Abstract
On 31 January 2020, World announced COVID-19 as an Emergency Public Health of International Concern. The number of patients in Indonesia continues to grow. Anti-viral in the COVID-19 Drug Information Laboratory in Indonesia are Lopinavir/Ritonavir, Favipiravir, Remdemsivir, Oseltamivir, Chloroquine Phosphate and Hydroxychloroquine Phosphate. Therefore, it is necessary to know the basis and management of its use. An online systematic search was performed on articles published until 30 March 2020. We use search keywords that are tailored to the purpose of writing. All six antivirals were used for the treatment of RNA virus. Chloroquine, Hydroxychloroquine and Remdesivir effectively control the SARS-CoV2 virus invitro. Lopinavir/Ritonavir, Hydroxychloroquine and Oseltamivir have been used clinically for the treatment of SARS-CoV2 virus. In 2020, there are 42 clinical trials of six antivirals. Guidance of the antivirus are from China, Belgium and Indonesia. Its differences are based on the patient’s condition. There is a lack of evidence of six antiviral effectiveness against the SARS-CoV2 virus. It has been used for other RNA viruses. It is supported by a safety profile. In a pandemic situation and the absence of a specific antivirus, the use of the six antiviruses can be done and be useful.
Introduction
In December 2019, cases of pneumonia were discovered in Wuhan, China caused by a novel betacoronavirus. The overall case fatality rate is around 2.3%. This fatality increased to 8.0% in patients aged 70–79 years and to 14.8% in those aged >80 years (Wu & McGoogan, 2020 The average time from the first symptom appearing to becoming acute respiratory distress syndrome (ARDS) was 8 days (Wang et al., 2020a). This transition occurs in many severe cases of COVID-19. This state is due to the occurrence of cytokine release syndrome, or ‘cytokine storm’, overproduction of immune cells and cytokines that lead to failure of multi-organ systems (lungs, kidneys and heart) which is fast (Shimabukuro-Vornhagen et al., 2018). As of 31 January 2020, the World Health Organization (WHO) announced that COVID-19 is registered as an Emergency Public Health Emergency of International Concern (PHEIC), which means that it can pose risks to many countries and requires coordinated international responses. As of 31 January 2020, the WHO announced that COVID-19 is registered as an Emergency PHEIC, which means that it can pose risks to many countries and requires coordinated international responses.
The number of COVID-19 patients in Indonesia has also grown. Therefore, immediate efforts are needed to provide effective treatment and to reduce the risk of transmission. Indonesian Drug and Food Control Agency (BPOM) has issued Drug Information Laboratory for COVID-19. There are six antivirals listed, namely Lopinavir/Ritonavir, Favipiravir (FPV), Remdemsivir, Oseltamivir, Chloroquine Phosphate and Hydroxychloroquine Phosphate. Because of the pandemic, it needs to study the articles that aimed at finding out the results of the six antivirus tests on virus infections other than SARS-CoV2 virus, knowing the results of the six antivirus tests due to SARS-CoV2 virus, knowing the six clinical trial plan of antivirus due to SARS-CoV2 in 2020 and knowing the guidance for using antivirus to treat SARS-CoV2 virus.
Methods
Online systematic searches were carried out in studies published until 30 March 2020. According to indexes from various databases, we used search terms with keywords: antiviral for SARS CoV2, antiviral for COVID-19, COVID-19 treatment, clinical for COVID-19 patients and its co-morbid. Antiviruses here include Chloroquine, Hydroxychloroquine, Lopinavir-Ritonavir, FPV, Remdesivir and Oseltamivir. We have checked the reference list for each selected article using the snowball method applied to the reference articles taken. This aims to broaden the references obtained. We also searched the Clinical Trial Registry for the Antivirus. It means to identify ongoing trials. We do not register as a systematic review protocol because of the limited available evidence and the urgency of this problem.
