Abstract
Background
Ethacridine have anticancer effects by inhibiting regulatory transcription factors and cell viability in various cancer cells. To investigate the effect of ethacridine on colorectal cancer cell lines, SW620 was studied via regulation of JAK/STAT/ERK signaling pathway.
Materials and Methods
Different doses of ethacridine (5–35 µM) expressed antiproliferative effects by decreasing the viability in a dose-dependent manner and the IC50 value was found to be 10 µM.
Results
Subsequent treatment with 10 µM of ethacridine showed that it induced mitochondrial dysfunction and reactive oxygen species generation. DAPI and PI staining assays revealed prominent apoptotic cells under the microscope when treated with 10 µM of ethacridine. In the mRNA expression study performing RT-PCR of apoptotic markers, cyclin-D1, Bax, Bcl-2, caspase 3, C-Myc, and surviving, enhanced levels of these markers were suppressed, which was inversely proportional to the levels of apoptotic enhancers namely Bax and caspase-3. It was also observed that increased NF-κB, IL-6, TNF-α, and COX-2 in colorectal cancer are suppressed by ethacridine. The expressions of JAK/STAT/ERK were also significantly suppressed after ethacridine treatment in SW620 cell lines.
Conclusion
In summary, it was corroborated that ethacridine promoted apoptosis in colon cancer cells by inhibiting quite a few cell signaling factors.
Introduction
Colon cancer is reported as the fourth leading cause of cancer death in both sexes worldwide, with 90.3% and 12.5% early-stage and advanced-stage survival rates, respectively, in diagnosed patients. Clinicopathologically colon tumor prognosis assessment was diagnosed principally as tumor location, size, nodal invasion, and metastatic disease. 1 Colorectal carcinoma (CRC) is the fourth leading cause of death in patient of both genders and is the third most diagnosed form of cancer in the world. Due to varied changes in human lifestyle and food habits, there has been a large occurrence of cancer-related deaths across the globe. Probably 4% to 5% of cancer is found to be associated with colorectal cancer and factors, such as lifestyle, age, and disease history, influence the disease. 2 Despite the enhanced developments in the treatment of colorectal cancer, the prognosis of the disease is still insignificant. 3
At present, the only practiced medical treatment method for colon cancer is surgery followed by chemotherapy. 4 New therapeutic strategies should be devised to overcome the underrated prognosis and challenges faced in the treatment of CRC. In that screening for an effective drug, ethacridine has shown predominant results in the treatment of various types of cancers by specifically targeting the cancer cells. 5 In general, ethacridine is known for its antiseptic properties. In a research study on the effect of ethacridine on thyroid follicular cells, cellular viability decreased along with clonogenicity and also induced apoptotic and differentiation effects.
Ethacridine revealed that it activates transcription cofactors of binding sites of cellular differentiation in a detailed chemical screening process. 6 Ethacridine (2-ethoxy-6,9-diaminoacridine) resulted from the attempted combination of the activities of acriflavine and hydrocupreine in the 1920s. Huang et al., 7 demonstrated that ethacridine acts as an inhibitor of poly(ADP-ribose) glycohydrolase (PARG) and an activator of the transcriptional coactivator with PDZ-binding motif (TAZ). Furthermore, ethacridine suppressed cell growth and clonogenic ability of thyroid cancer cells in a time- and dose-dependent manner. The PAX8 and NIS expressions were significantly increased so that ethacridine appeared to induce apoptotic cell death in thyroid cancer cells and promote differentiation in a subset of thyroid follicular cells. Recent studies have proven that ethacridine was pharmacologically effective against malignancies and further, it possesses more pharmacological potential. These findings correlate, and this study proposes that ethacridine might have an anticancer potential against colon cancer cells.
Colorectal cancer is the second most highly observed mortality among all cancer-related deaths, thereby creating a necessity for the exploration of unique molecules that would be cost-effective with less toxic effects. 8 Apoptosis has been a trigger factor in several chemotherapeutic studies. Both intrinsic and extrinsic factors should be targeted by a chemotherapeutic agent. In cancer, these apoptotic factors are either down-regulated or up-regulated, thereby facilitating pores and release of cellular proteins. Hence, in our study, to investigate the effect of ethacridine on these apoptotic factors, inflammatory signals, and through regulation of JAK/STAT/ERK signaling pathways related to cellular growth is experimentally studied in colon cancer SW620 cells.
