Abstract
Background
Antiepileptic drugs (AEDs), the predominant treatment for epilepsy, are also utilised for migraine, neuropathic pain, and bipolar disorders, accounting for 1% of usage among the common population. There is a greater risk of stroke, heart attack, and arrhythmia among AED users particularly those with enzyme-inducing properties.
Summary
This systematic review aimed to look into the ischemic stroke risk among AED users, particularly in patients who had never had a cerebrovascular accident before. English-language literature was searched in the databases of PubMed, EMBASE, and Google Scholar between 2000 and 2021. Studies should report the association between AEDs and the ischemic stroke risk. The outcomes for assessing efficacy and safety were: event ratio, hazard ratio, odds ratio, and risk ratio. Seven of the eight studies showed an increased risk of ischemic stroke associated with AEDs among individuals without a history of cardiovascular accidents Phenytoin, sodium valproate, oxcarbamazepine, levetiracetam, phenobarbital, and carbamazepine were all linked to an increased risk of stroke. Adequate data on the association between the duration of AED exposure and stroke was missing.
Key message
Not all AEDs are enzyme inducers. We conclude significant stroke risk with AEDs (mainly enzyme inducers), and dose dependency need to be assessed. The risks and benefits of AEDs should be weighed among the patients, especially with vascular risk factors to limit the risk of ischemic stroke.
Introduction
Stroke ranks second in global mortality and third in disability. 68% of strokes are ischemic, while 32% are haemorrhagic. 1 Stroke, which accounts for 10% of all epilepsies in adults and the elderly, is the leading cause. 2 Vascular risk factors are very common in patients with stroke and epilepsy.3, 4 Antiepileptic drugs (AEDs), the predominant treatment for epilepsy, are also utilized for migraine, neuropathic pain, and bipolar disorders, accounting for 1% of usage among the general population. 5 With the growing use of AEDs comes an increased risk of stroke, heart attack, and arrhythmia.6–8 In particular, the enzyme-inducing property of some AEDs is associated with accelerated atherosclerosis, which increases the risk of stroke. 9 This systematic review aimed to look into the ischemic stroke risk among AED users, particularly in patients who had never had a cerebrovascular accident before and know about risk among enzyme inducers, dose-dependent effects along with exposure duration.
Methods
This systematic review follows the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) checklist and flow diagram for manuscript format development and is reported in accordance with the PRISMA Declaration 2020. 10 Our research looked into whether the first-time use of AEDs increased the risk of ischemic stroke in epilepsy patients who had no history of cerebrovascular disease. Second, we studied the stroke risk among multiple AEDs (enzyme inducers or not) along with their dose-dependent effect and exposure duration We designed the eligibility criteria for our study which are as follows:
Study type(s): Studies that were either prospective or retrospective and published in English were eligible to be included in this review. Study subjects(s): Subjects diagnosed with any neurological disease-causing seizures/epilepsy episodes taking AEDs of any age and gender. Study treatment(s): Subjects taking anti-epileptic drugs prescribed for seizures/epilepsy at least once in their medical history, allowing for studies to measure ever, past, current, and/or cumulative use of AEDs were used in our study. Objective outcome(s): Studies should report the link between AEDs and ischemic stroke risk. The outcomes for assessing efficacy and safety were: event ratio, hazard ratio, odds ratio, and risk ratio.
Search Methods and Study Selection
English-language literature was searched in the databases of PubMed, EMBASE, and Google Scholar between 2000 and 2021. Boolean search operators ‘AND’ and ‘OR’ were used to link search terms and Boolean logic was used for conducting a database search: (‘Cerebrovascular Disorders’[MeSH Terms] OR ‘Acute Ischemic stroke’[All Fields] OR ‘stroke’[All Fields]) AND (‘Anti-epileptic drugs’[All Fields] OR ‘Anti-seizure’[All Fields] OR ‘AED’[All Fields]) AND (‘humans’[MeSH Terms] AND ‘English’[Language])) AND ((humans[Filter]) AND (english[Filter]) AND (2000:2022[pdat])). The detailed search approach is provided in the supplementary file. We searched the reference lists of each included article for additional studies. Following that, a manual check was performed to remove any remaining duplicates. Papers were initially reviewed independently by two reviewers (SK and KB) based on title, keywords, and abstract, and then confirmed by a third reviewer (RB). Articles that passed the initial screening were then read in full by two reviewers (SK and KB). By consulting with a third reviewer (RB), we were able to reach an agreement on discrepancies in the final research selection between the two primary reviewers (SK and KB).