Result
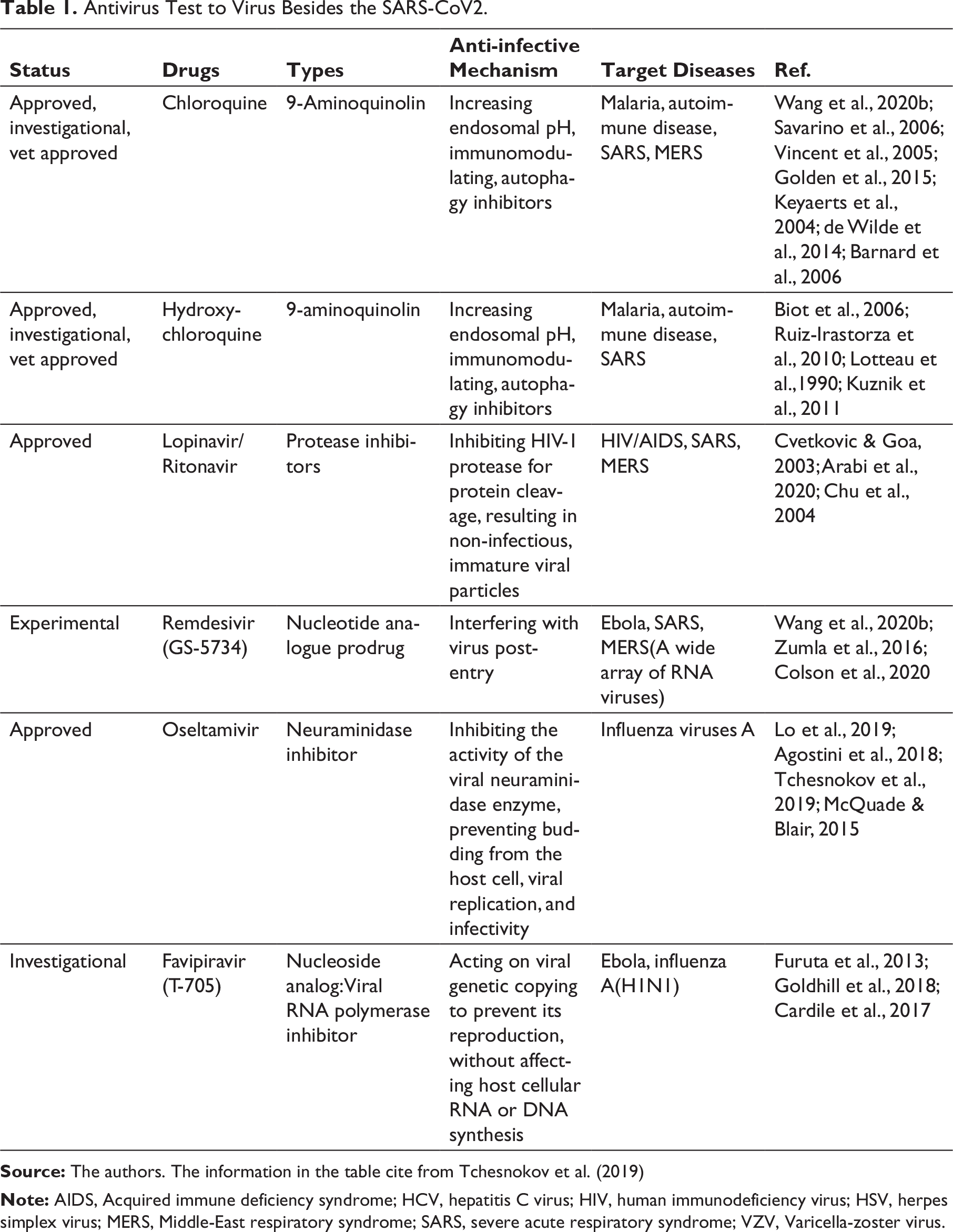
Based on Table 1, the six antiviruses have mode of action and mechanism against viruses. Chloroquine and Hydroxychloroquine have the same mode of action and effective virus mechanism because hydroxychloroquine is a derivative of chloroquine. Hydroxychloroquine is synthesised by inserting hydroxyl groups into chloroquine. Other antiviruses have different mechanisms. Dengue virus, SARS-CoV1, HIV, MERS, Ebola and Influenza A (H1N1) viruses are kind of RNA viruses. The SARS-CoV2 virus is also an RNA virus. Therefore, these antiviruses are also expected to be used for the SARS-CoV2 virus.
Antivirus Test to Virus Besides the SARS-CoV2.
