Abstract
Background
Contemporary cancer care primarily focuses on advanced biomedical treatments, often overlooking the psychological and social challenges associated with the illness (psychosocial factors). This oversight can undermine the efficacy of healthcare and subsequently impact the overall well-being of cancer patients. There is a widespread consensus among medical professionals that psychological factors play a crucial role in the care and treatment of cancer patients.
Purpose
The main aim of this study was to examine the effects of treatment as usual (TAU) and a combined intervention (CI) of TAU and psychological intervention (PI) on stress, coping strategies and quality of life among adult cancer patients.
Methods
The present study employed a pretest–post-test control group design comprising two groups—Group I (a control group involving TAU only) and Group II (an experimental group involving TAU and PI). In Group I (n1 = 30), the patients with cancer were randomly selected from a pool of 105 cancer patients from three cancer types, lung cancer (LC), breast cancer (BC) and head/neck cancer (HNC), and were administered only TAU (e.g., surgery, chemotherapy and radiation‑hormonal therapy). In Group II (n2 = 30), the participants were administered not only the regular TAU but also the PI involving psychoeducation, cognitive-behavioural therapy (CBT) and guided imagery. Three research instruments, namely QSC-R23, Brief Cope Inventory and EORTC QLQ-C30 version 3.0, were used for the present study.
Results
The study findings revealed that both TAU and CI resulted in positive outcomes across the measured variables. Notably, there was a decrease in stress levels and an improvement in coping strategies, encompassing problem-focused, emotion-focused and adaptive coping. Furthermore, participants showed a decreased reliance on maladaptive coping strategies and reported an enhanced quality of life, as evidenced by improvements in symptom scales.
Conclusion
The study indicated that combining PI with TAU resulted in better outcomes, highlighting the effectiveness of CI (TAU with PI) for cancer patients compared to TAU alone.
Keywords
Introduction
Contemporary cancer care primarily focuses on advanced biomedical treatments, often overlooking the psychological and social challenges associated with the illness (psychosocial factors). This oversight can undermine the efficacy of healthcare and subsequently impact the overall well-being of cancer patients. There is a widespread consensus among medical professionals that psychological factors play a crucial role in the care and treatment of cancer patients. Cancer diagnosis and treatment often lead to psychological distress. Research shows that individuals with lower distress levels have better treatment adherence and cope more effectively.1–3 To address these psychosocial effects, interventions tailored to cancer patients have been developed, including supportive-expressive group therapy4, 5 and psychoeducational interventions.6, 7 Cognitive-behavioural therapies such as progressive muscle relaxation, hypnosis and guided imagery have also proven effective in managing chemotherapy side effects and anticipatory nausea/vomiting. 8 Our study aims to develop a psychological intervention (psychoeducation, relaxation [guided imagery] and cognitive-behavioural therapy) that complements medical treatment and provides support to patients with cancer. Building upon previous studies that have effectively utilised complementary psychological interventions to enhance the well-being of cancer patients, our study aims to use psychoeducation, cognitive-behavioural therapy and relaxation therapy (guided imagery). The primary objective of this study was to examine the effects of treatment as usual (TAU) and a combined intervention (CI) of TAU and psychological intervention (PI) on stress, coping strategies and quality of life among adult cancer patients.