Data Extraction
Two different authors (BPG and SK) conducted a thorough analysis of selected studies for a systematic review that matched our inclusion criteria and extracted detailed data on headings: Author, publication year Period of study, Study design, Location of study, Population included details, Participants, mean age (years), sex (male/female), AEDs used, End assessment outcomes and Confounders were recorded in Microsoft Excel 2013 (Microsoft Corp, Redmond, USA). The corresponding authors of the relevant studies were contacted via email for clarification if the necessary data were absent, not published in the article, or reported in an unusual form. In such circumstances, supplementary material to the main publication was also investigated.
Results
Literature Search and Data Extraction
We retrieved 493 studies through electronic database searches and four additional studies through manual searches of reference lists and relevant systematic reviews. After removing duplicates, we evaluated 170 articles based on their titles and abstracts. Following screening, 57 full-text publications were obtained and evaluated against the established inclusion criteria, leaving only eight articles eligible for inclusion in the review. Figure 1 depicts the PRISMA diagram illustrating the identification and selection procedure.
The PRISMA Diagram Illustrating the Identification and Selection Procedure. 10
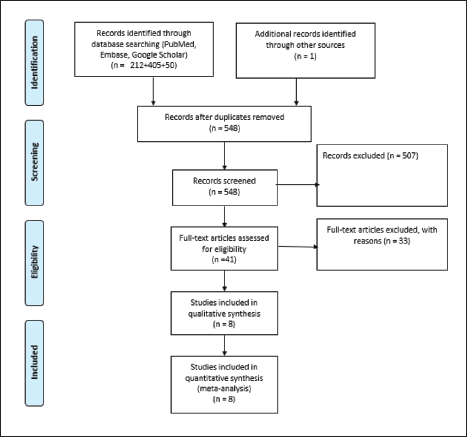
Study Characteristics
Study Design, Publication Year, and Study Site
Eight studies published from 2011 to 2014 were included in our review.7, 9, 11–16 Five were cohort studies7, 9, 11, 15, 16 two were nested case-control studies12, 13 and only one was a case-control study. 14 Two studies each were from the UK12, 13 and Taiwan7, 9 while single studies were from Denmark, 11 Spain, 14 the USA 15 and Finland. 16 The participants’ number, the mean age of diagnosis, and the Male/Female number are described in Table 1.
The Details of Patients and Studies in our Systematic Review.
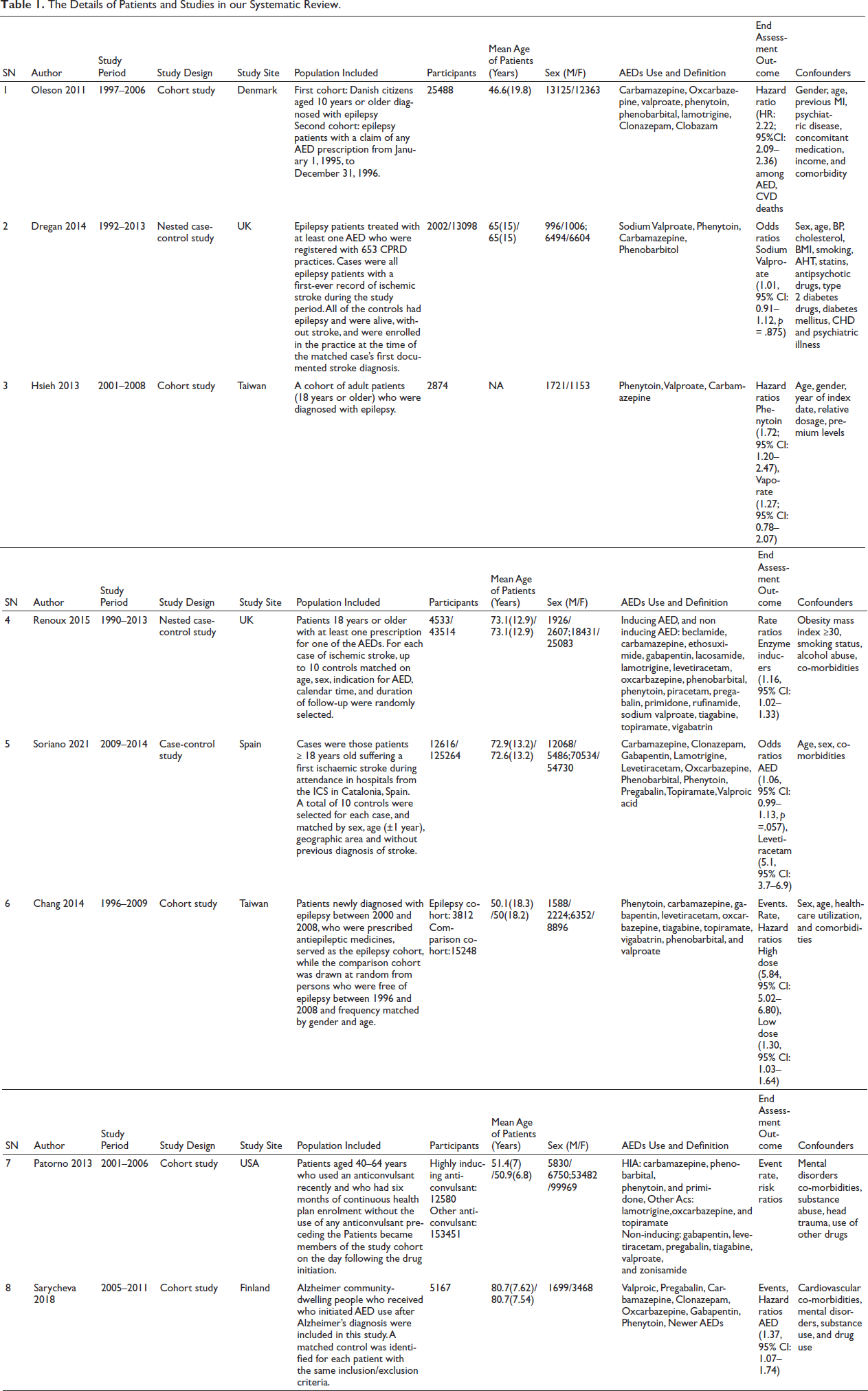
Population Included
The included studies had diverse samples from the hospital and population-based studies.