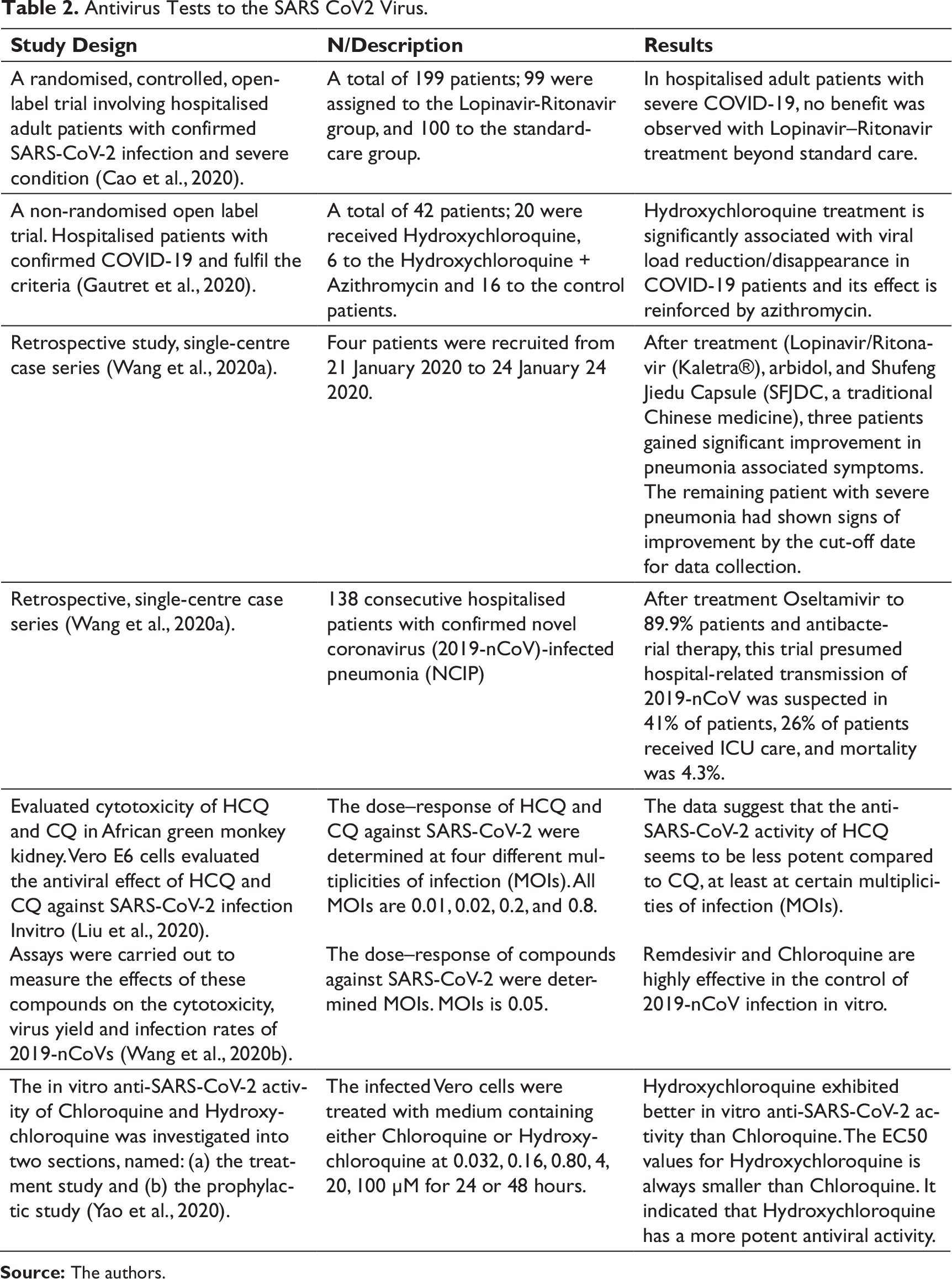
Based on Table 2, there are currently three invitro tests—Chloroquine, Hydroxychloroquine and Remdesivir. The results can be effective for controlling SARS-CoV2 virus. Based on Table 1, three antiviruses have been used for patient with SARS-CoV2 virus infection. These are Lopinavir/Ritonavir, Hydroxychloroquine and Oseltamivir. The use of Lopinavir/Ritonavir for severe patients was no different compared to standard care patients. Another study said the use of Lopinavir/Ritonavir combined with Arbidol and traditional Chinese medicine (Shufeng Jiedu Capsule) had a significant effect on patients with pneumonia. Studies show that the use of Hydroxychloroquine also had a significant influence to the improvement in patients. The addition of Azithromycin had a positive influence to the improvement in patients. The combination of Oseltamivir with antibacterial affects the transmission in hospitals. It is suspected to occur in 41% of patients, 26% of patients receiving ICU care, with 4.3% mortality.
