Abstract
Background
Stroke is a major leading global health complication. Identification and management of risk factors associated with stroke can help in prior detection, prevention, and improvement in patient care.
Purpose
To investigate the prevalence of hyperhomocysteinemia (HHcy) and Vitamins B6, B12, and folate deficiency in stroke patients and also to assess other risk factors associated with ischemic and hemorrhagic stroke.
Methods
Detail history of all the subjects in the study including history of hypertension, anemia, fasting glucose, carotid artery thickness, smoking, alcohol, and dietary intake was recorded. Standard assays for homocysteine (Hcy), Vitamins B6, B12, and folate estimation were done. Lipid and renal profile tests were also performed. The prevalence and odds of having HHcy, Vitamins B6, B12, and folate deficiency, and other risk factors in ischemic and hemorrhagic stroke patients were evaluated. Student’s t-tests and chi-square tests were done for statistical validation of the data.
Results
Prevalence of HHcy and Vitamins B6, B12, and folate deficiency was not observed in ischemic cases. HHcy and folate deficiency was found to be prevalent in hemorrhagic stroke patients. The odds that a person with HHcy and folate deficiency has hemorrhagic stroke was found to be significantly high.
Conclusion
In our study, high Hcy and low folate levels emerged as risk factors for hemorrhagic stroke.
Introduction
Stroke is an acute clinical event and a major global health problem. It is one of the foremost causes of mortality, morbidity, and long-term disability throughout the world.1, 2 Stroke is the fourth leading cause of death and the fifth leading cause of Disability Adjusted Life Years (DALY) in India. The impact of stroke is such that the survivors have to live with physical disabilities and emotional strain and become dependent on families for long-term care and rehabilitation. Various types of risk factors have been identified that are associated with stroke. 3 This includes modifiable risk factors which comprise hypertension, diabetes, obesity, smoking, alcoholism, diet, and lack of physical activity, and non-modifiable risk factors such as age, gender, family history, and race/ethnicity. 3 Management of these risk factors is crucial for primary and secondary prevention of stroke and functional outcomes post stroke.
Hyperhomocysteinemia (HHcy) is an established modifiable risk factor that may contribute to stroke. 4 Several studies have been conducted that showed the association of plasma total homocysteine (tHcy) levels with stroke.5–10 Elevated Hcy levels may be caused by a Vitamin B6 or B12 or folate deficiency which in turn may be due to either their low dietary intake or a decrease in intestinal absorption of food elements with aging or due to genetic polymorphisms.11–13 However, data from such kinds of studies from the Indian subcontinent are relatively sparse.
Therefore, this study aimed to assess the prevalence of HHcy and Vitamins B6, B12, and folate deficiency, and various other risk factors in Indian stroke patients from the Northeastern part of the country. Such a study will enable the design and development of different strategies for the prevention, early detection, treatment, and rehabilitation of stroke patients.
Methods
Study Design
A case-control study was designed to investigate the association of plasma tHcy and Vitamins B6, B12, and folate levels with ischemic and hemorrhagic stroke and to estimate the odds ratio of stroke with various other factors. This study was conducted from 2002 to 2005 in GNRC. It is the first super specialty tertiary health care center in Northeast India. All experimental protocols followed standard procedures and the study was approved by the ethics committee of the hospital. Written informed consent was obtained for all the subjects involved in the study.
Patients and Controls
Patients from Guwahati in Assam who suffered from acute ischemic and hemorrhagic stroke and were admitted to GNRC hospital were selected for the study. A total of 110 patients from each stroke and with ≥35 years of age were included. Since HHcy is found in patients with major systemic illnesses including myocardial infarction, peripheral vascular disease, hepatic disease, thyroid disease, and renal disease; pregnant women and patients on anticonvulsant medications were excluded from the study. One age and gender-matched healthy volunteer without any history of stroke from the neighboring areas of the hospital was included. The subjects were given detailed information about the objectives and the methodology of the study. Data on demographic characteristics, environment, dietary habits, and lifestyle factors were collected based on a pre-designed proforma.
Clinical Evaluation
Physical examination was carried out for all the subjects in the study. The stroke patients were further diagnosed using the neuroimaging techniques such as brain Computed Tomography (CT) scans and Magnetic Resonance Imaging (MRI). Electrocardiography (ECG) to assess heart rate and rhythm was performed for all the subjects. The Carotid Doppler test was performed to examine the presence of intima-media wall thickening of the carotid arteries. The blood pressure of each subject was measured and recorded. A detailed analysis of lipid and renal profiles along with routine biochemical and hemogram tests was performed for the subjects.