Olesen et al. included Danish hospital-based two cohorts of epilepsy patients. The first cohorts reported alive and aged 10 years or older with a diagnosis of epilepsy before 1997. The second cohort had a patient with prescription of any AED from 1995 to 1996. 11
Dregan et al. included the United Kingdom hospital-based cohorts of patients suffering from epilepsy who were treated with at least one AED between 1992 and 2013. The cases had their first event of ischemic stroke during the study duration, while the controls were epilepsy patients who did not have a stroke during that time. 12
A population-based cohort of Taiwanese adult patients (18 years old) with epilepsy who received novel AED treatment between 2001 and 2008 was included by Hsieh et al. 9
Renoux et al. included a cohort of incident AED users aged greater than 18 years old between 1990 and 2013. Up to 10 controls were randomly chosen from among the cohort members in the risk sets identified by each case of ischemic stroke during the study period. The controls were matched on age, sex, the need for AED, the calendar year, and the length of follow-up. 13
Soriano et al. included population-based cohorts from Spain of patients greater than 18 years old suffering a first ischemic stroke between 2009 and 2014 who attended hospitals. For each case, a total of 10 controls were chosen, matched by sex, age (±1 year), region, and the absence of a prior stroke diagnosis. 14
Chang et al. studied a population-based sample of Chinese people who were newly diagnosed with epilepsy and were prescribed antiepileptic medicines between 2000 and 2008 as cases and non-epilepsy people who were frequency-matched by sex, age, and index year as controls. 7
Patorno et al. used population-based cohorts from the United States that comprised people aged 40–64 who had started using AED between 2001 and 2006 and had no severe coronary or cerebrovascular conditions in the six months prior to starting medication. 15
Finally, Sarycheva et al. studied Alzheimer’s disease in community-dwelling Finnish cohorts from 2005 to 2011. Each incident AED user was paired with one nonuser based on gender, age, and duration since AD diagnosis. 16
Anti-epileptic Drugs (AED) Used and Usage Duration Definition
These studies included multiple AEDs and the usage duration of AEDs was defined which is summarised in Table 1.