Antivirus Tests to the SARS CoV2 Virus.
Based on Table 3, five clinical trials of Remdemsivir are present against SARS-CoV2. Ten clinical trials of Lopinavir/Ritonavir present against SARS-CoV2 virus. Four FPV clinical trials against SARS-CoV2 virus. Two clinical trials of Oseltamivir against SARS-CoV2 virus. Sixteen Chloroquine clinical trials against SARS-CoV2 virus. Five Hydroxychloroquine clinical trials against SARS-CoV2 virus. It has various purposes, for example, length of stay in hospital and so on.
Ongoing Antivirus Test to SARS CoV2 Virus (WHO’s list of studies for the treatment of COVID-19, 2020).
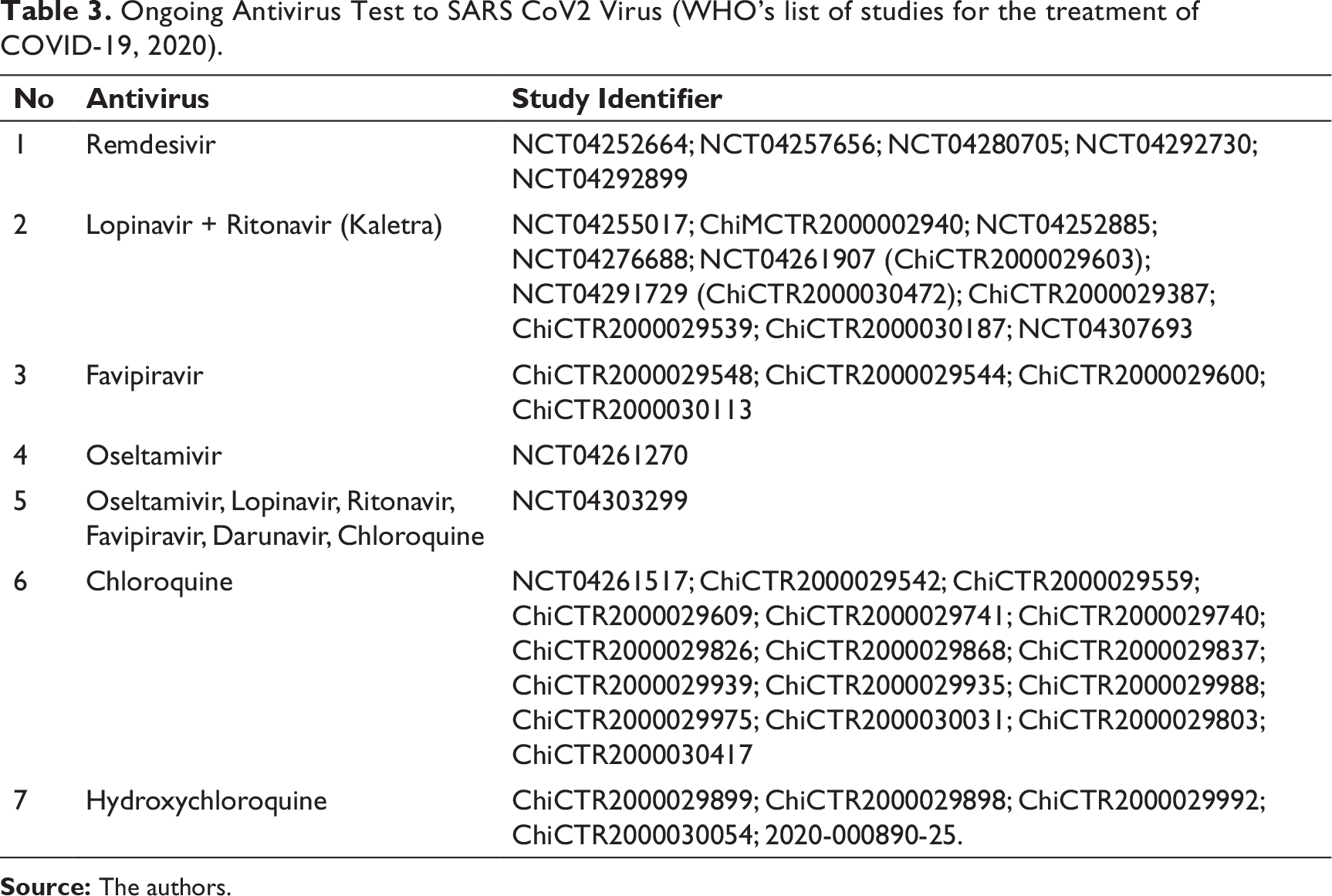
Table 4 contains three existing antiviral management for COVID-19. The first treatment is based on clinical experience of the first hospital affiliated with the medical school of Zhejiang University in China. They use Arbidol (Umifenovir) tablets, Lopinavir/Ritonavir tablets, interferon spray, immunoglobulin IV and thymic peptides inhalation. The second management treatment is an interim guideline in Belgium. They use Hydroxychloroquine tablet, Chloroquine tablet, Lopinavir/Ritonavir tablet and Remdesivir tablet. The third management treatment is based on compilation protocol that was made by representatives of the five Indonesian Doctors Associations. They use Chloroquine Phosphate tablet, Hydroxychloroquine tablet, Oseltamivir tablet and FPV tablet. There are differences in the use of antivirus. Differences in the clinical severity of patients cause differences in the use of antivirals and additional necessary support medications. The use of antivirus is also influenced by adverse effects and availability in hospitals.
