Abstract
Background
Parkinson’s disease (PD) is a progressive neurodegenerative disorder that mainly affects the aged population. Transcranial magnetic field (MF) stimulation has shown to provide temporary motor recovery in neurological disorders.
Purpose
The aim of this study was to understand the cellular and molecular mechanism of low-intensity MF stimulation (17.96 µT; 50Hz; 2 h/day, four weeks) in a rat model of severe PD.
Methods
A clinically relevant, bilateral striatal 6-hydroxydopamine (6-OHDA) lesioned rat model of severe PD was employed to test the efficacy of low-intensity MF stimulation in the management of motor symptoms. The mechanism of action of MF was dissected by assessing the microglial activation, tissue ultrastructure, and cerebrospinal fluid (CSF) metabolomics using microdialysis.
Results
We observed a significant improvement in the postural balance and gait after MF exposure with a significant reduction in the number of activated microglia. There was an improvement in striatal dopaminergic innervation and glutamate levels but it did not reach a level of statistical significance.
Conclusion
MF stimulation helped ameliorate the motor deficits and reduced inflammation but was unable to provide a significant change in terms of dopaminergic innervation and metabolic profile in the severe 6-OHDA PD rat model.
Keywords
Introduction
Magnetic field (MF) stimulation, owing to its inherent noninvasive nature finds immense use in health and neurological disorders. In a healthy individual, it can be used to study the integrity of motor tracts, measure conduction velocity, functionally map the sensory/motor cortex, and evaluate the cortical plasticity. 1 It has been approved by the Food and Drug Administration (FDA) for various psychiatric and chronic pain disorders. MF stimulation of the prefrontal cortex provides relief to patients with drug-resistant depression,2, 3 which may be because of the increase in turnover of dopamine. 4 In healthy human subjects, positron emission tomography (PET) and [(11)C]raclopride studies have shown to increase extracellular dopamine concentration in the ipsilateral caudate nucleus after repetitive transcranial magnetic field stimulation (rTMS) exposure to the dorsolateral prefrontal cortex. 5 This makes it an interesting candidate for the treatment of Parkinson’s disease (PD). Significant amelioration of bradykinesia was observed in PD patients exposed to rTMS over the motor hand area (M1). 6 However, in a contradictory report, 5-Hz rTMS was unable to improve fine coordinated finger movement as assessed by the Grooved Pegboard test. 7 The improvement in fine motor function by rTMS therefore seems to depend upon the on/off state of PD, 8 also recent meta-analysis studies suggest that the therapeutic outcomes also depend on the intensity, duration, and frequency of stimulation. 9 The major shortcoming in the application of rTMS therapy for any neurodegenerative disorder is the lack of understanding of the underlying cellular and molecular mechanisms involved. Substantial animal studies are required to elucidate the mechanism of action of MF and to standardize the effects of different stimulation paradigms before it can be translated into clinical applications. 10 Further, most of the studies use coils that generate high-intensity MFs, which increase the risk of seizures especially in sensitive individuals.
Uniform MFs of low intensity are able to penetrate physiological barriers and have low or no seizure-inducing capability. 11 We have observed that when spinal-cord-injured rats (T12-13 vertebral level) were exposed to MFs in a modified Lee-Whiting coil, which produces a uniform MF of 17.96 µT at 50 Hz, led to an improvement in hind limb motor function, increased axonal sprouting, reduced oxidative stress, inflammation, and scarring at the site of injury. 12 Rats with a striatal injection of 6-hydroxydopamine (6-OHDA) when exposed to MF for seven days, showed sparing of dopaminergic terminal in the striatum and reduced oxidative stress and mitochondrial dysfunction 13 as compared to the disease control rats.
In this study, our aim was to emulate the motor stage of human PD in rats, where clinical diagnosis is usually possible, and test the efficacy of our treatment paradigm. To achieve this goal, we administered 6-OHDA bilaterally into the striatum and the disease was allowed to progress for three weeks. Intervention in the form of MF exposure was given for four weeks thereafter, and its effect on motor function, dopaminergic innervation and metabolomics of striatum, microglia, and mitochondrial ultrastructure was studied.
