Abstract
Objectives
Methods
The total phenolic and total flavonoid contents were measured by colorimetric assays, while the widely targeted metabolomics was applied to reveal leaf and root
Results
The results showed the significant content of phenolics and flavonoids in plants collected from mining soils of Thai Nguyen province, Vietnam, and their potential ABTS scavenging activity. Additionally, the plant's metabolome characterized 82 compounds, mainly secondary metabolites. The multivariate analysis indicated the positive correlation among quercetin derivatives and bioactivity, suggesting the contribution of their combination, not individually, to the antibacterial effect of
Conclusion
Hyperaccumulators in metalliferous soils are potential reservoirs for exploring the antibacterial agents against multi-drug resistant strains, and this effect may be exerted by additive or synergistic mechanisms of action of multiple compounds.
Keywords
Introduction
Heavy metals are toxic to plants in many ways, depending on their different states. At the cellular and molecular levels, these agents can impact various plant physiological processes, for example, disrupting the normal function of macromolecules such as enzymes and proteins, making chelates with functional groups of metabolically critical small molecules, and modifying the conformational of membrane integrity. 1 As a consequence, plant metabolism could change, leading to variation in the metabolite profile of plant samples.2–5 In addition, heavy metals can disrupt the redox-oxidative balance of plant cells by inducing the formation of free radicals and reactive oxygen species (ROS). 6 However, several plants could survive in the metalloid areas and still grow and mature normally. There are two main ways to resist heavy metal stress: avoidance or tolerance. The former modality induces the restriction of metal uptake, and the latter activates many procedures that allow plants to adapt in the presence of heavy metal inside. Indeed, plants can produce a range of metabolites or binding ligands that are intracellularly chelated with heavy metals, upregulating the antioxidant defense against the negative impact of ROS. 7 Phenolics, known as the most important secondary metabolites, play an essential role in the oxidative defense system of the plant kingdom. 8 Increased phenylpropanoid metabolism and phenolics level can be observed in some species under heavy metal contamination. 9 This group, especially flavonoids, can be considered the chemical barrier for oxidative stress caused by elements.
Besides the effect on plants, heavy metals also put critical pressure on bacterial communities in metalliferous soils.
5
In long-term exposure to these agents, bacterial strains become more and more resistant to available biocides. This phenomenon was explained partly by horizontal gene transfer of heavy metal resistance genes and antibiotic resistance genes induced by high levels of metals in soils.
10
Also, efflux pumps are found to have a higher frequency in pollution ecosystems because of their effectiveness in the detoxification of exogenous elements.11–17 Indeed, this is an effective mechanism for microorganisms to extrude various heavy metals and compounds with diverse structures, not limited to antibiotics used in clinical activities, thus representing multidrug-resistant (MDR) phenotypes. A wide range of bacterial species was reported to express efflux systems, and exposure to multiple heavy metals induced their over-expression more effectively.
18
Interestingly, plant metabolites may be responsible for the expression or the dispersion of MDR phenotypes, thus, possibly involved in bacterial growth. Indeed, i) plant antimicrobial compounds, eg, berberine, are sometimes substrates of MDR pumps, 19 and many pieces of evidences indicate the structural similarity between plant compounds and substrates of bacterial MDR pumps (eg, polyphenols and polycyclic aromatic hydrocarbons)25–27; ii) plant produce compounds able to induce the expression of MDR pumps in rhizosphere bacteria 28 ; and iii) plant produce compounds able to inhibit bacterial MDR pumps (Efflux Pump Inhibitors- EPIs), allowing to reduce the MIC of antimicrobial compounds that are the substrate of these pumps.29–31 For all the above-mentioned reasons, plants growing in metalliferous soil have been considered a potential source for screening the antioxidant and antibacterial effects because of the deep interaction with heavy metals and MDR phenotypes in the ecosystem.