Estimation of Plasma Analytes Levels
A simple, rapid, and highly specific assay was followed for the determination of plasma analyte levels.14, 15 Fasting levels of tHcy were estimated by collecting 2 ml of blood along with the anticoagulant EDTA. The collected blood samples were immediately centrifuged to avoid any erroneous elevation of tHcy levels. The separated plasma was then preserved at –80°C and analyzed the next day. Plasma tHcy (the sum of Hcy, homocysteine [homocysteine-homocysteine disulfide], and cysteine-homocysteine disulfide both in free and radical forms) and Vitamin B6 were then estimated by using High-performance Liquid Chromatography (HPLC) with fluorescence detector. 16 Vitamins B12 and folate were estimated by following the chemiluminescence illumination system.
Statistical Analysis
The statistical analyses were performed using the SPPS software version 17. 17 Further statistical analysis was carried out using student’s t-test and the chi-square test. The odds ratio was calculated for the different variables included in the study. Descriptive statistical values were expressed as mean ± standard deviation (SD). Statistical significance was considered when p-values were <.05.
Results
The baseline characteristics of all the subjects involved in this study are shown in Table 1. A total of 220 patients from both stroke types and 220 controls were included in the study. The subjects comprised of 21.8% (n = 24) females and 78.2% (n = 86) males. Age-wise distribution of cases and controls is shown in Figure 1. Majority of the ischemic (85.5%; n = 94) and hemorrhagic (91.8%; n = 101) stroke patients were diagnosed with hypertension. Elevated fasting blood sugar levels were also observed in both ischemic (36.4%; n = 40) and hemorrhagic (41.8%; n = 46) stroke patients. Anemia was seen in a lesser number in both cases and controls. The majority of the subjects involved in the study followed a non-vegetarian diet.
Clinical Profile of Cases and Controls Included in This Study.
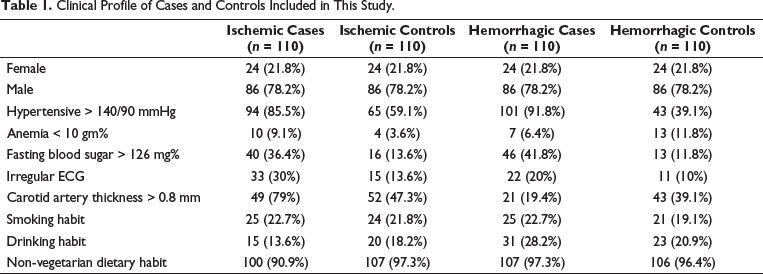
Age-wise Distribution of Ischemic and Hemorrhagic Cases and Controls Included in This Study.
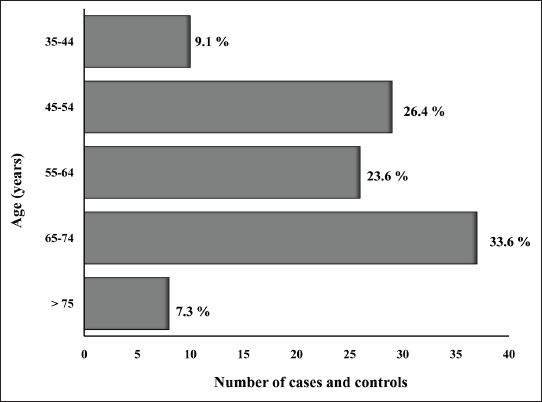
Table 2 describes the levels of different plasma analytes in cases and controls. No significant difference was observed in mean plasma tHcy levels in both ischemic cases and controls. High plasma tHcy levels were seen in 59.1% (n = 65) ischemic cases and 56.4% (n = 62) controls. However, mean plasma tHcy levels were significantly high in hemorrhagic cases (p =.00) as compared to the controls. 71% (n = 78) hemorrhagic stroke patients had high plasma tHcy levels as opposed to 56.4% (n = 62) controls. The correlation between plasma tHcy level and hemorrhagic cases was statistically significant on chi-square test (df = 1; p =.025). This finding supported our hypothesis that HHcy was associated with hemorrhagic stroke.
Plasma Analytes Levels in Ischemic and Hemorrhagic Cases and Controls Included in This Study.