Methods
Animals
Adult male Wistar rats weighing 270 gm to 320 gm, procured from the central animal facility, All India Institute of Medical Sciences (AIIMS), New Delhi were used for this study. They were provided with laboratory food pellets (Ashirwad Industries, India) and clean drinking water ad libitum. Rats were housed individually in polycarbonate cages at an ambient room temperature of 24 ± 2°C, relative humidity of 50% to 55%, and in 14:10-h light-dark cycles. All experiments were performed under strict ethical guidelines, after approval from the Institutional Animal Ethics Committee (File No. 797/IAEC/12).
Experimental Design
The rats were divided into two groups – Parkinson’s disease group (PD) and Parkinson’s disease group exposed to magnetic field (PD + MF) – containing nine rats in each. In all the rats, baseline recording of motor behavior tests was followed by administration of 6-OHDA to induce PD. In the PD + MF group, MF exposure was initiated from the end of the third week until the seventh week of 6-OHDA administration. At the end of the study, motor behavioral tests were repeated and microdialysis was performed. Rats were sacrificed by transcardial perfusion first with cold saline and then 4% paraformaldehyde to fix the brain for immunohistochemistry. For transmission electron microscopy (TEM), the brain was dissected out, subdivided into areas with substantia nigra and postfixed in 2% paraformaldehyde and 2.5% glutaraldehyde in 0.1-M phosphate buffer (pH 7.4) for 6 h to 8 h at 4°C.
6-OHDA Administration to Develop Bilateral PD Model
A surgical procedure similar to that followed by Umarao et al. 13 was used. In brief, anesthetized rats (50 mg/kg thiopentone i.p.) were fixed on the stereotaxic apparatus, (Model-1404, David Kopf, USA) in a “flat-skull” position and bregma was identified as the reference. Two holes each were drilled on either side of the skull to target the striatum bilaterally at the following coordinates: AP: 0.0 mm, ML: ±3.2 mm, DV: -6.0 mm and AP: 1.0 mm, ML: ±2.5 mm, DV: -5.5 mm. 14 Bilateral lesions were achieved by injecting 2 µL 6-OHDA (7.5 µg/µL in 0.2% ascorbate saline) at a speed of 2 µL/min (Harvard Nanomite pump, USA) and i.p. injection 20 mg/kg body wt. of desipramine was given 30 min before 6-OHDA infusion to preserve other monoamine neurons. The syringe was held in place for 5 min and retracted slowly. The wound was sutured and postoperative care was taken.
Magnetic Field Exposure
Three weeks post 6-OHDA insult, the rats were exposed to low-intensity MFs until the end of the study period (2 h/day for four weeks) in an MF chamber. The latter is a modified Lee-Whiting coil, where coils are connected in a series and generate uniform MF of 17.96 µT, 50 Hz at the central volume. 15 The MF chamber is composed of two pairs of Helmholtz coils mounted on a stand, a movable platform for placement of the semi-restrained rats in specially designed polypropylene boxes. Out of the four coils, the outer two coils have 18 turns and the inner two coils have 8 turns and the current ratio between the outer and inner pairs of coils is 2.2604. Distances of outer and inner coils from the center are 470 mm and 122 mm, respectively. The MF is fine adjusted to 17.96 µT before exposing rats by using a magnetometer (Walker Scientific Inc., Auburn Hills, MI, USA).
Assessment of Motor Function
Spontaneous Locomotor Activity
Open field test (OFT) is a measure of the exploratory behavior and general motor activity of rats. The quantification of the activity was performed by the live video tracking software from Noldus (Ethovision XT software), Holland. The average speed of movement (velocity, cm/s) and the total distance moved by the rat were recorded for 300 s. The experiments were conducted every day between 21:00 h and 00.00 h in a quiet, dimly lit, air-conditioned room.
Postural Balance
This test assesses the rats’ ability to balance on a rotating rod/drum. In this study, a modified protocol described by Monville (2006) was used which combined fixed and accelerating speed protocol and determined the degree of loss/recovery or the progression of the damage to the basal ganglia.