Methods
Participants
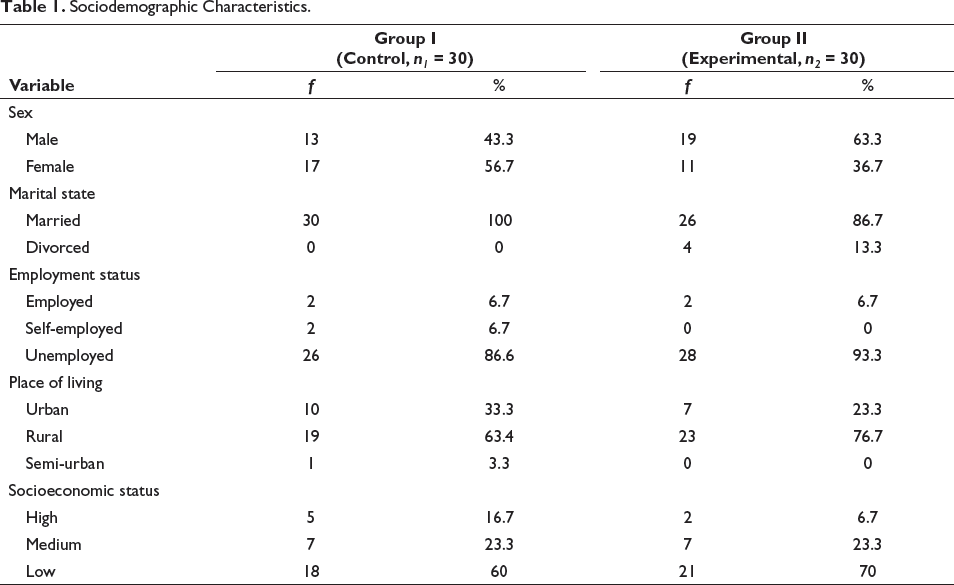
The present study applied a pretest–post-test control group design comprising two groups—Group I (a control group involving TAU only) and Group II (an experimental group involving CI: TAU and PI). Group I (control group) comprised 30 cancer patients (13 men and 17 women) randomly selected from a pool of 105 cancer patients from three cancer types (10 patients in each type), lung cancer (LC), breast cancer (BC) and head and neck cancer (HNC), who were administered TAU. Group II (experimental group) consisted of 30 cancer patients (19 men and 11 women) from three cancer types (LC, BC and HNC; 10 patients in each type). They were selected through convenient sampling from different cancer hospitals in Hyderabad, India, and administered CI. The age of participants ranged from 27 to 65 years. Inclusion criteria for recruitment included being newly diagnosed with LC, BC and HNC up to stage IV, having a score below IV on the Eastern Cooperative Oncology Group (ECOG) 9 Performance Status scale and no history of psychiatric illness (confirmed through a self-report soft sign test). Participants were included regardless of their occupation, socioeconomic status or place of residence. The exclusion criteria were patients under the age of 27 or over the age of 65, those with psychiatric illnesses, individuals receiving palliative care, those who had previous exposure to psychological therapies or interventions and those with severe physical comorbidities. The treatment response was evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST) in accordance with the guidelines provided by the World Health Organization (WHO). 10
Research Tools
Sociodemographic Data Sheet
Essential information was collected from the patients comprising the sociodemographic details of the patients with cancer that include age, gender, marital status, occupation, place of living, socioeconomic status and residential address.
Questionnaire on Stress in Cancer Patients-Revised Version (QSC-R23) 11 was used to assess psychological stress in cancer patients. The QSC-R23 consisted of 23 items with five domains—psychosomatic complaints, fears, information deficits, everyday life restrictions and social strains. Responses were rated on a 6-point scale (0 = the problem does not apply to me, 5 = the problem applies to me and is very big problem). The scores ranged between 0 and 115, where a higher score represented a higher stress level. The Cronbach’s alpha of the scale was 0.89.
Brief Cope Inventory 12 was used to measure effective and ineffective ways to cope with stressful events after disease diagnosis. It consisted of 14 scales; each analysed the degree to which the participant used a specific coping strategy. The scale was grouped into four domains—problem focused, emotion focused, adaptive and maladaptive coping strategies. Patients rate their answers on a 4-point Likert scale (1 = I haven’t been doing this at all, 4 = I’ve been doing this a lot). The inventory possessed adequate reliability.
The European Organization for the Cancer QoL Questionnaire version 3.0 (EORTC QLQ-C30 version 3.0) 13 was used to measure the patient’s health-related quality of life (QoL). The EORTC QLQ-C30 consisted of five functional scales (physical, role, emotional, cognitive and social), three symptoms scales (fatigue, nausea/vomiting and pain), a global health status and QoL (GHS/QoL) scale and a few single items measuring additional symptoms (dyspnoea, insomnia, appetite loss, constipation and diarrhoea) and perceived financial impact. Each item was measured on a 4-point Likert scale (1 = not at all, 4 = very much), except for the two GHS/QoL items, which were rated on a 7-point Likert scale (1 = very poor, 7 = excellent). While a higher score referred to better functioning in the functional and GHS/QoL scales, a higher score for the symptom scales and items implied the presence of higher levels of symptoms.