This research focuses on the most dominant fern,
Material and Methods
Chemicals
For TPC, gallic acid, Na2CO3 solution, Folin–Ciocalteu reagent were purchased from Merck (Darmstadt, Germany). To determine TFC, NaNO2, AlCl3, NaOH, and ethanol were obtained from Aladdin (Shanghai, China); quercetin was purchased from Vietnam National Institute of Drug Quality Control (Hanoi, Vietnam). In the ABTS assay, Trolox, methanol, and K2S2O8 originate from Merck (Darmstadt, Germany). Distilled and ultrapure water were prepared using a Millipore Milli-Q purification system (Millipore GmbH, Schwalbach, Germany).
Plant Materials
35 plant individuals of
Bacterial Materials
The tested bacteria were six different
Methods
Sample Preparation
For plant treatment, the whole plant was divided into aerial and belowground parts, washed to remove contamination, and then rinsed in distilled water. All tissues were cut down into small parts and dried in the oven at 55 °C for 48 h until their weight did not change. Dry tissues were then ground into powder. Subsequently, 100 mg of each sample were extracted with 80% methanol at room temperature in a sonic bath for 30 min. 38 The extracts were concentrated in the vacuum concentrator to obtain dry extracts. These extracts were subjected to all chemical tests in further experiments.
Total Phenolic Content
The TPC was measured using the Folin-Ciocalteu assay with gallic acid as a standard. 39 The extracts were weighed and dissolved in methanol at different concentrations. Subsequently, 40 μL of the diluted sample was mixed with 480 μL of Folin-Ciocalteu reagent and 480 μL 6% Na2CO3. The mixture was then incubated at 40 °C for 15 min and measured the optical absorption at the wavelength λ = 765 nm. For the standard curve, a series of gallic acid (C7H6O5) solutions from 100 to 1000 μg/mL was prepared, and the same reaction with the tested sample was followed. The TPC was measured as mg gallic acid equivalent per g extract (mg GAE/g).
Total Flavonoid Content
For the TFC measurement, a colorimetric assay followed the procedure of Nurcholis et al 40 The samples were similarly weighed and dissolved in methanol at different concentrations. A mixture including 240 μL of the initial solution and 40 μL 5% NaNO2 was incubated at 25 °C for 6 min. Then, 40 μL 10% AlCl3 was added to the mixture and put at the same temperature and duration as the previous step. Finally, 400 μL NaOH 1 M and 280 μL 30% ethanol were mixed with, incubating for 15 min at 25 °C. The solution was measured at the wavelength λ = 510 nm. A calibration curve was built by a series of standard solutions of quercetin (C15H10O7) from 0 to 200 μg/mL. The TFC was calculated as mg quercetin equivalent per g extract (mg QE/g).
Metabolite Profiling
In our previous investigation, UPLC-QqQ-MS was used for the rapid dereplication of a wide range of phytochemicals in leaf and root extracts of
2,2-Azinobis 3-Ethylbenzothiazoline-6-Sulfonic Acid (ABTS) Scavenging Assay
ABTS assay is commonly used to determine the antioxidant capability of samples. 41 The crude extracts were dissolved in methanol at the stock concentration of 10 mg/mL and then diluted in 0.25 and 0.5 mg/mL as two tested concentrations. To prepare the standard curve, the stock of 5 mg/mL Trolox in methanol was used to make a series of 1/64, 1/96, 1/128, 1/192, and 1/256 as dilution factors. The absorption of these solutions is measured in 96-well plates at 734 nm by a microplate reader. The ABTS scavenging activity was calculated according to the formula: % scavenging capacity = (Ac-As)/Ac×100, where Ac and As are the absorbance of the standard and the tested sample, respectively.
