and TFC values. MAE is compared with other conventional extraction techniques for the mentioned criteria.
Materials and Methods
Standards and Reagents
Quercetin, Folin-Ciocalteu reagent, ascorbic acid, and aluminium chloride were procured from HIMEDIA Co. Ltd. (India). Solvents used throughout the process of extraction and chromatography were procured from Fischer Scientific (India). Gallic acid and 2,2-diphenyl-1-picrylhydrazyl-hydrate (DPPH) were purchased from Siga-Aldrich Co. (St. Louis, MO, USA).
Plant Collection
O. indicum commonly known as “Sonpatta” was collected from the Birla Institute of Technology, Mesra Campus, Ranchi, Jharkhand, India. The voucher specimen number Bot/GGV/2022/15 has been assigned after depositing O. indicum specimen to the Department of Botany, Guru Ghasidas Viswavidyalaya, Chhatisgarh. The collected leaves were dried in the shade, and then the size reduction was conducted to obtain a uniform plant matrix through mesh size 60. The obtained powder was kept in air-tight pouches and protected from sunlight.
Apparatus
MAE was performed with Anton Paar Multiwave 3000 (USA) with a maximum capacity of 1400 W. About 1 g of leaf powder for MAE was weighed accurately and then extracted with 25 mL methanol. Around 4 min, an initial pre-leaching time was kept before the extraction process. The preliminary work extraction vessel was exposed to microwave irradiation under different experimental conditions and according to the Taguchi design extraction parameters optimization process. The microwave extractor was optimized, that is, irradiation–cooling–irradiation in alternation. The irradiation time and cooling time were kept to 1 min for the sample solution. Throughout the extraction process, condensation was carried out in ice-cold water. After completion of the extraction, filtration of the extract was done through No. 1 Whatman filter paper and allowed for centrifugation at 4000 rpm followed by evaporation of supernatant to obtain the dried residue. Then reconstitution of obtained residue was done in an appropriate solvent to perform further quantitative analysis.
Ultrasound-assisted extraction (UAE) was performed in an ultrasonic bath (Takashi, Japan) by taking 1 g of the powdered sample, which was placed into a beaker (250 mL), volume made up with extracting solvent (methanol) and sonicated for 60 min at 25˚C temperature with 15 min interval for cooling purpose. After the extraction, the contents are filtered, concentrated, and stored.
Conventional Extraction Techniques
Classical Soxhlet apparatus was chosen for the process; about 1 g of the powdered sample was extracted for 48 hr to ensure exhaustive extraction of drug samples. At the same time, maceration was also done by taking 1 g of the plant sample. After every 12 hr, the macerating solvent (methanol) was replaced to obtain the maximum constituents in the extract and then pooled together to obtain the final extract.
Scanning Electron Micrographs
Examination of the specimens was done with a JEOL JSM-6700F (Akishima, Tokyo, Japan) scanning electron microscope. The vacuum condition was kept too high at 5 kV of accelerating voltage. SEM analysis of the left-over marc was performed to elucidate the reason behind the extraction acceleration and the effects of microwave heating on the plant matrix.