Procedure
Approval was obtained from the Institutional Ethics Committee at the University of Hyderabad, with Review Letter No. UH/IEC/GP/11/8. Written informed consent was obtained from each participant of the study. The participants were briefed about the study and recruited after they consented to participate. Participants were also given full freedom to withdraw at any point in time if they were not comfortable with the entire study. The study was done on two groups of patients—Group I and II. In Group I (control group, n1 = 30), patients with cancer from three cancer types (BC, LC and HNC) were administered only TAU (e.g., surgery, chemotherapy and radiation‑hormonal therapy). In Group II (experimental group, n2 = 30), the participants were administered not only the regular TAU but also the PI, which included psychoeducation, cognitive-behavioural therapy and guided imagery (see Figure 1). When TAU and PI were used together, it was called CI. The study involved administering the intervention to a total of 30 patients, with 10 patients each from LC, BC and HNC, over six weeks. Prior to their participation, each participant was informed about the study and recruited after providing their consent. Research tools were administered to all participants under the supervision of a senior medical oncologist and a clinical psychologist in both groups. Each participant underwent pre- and post-assessment with the administration of intervention along with the standard TAU. Following the completion of the study, all participants were debriefed and thanked. The responses of patients to the CI were measured and analysed using descriptive statistics and one-way ANCOVA to examine any variations in treatment response based on the intervention’s variation. Statistical analysis was performed using SPSS 16.0.
Flow Chart of Psychological Intervention.
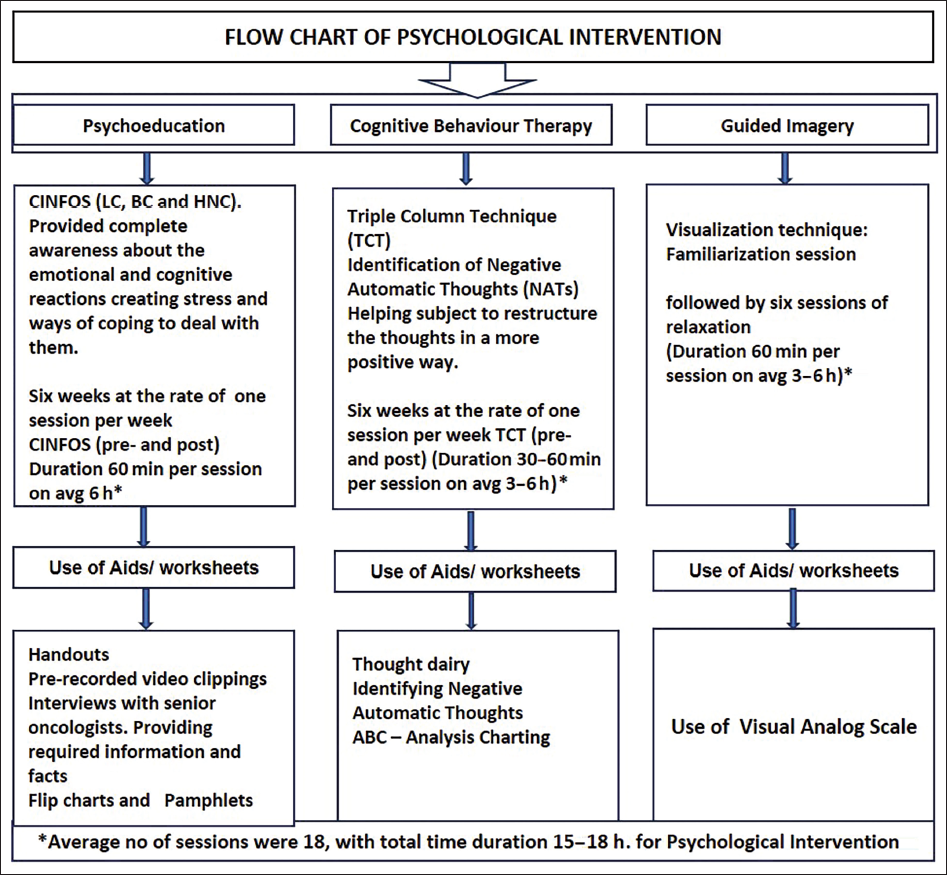
Results
The sociodemographic details of both control and experimental groups are presented in Table 1. Further, both groups did not differ significantly in terms of age; t(58) = 0.18, p = .86. This indicated that both control (M = 52.10, SD = 10.84) and experimental (M = 51.60, SD = 10.83) groups were matched on age.
