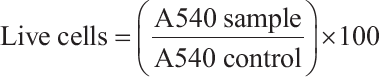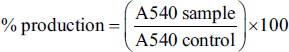Abstract
Background
The oxidation of low-density lipoprotein (LDL) by reactive oxygen species (ROS) causes inflammation, which results in the expression of adhesion molecules and monocyte adhesion and migration. This eventually leads to the progression of atherosclerosis. Erythroxylum cuneatum (EC), locally known as “Chinta mula,” is used as traditional medicine in certain countries. However, the scientific evidence of its medicinal properties, particularly related to cardiovascular disease (CVD) is still limited.
Objectives
This study was designed to evaluate the antioxidant and anti-inflammatory properties of EC leaf extract in protecting against atherosclerosis in vitro.
Materials and Methods
Human aortic endothelial cells (HAoECs) induced by oxidized LDL (oxLDL) were treated with comparable concentrations (40 and 80 µg/ml) of EC ethanol and acetone leaf extracts. The antioxidant activities were determined by thiobarbituric acid reactive substances (TBARS), ROS, and nitric oxide (NO) production assays. The anti-inflammatory effects of EC leaf extract were evaluated using monocyte adhesion and migration assays and the expression of adhesion molecules, namely, intracellular adhesion molecule-1 (ICAM-1) and human vascular cell adhesion molecule-1 (VCAM-1).
Results
Both EC extracts possess antioxidant and anti-inflammatory activities against oxLDL-induced HAoECs, which were concentration-dependent. Acetone extract showed significant reduced TBARS levels and increased NO production compared to ethanol extract. It also caused a significant decrease in monocyte adhesion and expression of ICAM-1 as compared to ethanol extract.
Conclusion
These findings suggest that EC leaf extract is valuable in preventing atherogenesis. The superior effect of EC acetone extract warrants future studies to elucidate its mechanisms in the prevention of CVDs, particularly atherosclerosis.
Introduction
For numerous years, Cardiovascular diseases (CVDs) have remained the leading cause of death globally. According to the World Health Organization (WHO), CVDs account for around 32% of all deaths worldwide, or 17.9 million people. 1 About 85% of the fatalities were due to heart attack and stroke, which were triggered by blood vessel blockage.
Atherosclerosis, the major cause of CVDs, occurs when there is a hardening and narrowing of arteries that ultimately reduces the blood flow through the body. 2 It is an inflammatory condition that is caused by oxidative stress. The initial steps involve endothelial dysfunction, oxidative damage, inflammation, and the interaction between platelets and endothelium. 3 The increase of lipid and low-density lipoprotein (LDL) cholesterol in the intima as well as endothelial activation are hallmarks of the inflammatory process. An increase in reactive oxygen species (ROS) causes excess LDL to be oxidized, resulting in oxidized LDL (oxLDL). 4 Monocytes differentiate into macrophages that take up the oxLDL, causing foam cell formation, which eventually leads to plaque progression. This eventually triggers the atherosclerotic wall thickening and proliferation of smooth muscle cells, leading to vessel narrowing.5, 6
Despite the development of atherosclerosis therapy, its mortality rate remains high. 7 There are also synthetic drugs accessible as a cure for oxidative damage and inflammation, yet they are hindered due to the side effects. Synthetic antioxidants, namely, tertiary butyl hydroquinone, butylated hydroxyanisole, and butylated hydroxytoluene, might be toxic and eventually carcinogenic. 8 Moreover, nonsteroidal anti-inflammatory drugs such as celecoxib and aspirin are widely used and are well-known for their actions prompting hospitalization. 9 Therefore, a large number of the world’s population have been using alternative medicines for centuries and their use is widely integrated into the public health scheme in some countries. 10 Several medications based on natural products were proven to have a progressive effect in treating oxidative damage and inflammation. 11
Anti-inflammatory and antioxidant properties have been discovered in Erythroxylum cuneatum (EC), popularly known as “Chinta mula.” 12 It is utilized in Thai traditional medicine to treat fevers and as an anti-inflammatory agent. 13 Phytochemicals such as alkaloids have been isolated from EC leaves more than 20 years ago. 14 Our previous study has also identified several phytochemicals and antioxidant properties of EC in different types of crude extracts. 15 These findings showed the potential of EC to be a complementary medicine in the treatment of various diseases including CVDs. However, its effectiveness and the associated mechanisms are still unexplored. Hence, this study was conducted to explore the prevention of EC leaf extract against oxidative damage and inflammation in aortic endothelial cells.
