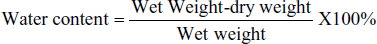Abstract
Background
Cerebral ischemic stroke is the second major cause of mortality worldwide that results in persisting disability and mental agony. Ischemic stroke is induced by the diminished blood circulation to the brain, which can be due to obstruction by arteriosclerosis, fibromuscular dysplasia, or thrombosis. Triptonide is a diterpene triepoxide, purified out of extracts of Tripterygium wilfordii Hook F, and is an emerging target against, for example, cancers and inflammatory insults.
Materials and Methods
Taking this into consideration, this study was designed to analyze the effect of triptonide on ischemic/reperfusion (I/R) cerebral infarction stroke rats.
Results
Our study showed that triptonide decreased the infarct volume and brain edema produced by I/R. Moreover, triptonide protected the rats from any neurological deficits, which were analyzed using a five-point scoring system, augmented antioxidant enzymes like superoxide dismutase, catalase, glutathione peroxidase, reduced glutathione content, and lowered the activity of acetylcholinesterase. Triptonide also decreased the levels of pro-inflammatory cytokines such as interleukin-1β (IL-1β), TNF-α, and IL-6, while it augmented anti-inflammatory cytokines IL-10 and vascular endothelial growth factor. In this study, cerebral infarction stroke rats showed an increase in malondialdehyde levels. Triptonide preserved the normal brain architecture from various neurotoxic effects.
Conclusion
Thus, triptonide can be targeted for drug discovery in the future to protect against cerebral infarction stroke.
Introduction
Stroke or cerebrovascular accident is the second common cause of death worldwide, accounting for about 11.6% of total deaths, and the third common cause of death together with disability in 2019. Although there is a gender bias in the incidence of stroke, no such difference was observed in the case of stroke-related deaths. 1 Even though stroke occurs generally in old people, it also affects younger age groups. Stroke in younger patients displays a wide number of possible risk factors and causes.
Of the three pathological categories of stroke, ischemic, intracerebral hemorrhage, and subarachnoid hemorrhage, the first one is predominant and constituted 62.4% of all novel cases of stroke in the year 2019. 1 Ischemic stroke (also known as cerebral infarction or brain attack) is a prevalent source of chronic disability, which poses limited treatment options. Ischemic stroke can be further classified into types based on etiology such as cardioembolism, small vessel obstruction, large artery arteriosclerosis, and stroke of indeterminate cause. 2 It can also be due to an embolism that plugs the blood flow to the brain.3, 4 Researchers demonstrated that cerebral ischemic/reperfusion (I/R) insult leads to edema in the brain, disrupted blood–brain barrier (BBB), and improved vascular permeability.5, 6 Any focal neurological debt prolonging more than 24 hr can lead to cerebral infarction stroke. Hence, it is of great significance to design a novel drug for the treatment of cerebral ischemic stroke.
Complementary or traditional Chinese medicines (TCM) have been employed in the treatment of many diseases, as they have few side effects. One such TCM is the Chinese herb Tripterygium wilfordii, which has been widely used for curing various inflammatory ailments in complementary medicine since ancient times. 7 Triptonide is purified from the extracts of T. wilfordii Hook F8, 9 and was originally identified as a promising non-hormonal male contraceptive agent. Chemically, it is a cyclic ketone, an organic heteroheptacyclic compound, a diterpene triepoxide, and a butenolide. Triptonide displays a variety of functions, including anti-neoplastic activity, immunosuppression, and protection against inflammation. It has also been shown to induce sperm deformity and decrease sperm motility in mice. Recently, researchers indicated triptonide to be involved in the anti-carcinogenic activity on cancers of the stomach, lung, pancreas, and leukemia,10–16 by altering signaling molecules involved in cancer induction and advancement. Triptonide was shown to inhibit triple-negative breast cancer in terms of migration, invasion, and angiogenesis in vitro and tumor proliferation and metastasis in vivo.17, 18 Triptonide also demonstrated contraceptive effects, 19 anti-leukemia activity, 20 and anti-inflammatory 21 activities. In ischemic stroke, different types of inflammatory cells induce reactive oxygen species (ROS)-activating pro-inflammatory genes, including cytokines. The current study was planned to investigate the therapeutic effects of triptonide on cerebral ischemia-induced rats by alleviating the oxidative stress and inflammatory responses.