Olesen et al. included Carbamazepine, Oxcarbazepine, valproate, phenytoin, phenobarbital, lamotrigine, Clonazepam, and Clobazam in their study. The duration of AED use was followed for 5–10 years after initiating the AED, and antiepileptic medication persistence was determined by identifying the proportion of patients who were alive and claimed a prescription for their AED prescription during the follow-up. 11
Dregan et al. included sodium valproate, carbamazepine, phenobarbital, and phenytoin treatments in their study. The duration of AED usage was followed through stroke incidence after the study start date. It was further divided into four categories: Ever treated denied, pre-stroke year treatment, cumulative number of prescriptions, and time on treatment. Ever treated denied was defined as the start of an AED during the study period to the index stroke, pre-stroke year treatment as the start of an AED one year prior to the index stroke, and time on treatment as the total number of days from the study start date (mostly the epilepsy index date) to the stroke. 12
Hsieh et al. included phenytoin, sodium valproate, and Carbamazepine in their study which was newly used within 1 year of the study period. The use of AEDs was monitored until a stroke occurred, and switching from one AED to another, the addition of another AED, and the discontinuation of the AED regimen (defined as no AED prescription for >30 days after the end date of the previous prescription), no enrolment from the study registry, and the completion of the study period. The prescribed daily dosage (PDD) was determined using prescription data from hospital visits, and the defined daily dose (DDD) assignment was based on dose information collected from the World Health Organization Collaborating Centre. The PDD/DDD ratio of an AED provided information about a drug’s relative dose in relation to recommendations, and the ratio also provided information about the severity of epilepsy. 9
The duration of each prescription was determined in the study by Renoux et al., and AEDs were grouped into three categories: those that inhibit enzymes (sodium valproate), those that induce them (carbamazepine, phenytoin, phenobarbital, and primidone), and non-inducing AEDs (all other AEDs). Initiators were those who had no AED prescriptions written in the year before the index date other than during the 90 days prior to the index date, and current users were those whose recent AED exposure began earlier than the previous 90 days. Patients who had their last AED prescription expire between 365 and 90 days prior to the index date were considered past users. 13
Soriano et al. included Carbamazepine, Clonazepam, Gabapentin, Lamotrigine, Levetiracetam, Oxcarbazepine, Phenobarbital, Phenytoin, Pregabalin, Topiramate, Valproic acid in their study. The administration of at least three packages of AED during the study period was considered AED exposure. If the dispensation occurred more than one year before the index date, it was considered past exposure. Patients were considered to have current exposure if they had at least one dispensation within three months of the index date. 14
Chang et al. included phenytoin, levetiracetam, oxcarbazepine carbamazepine, gabapentin, vigabatrin, phenobarbital tiagabine, topiramate, and valproate in their study. The average defined daily doses (DDDs) of AEDs were used to calculate the duration of AED use, which was then divided by the duration of the entire follow-up period (DDD/year). 7
Patorno et al. classified anticonvulsants into two groups: those that strongly induce cytochrome P450 enzyme system activity (such as carbamazepine, phenobarbital, phenytoin, and primidone) and those that minimally induce (oxcarbazepine, topiramate, and lamotrigine) and that do not induce (such as gabapentin, levetiracetam, pregabalin, valproate, and zonisamide). The duration of the participant’s use of an AED was determined by following them until the conclusion of their exposure risk window, switching to a different anticonvulsant, the occurrence of a study event, death from a cause other than one covered by the study outcome, the termination of continuous health plan enrolment, the conclusion of the study period, or the end of the study period, whichever came first. 15
Finally, Sarycheva et al. categorized AEDs into older and newer. Valproic acid, carbamazepine, clonazepam, and phenytoin were among the older AEDs, while pregabalin, gabapentin, lamotrigine, oxcarbazepine, topiramate, and levetiracetam were among the more recent ones The duration of AED use has been recorded from the index date of AED use or the corresponding matching date for nonusers until the incidence of stroke, death, AED use discontinuation for users, AED initiation for nonusers, continuous hospitalization/institutionalization lasting more than 90 days, after three years of follow-up, or the end of the study. 16
Confounders Present
Age, sex, body mass index, hypertension, diabetes mellitus, dyslipidaemia, smoking status, heart failure, alcoholism, income, concurrent medications, prior myocardial infarction, and psychiatric illness were the main confounders in these studies.