Materials and Methods
Preparation of Plant Material and Extraction
EC leaves were gathered from Bukit Broga in Selangor, Malaysia, and a voucher specimen (SK 2100/12) detail was obtained from the Institute of Bioscience, University Putra Malaysia. The leaves were air-dried before being pulverized into a fine powder. The powdered material was then extracted with solvents of different polarities, that is, ethanol and acetone, by Soxhlet extraction for 8 h per day for 6 to 7 days at the solvent’s boiling point until they turned colorless. Then, the solvents were evaporated to yield a concentrated crude extract. The crude extracts were dried in a fume hood to obtain a concentrated extract and stored at 4°C until further use. The percentage of extract yield for ethanol and acetone extracts was 17.60% and 18.55%, respectively.
Probucol was utilized as a positive control since it has antioxidant and anti-inflammatory effects.16, 17 10 µM probucol was prepared by dissolving it in ethanol to be used in the study.
Cell Culture
The primary cell, the human aortic endothelial cell (HAoEC) (PromoCell, Heidelberg, Germany) was cultured according to the manufacturer’s instruction in T-25 flasks using endothelial cell growth medium MV (PromoCell, Heidelberg, Germany) under humidified atmosphere (5% CO2) at 37°C. The cells were sub-cultured every 4 days once and used in passages 4 to 6. The cells were cultured as monolayers and harvested from the primary culture after 6 to 8 days. A human monocytic cell line U937 (ATCC, Virginia, USA) was cultured according to the manufacturer’s instruction in T-25 flasks using RPMI-1640 medium (ATCC, Virginia, USA) supplemented with 1% penicillin-streptomycin (Biowest, Nuaillé, France) and 10% fetal bovine serum (Biowest, Nuaillé, France) under humidified atmosphere (5% CO2) at 37°C.
Cell Viability Test
The cell viability test was conducted using an 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay.
18
The HAoECs were seeded into a 96-well plate and cultured for 24 h at 37°C with various concentrations of EC ethanol and acetone leaf extracts ranging from 6.25 to 100 µg/ml. Each well was filled with MTT solution. The medium was withdrawn after 4 h of incubation and replaced with 100 µL of 100% of dimethyl sulfoxide (DMSO) (Amresco Chemical Co., Ohio, USA). The plate was shaken gently to dissolve formazan crystals. Using a microplate reader, the absorbance of each well was measured at 540 nm. The percentage of live cells was then calculated using this formula:
Antioxidant Activity
Thiobarbituric Acid Reactive Substance (TBARS) Assay
The assay was carried out by seeding cells onto 96-well plates and pre-treating them for 24 h with 40 and 80 g/ml EC ethanol and acetone leaf extracts, respectively, before adding 30 g/mL oxLDL for 1 h. Cell culture medium was collected and heated with TCA-TBA-HCl reagent for 15 min. After incubation, the sample was cooled down and centrifuged at 1,000 g for 10 min. The absorbance was recorded at 535 nm against blank using a microplate reader (VERSAmax tunable, California, USA). Using an extinction coefficient of 1.56 × 105 M−1 cm−l, the amount of TBARS was determined. 19
ROS Assay
The effect of EC leaf extract on ROS was determined based on the modified method reported by Yang et al. 20 The cells were seeded onto 96-well plates and pre-treated for 24 h with 40 and 80 µg/ml EC ethanol and acetone leaf extracts, respectively, followed by 1 h with 30 µg/mL oxLDL. The intracellular amount of ROS was examined by staining the HAoEC for 30 min at 37°C with DCFH-DA solubilized in DMSO. At 485 nm excitation and 530 nm emissions, the fluorescence intensity was measured using a fluorescence microplate reader (Tecan M200 Infinite, Mannedorf, Switzerland). The results were calculated as a percentage of ROS inhibition.