Materials and Methods
Chemicals
Triptonide and other chemicals were obtained from Sigma Aldrich, USA. The assay kits were procured from Thermo Fisher Scientific, USA. All the reagents used were analytical grade.
Experimental Animals
Male Sprague–Dawley (SD) rats (10–14 weeks, 250–300g body wt.) were used in the experiments. Before commencing the experiments, all animals were kept under standard conditions (12 hr dark–light cycle, 50–60% relative humidity, 37°C). The rats were supplied with normal rat feed and water.
Experimental Design
A total of 24 rats were split into four groups, 6 animals in each group. Group I had sham-operated rats, group II had I/R cerebral infarction stroke rats, and groups III and IV included I/R cerebral infarction stroke rats treated with 10 mg/kg b.w. triptonide and 20 mg/kg b.w. triptonide, respectively.
Induction of the I/R Cerebral Infarction Stroke Model
The current study employed the I/R model by inducing middle cerebral artery occlusion (MCAO). 22 In brief, SD rats were first anesthetized using 10% chloral hydrate (i.p.). Then, the right common carotid arteries, external carotid arteries, and internal carotid arteries were exposed. This was followed by occluding the middle cerebral artery by a nylon filament (0.26 mm diameter) with a blunted silicone tip (0.34mm diameter). This ischemic condition lasted for 2 hr, and then the filament was gently pulled out to admit reperfusion for 24 hr. Positive MCAO was defined as reduced cerebral blood flow to less than 30%. All surgical procedures excluding MCAO were performed on sham-operated rats.
Rats were euthanized, and serum and tissue samples were separately collected for further experiments. 10% tissue homogenate was made with homogenizing buffer (0.1M Tris-HCl buffer, pH 7.4), which was spun at 12,000 rpm for 10 min. The supernatant was used for measuring inflammatory cytokines.
Measurement of Infarct Volume
Following I/R, the brain tissues were immediately excised and cut at 0.2 cm intervals. The tissue slices were stained using 2% 2,3,5-triphenyltetrazolium chloride solution for half an hour at room temperature, followed by imaging of the stained tissues. Relative infarct volume was calculated by comparing nonstained areas (infarct) to the total area, and results were expressed in terms of percentage.
Measurement of the Brain Edema
The water content was calculated to assess the degree of cerebral edema (CED, wet–dry method). In short, brains were excised and weighed. The dry weight of the brains was obtained by dehydrating the samples at 100°C for 24 hr using an oven. The amount of water in the brain was expressed as a percentage using the following formula:
Assessment of Neurological Deficits
Neurological deficits were analyzed using a 5-point scoring system 24 hr after recanalization. In brief, scores were allotted as follows: (a) 0 point—rats with a normal walk and no observable neurologic deficits; (b) 1 point—mild focal neurological deficit, with flexion of the contralateral torso and the forelimb upon lifting of the animal by its tail; (c) 2 points—moderate focal neurologic deficit, circling to the contralateral side but normal posture at rest; (d) 3 points—severe focal neurological deficit, falling to the contralateral side at rest; (e) 4 points—very severe, no spontaneous motor activity and depressed consciousness level.
Determination of Malondialdehyde
The malondialdehyde (MDA) level in rat serum was estimated using the Thiobarbituric Acid Reagent (TBARS) method. 23 Lipid peroxidation releases MDA, which forms a pink-colored adduct with TBARS, and the color formed was measured at 532 nm using a spectrophotometer. The levels of MDA were expressed in terms of nmoles of MDA/milligram of protein.