Antibacterial Activity
The inhibition potentials of
Statistical and Multivariate Analysis
For statistical analysis, t-tests and one-way ANOVA were applied to determine whether the difference between the two variables is significant using Graphpad Prism 9.2.0. The univariate analysis, ie Pearson correlation and the multivariate analysis was done by using RStudio version 3.5.3. (http://www.R-project.org/). Peak intensity was normalized by taking their logarithm base 10. The correlation between the whole metabolome and bioactivities was calculated by the
Results and Discussion
The results were presented in three main parts. The metabolite profiles reported the TPC, TFC and metabolomes of 35 leaf and 35 root methanolic extracts. In parallel, the bioassay part demonstrates the antioxidant and antibacterial characteristics of these 70 extracts. Finally, univariate and multivariate analysis integrated chemical and biological profiles to reveal the relationship between metabolites and effects.
Total Phenolic Content and Total Flavonoid Content
Figure 1 shows the distribution of TFC and TPC in the methanolic extract of 35 aerials and 35 belowgrounds. It was indicated that TPC varied a lot in both tissues, ranging from 5.78 to 29.53 and from 7.37 to 33.17 mg GAE/g dry weight for those leaf and root extracts, respectively. All the samples showed remarkable TPC, suggesting that phenolics represented as major components in the extracts. The TFC of

TPC (mg GAE/g dry extract) and TFC (mg QE/g dry extract) of 70 samples: 35 aerial parts (from A1 to A35, in orange color) and 35 belowground parts (from S1 to S35, in blue color).
Metabolome Profiling
By the widely targeted metabolomics analysis, 70 extracts were profiled and a total of 82 metabolites were selected according to the criteria mentioned in the method section (Table 1). It is indicated that amino acids and carboxylic acids dominate the metabolome of both aerial and belowground parts of
Metabolite Profiling of P. vittata Methanolic Extracts by Widely Targeted Metabolomics.

Each metabolite has its code (ID), annotation, precursor and product, retention time and the group that it belongs to.
Naturally, flavonoids have a significant role in plant defense and adaptation, ie, regulating the development, pigmentation, flavoring, and UV protection. 45 These functions are supposed to be emphasized in metalliferous soils. 46 As an overdose of metals can generate ROS; plants produce more phenolics and their subgroup, flavonoids, the most important components in antioxidative systems. Additionally, these compounds possess a wide range of health benefits such as anticancer, antimicrobial, antiviral, and neuroprotective activity thanks to their diverse structures.47–50
Quantitatively, the intensity of selected peaks could be valuable data. The table gathers the peaks’ intensity normalized by internal standards, which can be seen in the Supporting Information, Table S1. Indeed, the metabolome is the most time-sensitive parameter in system biology because metabolites’ levels can change rapidly during the life span of an organism. Even though plant individuals grow in the same region, their metabolome varies, leading to their differences in bioactivities.51,52 Therefore, we recorded the variation in the intensity of selected peaks among 35 leaf and 35 root extracts to correlate them to the bioactivities data.
The Capability to Scavenge the ABTS Radical
The scavenging capability of ABTS radical of 70 extracts of

The inhibition percentage (%) of ABTS scavenging activity of 70 samples: 35 aerial parts (from A1 to A35, in orange color) and 35 belowground parts (from S1 to S35, in blue color).
Antibacterial Activity
To identify the most sensitive isolates of

Inhibition percentage of six mixed extracts (pool 1,2,3 - aerial parts, pool 4,5,6 - belowground parts) on different strains of S.
For further experiments, we examined the activity of 70 crude extracts on these isolates to find out the relationship between the whole metabolome profile and antimicrobial activity. Supporting Information Table S2 illustrates the inhibition percentage of 70 samples against 0366 isolates, as an input for multivariate analysis.