After initial acclimation to the equipment, on the first day, rats were placed on the moving wheel for three trials at three different speeds: 12 rpm, 20 rpm, and 28 rpm, given at an intertrial interval of 20 min. On the third day, the rats were made to perform at an acceleration of 4 rpm to 40 rpm over 300 s, with a cut-off of 300 s and intertrial interval of 20 min. An average time of stay on the rod for three trials was recorded. 16
Gait
Gait was analyzed using footprint patterns (walking tracks). It was a three-day experiment with the first two days allotted for training and the last day for recording. The rat’s paws were coated with nontoxic ink/dye (red and green color ink for hind and forelimbs, respectively) and they were made to walk along a brightly lit narrow alley (100 cm long with 20 cm side walls) with a dark goal box of 20 × 20 × 20 cm3. Three footprints, present in the central region of the alley, excluding the ones close to the entry-point and the goal box, were considered for analysis of stride length, ratio of the distance between the fore base and hind base, and overlap between hind limb and fore limb. 17
Immunohistochemistry
Sections of the striatum with a thickness of 20 µm thickness were mounted on poly-l-lysine coated slides. Antigen retrieval was done in 10 mM sodium citrate (pH 6, 20 min, 95 ºC) and quenching off endogenous peroxidase activity with 3% H2O2. Blocking was performed with 10% normal goat serum and 3% bovine serum albumin in phosphate buffer saline triton (PBSTx) (2 h) followed by incubation with rabbit-anti-rat-tyrosine hydroxylase antibody (cat# AB152 Millipore, 152, 1:200) for 48 h at 4°C and incubated with horseradish peroxidase (HRP) conjugated antirabbit secondary antibody overnight at 4°C. The color was developed with diaminobenzidine and visualized under a microscope (Nikon, Eclipse). Densitometric analysis was performed using ImageJ 1.47.
For immunofluorescence, sections were incubated in anti-OX-6 (cat# MCA2687GA Biorad, 1:200) and OX-42 antibodies (cat# MCA275R, Biorad, 1:200), and then in secondary antibodies conjugated to Alexa-488. The sections were mounted with DAPI containing fluroshield and viewed under Nikon Ni with NIS elements fluorescence microscope. Quantification was performed by counting the number of positive cells under 40X objective at five fields of the sections from the striatum, taking every third section into count.
Transmission Electron Microscopy
To study the ultrastructure of the striatal tissue, fixed brain samples were osmicated and embedded in Araldite CY212. Sections of 60 nm thickness were cut, picked on copper grids and stained with uranyl acetate and lead citrate, and visualized under a Tecnai G2-20 microscope. 12
Microdialysis
Extracellular Lysate Extraction
At the end of the seventh week, a pair of CMA-12 guide cannula was inserted into the skull to a depth of 5 mm below the skull into each hemisphere at: (a) AP: 0.0, ML: 3.2; (b) AP: 0.0, ML: -3.2, and secured by anchoring screws with self-curing acrylic. On the day of the experiment, the rats acclimatized to the freely moving animal system following which extracellular fluid was extracted from each hemisphere for 90 min using CMA 12 Probe (1 mm tip). Samples collected from 30 min to 60 min were freeze-dried. 18
Nuclear Magnetic Resonance (NMR) Analysis of Microdialysis Lysate
The lysates were reconstituted in 110 µL of D2O keeping pH at seven with trimethylsilyl propionate (TSP) as an external reference.
Proton 1D nuclear magnetic resonance (NMR) was carried out on the 700 MHz high-resolution NMR spectrometer (Bruker, USA) in 3 mm NMR tubes. Water suppression and sculpting were performed with a presaturation pulse to reduce the water peak at 4.7 ppm. The following parameters were used: (a) relaxation delay: 14 s, (b) spectral width: 12 ppm, (c) number of scans: 256, and (d) data points: 32 K.
Postacquisition processing of the spectra was done by fast Fourier transformation, phase correction, and baseline correction. Peak selection was based on previous literature and maintained for all spectra. 19 Normalization of raw data for each metabolite was done with respect to the TSP standard.