Total Phenolic Content (TPC)
TPC was determined by the Folin-Ciocalteu method. 11 The standard drug of gallic acid was taken to plot a calibration curve by preparing the dilutions of 10, 20, 30, 40, and 50 µg/mL in methanol from the standard solution of gallic acid, that is, 0.01 g of gallic acid in 100 mL of methanol (100 µg/mL). About 100 µL of each of the dilutions was mixed with 500 µL of water and then with 100 µL of Folin-Ciocalteu reagent, which was allowed to stand for 6 min. Then, the addition of 500 µL of distilled water and 1 mL of 7% sodium carbonate was done to the reaction mixture. Through the spectro-photometrical technique, the absorbance of the reaction mixture was recorded at 760 nm after 90 min. This procedure was fixed as a standard for the extracts obtained from all the extraction techniques. By using the calibration curve (y = 0.015x +0.011, R 2 = 0.997), the TPC was calculated as milligram of gallic acid equivalents per gram (mgGAE/g) of the dry sample using the following equation
T = (C × V)/M
T = TPC in mg/g
C = concentration of gallic acid established from the curve in mg/mL
V = volume of the extract solution in mL
M = weight of the extract in g
Total Flavonoid Content (TFC)
The Aluminum chloride complex method was used to determine the TFC of the extracts. 11 Here quercetin was used as a standard drug for this study. TFC was determined as quercetin equivalent. Dilutions of 10, 20, 30, 40, and 50 µg/mL concentrations of quercetin were made to plot a calibration curve. Dilutions were prepared in methanol by using a standard solution of quercetin, that is, 0.01 g in 100 mL (100 µg/mL). About 3000 µL of methanol and then with 200 µL of 10% aluminium chloride and 200 µL of 1 M potassium acetate were spiked with 1000 µL of each of the quercetin dilution and mixed well and allowed to stand for 6 min. Then, 5.6 mL of distilled water was added and absorbance was read at 420 nm by a UV spectrophotometer. The same procedure was repeated with the different extracts. By using the calibration curve (y = 0.006x − 0.019, R 2 = 0.998), TFC was calculated as milligram of quercetin equivalents per gram of (mgQE/g) of dry weight samples through the equation:
C = (c1 × V)/N
C = concentration of quercetin in mg/g
c1 = concentration of quercetin established from calibration curve in mg/mL
V = volume of the extract in mL
N = weight of the plant extract in g
Total Antioxidant Capacity (TAC)
The widely used compound for the testing of antioxidant activity is DPPH. At 517 nm, DPPH shows maximum absorption and shows purple color. A free radical pair up with a hydrogen atom and transforms into DPPH-H (yellow color) when antioxidant extracts interact with DPPH, lowering the absorbance value.
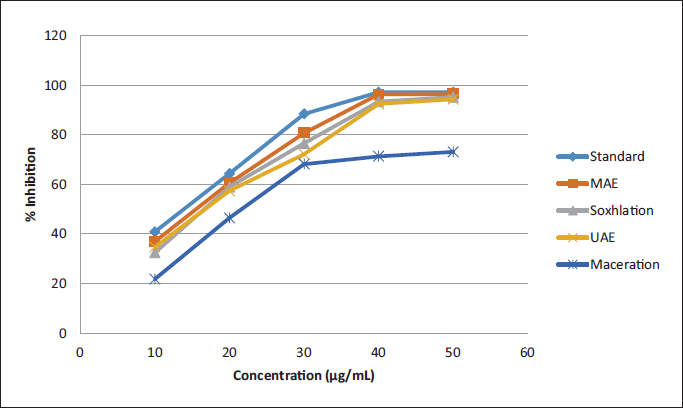
The antioxidant-free radical scavenging activity of various leaf extracts was carried out using a DPPH assay. The respective decrease in absorbance was gradually measured at 517 nm. With little modification, the procedure is portrayed by Kant et al. 12 The gradual decrement in absorbance was then observed through a UV-Vis spectrophotometer at 517 nm with respect to a blank (300 µL of ethanol in 2 mL of DPPH solution). Triplicate measurements were performed considering the statistics. The percentage inhibition of DPPH was calculated using the Eqn. I y = 1.546x + 27.78, where as is the absorbance of the test extracts and Ab is the absorbance of the control.
Taguchi Design
The Taguchi-based optimization technique is one of the best and was adopted to optimize the MAE of phytoconstituents like phenolics and flavonoids from O. indicum. One of the powerful optimization strategies called orthogonal array design was given by Taguchi, which explains how individual factors influence the final output and how they interact among themselves in a minimum number of experimental trials with less waste generation. 13
An experiment that may require 81 trials to evaluate four factors with three levels each can be achieved with nine trials only by applying the Taguchi L9 array. Hence, orthogonal design conditions were performed for the extraction process.
All the results are taken as the mean result of the sample in triplicate. When the individual parameters are optimum, the one with the highest value needs to be chosen as it is the best to maintain the maximum production efficiency. With the help of this concept, obtained final conditions for the experiment operation were found to be A1 (microwave power 160 W), B1 (irradiation time 4 min), C1 (57˚C), and D1 (soak time 4 min). 14
Factors Affecting the MAE Process
Solvent: The most crucial factor affecting MAE is the selection of solvent. It is done by solubilizing the compound of interest in it, penetrating of solvent, interacting with the sample matrix, dielectric constant, and mass transfer kinetics of the process. For phenolic compounds, methanol was considered a more efficient solvent to extract than ethanol. Ethanol provides low solvation property due to the presence of ethyl radicals. Structurally, ethyl radicals are longer than methyl radicals, resulting in lesser solubilization of phenolic molecules. 15
Duration of extraction: A longer extraction duration is essential to get more yield. However, it may increase the risk of degrading the essential components that are thermolabile. Hence, a proper method should be used in MAE to ensure the stability of essential nutrients and the maximum component yield.