Concomitant Medications Used
Multiple concomitant medications were used in these studies for medical co-morbidity other than epilepsy. While summarizing all the studies; anticoagulants, antiplatelets, diabetes and cardiac therapy, lipid-modifying agents, hormonal contraceptives, non-steroidal anti-inflammatory drugs (NSAID), analgesics, and psychotropic drugs were currently used as co-medications.
Anti-epileptic Drug Use and Risk of Stroke
Olesen et al. found that AED-treated epilepsy patients who had never had a stroke (hazard ratio: 2.22; 95% CI: 2.09–2.36) had a significantly greater risk of stroke than non-AED-treated epilepsy patients (hazard ratio: 1.58; 95% CI: 1.47–1.70). The use of carbamazepine was found to be associated with an increased risk of stroke (hazard ratio: 1.21; 95% CI: 1.10–1.34), whereas valproate and lamotrigine exhibited a non-significant lower risk of stroke (p value: .12). The 5-year and 10-year mean rates of AED treatment persistence were 62.7% and 53.2%, respectively. 11
Dregan et al. showed that pre-year stroke sodium valproate therapy was related to a greater risk of stroke (odds ratio: 1.22, 95% CI 1.09–1.38). While no link was found between having ever been administered Sodium Valproate and having an ischemic stroke (odds ratio: 1.01, 95% CI: 0.91–1.12, p = .875). Patients who received the highest quarter of Sodium Valproate had decreased odds of ischemic stroke (odds ratio: 0.57, 95% CI: 0.44–0.72, p = .001). In stroke patients, the average number of years on sodium valproate therapy was 0.66 years. 12
When compared to carbamazepine, Hsieh et al. discovered that patients receiving phenytoin had a significantly greater stroke risk (adjusted hazard ratio: 1.72; 95% CI: 1.20–2.47), followed by valproate (adjusted hazard ratio: 1.27; 95% CI: 0.78–2.07). The risk of stroke rose by 3% every year of age and by 70% per unit rise in the PDD/DDD ratio of AEDs. The average time between starting an AED and having a stroke was 365 days for carbamazepine, 256 days for phenytoin, and 268 days for valproic acid. 9
In contrast to non-inducing AEDs, Renoux et al. discovered that enzyme-inducing AEDs had a higher risk of ischemic stroke (relative risk 1.16, 95% CI: 1.02–1.33). Comparing the current use of inhibiting AED to the current use of non-inducing AED, there was no difference in the incidence of ischemic stroke (relative risk 1.13, 95% CI: 0.96–1.33). Longer current usage of both inducing and inhibitory AED did not appear to significantly increase the risk of ischemic stroke when use was stratified by length of use. The average number of years until the index myocardial infarction or stroke was the follow-up period. 13
According to Chang et al., the epilepsy patient with the lowest dose of AED exposure had a 1.30-fold higher risk of having a stroke compared to the comparison cohort, and the epilepsy patient with the highest dose of AED exposure had the highest increased risk of stroke (5.84, 95% CI: 5.02–6.80). The findings also showed that AED exposure increased the probability of having an ischemic stroke, a haemorrhagic stroke, or both (P for trend .0001). From the beginning of the trial, the duration of AED use and the occurrence of stroke were monitored for eight years. 7
Soriano et al. concluded that levetiracetam monotherapy had a significant risk of ischemic stroke (odds ratio: 5.1, 95% CI: 3.7–6.9), while global exposure of levetiracetam, phenytoin, and valproic acid also have increased risk of ischemic stroke. The global exposure to AED also showed an increased risk (odds ratio: 1.06, 95% CI: 0.99–1.13, p = .057) of ischemic stroke. The global exposure to phenytoin, valproic acid; past exposure to phenytoin, phenobarbital, and current use of valproic acid showed the risk of ischemic stroke comparatively less than levetiracetam. The AED exposure was followed through study time, that is, five years. 14
Sarycheva et al. concluded that AED use was related to an increased risk of stroke (hazard ratio 1.37, 95% CI: 1.07–1.74) at 90 days, regardless of AED type. There were no statistically significant differences between the stroke risks of users of older AEDs and those of users of newer AEDs (adjusted hazard ratio 1.04, 95% CI: 0.71–1.53). AED exposure had a mean follow-up period of 351.7 days as opposed to 728.6 days for nonusers. 16
Finally, Patorno et al. found no statistically significant evidence of AEDs (enzyme inducers and non-inducers) and increased risk for stroke. 15
Discussion
In our systemic review, seven of the eight studies showed an increased risk of ischemic stroke associated with AEDs among individuals without a history of cardiovascular accidents. AEDs mainly carbamazepine, oxcarbamazepine, phenytoin, valproic acid, levetiracetam, phenobarbital, and primidone showed an increased stroke risk. The stroke risk was associated with phenytoin, sodium valproate, oxcarbamazepine, levetiracetam, phenobarbital, and carbamazepine.7, 9 No association was noted between the duration of AED exposure and stroke.12, 13 The mean duration of AED exposure was heterogeneous and thereby could not be estimated.