Statistical Analysis
All analyses were carried out by an investigator blinded to treatment. The results of behavioral data are presented as mean (M) ± standard error of mean (SEM). The differences in the mean of metabolites and histological data are compared using student’s t-test and behavioral data using one-way repeated measure ANOVA with Holm-Sidak post hoc test. The data were analyzed using sigma plot 13 and the graphs prepared using Graphpad Prism 5.
Results
Rats (n = 29) were divided into two groups, 15 in PD and 14 in the PD + MF group. Six rats in the PD and five rats in the PD + MF groups died within three weeks of 6-OHDA insult. The mortality was mainly associated with adipsia and aphagia associated with 6-OHDA-induced dopamine loss. No significant difference in pre-6-OHDA insult baseline data was observed among the groups. Keeping in mind the objective of the study, the behavioral study data are represented to focus on the effect after MF exposure.
Postural Stability
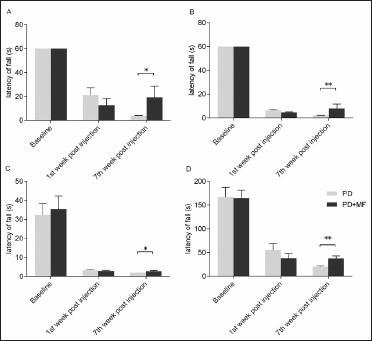
There was a statistically significant difference (P = .001) in the latency of fall(s) between the two groups at all speeds and accelerating rpm protocol after MF exposure. Post hoc analysis to assess effect of MF exposure as compared to the PD revealed a significant improvement at 12 rpm (P = .03), 20 rpm (P = .01), 28 rpm (P = .02), and accelerating rpm (P = .002) post MF exposure (Figure 1).
Amelioration of Postural Instability After MF Exposure. A Significant Improvement was Observed in the Latency of Fall in Seconds from the Rotarod in the PD Rats After Exposure to MF at all Speeds i.e., (A) 12 rpm (Low), (B) 20 rpm (Intermediate), (C) 28 rpm (High Speed), and(D) Accelerating 4 rpm to 40 rpm Protocol. Values are Expressed as the Mean ± SEM. *P < .05, **P < .01 Significant Difference Between the Two Groups, n = 6.
Spontaneous Locomotor Activity
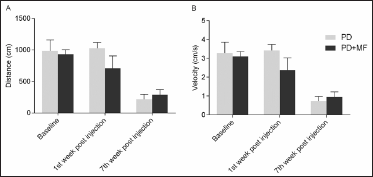
A comparison of pre and postsurgery data showed a significant difference in all the groups for both distance covered and velocity (P = .05). However, post hoc analysis showed no significant change after MF exposure at the seventh week (P = .93). No significant improvement (P = .93) was observed in the velocity at the end of the study (Figure 2).
The Effect of MF on the Spontaneous Locomotor Activity (A) Distance Covered (cm) and (B) Velocity (cm/s) of Movement by PD Rats in an Open Field Arena in 300 s. A Decrease in Both the Distance and Velocity was Observed. Values are Expressed as the Mean ± SEM, n = 6.
Gait Pattern
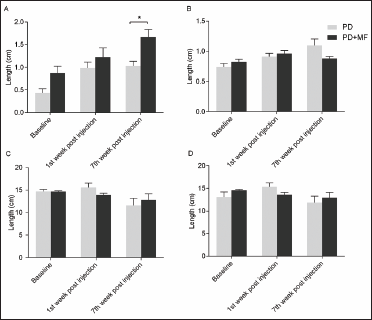
There was an overall significant difference (P ≤ .05) in overlap, ratio of forebase to hindbase, and forelimb length. Post hoc analysis of the data for overlap demonstrated a significant increase in the MF group as compared to their basal (P = .009) and a significant reduction as compared to the PD group (P = .04). No significant change was observed in the forebase-to-hindbase ratio after MF exposure at the end of the study (P = .49). Forelimb stride length showed a significant decrease at 7 weeks post injury in the PD group as compared to basal (P = .03), no significant difference (P = .15) was observed in hindlimb stride length. No significant effect of intervention was evident on stride length.