Assay of Antioxidant Enzymes
The antioxidant enzymes in serum were estimated as follows: superoxide dismutase (SOD) was estimated by the method reported by Marklund and Marklund, 24 catalase (CAT) by the method reported by Sinha, 25 glutathione peroxidase (GPx) by the method reported by Rotruck et al., 26 and glutathione (GSH) levels by using the method reported by Hu. 27
Determination of Acetylcholinesterase Activity
Acetylcholinesterase (AchE) activity was analyzed by the standard method. 28 When acetylthiocholine iodide is cleaved by AchE, a thiocholine substrate is formed. Thiocholine reacts with 5,5-dithiobis-(2-nitrobenzoic acid) reagent to yield yellow-colored thionitrobenzoic acid. The concentration of this product can be analyzed by measuring at 412 nm spectrophotometrically. The AchE activity can be expressed as moles substrate hydrolyzed/min/mg protein.
Measurement of Inflammatory Cytokines
IL-1β, tumor necrosis factor-α (TNF-α), IL-6, IL-4, IL-10, and vascular endothelial growth factor (VEGF) in serum and brain tissues were measured using commercially available kits as per manufacturers’ protocols (Thermo Fisher Scientific, USA).
Histological Analysis
Brain tissues were taken from SD rats after the treatment period and were rinsed immediately with physiological saline. For histological studies, brain tissues were first fixed in a 10% formalin fixative solution. Then the tissues were embedded in paraffin and then sectioned at 5 µm thickness using a rotary microtome. The sections were then stained with hematoxylin and eosin (H&E), and morphology was analyzed using a light microscope (Nikon, Japan).
Statistical Analysis
Data were investigated by one-way analysis of variance, followed by students’ Newman Keuls test using SPSS software. Data were represented as mean ± SD of six observations.
Results
As shown in Figure 1A, infarct or lesion was not observed in sham-operated rats, as there was no cerebral ischemia. In the I/R cerebral infarction stroke model, the lesion was observed at approximately 50%, while triptonide decreases the infarct volume at both doses. When compared to the sham-operated rats, I/R cerebral infarction stroke rats showed a significant (p < 0.01) increase in CED. Triptonide (10 mg/kg b.w.) brought back the water content to normal levels, while a high concentration (20 mg/kg b.w.) significantly (p < 0.05) lowered CED (Figure 1B).

By a 5-point scoring system, neurological deficits in cerebral infarction stroke rats were analyzed. Sham-operated rats (group I) showed no significant observable neurological deficits. Group II rats showed mild to moderate focal neurological deficit. In triptonide-treated (group III) rats, these neurological deficits were not observed; triptonide at 20 mg/kg b.w. dose worked significantly (p < 0.05) better on them (Figure 2).
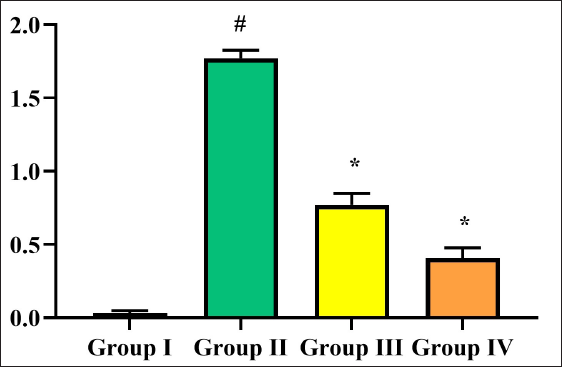
When compared to sham-operated rats (group I), cerebral infarction stroke rats (group II) showed an increase in MDA levels. This increase was restored to a normal level by treatment of triptonide at 10 mg/kg b.w. dose, while the levels further decreased significantly (p < 0.05) at 20 mg/kg b.w. dose of triptonide (Figure 3A). SOD and CAT activities declined significantly in cerebral infarction stroke rats. On the other hand, triptonide treatment (groups III and IV) significantly (p < 0.05) improved the levels of these antioxidant enzymes and brought them back above normal (Figure 3B and C). A similar mode was noticed in the case of GPx and reduced glutathione content. In this case, triptonide treatment augmented these levels to near-normal levels (Figure 3D and E).