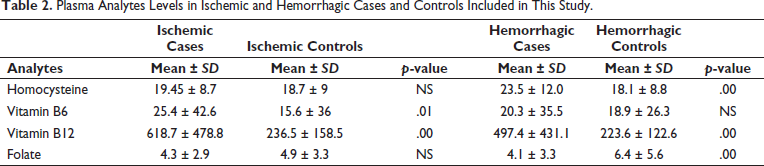
Interestingly, ischemic stroke patients showed significantly high mean Vitamin B6 levels (p =.01) in comparison to the controls while no difference was observed in hemorrhagic cases and controls. Similarly, significantly elevated Vitamin B12 levels were seen in both ischemic and hemorrhagic cases (p =.00) as compared to controls. On the other hand, mean plasma folate levels were found to be significantly less in hemorrhagic stroke patients (p =.00) when compared with the controls while no significant difference in folate levels was seen for ischemic cases and controls. Folate deficiency was seen in 42.3% (n = 33) hemorrhagic stroke patients and only 16.1% (n = 10) controls showed deficiency of folate. A statistically significant correlation of folate levels with hemorrhagic cases was seen in chi-square analysis (df = 11.12; p =.001). This in turn supported our hypothesis that low folate level is associated with hemorrhagic stroke.
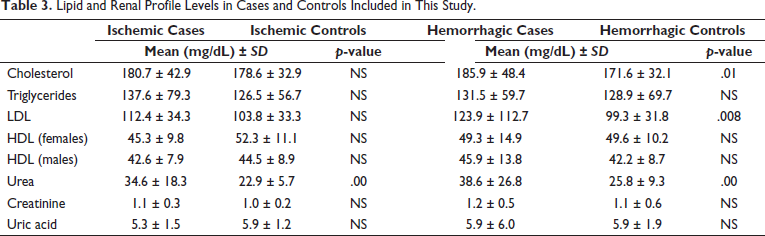
A thorough lipid and renal profile analysis for all the subjects involved in the study is shown in Table 3. The ischemic stroke patients and the controls shared similar lipid profiles and statistically no significant differences were seen. However, the hemorrhagic stroke patients showed a significant increase in their cholesterol (p =.01) and LDL levels (p =.008) in comparison with the controls. Only the levels of urea were found to be significantly high in both ischemic (p =.00) and hemorrhagic (p =.00) stroke patients as compared to the control group.
Lipid and Renal Profile Levels in Cases and Controls Included in This Study.
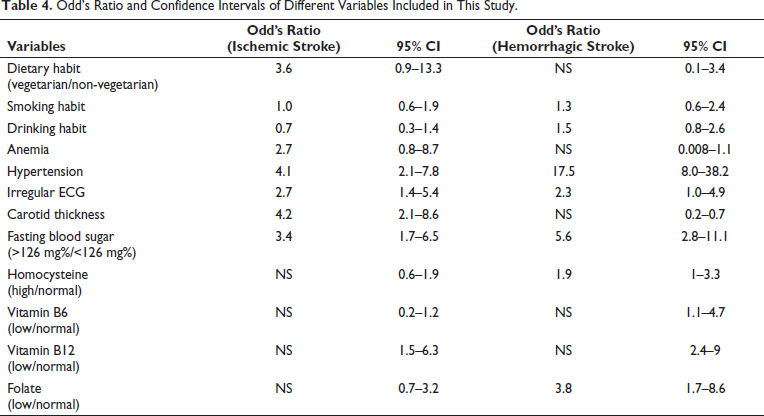
A multivariate regression analysis was performed to assess the effect of various factors including HHcy and folate deficiency on the risk of ischemic and hemorrhagic stroke (Table 4). The risk profile for ischemic stroke in our study included vegetarian dietary habit (OR = 3.6), anemia (OR = 2.7), hypertension (OR = 4.1), irregular ECG (OR = 2.7), carotid thickness (OR = 4.2), and diabetes mellitus (OR = 3.4). Risk factors for hemorrhagic stroke in our study included smoking (OR = 1.3), drinking (OR = 1.5), hypertension (OR = 17.5), irregular ECG (OR = 2.3), diabetes mellitus (OR = 5.6), high Hcy (OR = 1.9), and low folate (OR = 3.8).
Odd’s Ratio and Confidence Intervals of Different Variables Included in This Study.
Discussion
Less data is available from the Indian subcontinent about the prevalence of HHcy and vitamin deficiencies as modifiable risk factors for stroke. To our knowledge, this is the first study that highlights the relationship between plasma tHcy, Vitamins B6, B12, folate levels, and stroke among the population of Assam. In our case-control study, the prevalence of HHcy and folate deficiency was seen in hemorrhagic stroke patients. So far most of the case-control and prospective studies that have been conducted for patients of all age groups are limited to ischemic stroke, and there is a scarcity of information about the association of HHcy with hemorrhagic stroke. However, in our study, we did not find any correlation between HHcy and Vitamins B6, B12, and folate deficiencies with ischemic stroke. This result is consistent with some earlier prospective studies that did not show any association between HHcy and ischemic stroke.18, 19
Hcy is derived from the demethylation of the essential amino acid methionine. 7 Vitamins B6, B12, and folate act as cofactors in Hcy metabolism. 7 The normal range of fasting plasma tHcy concentrations lies between 5 and 15 µmol/L while moderate HHcy lies in the range of 15–30 µmol/L, intermediate HHcy is between 30 and 100 µmol/L, and severe HHcy is > 100 µmol/L. 20 We observed elevated mean plasma tHcy levels in hemorrhagic stroke patients in our study. Many factors can contribute to HHcy including acquired, dietary, ethnic, and genetic factors. 10 Thus, studies that have been done and published in Western countries might not apply to the Indian population. In developing countries, a high prevalence of vitamin deficiencies causes HHcy which in turn increases the risk of stroke. 21 In our study, vitamin deficiency was not seen among the ischemic cases. However, folate level was significantly low in the hemorrhagic stroke patients.