Sociodemographic Characteristics.
Comparison Between the Post-intervention Assessment of Scores of Stress, Coping and GHS/QoL on Treatments as Usual (Group I) and Combined Intervention (Group II)
The obtained quantitative data were analysed using one-way ANCOVA on post-test scores of stress, coping strategies and quality of life of patients who were administered only TAU and CI. The impact of CI among cancer patients is observed in terms of treatment response (TR). Following the assumptions, we performed one-Way ANCOVA on each of the dependent variables, and the results are presented in Tables 2, 3 and 4.
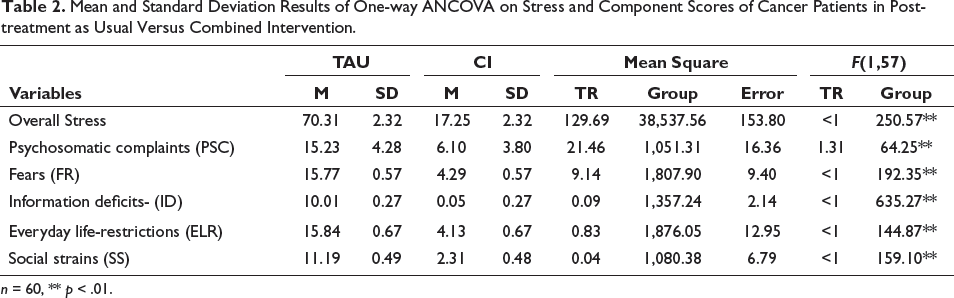
Mean and Standard Deviation Results of One-way ANCOVA on Stress and Component Scores of Cancer Patients in Post-treatment as Usual Versus Combined Intervention.
n = 60, ** p < .01.
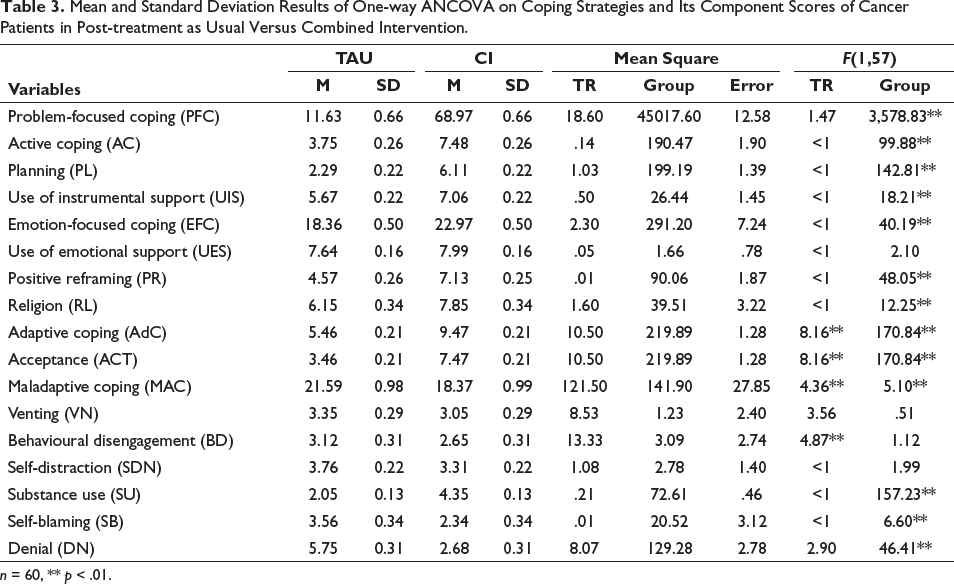
Mean and Standard Deviation Results of One-way ANCOVA on Coping Strategies and Its Component Scores of Cancer Patients in Post-treatment as Usual Versus Combined Intervention.
n = 60, ** p < .01.