Microwave power: Low or moderate microwave power is generally desirable with a longer extraction time to optimize an MAE protocol.
Matrix characteristics: The matrix characteristics, such as the nature of the material and particle size, affect the recovery of the compounds. For better penetration of microwaves generally, the finer particle size of the sample is larger in the surface area.
Statistical Data Analysis
All experiments (TFC, TPC & TAC) were carried out taking triplicate of each sample. The results were expressed as means ± standard deviation (n = 3), and the p values <.05 were considered significant. All the statistical data analysis was performed through Graph Pad Prism version 9.1.
Results and Discussion
Comparison of MAE with Other Conventional Techniques
To understand the true efficacy of MAE in the present study, the percentage of yield value (%w/w) of methanolic extract (1 g) of the plant by different extraction techniques such as maceration, soxhlation, UAE, and MAE was found to be 0.10 ± 0.008, 0.12 ± 0.009, 0.14 ± 0.03, and 0.17 ± 0.01, respectively. The moisture content of the collected material is 7.58% w/w, determined by loss on drying which is very important in MAE. The optimum operating conditions in MAE (Table 1) were considered for analysis of plant material as per Taguchi design.
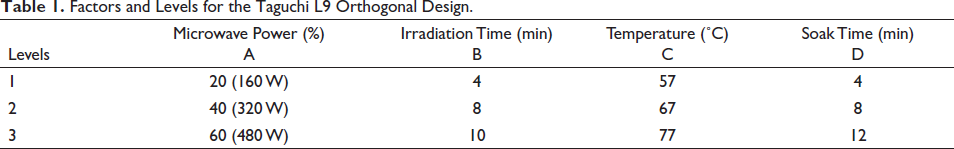
Factors and Levels for the Taguchi L9 Orthogonal Design.
Extraction Mechanism by SEM
SEM analysis of the remaining marc was performed for confirming the effect of microwave heating on the plant material and the reason for enhanced extraction. When the surface morphology was studied at 2000 magnification level same impression was observed for the marc of macerated, Soxhlet, and UAE extracted samples. On the contrary, significant disruption of surface morphology was seen in the marc of the microwave-extracted sample. To establish the actual effect of microwave heating, the SEM micrographs were studied at 4000 magnification which depicts the cellular channels through which the analyte leached out from the plant matrix.
Due to extreme internal thermal stress (by microwave heating), total breakdown of the plant cell wall occurred, which cause dehydration of cellulose, which lead to loss of integrity of the cell wall. It can be detected by the SEM image (Figure 1). The widening of the cellular channels formed an easy and straight exit path for the plant constituents (analyte) to come out into the extraction solvent from the inner matrix of the sample. 16

TPC, TFC, and TAC Values by Different Extraction Techniques
The yield of total phenolics and flavonoids obtained by MAE was 45.67 ± 0.13 mg GAE/g and 76.82 ± 0.18 mg QE/g which is higher than the conventional Soxhlet extraction yield of 48 hr process. The yield of TPC and TFC by MAE was 144% and 114% as compared to Soxhlet extraction.
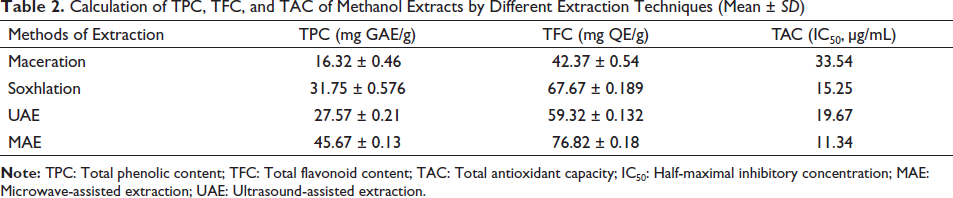
The TPC was found to be of encouraging amount in the case of MAE extracts in comparison to other methods. The TPC, TFC, and TAC of methanol extracts by different extraction techniques were determined and MAE was found to be having higher values with respect to other techniques (Table 2).