Materials and Methods
Chemicals
Ethacridine, FBS, and antibiotics were obtained from Merck, Germany. The antibodies against JAK2, p-JNK2, STAT3, p-STAT3, ERK, p-ERK, and β-actin were procured from Labome, USA.
Cell Culture
SW620 colon cancer cells collected from the Peking Union Cell Resource Center (Beijing, China) were cultured in DMEM nutrient medium added with penicillin (100 units/mL), 10% FBS, and streptomycin (100 µg/mL), at 37°C with 5% CO2, these cells were passaged by adding trypsin and ethylenediaminetetraacetic acid (EDTA) at a percentage of 0.25% and 0.02%, respectively, when the cells reach 80% confluence. For the experiment, cells that are in the logarithmic phase of the growth curve were collected.
Cell Viability Assay
In 96-well plates, SW620 cells were seeded at a mass of 1 × 10 5 cells/well and preserved for 1 day. Cells were then treated with ethacridine at concentrations of 2.5 to 20 µM/mL for 24 hr. MTT assay was performed based on the study by Velu et al., 9 and cell viability was calculated.
Mitochondrial Membrane Potential (MMP) Assay
Mitochondrial dysfunction is associated with all types of diseases. To assess the membrane potential of cancer cells, a cationic lipophilic dye namely Rh-123 staining is used. Red fluorescence is emitted when the dye gets concentrated with the polarized cellular mitochondrial membrane, similarly, when there is a decrease in membrane potential, Rh-123 dye cannot accrue with the matrix of mitochondria and emits green fluorescence as the dye exists as a monomer. This ratio of red/green fluorescence indicates the MMP level thereby evaluating the effect of 10 µM of ethacridine on mitochondrial dysfunction. MMP was evaluated based on the study by Li et al. 10
Reactive Oxygen Species (ROS) Assay
Intracellular ROS formation was measured in SW620 colon cancer cells using DCFH-DA staining technique of Jesudason et al. 11 Cells were treated with 10 µM of ethacridine followed by adding DCFH-DA (10 µM) and maintained at 37°C for 30 mins. Results were then read at 485/535 nm wavelength in a well plate reader.
Apoptosis Detection by DAPI and PI Staining
Apoptosis was examined by using 4,6-diamino-2-phenylindole (DAPI) and propidium iodide (PI) staining. Colon malignant cells were grown in dishes in the presence of 10 µM of ethacridine, and the stimulation of programmed cell demise was assessed after 1 day. Apoptotic cellular morphological alterations during DAPI staining were observed using the fluorescence microscope.12, 13
Real-time Polymerase Chain Reaction
The mRNA expressions of inflammatory, cell proliferation, and apoptotic markers (Cyclin D1, caspase-3, Bcl-2, Bax, C-Myc, survivin, NF-κB, COX-2, TNF-α, and IL-6) were analyzed by using real-time polymerase chain reaction (qPCR). Cells were added with 10 µM ethacridine and subjected to RNA sequestration consuming TRIzol reagent (EZ BioResearch, USA) and converted to cDNA using Reverse Transcriptase enzyme. qPCR conditions were followed based on the study by Huang et al. 7 Cyclin-D1, caspase-3, Bcl-2, Bax, C-Myc, survivin, NF-κB, COX-2, TNF-α, and IL-6 were normalized when compared to control β-actin and analyzed using 2–∇∇Ct method.
Western Blotting Analysis
Regulatory Protein marker expressions such as JAK-2, STAT-3, and ERK were analyzed by the Western blotting technique. Human colon cancer cells SW620 cells were treated with 10 µM of ethacridine and cultured for 24 hr. The cell lysates were prepared by using an ice-cold lysis buffer containing protease inhibitors and executed western blotting analysis. The protein content was estimated using the BCA Protein Assay Kit. About 20 µg of protein was electrophoresed by SDS-PAGE and shifted to nitrocellulose membrane. About 3% bovine serum albumin was added to block the membrane for 2 hr and primary antibodies (JAK-2, STAT-3 and ERK) were added in 1:1,000 dilution and kept overnight at 4°C. Followed by the addition of secondary antibody for 60 min, bands were detected using smearing chemiluminescence reagent, followed by blot densitometry analysis. Blots were detected with ImmunoStar LD (Wako Pure Chemical Ind., Tokyo, Japan). Immunoreactive bands were visualized by using FluorChem™ FC3 (ProteinSimple, San Jose, CA, USA). The quantification of protein bands was evaluated using densitometry with Image J software.