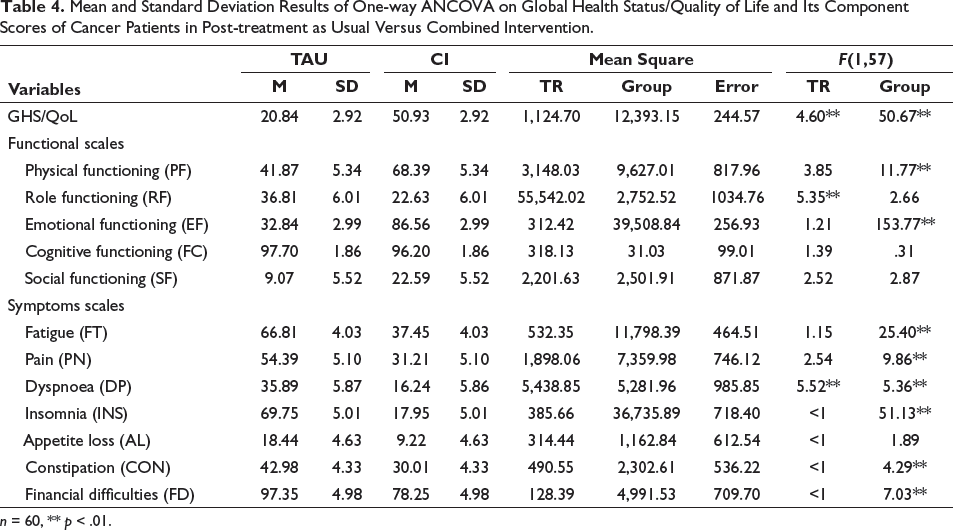
Mean and Standard Deviation Results of One-way ANCOVA on Global Health Status/Quality of Life and Its Component Scores of Cancer Patients in Post-treatment as Usual Versus Combined Intervention.
n = 60, ** p < .01.
Stress of Cancer Patients in Post-intervention Treatment as Usual and Combined Intervention
Table 2 shows one-way ANCOVA conducted on the post-test scores of interventions for overall stress. The post-test scores suggest that a reduction in scores corresponds to a reduction in symptoms, which was observed in CI. On overall stress, F(1,57) = 250.57, p < .01was significantly better in CI (M = 17.25, SD = 2.32) compared to only TAU (M = 70.31, SD = 2.32), and TR was not found to be a covariate in this analysis. When considering the stress domains: PSC, F(1,57) = 64.25, p < .01was significantly lower in CI (M = 4.28, SD = 3.80) compared to TAU (M = 15.23, SD = 4.28). The subdomain of FR, F(1,57) = 192.35, p < .01was significantly lower in CI (M = 4.28, SD = 0.57) compared to only TAU (M = 15.77, SD = 0.57). On ID, F(1,57) = 635.27, p < .01was significantly lower in CI (M = 0.05, SD = 0.27) compared to only TAU (M = 10.01, SD = 0.27).
On ELR, F(1,57) = 144.87, p < .01, which is significantly lower in CI (M = 4.13, SD = 0.67) compared to only TAU (M = 15.84, SD = 0.67). On SS, F(1,57) = 159.10, p < .01, which is significantly lower in CI (M = 2.31, SD = 0.49) compared to TAU (M = 11.19, SD = 0.49). The results also revealed that in all domains of stress, TR was not found to be a covariate.
Coping Strategies of Cancer Patients in Post-treatment as Usual and Combined Intervention
Table 3 shows one-way ANCOVA analysis was done on post-test scores of coping strategies and their subdomains. On PFC, F(1,57) = 3,578.82, p < .01, which is significantly better in CI (M = 68.97, SD = 0.66) compared to only TAU (M = 11.62, SD = 0.66). Regarding PFC, the subdomain of AC, F(1,57) = 99.89, p < .01, which is significantly better in CI (M = 7.48, SD = 0.26) compared to TAU (M = 3.75, SD = 0.26). On PL, F(1,57) = 142.81, p < .01, which is significantly better in MI/PI (M = 6.11, SD = 0.22) compared to TAU (M = 2.29, SD = 0.22). On the UIS, F(1,57) = 18.21, p < .01, which is significantly better in CI (M = 7.06, SD = 0.22) compared to TAU (M = 5.67, SD = 0.22). The results also revealed that in all domains of PFC, TR was not found to be a covariate.