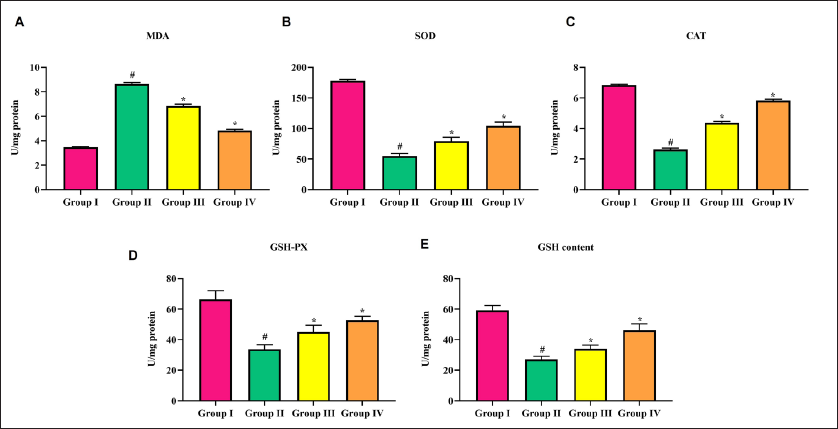
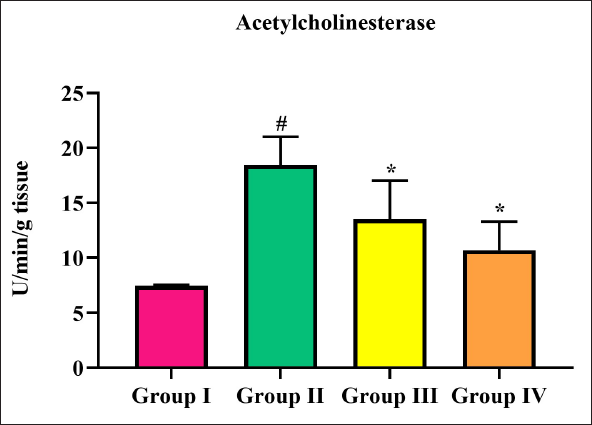
AchE activity elevated significantly in cerebral infarction stroke rats, in comparison with sham-operated rats. Triptonide treatment lowered the activity of AchE to normal at both doses (10 and 20 mg/kg b.w.), out of which the latter was significant (Figure 4).
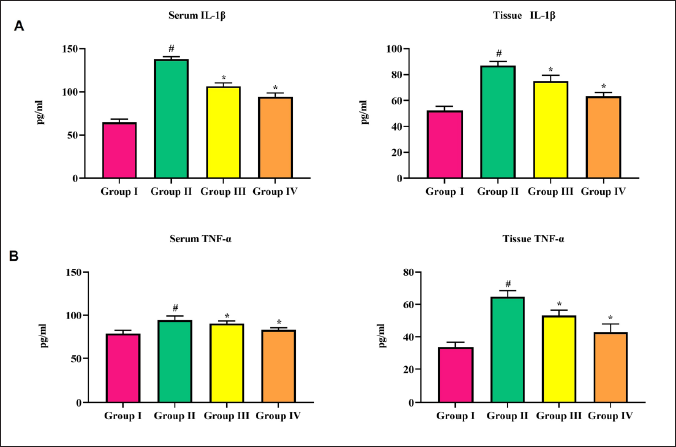
There were increases in serum and tissue interleukin-1β (IL-1β) levels observed in the cerebral infarction rat model. Triptonide treatment slightly lowered serum IL-1β levels and significantly (p < 0.05) decreased tissue IL-1β at both doses (Figure 5A and B). TNF-α showed a surge in the cerebral infarction rat model (group II) rats in both serum and brain tissue. However, this surge was reverted by triptonide to near-normal levels (Figure 5C and D).