There was approximately a 2.5-times (0.432 ± 0.09 mm to1.03 ± 0.10 mm) increase in the mean gap between consecutive fore and hindlimb overlap after 6-OHDA administration, whereas, the length of the stride for both the limbs decreased, indicating short shuffled stride, typical of PD.20, 21 The base of support of the rat was also altered and the forebase was found to be wider as compared to the hindbase. In normal rat, however, the forebase was always lesser than the hindbase (Figure 3).
Effect of MF on Gait Parameters (A) Overlap, (B) Base of Support, (C) Forelimb Stride, and (D) Hindlimb Stride Length. A Significant Increase (P < .05) in the Overlap Distance After MF Exposure was Observed. Values are Expressed as the Mean ± SEM. *P < .05 Significant Difference Between the Two Groups, n = 6.
Immunostaining
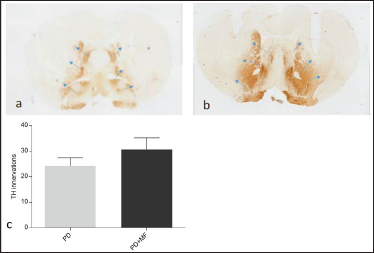
Tyrosine hydroxylase immunoreactivity was decreased in the striatum of 6-OH dopamine-lesioned PD rats. It demonstrated sparing of TH immunopositive dopaminergic terminals in the striatum after MF exposure, but did not reach a level of statistical significance (P = .06), as seen after quantification (Figure 4).
Representative Showing Dopaminergic Nerve Terminal Innervation of the Striatum as Evident from TH Immunostaining with the Blue Arrows Indicating the Boundary Between the Innervation and Denervated Striatum (A) PD Rat, (B) PD + MF Rat, and (C) Graphical Representation of the % Sparing of TH Innervation in the Striatum. Values are Expressed as the Mean ± SEM, n = 6.
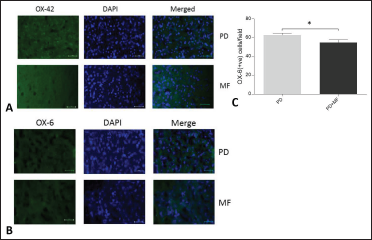
We observed a widespread increase in microglial number in the striatum, as indicated by the expression of OX-42 cells. Therefore, we proceeded to quantify the number of activated phagocytic form of microglia (OX-6). A significant decrease (P = .04) in the OX-6 positive microglia in the MF-exposed group as compared to the PD group was observed, suggesting an anti-inflammatory effect of MF exposure (Figure 5).
Representative Images Showing Effect of MF on the Microglia of Striatum (A) Anti-OX-42 and (B) Anti OX-6 Immunostaining, (C) Graphical Representation of the Number of Activated Microglia in the Striatum in PD and PD+ MF Groups. MF Stimulation Decreased the Microglial Number Significantly (P < .05). Values are Expressed as the Mean ± SEM. *P < .05, Significant Difference Between the Two Groups, n = 6.
Mitochondrial Ultrastructure
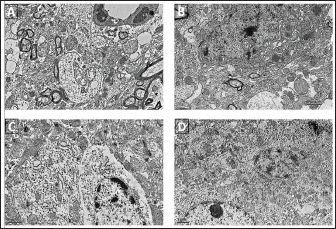
The striatal and nigral tissue was characterized by the presence of large swollen mitochondria which lacked clear cristae (Figure 6A and B). The matrix ultrastructure was dense. In addition, the presence of gliosis as well as large-scale damage to the neurons, as revealed by the loss of nuclear membrane, axonolysis, vacuolation, and loss of synaptic integrity, which were indicative of advanced PD, were observed. The MF-exposed group also showed similar damage comparable to the PD rats, with the exception that mitochondrial fission (Figure 6C and D) was observed, which is indicative of regenerative processes coming into play to combat the extensive damage.
Representative Images of the Ultrastructure of Striatum and Substantia Nigra in PD (A, B) and PD + MF (C, D) Groups, n = 3.