On EFC, F(1,57) = 40.19, p < .01, which is significantly better in CI (M = 22.97, SD = 0.50) compared to TAU (M = 18.36, SD = 0.50). On the EFC subdomain of PR, F(1,57) = 48.05, p < .01, which is significantly better in CI (M = 7.13, SD = 0.26) compared to TAU (M = 4.56, SD = 0.26). On RL, F(1,57) = 12.25, p < .01, which is significantly better in CI (M = 7.85, SD = 0.34) compared to only TAU (M = 6.15, SD = 0.34). Further, the results also indicated that in the domains of PR and RL within the EFC, TR was not found to be a covariate. On AdC, F(1,57) = 170.84, p < .01, which is significantly better in CI (M = 9.47, SD = 0.21) compared to TAU (M = 5.46, SD = 0.21). The results also revealed that the TR (covariate) was significantly related to the ACF, (1,57) = 8.16, p <.01. On ACT, F(1,57) = 170.84, p < .01, which is significantly better in CI (M = 7.47, SD = 0.21) compared to TAU (M = 3.46, SD = 0.21). The results also revealed that the TR (covariate) was significantly related to the ACT, F(1,57) = 8.16, p < .01. On post-tests scores of the MAC, F(1,57) = 5.10, p < .01, which is significantly reduced in CI (M = 18.37, SD = .99) compared to TAU (M = 21.59, SD = 0.99). The results also revealed that the treatment response (covariate) was significantly related to the MAC, F(1,57) = 4.36, p < .01. On the MAC subdomain of SU, F(1,57) = 157.23, p < .01, which is significantly increased in CI (M = 4.35, SD = 0.13) compared to TAU (M = 2.05, SD = 0.13). Sample being small, the effect of even very few people with SU had an overall impact on the estimated mean score. On SB, F(1,57) = 6.60, p < .01, which is significantly lower in CI (M = 2.34, SD = 0.33) compared to TAU (M = 3.56, SD = 0.33). On DL, F(1,57) =46.41, p < .01, which is significantly lower in CI (M = 2.68, SD = 0.31) compared to TAU (M = 5.75, SD = 0.31). Additionally, the findings indicated that for other domains of MAC, SU, and SB, TR was not found to be a covariate.
Global Health Status/Quality of Life in Post-intervention Treatment as Usual and Combined Intervention
Table 4 displays the post-test scores of the GHS/QoL results. After controlling for treatment rating scores, a significant effect of the between-subjects factor was observed (F(1,57) = 50.67, p < .01). Adjusted mean GHS/QoL scores indicate that the GHS/QoL was significantly higher in the CI group (M = 50.93, SD = 2.92) compared to the TAU (M = 20.84, SD = 2.92). The results also revealed that the TR (covariate) was significantly related to the GHS/QoL, F(1,57) = 4.60, p < .01, this implies that an improvement in scores also signifies an enhancement in QoL, which was observed after the CI. With reference to the functional scales of GHS/QoL on post-test scores of the PF, F(1,57) = 11.77, p < .01, which is significantly better in CI (M = 68.39, SD = 5.34) compared to TAU (M = 41.87, SD = 5.34). On RF, F(1,57) = 5.35, p < .01, which is significantly affected in CI (M = 22.63, SD = 6.01) compared to TAU (M = 36.81, SD = 6.01). On EF, F(1,57) = 153.77, p < .01, which is significantly better in CI (M = 86.56, SD = 2.99) compared to TAU (M = 32.84, SD = 2.99). The results also revealed that in subdomains of GHS/QoL with reference to PF, RF and EF, the TR was not found to be a covariate. On symptom scale of fatigue (FT), F(1,57) = 25.40, p < .01, which is significantly lower in CI (M = 37.45, SD = 4.03) compared to TAU (M = 66.81, SD = 4.03). On PN, F(1,57) = 9.86, p < .01, which is significantly better in CI (M = 31.21, SD = 5.10) compared to TAU (M = 54.39, SD = 5.10). On dyspnoea (DP), F(1,57) = 5.36, p < .01, which is significantly lower in TAU (M = 16.24, SD = 5.87) compared to CI (M = 35.84, SD = 5. 87).