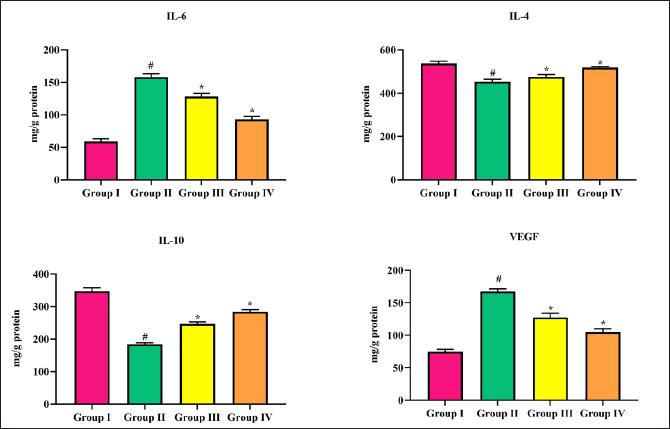
In comparison with group I sham-operated rats, cerebral infarction stroke rats showed a remarkable elevation in interleukins-1,-4,-10 and VEGF. Triptonide administration restored IL-6 levels to near-normal levels at both doses significantly (p < 0.05) at 20 mg/kg b.w. dose. On the other hand, triptonide treatment slightly increased the IL-4, IL-10, and VEGF levels at both doses (Figure 6).
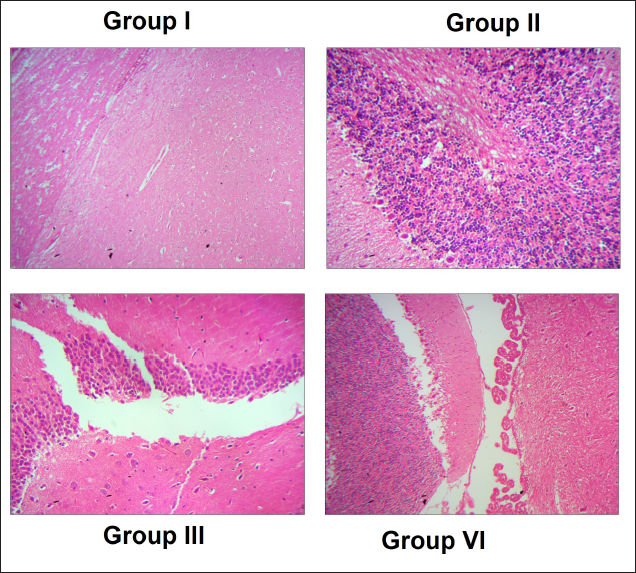
Group I sham-operated rats showed a normal brain architecture, whereas the I/R cerebral infarction rat model showed abnormal distorted morphology of the brain. Triptonide restored the brain morphology to normal, which was manifested at 20 mg/kg b.w. dose (group IV) (Figure 7).
Discussion
Cerebral infarction stroke is a distressing state leading to neurological debility, often followed by death. 29 An important factor that is employed in assessing the severity of stroke is lesion or infarct volume. It is used widely in predicting stroke consequences. In addition, infarct volume has been suggested as a substitute for classic disability or handicap scales. 30 In our study, sham-operated rats showed no infarct, as they were not subjected to cerebral ischemia. In the I/R cerebral infarction stroke model, a lesion was observed, which confirmed that cerebral infarction had occurred. The decrease in infarct size observed on treatment with triptonide may be due to triptonide-induced resolution of edema in the brain or inhibition of formation of an infarct.