Metabolic Profile
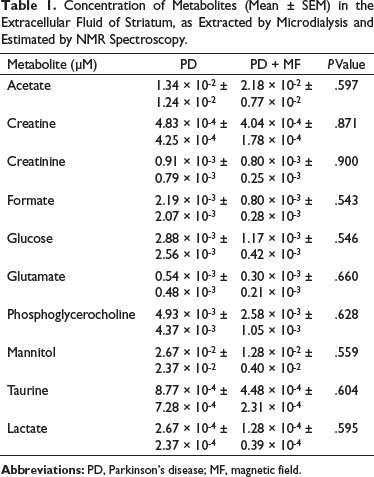
Microdialysis was performed to determine the metabolic profile of the striatal tissue. There was a decrease in the levels of glutamate in the striatum of PD + MF rats as compared to that in the PD group, although this decrease was not statistically significant (P = .66). The levels of the neuroprotective creatine and creatinine were not significantly altered by MF exposure (P = .87 and P = .90, respectively; Table 1).
Concentration of Metabolites (Mean ± SEM) in the Extracellular Fluid of Striatum, as Extracted by Microdialysis and Estimated by NMR Spectroscopy.
Discussion
PD is classically characterized by deficits in motor functions (bradykinesia, postural instability, rigidity, and tremors at rest) and cognition (dementia). To assess motor deficits, we performed open field, rotarod, and stride length tests. A significant decrease in the total distance covered and the velocity of the movement was observed following 6-OHDA administration in rats, suggesting bradykinesia. This reduction is consistent with previous studies.22, 23 A significant reduction in the latency of fall was observed at all speeds in fixed speed protocol as well as in the accelerating speed protocol following 6-OHDA administration, suggesting postural instability.24, 13 We also observed a reduction in stride length, an increase in the base of support, and a decrease of overlap between the forelimb and hind limb, which indicated the rats walking with short strides and wider stance for weight bearing. The behavioral data indicate an overall deficiency in motor functions of the PD rats which was consistent for the complete study period. The drug 6-OHDA mainly exerts its deleterious effects by oxidative stress, by the formation of quinones, p-quinones, and superoxides leading to mitochondrial dysfunction and inflammation. We therefore studied the mitochondrial ultrastructure, which showed round swollen mitochondria with loss of cristae. An increase in the expression of both OX-6 and 42 as the microglial markers in striatum was observed in PD rats. The loss of 76% dopaminergic innervations at the striatum coupled with the proliferation and activation of microglia was amply reflected by the altered metabolism as demonstrated by the metabolomics of extracellular fluid.25–27 Previous studies have also shown that an alteration in the neurotransmitter synthesis as a result of PD ultimately leads to reduced thalamic output, hence the manifestation of the motor symptoms, thus defining features of PD. 28
MF follows Faraday’s law and modulates the excitability of the neural tissue by inducing eddy currents. It also exerts its effect by nonpolarizing calcium-dependent pathways (independent of endoplasmic reticulum). We used low-intensity MF stimulation that could penetrate the skull and CSF to avoid the repercussion of high-intensity stimulations such as seizures. In this study, we used a modified Lee-Whiting coil, 15 which is a pair of Helmholtz’s coil that produces a uniform MF at the center of the coil, the intensity of which was 17.96 µT at 50 Hz. The rats were exposed for 2 h daily for four weeks, a paradigm that is consistent with previous studies in our laboratory. 12 We began MF exposure three weeks after the administration of 6-OHDA, as in most human PD cases diagnosis is only possible after frank motor symptoms are presented and therefore we presume that any disease-modifying effect of the interventions at this stage will be most beneficial. In striatal 6-OHDA rat model, the manifestation of motor symptoms takes approximately one to three weeks to develop.29, 30 The substantia nigra compacta (SNc) dopaminergic neurons have very high arborization and branching (200,000–300,000 connections) and thereby even a small number of neurons are able to innervate a large area of the striatum. 31 We therefore hypothesize that even a few surviving neurons of the SNc may be able to compensate for the functional loss by forming new connections. Thus this plastic nature of SNc neurons seems to be the key to the management of the disease (limiting factor) and not the number of neurons present. 32 In this study, we have delved into MF’s ability to facilitate the formation of new connections at the striatum by making the environment conducive to survival and formation of new connections. We observed that MF exposure of PD rats for four weeks resulted in a significant improvement in motor function in terms of postural stability and is consistent with the observations that rTMS can ameliorate the functional deficit in unilateral 6-OHDA rats, and decrease levels of proinflammatory cellular markers, i.e., COX 2 and TNF-α. 33 MF also exerts its beneficial effects by increasing the synthesis and turnover of dopamine and receptor reactivity of monoamine networks or their interaction with other networks. 