The results also revealed that the TR was significantly related to the DP, F(1, 57) = 5.52, p < .01, which indicates that when the scores are reduced, symptoms also reduced, which was seen after CI. On INS, F(1,57) = 51.13, p < .01, which is significantly better in CI (M = 17.95, SD = 5.01) compared to TAU (M = 69.75, SD = 5.10). On CON, F(1,57) = 4.29, p < .01, which is significantly better in C1 (M = 30.01, SD = 4.33) compared to TAU (M = 42.98, SD = 4.33). On FD, F(1,57) = 7.03, p < .01, which is significantly more in TAU (M = 97.35, SD = 4.98) compared to CI (M = 78.25, SD = 4.98). The results also revealed that in subdomains of GHS/QoL symptom domains with reference to FT, PN, INS, CON and FD, the TR was not found to be a covariate.
Discussion
A one-way ANCOVA was conducted to compare post-test scores between TAU and CI. The goal was to determine if there were differences and which set of post-test scores performed better.
Stress of Cancer Patients in Post-intervention Treatment as Usual and Combined Intervention
After instigating CIs, overall stress scores in the post-test decreased compared to those in the post-test following only TAU. This decrease in mean overall stress scores with CIs aligns with findings from other studies.14–16, Similarly, as indicated in Table 2, there was a significant difference in stress score domains such as psychosomatic and fear17, 18, information deficits, everyday life restrictions and social support. 19 The decrease in average stress scores within these domains resulting from CIs, compared to TAU, clearly indicates a stress reduction. Treatment was not found to be a covariate on overall stress scores and other domains of stress. The CI did have a modest yet strong impact after the intervention. However, there is a lack of research on preventive interventions, and the existing studies have not been adequately replicated. 20
Coping Strategy Scores of Cancer Patients in Post-intervention Treatment as Usual and Combined Intervention
During CIs, post-test scores of coping strategies, specifically problem-focused, emotion-focused and active coping strategies, increased based on the estimated mean values. In contrast, the overall scores on maladaptive coping decreased during CIs compared to TAU. Additionally, post-test scores on certain subdomains of these coping strategies also increased after CIs compared to TAU. Notably, there was a significant increase in problem-focused domains, including active coping, planning, and the utilization of instrumental support. 21 However, TR was not found to be a covariate for these problem-focused subdomains. Similarly, in emotion-focused domains, such as positive reframing and religion.
CI also showed significant improvement in adaptive coping and acceptance, as indicated by higher estimated mean values compared to TAU. The treatment response was found to be a covariate in adaptive coping and acceptance scores. The items related to accepting the reality of the illness and living with it were included in the acceptance domain under adaptive coping. Both TAU and CI seemed to contribute to the participants’ acceptance, with TAU playing a crucial role in facilitating realistic acceptance over time, which likely influenced their overall clinical status. Although acceptance increased during CI, the role of TAU and its impact on accepting reality appeared to be significant. 22, 23 Therefore, treatment is considered a covariate in adaptive coping and the acceptance domain.
Further, there was a significant decrease in estimated mean values of maladaptive coping strategies and their domains, self-blaming and denial compared to TAU. This indicates that participants relied less on maladaptive coping strategies such as self-blaming and denial during CI. However, the TR was not found to be a covariate on the usage of these specific coping strategies.
Global Health Status/Quality of Life and Its Component Scores of Cancer Patients in Post-intervention Treatment as Usual and Combined Intervention
The estimated mean values of quality of life significantly improved in CI compared to TAU, indicating an overall improvement in quality of life. This improvement was particularly observed in the domains of functional scales, specifically physical function and emotional function. The treatment response was not found to be a covariate for overall scores of QoL (see Table 4), except for role functioning. These findings align with a study conducted by Chung, 24 which also reported similar improvements in physical function and emotional function. On the domain of role function, the TR response was a covariate; this could be due to the statements in this particular domain that highlight limitations in work, daily activities and the ability to pursue hobbies and leisure activities.
Both TAU and CI may have limitations in role functions, as the sample consisted of newly diagnosed cancer patients undergoing various treatments. The treatment regimen and the adjustments patients have to make regarding their health condition, daily activities and work-related tasks are primarily focused on TAU. Therefore, the psychological intervention may not have significantly contributed to the improvement in post-test scores of the combined intervention compared to TAU. In other domains of the GHS/QoL symptom scales, there were significant differences in fatigue, pain, dyspnoea, insomnia and constipation between the post-test scores of CI.