Statistical Examination
A statistical study was achieved using Graphpad Prism software version 8.0.1, with comparison attained using ANOVA and subsequently Duncan’s test. A P value less than 0.05 is statistically significant.
Results
Ethacridine Exhibits Antiproliferative Effects on SW620 Cells
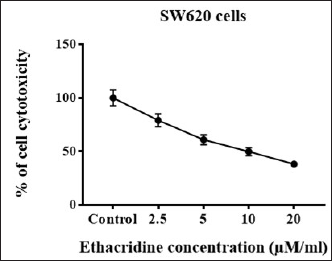
MTT test was used to study the antiproliferative effects of different doses (2.5–20 µM/mL) of ethacridine. It was observed from the outcomes that ethacridine caused a significant dose-dependent decrease in cell viability. The IC50 value was found to be 10 µM in SW620 cells. Thus, the results were in coincidence with the proposed anticancer activity of ethacridine (Figure 1).
Cell Viability Measurement at Different Concentrations of Ethacridine (2.5–20 µM/mL) in SW620 Cells. Samples Were Distributed as Triplicates and Expressed as Mean ± SD. Values with p < 0.05 Are Considered to be Statistically Significant.
Ethacridine Induces ROS in SW620 Cells
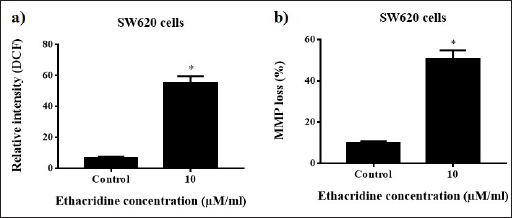
ROS levels were augmented and increased by adding 10 µM of ethacridine with an evident increase in DCF fluorescence, and the results observed are shown in Figure 2a. Fluorescent microscopic view shows prominent bright green color in cells treated with ethacridine when compared with the control. Data observed from the spectrofluorometric analysis were also validated with the increased microscopic ROS levels (Figure 2a).
ROS and Mitochondrial Membrane Potential Analysis in 10 µM Ethacridine-treated SW620 Cells. *p < 0.05 are Considered to be Statistically Significant.
Ethacridine Induces Mitochondrial Dysfunction in SW620 Cells
The mitochondrial membrane concentrated with Rh-123 emits green fluorescence; however, when there is a drop in the membrane potential, Rh-123 exists as a monomer without accumulation with the mitochondrial membrane and emits green fluorescence (Figure 2b). MMP level was estimated from the ratio of green fluorescence, thereby assessing the impact of ethacridine on mitochondrial dysfunction. After treating the cells with 10 µM of ethacridine, MMP levels decreased in a dose-dependent manner in both SW620 cells (Figure 2b). The results propose that ethacridine can induce mitochondrial dysfunction in cancer cells.
Ethacridine Stimulated Apoptosis by DAPI and PI Staining
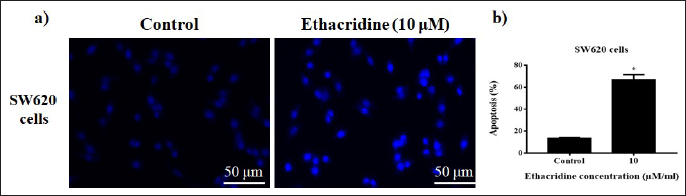
DAPI staining is used to evaluate apoptotic cells by using the dye to stain the nucleus. Results showed that DAPI-stained apoptotic cells were significantly high (p < 0.05) in cells added with ethacridine (10 µM) counter to untreated cells (Figure 3a,b).
(a) DAPI Stained Images of Cells Exposed to 10 µM Ethacridine SW620 Cells. Scale Bar = 50 µm. (b) *p < 0.05 Are Considered to be Statistically Significant.
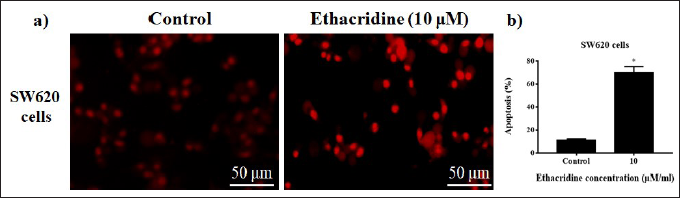
Apoptotic and necrotic cell differentiation is studied using PI staining. SW620 cells treated with 10 µM of ethacridine and subjected to PI staining revealed certain features of apoptotic cells, such as nuclei fragmentation. It was significantly evident from the results that 10 µM of ethacridine induced apoptotic changes in cancer cells against untreated cell lines (Figure 4a,b).