AEDs can be classified as inducing, non-inducing, or inhibitory AEDs based on their mode of action on liver enzymes. Only sodium valproate inhibits liver enzymes, while carbamazepine, phenobarbital, phenytoin, and primidone induce liver enzymes. Other second-generation AEDs are non-inducing. 17 AEDs that induce hepatic enzymes, which largely influence the cytochrome P450 system, can disrupt lipid metabolism. 18 Total cholesterol, low-density lipoprotein (LDL), cholesterol, triglycerides, lipoprotein, C reactive protein, and homocysteine levels in the blood have all been found to rise with the use of AEDs.19–22 These changes were also evident two to three months after the onset of treatment. 23 This notion is supported by the finding that changing non-inducing AEDs from inducing AEDs decreased the atherosclerosis risk.21, 24 The longer duration of AED exposure further accelerates atherosclerosis independent of risk factors like age, gender, and oxidative stress. 25 On the other hand, sodium valproate, an enzyme inhibiting AED is not considered to be associated with the above metabolic alteration; its net vascular effect is still unclear. Through the production of insulin resistance, body weight gain, metabolic syndrome, and increased oxidative stress, valproate may have a proatherogenic effect.26, 27 Overall, an increase in carotid artery intima-media thickness is observed among epilepsy patients with AED usage. 28 These findings support the mechanism behind the increased risk of stroke with AED exposure seen in our study.
Our hypothesis that AEDs can be a risk factor for the occurrence of ischemic stroke patients amid the confounders like concomitant medications used, co-morbidities, previous history, and other associated vascular risk factors; can make its implication very limited. Similarly, the possibility of epilepsy itself increasing the stroke risk cannot be ruled out. But, certain AEDs like enzyme-inducing drugs backed up by their mechanisms in the body in promoting proatherogenic factors could give some sort of evidence for our hypothesis.
Limitations and Strengths
To the best of our knowledge, this is the first study to examine the key studies in order to determine the link between AED use and ischemic stroke risk. The major limitation of our study is the heterogeneous usage of concomitant medications for other medical co-morbidity across the studies. We were not able to conduct a meta-analysis due to a lack of homogeneous data. We could not search other databases. Further ranking of probabilities required in terms of stroke risk with individual AED and additive/synergistic action of AED combinations were not clearly defined. Finally, AED exposure and dose were not well quantified in all the studies so the comparison was missing.
Conclusion
We conclude significant stroke risk with AEDs (mainly enzyme inducers), and dose dependency need to be assessed. The risks and benefits of AEDs should be weighed among the patients, especially with vascular risk factors to limit the ischemic stroke risk. To make firm conclusions on the vascular risk associated with the use of specific AEDs, large, prospective, and high-quality validated cohort studies adjusted for confounding factors are required.
Abbreviations
AEDs: Antiepileptic medications
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Authors’ Contribution
Study Conception and Design: BG, SK, & RB.
Data Collection: SK, KB, & DR.
Analysis and Interpretation of Results: BG, SK, RG, & RB.
Writing & Revision of Manuscript: SK, RB, DR, RG, KB & RB.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