Nitric Oxide (NO) Production Assay
The NO production assay was determined by Griess reaction.
21
The HAoECs were seeded onto 96-well plates and treated for 24 h with 40 and 80 µg/ml EC ethanol and acetone leaf extracts, respectively, followed by 1 h with 30 µg/mL oxLDL. A microplate reader (VERSAmax tunable, California, USA) was used to measure the absorbance at 540 nm after an equal amount of Griess reagent was added to the cell culture medium. The concentration of nitrite was determined using sodium nitrite as a reference. The following formula was used to compute the percentage of NO production;
Anti-inflammatory Activity
Monocyte Adhesion Assay
This assay was conducted based on the revised method reported by Babu et al. 22 The HAoECs were seeded in 48-well plates and allowed to develop until they reached monolayer confluence. They were subsequently pre-treated for 1 h with 40 and 80 g/ml EC ethanol and acetone leaf extracts, respectively, before receiving 30 g/mL oxLDL. The cells were rinsed in serum-free media before being inoculated with calcein-AM labeled U937 cells (Calbiochem, San Diego, USA). The HAoEC monolayer was rinsed with endothelial medium after 1 h of incubation. The monocyte adhesion was measured using a fluorescence microplate reader (Tecan M200 Infinite, Mannedorf, Switzerland) with 496 nm excitation and 520 nm emission. Results are illustrated as the fold difference, that is, the number of monocytes which adhered to the endothelial cells over the number of monocytes which attached to the endothelial cells without any EC extract (0 µg/ml).
Monocyte Migration Assay
The assay was performed based on the modified method by Ramirez et al. 23 One percentage gelatin (Nacalai Tesque, Kyoto, Japan) was used to coat 3 µM cell culture inserts. The HAoECs were seeded onto the gelatin-coated inserts at a density of 2 × 104 cells per insert until confluent. They were pre-treated with 40 and 80 µg/ml EC ethanol and acetone leaf extracts, respectively, followed by the addition of 30 µg/mL oxLDL for 1 h. The upper chamber was then filled with labeled U937 monocytes (Calbiochem, San Diego, USA), which were then incubated at 37°C for 2 h to facilitate monocyte transmigration to the lower chamber. The lower chamber’s cell culture media was collected and transferred to black opaque 96-well plates for fluorescence intensity measurement using a fluorescence microplate reader (Tecan M200 Infinite, Mannedorf, Switzerland) set to 492 nm excitation and 571 nm emission. Results are illustrated as the fold difference, that is, the number of migrated monocytes in the endothelial cells over the number of migrated monocytes in the endothelial cells without any EC extract (0 µg/ml).
Expression of ICAM-1 and VCAM-1
The determination of intracellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) level on HAoECs pre-treated with 40 and 80 µg/ml EC ethanol and acetone leaf extracts, respectively, and 30 µg/mL oxLDL was conducted using enzyme-linked immunosorbent assay kits in accordance with the manufacturer’s instructions (R&D Systems, Minnesota, USA).
Statistical Analysis
All data are presented as mean ± standard deviation. The distribution of the data was determined by using Kolmogorov–Smirnorv test. All data were normally distributed and analyzed by using analysis of variance, followed by post hoc Tukey’s test in Statistical Package for the Social Sciences software version 22.0. The value of p < 0.05 was considered statistically significant.
Results and Discussion
Cell Viability Test
The percentage of viability for each concentration was determined by plotting a graph against the concentration of EC leaf extracts (Figure 1). A dose-dependent decrease in the viability of HAoEC in both EC acetone and ethanol leaf extracts was observed. It was also found that the inhibition of cell viability by acetone extract was more potent than the ethanol extract in each concentration.