The estimated mean values of all these domains demonstrate a decrease in symptom scales in the post-test scores of combined interventions, indicating an improvement in quality of life.20, 25 Concerning dyspnoea (shortness of breath), the treatment response was found to be a covariate. Despite the improvement in post-intervention scores in CI, suggesting a reduction in dyspnoea (as indicated by the decrease in estimated mean scores), it is important to note that dyspnoea is a symptom that requires medical attention. This could explain why the treatment response was a covariate in this particular domain. For all other domains of symptom scales, the treatment response did not have a significant impact.
Though the findings need to be interpreted with caution, this study has several strengths. The results of the study draw our attention to the importance of psychological intervention in the treatment procedure of cancer patients. The findings establish the effectiveness of CI (medical treatment and psychological intervention) in positively addressing the stress, coping and quality of life of three types of cancer patients—LC, BC and HNC. The study stands as a guide for developing holistic psychoeducational, relaxation and cognitive-behavioural therapy intervention packages and tailor-made programmes for patients with specific types of cancer.
However, one of the limitations of this study was its inclusion of only three types of cancers with participants of a wide age range. The future study should include more homogenous participants with more types of cancers. Further, the study did not evaluate the longer-term impact of CI.
Conclusion
The study findings indicate that both TAU and CIs were effective in improving the measured variables, including reduced stress, enhanced coping strategies and improved quality of life. When comparing all the components and subcomponents of stress, coping strategies and quality of life, five components showed the treatment response as a significant covariate. These components are adaptive coping, acceptance, maladaptive coping, GHS/QoL and dyspnoea. Furthermore, the results demonstrate significant improvements in almost all domains, except for eight components such as the use of emotional support, venting, behavioural disengagement, self-distraction, cognitive functioning, social functioning, nausea and vomiting, and diarrhoea. This highlights the effectiveness of CIs, where psychological intervention alongside TAU yields better outcomes. This signifies the effectiveness of TAU and psychological intervention for patients with cancer when compared to the administration of only TAU.
Footnotes
Abbreviations
TAU Treatment-as-usual
PI Psychological intervention
CI Combined intervention
CBT Cognitive-behavioural therapy
ANCOVA Analysis of co-variance
M Mean
SD Standard deviation
TR Treatment response
PSC Psychosomatic complaints
FR Fear
ID Information deficits
ELR Everyday life restrictions
SS Social strain
PFC Problem-focused coping
AC Active coping
UIS Use of instrument support
EFC Emotion-focused coping
UES Use of emotional support
PR Positive reframing
RL Religious
AdC Adaptive coping
ACT Acceptance
MAC Maladaptive coping
VN Venting
BD Behavioural disengagement
SDN Self-distraction
SU Substance use
SB Self-blaming
DN Denial
GHS/QoL General health status/quality of life
PF Physical functioning
RF Role functioning
EF Emotional functioning
CF Cognitive functioning
SF Social functioning
FT Fatigue
PN Pain
DP Dyspnoea
INS Insomnia
AL Appetite loss
CON Constipation
FD Financial difficulties
Acknowledgement
The authors are grateful to the hospitals, doctors and patients who have participated in this study and contributed to the findings. The caregivers of the patients need special thanks for being supportive during entire our work.
Authors’ Contribution
VPB contributed to the study concept, performed the evaluation, intervention and data acquisition, and contributed to writing the article. PG contributed to the study design, data interpretation, discussion and critical write-up of the manuscript. SR contributed to data analysis and data interpretation. RKS contributed to allocating patients for study in the hospital setting and study design. SS contributed write-up, review and interpretation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Written informed consent was obtained from all participants of the study. Participants were briefed about the study and recruited after they consented to participate.
Statement of Ethics
The study protocol was approved by the University of Hyderabad, Institutional Ethics Committee (vide Review letter No. UH/IEC/GP/11/8 dated 07.01.2012.) All methods were carried out in accordance with ICMR and the World Medical Association Declaration of Helsinki relevant guidelines and regulations.