CED is considered an acute impediment of cerebral ischemic stroke, and it accounts for 5% of death in patients suffering from cerebral ischemia. 31 Brain edema is an outcome of disorganization of the tight junctions and BBB due to malfunction of the endothelial tissue layers. 32 Stroke severity can also be assessed by CED. CED takes place in three different phases, viz., the early cytotoxic phase, the middle ionic phase, and the vasogenic phase. 32 In our study, I/R cerebral infarction stroke model rats showed mild brain edema, which was not observed in triptonide-treated cerebral infarction rats. This showed that triptonide prevented the occurrence of CED and protected the normal BBB, the disruption of which causes edema.
A focal neurological deficit is defined as problems with nerves and the central nervous system and may affect any part of the body. Neurological deficits also include complications affecting speech, eyesight, and hearing and changes in movement, sensation, and coordination. In the present study, mild to moderate focal neurological deficits were observed in cerebral infarction stroke rats, which showed flexion to circling on the contralateral side. Triptonide-treated cerebral ischemic rats did not show such neurological deficits, demonstrating the protective action of triptonide on cerebral infarction.
MDA is the most commonly employed biomarker of oxidative stress to study many maladies such as cancer, asthma, and cardiovascular diseases. MDA is the final product of lipid peroxidation that induces cellular toxicity and hence is generally indicated as a measure of oxidative stress. 33 Hence, the analysis of circulating MDA levels will provide an insight into the severity and prognosis of ischemic stroke. In this study, cerebral infarction stroke rats (group II) showed an increase in MDA levels. This may be due to increased lipid peroxidation occurring in the brain due to cerebral ischemia, which ultimately leads to oxidative stress. Triptonide treatment for cerebral infarction stroke rats showed normal MDA levels, which demonstrates that triptonide protects the brain from oxidative stress and lipid peroxidation.
Superoxide radicals that are produced during I/R are scavenged by many antioxidant enzymes, including SOD, Gpx, and CAT. SODs comprise a family of metalloenzymes that defend against ROS-mediated oxidative stress to tissues. 34 These are enzymes that convert harmful superoxide anion (O2−) into O2 and H2O2. SOD protects the cell against inflammatory changes as well. CAT cleaves harmful H2O2 and detoxifies them into O2 and water. GPx converts harmful hydroxyl radicals to water and oxygen, making them non-toxic. In addition, other antioxidants such as glutathione, vitamin C, and vitamin E also eradicate toxic radicals.
In this study, cerebral infarction stroke rats exhibited a notable decline in the activities of SOD, CAT, Gpx, and reduced glutathione, which represents cerebral infarction, resulting in high oxidative stress and hence consumption of these antioxidants. Triptonide improved the activities of these antioxidants, thereby suppressing the oxidative stress built up by cerebral infarction. Ischemia and reperfusion are associated with endothelial tissue injury, leading to the generation of ROS, which can be overcome by overexpression of SOD. 35 Kim et al. 36 suggested that manganese SOD prevents MCAO. Weisbrot-Lefkowitz et al. 37 showed that higher expression of Gpx-1 protected mice against the effects of MCAO. A similar trend was seen in the case of GPx and reduced glutathione content. Triptonide treatment restored and protected the changes in the brain against oxidative insults.
AchE, a vital enzyme in the central nervous system, is required in the rapid hydrolysis of acetylcholine (ACh), leading to the cessation of cholinergic synaptic transmission. In the present study, cerebral infarction stroke rats showed a significant increase, while triptonide treatment decreased it. Fujiki et al. 38 also reported that inhibiting AChE diminishes cerebral lesion volume in experimentally stroke-induced rats. Thus, triptonide decreased infarct volume by diminishing AChE.
There exists an intimate association between interleukins and the incidence of ischemic stroke. After the initiation of ischemic stroke, an abnormal surge of inflammatory cytokines leads to many unfavorable outcomes such as inhibition of neurogenesis, increased inflammation, and suppressed integrity of the BBB, ultimately resulting in brain injury. Various inflammatory cytokines are involved in the pathogenesis of stroke, such as IL-1,-6, TNF-α, and so forth.