Some studies have shown a synergistic effect of elevated homocysteine and abnormal blood lipids on the onset of stroke. 22 In our study, we have seen that the hemorrhagic stroke patients had elevated blood cholesterol and LDL levels. However, both the factors were normal in ischemic cases and controls. Significant increase in urea levels was seen in both ischemic and hemorrhagic cases.
Some of the major risk factors for both ischemic and hemorrhagic stroke in our case-control study were similar to the long-recognized risk factors for stroke worldwide. 23 Hypertension is well known as a modifiable risk factor and the risk of stroke due to high blood pressure is as much as fourfold. Diabetes is also an established risk factor for stroke following hypertension. Heart diseases that include atrial fibrillation and atrial flutter are frequently associated with stroke. Smoking and chronic heavy alcohol consumption are known independent predictors of stroke risk. HHcy is a less well-documented risk factor for stroke.
Hypertension was one of the most common risk factor in our study followed by diabetes mellitus. Reports from previously published data had shown a relationship between hypertension and Hcy levels. It is seen that HHcy induces the production and secretion of serine elastase, thereby creating an elastolytic process in the arterial wall. This leads to stiffening of the arterial wall and finally results in a rise in arterial blood pressure. There can be other concomitant factors by which HHcy can contribute as a risk factor for stroke. 24 Elevated Hcy levels also cause oxidative damage to the vascular endothelial cells, depletes nitric oxide production from the endothelium, cause platelets adhesion to endothelial cells, promote vascular smooth muscle cells growth, trigger release of prothrombotic factors such as factor VII c, tissue plasminogen activator, and thromboglobulin and thus leads to thrombotic vascular disease. 25 The association of Hcy levels and diabetes mellitus has shown conflicting results in earlier studies. 26 Further investigations are required to show the association between Hcy levels and the traditional risk factors such as hypertension and elevated fasting glucose levels in the ischemic and hemorrhagic stroke patients of our study.
Thus, this study revealed the prevalence of high Hcy and low folate levels in hemorrhagic stroke patients. The main limitation of our study is the relatively small size of cases and controls. For the estimation of normal levels of tHcy among the general population, a large number of controls need to be considered. We were also unable to measure tHcy levels in different subgroups which was another limitation of our study. We could not study the correlation of the levels with the stroke outcome. Therefore, more studies are required on a large scale for further verification and our hospital plans to do the same in near future.
Conclusion
Our study is the first from Northeast India that showed the prevalence of HHcy and folate deficiency in hemorrhagic stroke patients. The mean tHcy and folate levels in hemorrhagic cases were found to be significantly high and low respectively than in the controls. Elevated levels of urea in both ischemic and hemorrhagic stroke patients and increased cholesterol and LDL levels in hemorrhagic cases were also seen in our study. Our study does support the hypothesis that HHcy and folate deficiency was associated with hemorrhagic stroke. A similar conclusion could not be drawn for ischemic stroke.
Footnotes
Authors’ Contribution
MG did the planning, data collection, and proofreading. MD did the study design, data collection, supervision, funding acquisition, and proofreading. RD did the supervision, performed literature survey, data analysis, and proofreading. ARB performed the literature survey, data analysis, and critically revised the work. PD did the literature survey, data analysis, and drafted the article. ApB did data analysis and proofreading. VL did data analysis, drafting of the manuscript, and critically supervised the work. DJC, JPS, and MM were involved in manuscript editing and overall supervision. NCB conceived the idea, study design, funding acquisition, manuscript editing, and overall supervision. NC did data collection, statistical analysis of the data, and drafting of the article. AB did the literature survey, data analysis, writing, and editing of the manuscript.
Statement of Ethics
Ethical clearance for the study was obtained from the ethics committee of GNRC Hospitals, Guwahati, Assam. The informed consent was taken from all the subjects for publication (as per our department policy we take such consent from all the patients at time of admission).
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Indian Council of Medical Research, New Delhi.