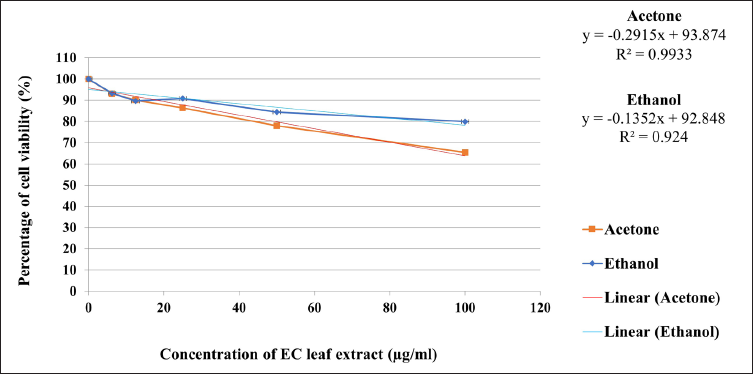
We believe this is the first study to reveal the viability of HAoECs-induced with EC leaf extract. Previous studies have shown that EC ethanolic leaf extract caused 50% toxicity in HepG2 (human liver carcinoma) cells and Vero (monkey kidney epithelium) cells at 64 µg/mL and 366 µg/mL, respectively. 24 An in vivo study revealed that EC aqueous and alkaloid leaf extract were toxic at 316.23 mg/kg and 416.86 mg/kg, respectively. 25 When comparing ethanol and acetone extracts, the former extract was shown to be less cytotoxic. In fact, at the highest concentration, that is, 100 µg/ml of EC ethanol extract, about 80% of the cells were still viable. Based on the MTT assay, the concentrations of 40 and 80 µg/mL of both extracts were selected for antioxidant and anti-inflammatory tests. oxLDL was used to trigger oxidative stress in the endothelial cells. Control group is the HAoEC without stimulation of oxLDL.
Antioxidant Activity
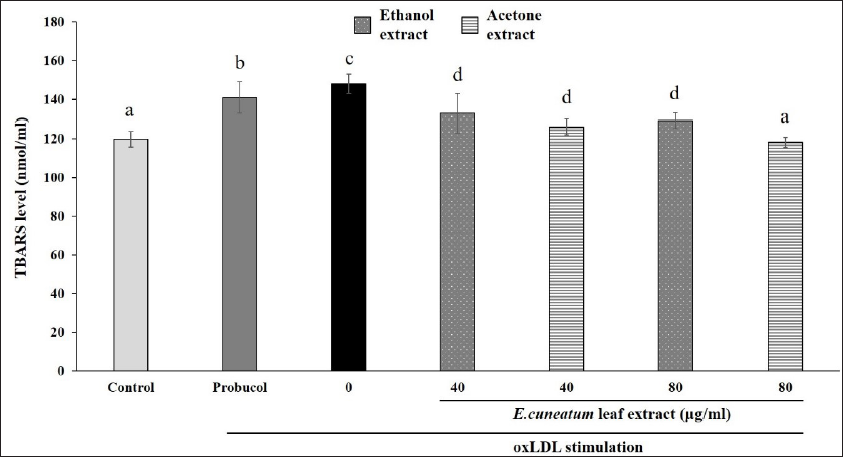
The addition of probucol or oxLDL alone to HAoEC (0 µg/ml) significantly increased TBARS levels compared to the control group (Figure 2). Treatment of both EC leaf extracts significantly reduced TBARS levels compared to no treatment. Acetone extract, at the higher concentration (80 µg/ml), showed a significant reduction of TBARS than other treatment groups, which was similar to control.
Probucol also inhibited about 85% ROS production in oxLDL-induced HAoEC (Figure 3). The treatment with both EC leaf extracts significantly inhibited the production of ROS, but was not as potent as probucol. EC ethanol extract at 80 µg/ml significantly suppressed ROS production (about 50%) compared to its lower concentration. Similarly, both concentrations of EC acetone extract reduced ROS production to almost 50%.