(a) PI Stained Images of Cells Exposed to 10 µM Ethacridine SW620 Cells. Scale Bar = 50 µm. (b) *p < 0.05 are Considered to be Statistically Significant.
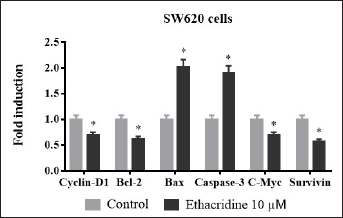
Effect of Ethacridine on the Expression of Apoptotic Markers
Cyclin D-1, Bcl-2, Bax, caspase-3, C-Myc, and survivin are recognized as the key regulators of apoptosis. Levels of these markers in the lysates of SW620 cells were determined through real-time PCR after exposure to 10 µM of ethacridine. Cyclin-D1, Bcl-2, C-myc, and survivin are the factors that inhibit apoptosis in cells (Figure 5). Exposure to ethacridine has significantly downregulated cyclin-D1, Bcl-2, C-myc, and survivin compared to control resulting in induction of apoptosis. However, levels of Bax and caspase-3, the coordinators in apoptosis are up-regulated by 10 µM of ethacridine. The expression of these markers significantly ascertains the anticancer effects of ethacridine and its sustainable role in cancer treatment (Figure 5).
Expression of cyclin-D1, Bcl-2, Bax, caspase-3, C-Myc, and survivin in terms of fold change in 10 µM ethacridine treated SW620 cells. Values *p < 0.05 are deliberated to be significant statistically.
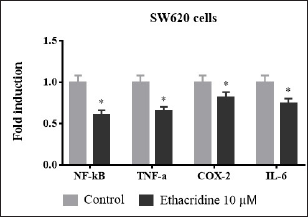
Effect of Ethacridine on Inflammatory Cytokines
Intensities of pro-inflammatory cytokines namely NF-κB, TNF-α, IL-6, and COX-2 are constantly elevated in all inflammatory diseases such as cancer (Figure 6). In our study, these levels were assessed in ethacridine-treated SW620 cell lines using qPCR. Treatment with 10 µM of ethacridine down-regulated the intensities of inflammatory cytokines when compared to control in both cell lines thereby in further compliance with the anti-inflammatory properties of ethacridine.
Fold inductionof inflammatory cytokines NF-κB, TNF-α, IL-6 and COX-2 in 10 µM ethacridine treated SW620 cells. Values *p < 0.05 are deliberated to be significant statistically.
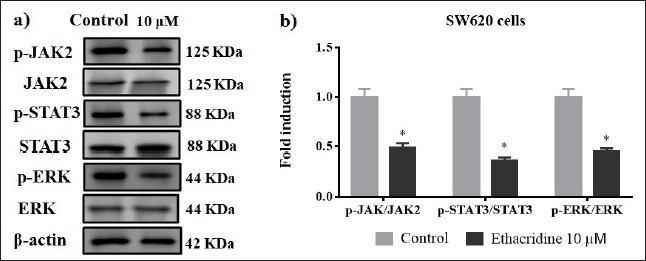
Impact of Ethacridine on JAK2/STAT3/ERK Signalling
Signal transducer and activator of transcription (STAT), extracellular signal-regulated kinase (ERK) and janus kinase (JAK), and their phosphorylated forms are over-expressed in colon cancer cell lines (Figures 7a and 7b). JAK2/STAT3/ERK is involved in tumor formation, cell adhesion, cell proliferation, and metastasis. 11 Phosphorylation of these proteins might activate downstream factors that are involved in the regulation of cell proliferation with enhanced clonogenic ability. Western blot results of JAK2/STAT3/ERK in 10 µM of ethacridine-treated SW620 cells revealed that these targets were significantly downregulated when compared with control cells.
(a) Western Blotting Images of JAK2, STAT3, and ERK in 10 µM Ethacridine-treated SW620 Cells. (b) *p < 0.05 Are Considered to be Statistically Significant.