Calculation of TPC, TFC, and TAC of Methanol Extracts by Different Extraction Techniques (Mean ± SD)
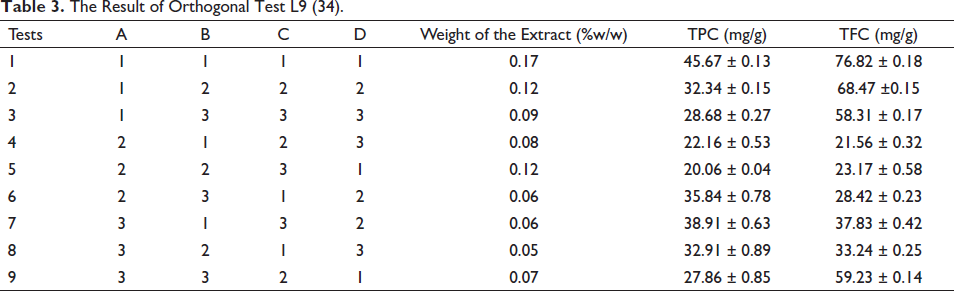
The TAC of various leaf extracts was carried out using a DPPH assay in which methanol extract was found to be having highest activity among all (Figure 2). The extraction method is considered to be the most critical parameter for the standardization of medicinal plants as well as for the true assessment of their nutraceutical and biological potential, which can be observed from the data. In terms of the yield of analytes, MAE has an excellent choice in comparison to conventional methods. It can be easily understood that maximum (exhaustive) production of bioactive from plant matrix (Table 3) is possible only when MAE is sued, it consumes much lesser time. The results of MAE were found to be significantly more than maceration and soxhlation (ANOVA followed by student’s t-test, p <.001, FExp 38.23; FCrit 2.26).
The Result of Orthogonal Test L9 (34).
% Radical Scavenging Activity of Different Extracts.
Taguchi Analysis
Taguchi analysis was used for the calculation of TFC by considering the various interactions such as TFC vs microwave power, TFC vs time, TFC vs temperature, and TFC vs soak time was determined. The optimal operating microwave extraction condition is; 160 W microwave power, 57˚C temperature, 4 min irradiation time, and pre-leaching time of 4 min.
Environmental Impact
Soxhlet extraction is a conventional extraction technique involving long heating hours; hence, it burns excess energy. We should make ways for environment-friendly, greener technology which uses fewer energy resources and protects the environment. In MAE, there is very little solvent consumption, and the extraction time is less, so it has a less negative impact on the environment by releasing CO2 and heat. 14
Conclusion
In this study, the percentage of yield of extract from MAE was found to be 141% concerning Soxhlation. It was also found that the extracts obtained by MAE process had TPC, TFC, and TAC values of 144%, 114%, and 134%, respectively, compared to extracts of the Soxhlation process. At the same testing concentration when compared with UAE, maceration, and Soxhlet, MAE extracts resulted in significantly more biological activity due to exhaustive extraction of bioactive in a thermally safe environment.
Ethnopharmacology research is currently considered to be the backbone for natural products and drug discovery research. Good extraction of phytoconstituents is the key to the success of good ethnopharmacological research. So greener technologies like MAE become a must in this regard which leads to a higher yield of the phytoconstituents and is also environmental friendly.
Footnotes
Abbreviations
MAE: Microwave-assisted extraction; TFC: Total flavonoid content; TPC: Total phenolic content; TAC: Total antioxidant capacity; UAE: Ultrasound-assisted extraction; ˚C: Degree Celsius; Min: Minute; W: Watt; g: Gram; mL: Milli liter; rpm: Round per minute; SEM: Scanning electron microscope; µL: Microliter: mg; GAE/g: Gallic acid equivalents, mg; QE/g: Quercetin equivalents per gram; nm: nanometer; DPPH: 2, 2-Diphenyl-1- picrylhydrazyl-hydrate; P: probability.
Acknowledgment
The authors are very much thankful to the Department of Pharmaceutical Sciences and Technology, Birla Institute of Technology, Mesra, Ranchi, for providing the necessary resources to complete this research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