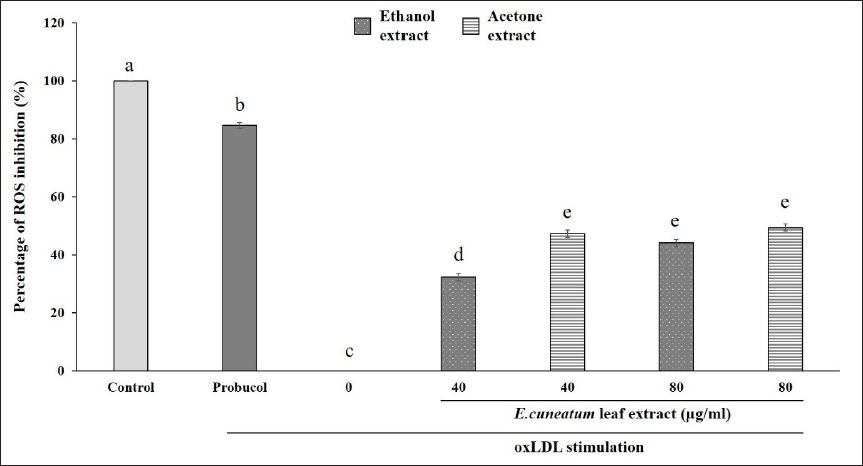
On the other hand, probucol caused about 85% of NO production in oxLDL-induced HAoEC (Figure 4). Both EC ethanol and acetone extracts increased the production of NO as compared to those without EC treatment (0 µg/ml). The effect of EC ethanol extract for both concentrations was similar to probucol. In contrast, treatment with EC acetone extract at both concentrations significantly boosts NO production by more than 90%, which is higher than probucol and ethanol extract.
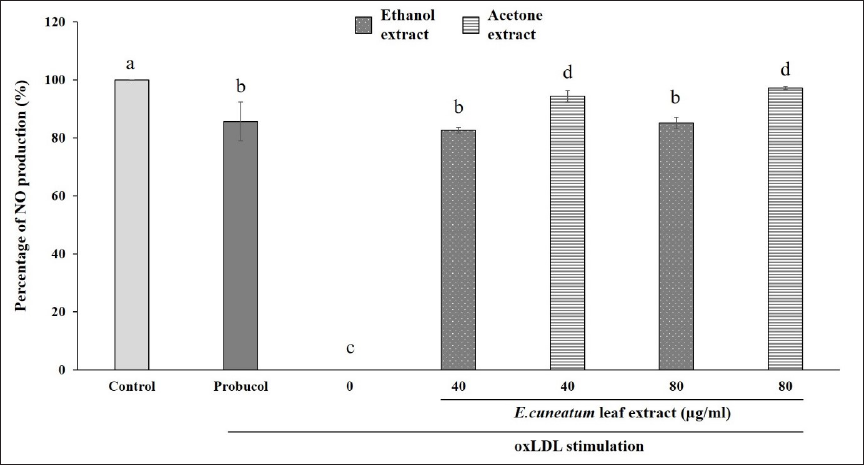
In general, both EC extracts inhibited lipid peroxidation by increasing TBARS content, reducing ROS production, and increasing the NO level, which suggested that they possess antioxidant properties. ROS produced by NADPH oxidases in endothelial and smooth muscle cells is involved in endothelial activation. During activation, endothelial cells take up oxLDL, which leads to a variety of biological effects such as reduction of endothelial NO production and upregulation of leukocyte adhesion molecules. 26 In this study, we discovered that EC extracts could protect against the initiation and propagation of atherosclerosis based on the antioxidant activities. A previous study has reported that methanolic EC extract had a metal chelating activity, which may reduce oxidation and scavenge free radicals.27, 28 The antioxidant properties of EC may be due to the presence of phytochemicals such as flavonoids, phenols, and alkaloids in ethanol and acetone extracts. 15