In the current study, an increase in serum and tissue interleukin-1β (IL-1β) levels was observed in the cerebral infarction rat model. This result was supported by another study showing that a high IL-1β surge after ischemic stroke leads to activation of phospholipase A2 for degrading arachidonic acid and destroying the phospholipid bilayer. 39 The metabolites of arachidonate, which are prostaglandin and leukotriene, can lead to disruption of the BBB, resulting in the incidence of CED, the hallmark of cerebral ischemia. 40 TNF-α also showed a surge in the cerebral infarction rat model in both serum and brain tissue in this study. It is to be noted that inflammatory reaction leads to brain injury by liberating neurotoxic molecules like TNF-α, and hence, the increase was observed in the present study in the cerebral ischemic model. Triptonide treatment lowered these IL-1β levels and protected the brain tissue from inflammatory insults.
Several studies suggest that IL-4 is crucial for maintaining proper brain functions. Deregulation of IL-6 leads to the pathogenesis of disorders such as coronary heart disease, leukemia, high blood pressure, ischemic stroke, and so forth. Group II rats showed a noticeable rise in interleukins-6,-4,-10 and VEGF levels. Mechtouff et al. 41 reported that high IL-6 levels were observed in ischemic stroke patients after 24 hr of reperfusion. IL-6 is considered to be an inflammatory marker following stroke, and high IL-6 levels are due to its secretion from neurons and microglia in the infarct area. 42 IL-10 is a crucial anti-inflammatory cytokine that decreases inflammation and prevents cerebral ischemia. In this study, triptonide treatment slightly increased the IL-10 levels at both doses. Overexpression of IL-10 leads to a neuroprotective effect and anti-inflammatory regulation, leading to enhanced survival of neurons during ischemia. 43 Although VEGF is implicated in the formation of arteriosclerosis, the root cause of stroke, it can also protect the ischemic brain from infarction and aid in recanalization of blocked cerebral arteries and brain repair. Hence, VEGF is neurogenic, angiogenic, and neuroprotective. In this study, triptonide augmented VEGF expression, leading to brain repair.
Finally, we were interested in analyzing the brain tissue morphology by H&E staining in sham and I/R groups. Sham-operated rats showed a normal brain tissue architecture. In the cerebral infarct model, many shrunken and differently shaped neurons with nuclei shrinkage with loose or sparse tissue were observed. Triptonide (20 mg/kg b.w.) restored the brain morphology to normal intact cells.
Conclusion
To conclude, triptonide protects the brain against various inflammatory insults and helps in the restoration of toxic effects after a cerebral ischemic episode. This study is the first of its kind, which demonstrates that a novel molecule, triptonide, exhibits neuroprotective effects. Therefore, it was clear that the triptonide has neuroprotective effects against cerebral ischemic stroke in an animal model. However, additional studies are still required in the future to confirm the neuroprotective effects of triptonide against stroke.
Footnotes
Abbreviations
H&E: Hematoxylin-eosin; I/R: Ischemic/reperfusion; IL-1β: Interleukin-1β; VEGF: Vascular endothelial growth factor; TCM: Traditional Chinese medicines.
Acknowledgment
This work was supported by Weifang Nursing Vocational College, No. 9966, Yunmenshan South Road, Qingzhou City, Shandong Province 262500, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
All the animal experiments were conducted according to the animal ethical guidelines.
Summary
Cerebral ischemic stroke is the second major cause of mortality worldwide, which results in persisting disability and mental agony.
Triptonide protected the rats from any neurological deficits, which were analyzed using a 5-point scoring system, augmented antioxidant enzymes, and lowered the activity of AchE.
Triptonide also decreased the levels of pro-inflammatory cytokines such as interleukin-1β (IL-1β), TNF-α, and IL-6, while it augmented anti-inflammatory cytokines IL-10 and VEGF.