Discussion
Apoptosis induction is important in controlling the spread of cancer cells. 14 Chemotherapeutic agents should target the factors that induce apoptosis and finding out these therapeutic agents is vital. Genetic mutations are one of the key factors of colorectal cancer. Mutations of oncogenes are genes related to DNA repair and tumor suppressor genes. Based on the mutational origin, colorectal cancers can be divided into familial, ancestral, and periodic. 15 The chance of people getting colorectal cancer is merely 4% to 5% mainly because of personal behavior and lifestyle. Another major risk factor is age as people younger than 50 years are less susceptible to CRC unless it is inherited. 16 JAK group of proteins fall under non-receptor protein tyrosine kinases that modulate several signaling pathways related to cytokine receptors such as leptin, erythropoietin, interferon-γ, and interleukin-6 receptors (IL-6) by activating phosphorylated STAT molecule.17–19 Several studies have shown that JAK2/STAT3 pathway is activated in different cancers by upregulating the synthesis of IL-6. JAK2 also acts as a key mediator of metastasis in cancer and targeting this molecule is eminent in cancer therapy. 13
Ethacridine is an antiseptic that in several studies proved to be a potent chemotherapeutic agent that inhibits phosphorylation of factors that initiate cell proliferation and metastasis.20, 21 In cancer-related studies on ethacridine, the compound exhibited inhibition of apoptosis and cell proliferation in cancer cell lines.22–24 In thyroid cancer, ethacridine proved to induce apoptotic changes with no prevailing changes in the cell-cycle distribution. 11 Based on these findings, our study evaluated the efficacy of ethacridine in targeting various factors of cell signaling pathways.
In our study, ethacridine showed antiproliferative effects by decreasing cell propagation in a dosage-reliant mode (2.5–20 µM/mL) for 24 hr. IC50 value was found to be 10 µM. Intracellular ionic imbalance and increased ROS generation affect mitochondrial permeability thereby stimulating cell apoptosis. 25 Assessing MMP is important in assessing mitochondrial dysfunction, 26 Rh-123 staining was used to measure MMP levels in SW620 cells. Ethacridine induced a notable decrease in MMP levels, increasing mitochondrial membrane permeability thereby inducing mitochondrial dysfunction.
ROS plays a major role in chemoprevention, hence it should be one of the features to be considered for a therapeutic agent. Downregulated GSH level followed by an increase in free radicals promotes cellular apoptosis.27, 28 In our study, we determined the ROS levels using DCFH-DA staining to assess the oxidative stress intensity in SW620 cells after treating the cells with ethacridine (10 µM). The observed results established that ethacridine can up-regulate ROS synthesis in both cell lines, thereby promoting apoptosis in cells.
DAPI and PI staining assays provide promising results in differentiating nuclear fragmentation and condensed nuclei between apoptotic and necrotic cells. Apoptotic induction is experimentally proven in several studies by mediating caspase enzymes and other factors. 29 In our study, we have proved that treatment with ethacridine induced apoptosis by showing distinct nuclear morphology in both PI and DAPI staining assays.
Cyclin-D1 enhances cell progression by binding to the cycle-dependent kinases and other factors. 30 Pro- and antiapoptotic Bcl-2 markers regulate mitochondria-based apoptotic pathways. 31 Bax and Bcl-2 ratios help in determining cell survival. C-Myc and survivin are the key factors inhibiting apoptosis. Hence, C-Myc, survivin, and caspase-3 expressions are inversely proportional to each other. 32 In our study, we observed the expressions of these apoptotic markers, cyclin-D1, Bcl-2, Bax, caspase-3, C-Myc, and survivin which showed that apoptotic inhibitors, cyclin-D1, Bcl-2, C-Myc, and survivin expression were down-regulated, whereas apoptotic enhancers, Bax, and caspase-3 expressions were augmented by ethacridine (10 µM). The levels of pro-inflammatory cytokines of NF-κB, TNF-α, IL-6, and COX-2 were also decreased after ethacridine treatment. A gene profiling study has revealed that the expression of JAK2 and STAT3 pathways have been activated in colorectal cancer. 33 ERK is also important in cell differentiation, cell growth, and apoptosis. 34 Ethacridine showed prominent results by inhibiting JAK, STAT, and ERK in our study.
Conclusion
Overall results conclude that ethacridine have inhibition of cell viability, induces ROS synthesis mitochondrial depolarization, and regulation of cell proliferation, and induces apoptotic. Taken together, this study proved that ethacridine act as a chemotherapeutic agent by suppressing various apoptotic markers, cell signaling molecules, pro-inflammatory cytokines, and also regulate JAK2, STAT3, and ERK signaling pathways in CRC cell lines.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by Hanzhong Central Hospital (2021–723).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Summary
Ethacridine suppressed cell proliferation of colon cancer cells. Ethacridine inhibits JAK, STAT, and ERK signaling on colon cancer SW620 cells.