Anti-inflammatory Activity
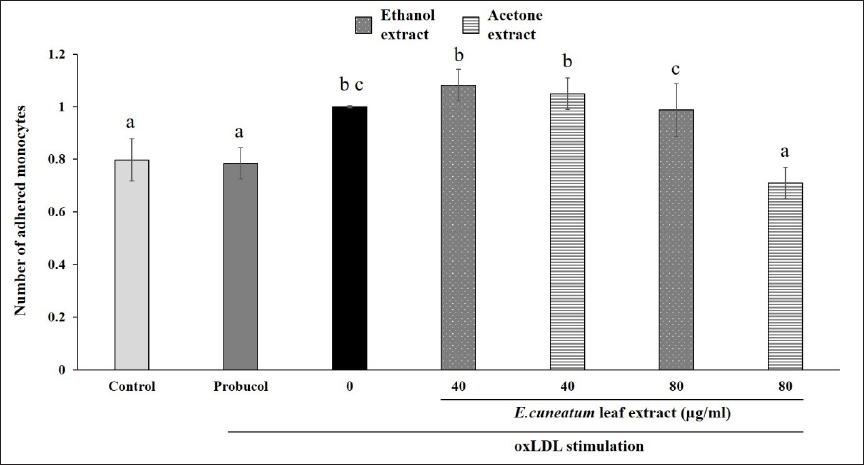
Figure 5 shows the effect of EC extracts on monocyte adhesion. The quantity of adhered monocytes in HAoEC alone (control) was significantly lower than oxLDL-stimulated HAoEC. Likewise, probucol significantly reduced monocyte adhesion in comparison to oxLDL-stimulated HAoEC (0 µg/ml). Both EC ethanol and acetone extracts did not significantly inhibit monocyte adhesion at 40 µg/ml concentration. In contrast, the acetone extract at 80 µg/ml showed the lowest number of adhered monocytes as compared to other treatment groups.
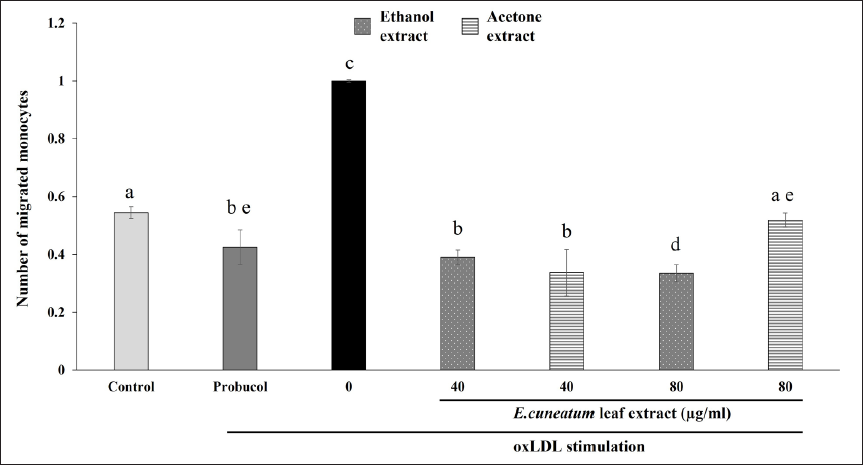
The quantity of migrated monocytes in HAoEC alone (control) was significantly lower than oxLDL-stimulated HAoEC without any treatment (0 µg/ml) (Figure 6). Probucol reduced the migration rate significantly in comparison to 0 µg/ml group. Treatment with EC ethanol extract at a higher concentration (80 µg/ml) significantly reduced monocyte migration, which is better than probucol. EC acetone extract also caused a reduction of monocyte migration; however, it was found that the effect was lessened at the higher concentration.