Correlation among Chemical Profile and Bioactivities of P. vittata Extracts
Univariate analysis does not show any significant correlation between TPC, TFC, and the scavenging activity ABTS and inhibition percentage against
Multivariate analysis, ie, partial least square regression (PLSR), were then conducted to reveal correlations between the whole profile and antioxidant/antimicrobial effects. This model was generally illustrated by a circle plot (Figure 4). These metabolites are located inside a 2-components space spanned by a circle of radius 1 (correlation coefficient 1) and a radius 0.5 (correlation coefficient 0.5). The relationship between the two variables is measured by the angle created by the endpoint of the variable vectors. The positive correlation is found if the angle is sharp, negative in the case of an obtuse angle, and null (having no meaning) if the angle is right, 90°. Therefore, following the cross-arrows quadrant separation, metabolites that fall into quadrant II can be predicted as having a positive correlation with antibacterial activity (Y). In contrast, quadrant IV predicts a negative relationship with Y, while quadrants I and III present no correlation at all.
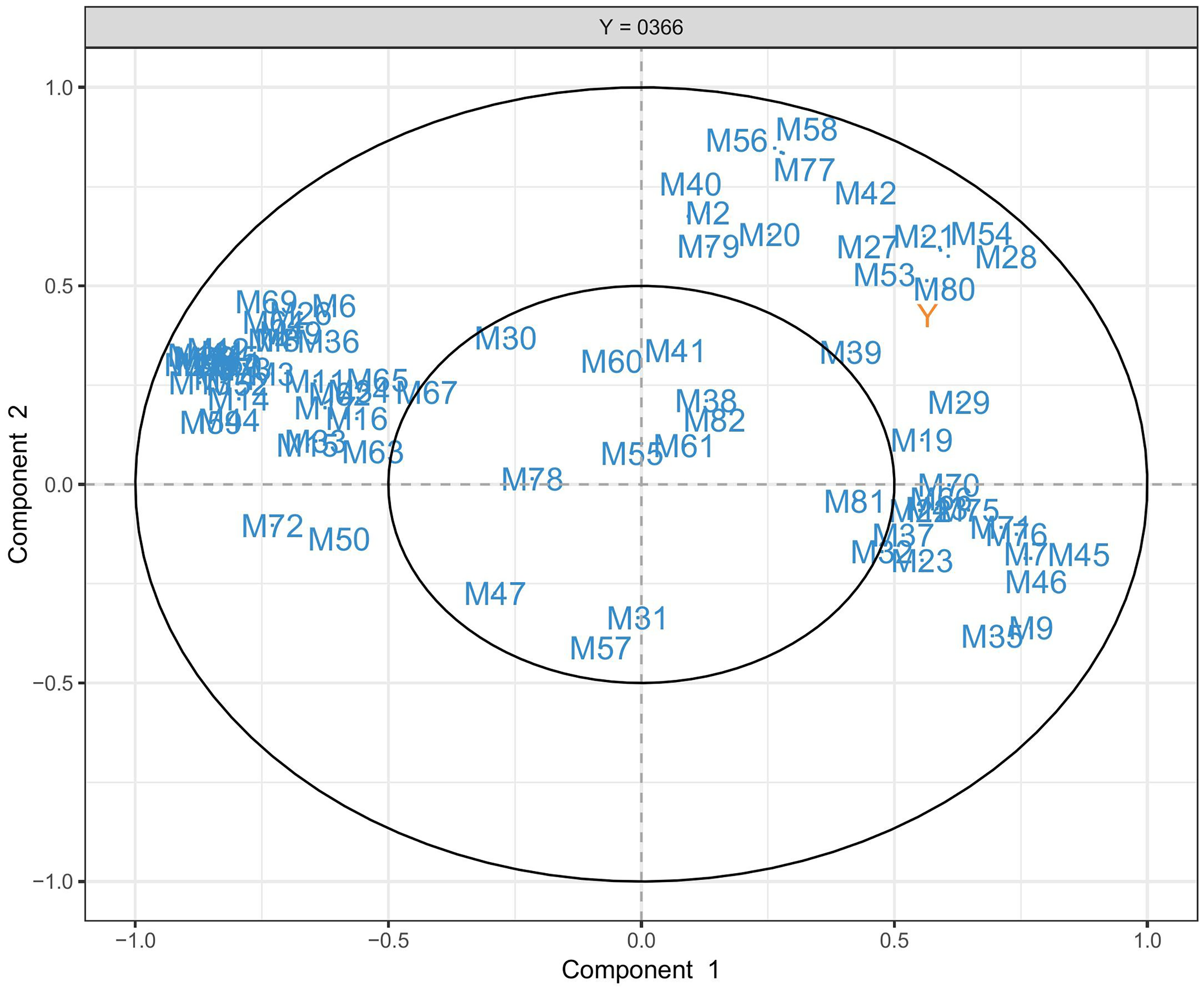
PLSR-based loading plot illustrates the correlation between the whole profile and the antimicrobial effect of leaf extracts.