4 Even prenatal exposure to MF has been shown to increase the dopamine level in the pup striatum, 34 indicating a positive influence of MF exposure on dopamine content. MF exposure was able to alter the metabolite concentrations. There was a trend for a decrease in the levels of all metabolites as compared to PD group namely; glutamate, glucose, creatinine, lactate, mannitol, and creatine in the lysate However, effects of MF are related to the severity of the disease; it becomes ineffective as the disease progresses and is unable to provide any beneficial effect once drug-induced dyskinesia sets in. 35 Human brain as well as the brain of other vertebrates possesses tiny membrane-bound vacuoles containing single domain magnetite crystals held within the lipid bilayer, known as magnetosomes. These structures render the plasma membrane and the mechanically sensitive ion channels present in them highly susceptible to external MF which alters the permeability of membranes and ion channels, thereby modulating the secretion and synthesis of various neurotransmitters and metabolites.36, 37
A greater sparing of dopaminergic terminals after MF exposure (31%) as compared to PD was observed. This could be because of the reduction in oxidative stress, inflammation, or release of neurotrophic factors. MF exposure has been shown to induce expression of various early immediate genes (c-fos and c-jun) and secretion of neurotrophic factors like BDNF, NGF, and other growth-promoting factors,38, 39 thereby creating neuroregenerative/protective trophic environment. Umarao et al. 13 have shown a decrease in the levels of the free radicals in the striatum in an acute bilateral 6-OHDA rat model of PD following MF exposure for seven days. MF has the ability to quench free radicals generated within the tissue. In this study, we observed a significant reduction in the number of activated microglia in PD + MF rats as compared to that in the PD group, but the effect of MF on mitochondria was not significant. This indicates that in advanced stages of PD, the mechanism for the beneficial role of MF in PD severity is through reduction of inflammation.
Morphological analysis of other brain areas, especially substantia nigra or frontal cortex, could have substantiated the results if it had been included as a part of the study. This study was an interventional one; the aim being to evaluate the efficacy and mechanism of action of MF, hence we compared the behavioral and cellular outcomes of intervention as compared to the disease group (without intervention) which we thought would serve as a control. We therefore did not include a non-PD sham group in the study design or protocol, which as it turns out is a major limitation. In retrospect, we feel that data of a sham group would have added to the interpretation of the results and helped us reach a definite conclusion.
Conclusion
We have developed a clinically relevant model of PD with robust motor symptoms. We confirm that MF exposure is able to reverse the postural instability and microglial activation in the same. Owing to the advanced form of the disease as evidenced by the ravaged cellular architecture, we were unable to reverse the TH innervation to the striatum. We hope to devise strategies based on nanomedicine that can enhance the effect of MF in PD. In conclusion, we can state that MF stimulation may be beneficial in mild or early stages of PD.
Footnotes
Acknowledgement
We thank Mr Pursottam Samal for histology slide preparations and brain sectioning.
Authors’ Contribution
SJ: Conceived the project and the majority of the laboratory work has been performed in her laboratory.
SB: Performed all experimental work and wrote draft.
TCN: Supported and interpreted histology data.
MS: NMR analysis and interpretation was done in her laboratory.
SD: Made graphs and drafted the manuscript.
Statement of Ethics
The study was approved by the Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), New Delhi, Animal Welfare Division under the Ministry of Environment, Forest and Climate Change (Government of India), and approved by the Institutional Animal Ethics Committee (Ethics No. 797/IAEC/12).
Declaration of Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Indian Council of Medical Research by a Senior research fellowship grant.