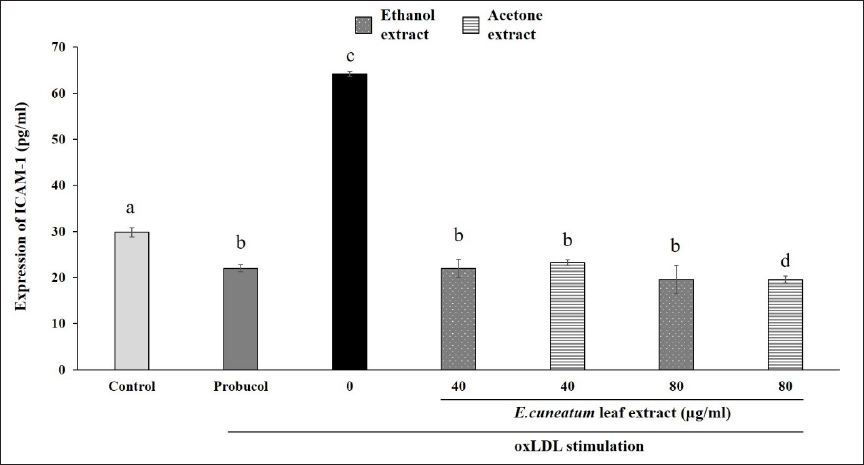
Subsequently, the oxLDL-stimulated HAoEC (0 µg/ml) group showed a significant increase in ICAM-1 expression compared to the control (Figure 7). Probucol was able to reduce ICAM-1 expression significantly compared to the control and 0 µg/ml group. Pre-treatment of EC acetone and ethanol extracts inhibited the expression of ICAM-1 in oxLDL-stimulated HAoEC, similar to probucol. EC acetone extract at 80 µg/ml was able to reduce the expression of ICAM-1 significantly as compared to other treatment groups.
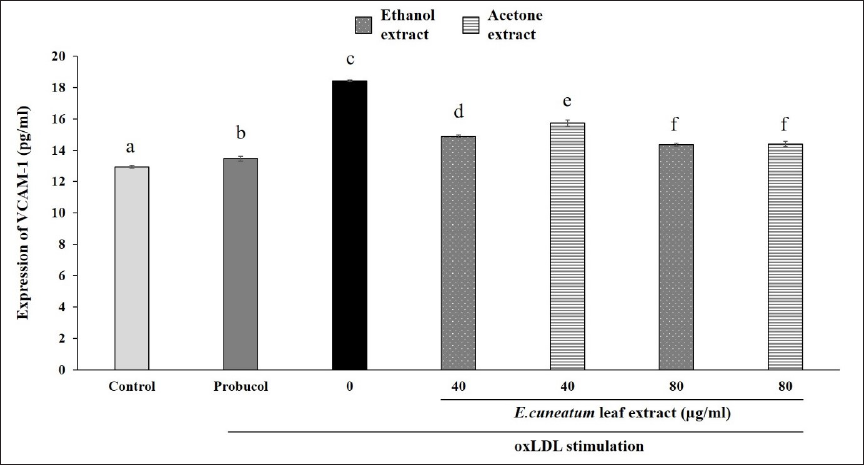
Similarly, the oxLDL-stimulated HAoEC (0 µg/ml) group showed a significant increase in VCAM-1 expression compared to the control (Figure 8). Probucol was able to reduce VCAM-1 expression significantly compared to oxLDL-stimulated HAoEC without treatment (0 µg/ml). Both EC ethanol and acetone extracts also decreased the expression of VCAM-1 in a dose-dependent manner. However, their effects were not as potent as probucol. EC ethanol and acetone extracts at higher concentrations showed similar effects on reducing VCAM-1 expression.