Figure 4 illustrates the circle plot of the PLSR model between 82 metabolites and the inhibition percentage of 35 leaf extracts of
In addition, the cluster of flavonoids is located near vector Y, highlighting that these compounds correlated significantly to bioactivity and are thus responsible for the effect. Note that no single correlation was found through univariate analysis, while the multivariate can reveal strong coefficients, reaching around 0.9 (nearly the cycle of radius 1) among flavonoids and bioactivity. Therefore, these compounds may work additionally or synergistically to contribute to the inhibition percentage of plant extracts against
In this study, the prediction power for the inhibition percentage of the metabolome profile has provided a good example of how to identify bioactive compounds in combination. At the same time, conventional strategies, such as bio-guided isolation, cannot detect this mechanism. However, our study still has limitations. The extracts obtained are crude extracts that provide moderate effects on a single bacterial strain. While our findings provide initial insights on the anti-
Conclusion
In a nutshell, this study demonstrates for the first time the potential of
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251322868 - Supplemental material for Antioxidant and Antibacterial Activities of Pteris vittata L. Extracts from Metalliferous Soils: Correlations with Phenolic Compounds
Supplemental material, sj-docx-1-npx-10.1177_1934578X251322868 for Antioxidant and Antibacterial Activities of
Supplemental Material
sj-xlsx-2-npx-10.1177_1934578X251322868 - Supplemental material for Antioxidant and Antibacterial Activities of Pteris vittata L. Extracts from Metalliferous Soils: Correlations with Phenolic Compounds
Supplemental material, sj-xlsx-2-npx-10.1177_1934578X251322868 for Antioxidant and Antibacterial Activities of
Supplemental Material
sj-xlsx-3-npx-10.1177_1934578X251322868 - Supplemental material for Antioxidant and Antibacterial Activities of Pteris vittata L. Extracts from Metalliferous Soils: Correlations with Phenolic Compounds
Supplemental material, sj-xlsx-3-npx-10.1177_1934578X251322868 for Antioxidant and Antibacterial Activities of
Supplemental Material
sj-xlsx-4-npx-10.1177_1934578X251322868 - Supplemental material for Antioxidant and Antibacterial Activities of Pteris vittata L. Extracts from Metalliferous Soils: Correlations with Phenolic Compounds
Supplemental material, sj-xlsx-4-npx-10.1177_1934578X251322868 for Antioxidant and Antibacterial Activities of
Footnotes
Acknowledgements
The authors would like to thank the UMR 5557 Ecologie Microbienne (Eq Bacterial Efflux & Environmental Resistance), Lyon, France and the International Joint Lab on “Drug Resistance in Southeast Asia” (LMI-DRISA) to donate & store the bacterial strains for this research.
Author Contributions
NLN and PNN are co-first authors of this article. TKON and PNN conceived of the presented idea. TKON conducted the widely targeted analysis. NLN, HNP, and PKN carried out the experiments related to TPC, TFC, ABTS and antibacterial activities. PNN and TKON conducted the multivariate analysis. All authors discussed the results and contributed to the final manuscript.
Data Availability
All data was included in the article or supporting information.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Science and Technology of Hanoi [grant number USTH.LS.01/22-24].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