Monocytes adhesion and migration into the sub-endothelial space are the early occurrences in the progression of atherosclerosis.5, 6 It is proven in this study that the induction of oxLDL to the endothelial cells caused an increase in monocyte adhesion and migration. This is in agreement with a study which found that the monocyte adhesion rate was increased by oxLDL-induced human umbilical vein endothelial cells (HUVECs) and decreased when treated with isothiocyanates. 29 Intracellular signaling processes mediate the process, which ultimately upregulates the expression of pro-inflammatory chemokines, such as monocyte chemotactic protein-1 and interleukin-8.30, 31 It is also facilitated by adhesion molecule upregulation including ICAM-1 and VCAM-1, which is observed in the present study. Additionally, ROS is involved in the expression of adhesion molecules, which leads to monocyte adhesion and migration.32, 33 Zhao et al. have found that Danshenol A, a compound extracted from Salvia miltiorrhiza Bunge, inhibited ROS-mediated monocyte adhesion in TNF-α-stimulated HUVEC, which is attributed to its antioxidant properties. 34 Similarly, bilirubin, which is a potent antioxidant, effectively restricts monocyte migration through disruption of adhesion molecule-dependent ROS signaling. 35
Our study revealed that both EC extracts were able to reduce monocyte migration and expression of ICAM-1 and VCAM-1 in oxLDL-stimulated HAoECs, even at the lower concentration. On the other hand, only a higher concentration of the extracts was able to decrease monocyte adhesion. This may be the consequence of the inhibition of ROS production demonstrated in this study. Ethanol and acetone extracts from various plants have been proven to inhibit the expression of adhesion molecules. An earlier study on Ginkgo biloba ethanol and acetone extracts showed comparable downregulation of ICAM-1 and VCAM-1 expression in human endothelial cells. 36 This effect was related to the impairment of the NF-κB pathway due to the increase of the intracellular cyclic adenosine monophosphate (AMP) second messenger level in human endothelial cells, which is critical in the prevention of atherogenesis. The anti-inflammatory properties of EC leaf extract have also been reported in vivo using the carrageenan-induced paw edema model.12, 37 The studies proved that EC leaf extract may also reduce local and acute inflammatory responses. The presence of various bioactive compounds and antioxidant properties in the extract may be responsible for reducing inflammation.
It was observed that the effects of EC leaf extracts were concentration-dependent. In the majority of the antioxidant and anti-inflammatory assays, both ethanol and acetone extracts showed similar effects at the lower concentration. However, it was apparent that EC acetone extract resulted in better effects in reducing lipid peroxidation, monocyte adhesion, and ICAM-expression accompanied by increased NO production. Truong et al. reported that different solvents used in extracting bioactive compounds from plant materials produced various extraction yields. 38 This is because the level of bioactive chemicals in the extract may vary depending on the polarity of the extraction solvents. They found that ethanolic extract of Severinia buxifolia had a higher extraction yield than acetone extract, which corresponded to the content of bioactive compounds and their biological activity. Interestingly, a study on different types of extracts from 37 types of raw vegetables demonstrated that acetone was the most efficient solvent for extracting polyphenols and antioxidants. 39 This supported our previous findings that more bioactive compounds were detected in EC acetone than ethanol extract. 15 In the present study, it was also observed that acetone extract, which had a higher percentage yield, showed higher antioxidant activities than ethanol extract. The acetone extract had a higher total phenolic content and was more potent in terms of IC50 values of DPPH scavenging activity compared to ethanol extract. Those preliminary findings validated that EC acetone extract has superior effects than ethanol extract found in the present study.
Conclusion
Our findings provided considerable evidence that EC leaf extracts possess antioxidant and anti-inflammatory properties. Their antioxidant effects were observed by reducing lipid peroxidation and ROS production as well as increasing NO production. Moreover, their anti-inflammatory properties in downregulating the expression of adhesion molecules alongside monocyte adhesion and migration proved that EC leaf extract has the potential to prevent the progression of atherosclerosis. We found that the acetone extract displayed better antioxidant and anti-inflammatory activities, which confirmed that acetone is an efficient extraction solvent for complementary medicine. This is the first study to exhibit that EC leaf extract may have anti-atherosclerotic properties. Therefore, future in vitro and in vivo studies are recommended to elucidate the mechanisms involved.
Footnotes
Abbreviations
Acknowledgment
The authors would like to thank all the supporting staff in the Department of Human Anatomy, FMHS UPM, and Faculty of Pharmacy, CUCMS, for their technical assistance in this research project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by Universiti Putra Malaysia Putra Grant Scheme (GP-IPS/2017/9538900) and CUCMS Research Grant Scheme (CRGS/03/02/2013).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
EC ethanol and acetone extracts reduced TBARS and ROS production levels in oxLDL-induced HAoECs.
EC ethanol and acetone extracts increased NO production in oxLDL-induced HAoECs.
Only EC acetone extract at 80 µg/ml concentration caused a significant reduction of monocyte adhesion in oxLDL-induced HAoECs.
EC ethanol and acetone extracts reduced monocyte migration and expression of ICAM-1 and VCAM-1 in oxLDL-stimulated HAoECs.