Comparison of Guidance of Antiviral Treatment for Confirmed COVID-19 Patients (As of March 2020).

Risk group : age >65 years AND/OR underlying end organ dysfunction (lung, heart, liver, etc.), diabetes, coronapathy, chronic obstructive pulmonary disease and arterial hypertension Clinical clasification Mild/uncomplicated Uncomplicated patients with viral respiratory infections have non-specific symptoms such as fever, weakness, cough (with or without sputum production), anorexia, malaise, muscle aches, sore throat, mild tightness, nasal congestion and headaches. Although rare, patients can complain of diarrhoea, nausea or vomiting. Elderly and immunocompromised patients with atypical symptoms. Moderate Adolescent or adult patients with pneumonia but no sign of severe pneumonia and no need for oxygen supplementation. or children with non-severe pneumonia with coughing or breathing difficulties accompanied by rapid breathing. Severe/Severe pneumonia Adolescent or adult patients with fever or under inspection of airway infection/pneumonia, plus one of: breath frequency >30×/min, severe respiratory distress, or oxygen saturation (SpO2) <93% in room air or PaO2/FiO2 ratio <300. Or paediatric patients with coughing or breathing difficulties, plus at least one of the following: Central cyanosis or SpO2 <90%; Severe respiratory distress (such as snoring, heavy chest wall pull); Signs of severe pneumonia: inability to breastfeed or drink, lethargy or loss of consciousness or seizures. Other signs of pneumonia are: chest wall traction, tachypnoea: <2 months, ≥60×/minute; 2–11 months, ≥50×/minutes; 1–5 years, ≥40×/minutes; >5 years, ≥30×/minutes. Critical Patients with respiratory failure, ARDS, septic shock and/or multiple organ failure.
There are differences in the use of antivirus. Differences in the clinical severity of patients cause differences in the use of antivirals and additional necessary support medications. The use of antivirus is also influenced by adverse effects and availability in hospitals.
Discussion
Coronavirus is a RNA blanket virus from the genus Betacoronavirus. It can be found in birds, humans and other mammals. Six coronavirus species cause infections in humans, four of which (229E, OC43, NL63 and HKU1) cause flu symptoms. However, some authors claim that SARS-CoV2 is associated with SARS-CoV and MERS-CoV, which have zoonotic origins associated with significantly severe disease and higher mortality (Zhu et al., 2020).
Wang and Cheng’s (2020) report results research that SARS-CoV and MERS-CoV use ACE2 cell surface receptors in lung, heart, kidney and intestinal tissue. This process can accelerate the virus replication and spread. This binding triggers conformational changes in glycoprotein S that allows division by the transmembrane protease serine protease type-II (TMPRSS2) of the S protein and the release of S fragments into the cellular supernatant which inhibits the virus neutralisation by antibodies (Glowacka et al., 2011).
Coronavirus therapy can be divided into several specific pathways: some work on important enzymes or functional proteins viruses, prevent the synthesis and replication of viral RNA; some act on the structural protein of the virus, blocking the virus from binding to human cell receptors, or inhibiting the virus’s own assembly process; several virulence-producing factors to restore the innate host immunity; some act on host-specific receptors or enzymes, preventing the virus from entering the host cell (Wu et al., 2020).
Based on previous experiences in combating the SARS-CoV and MERS-CoV epidemics, we can learn several lessons for treatment strategies for coronavirus (Zumla et al., 2016). An efficient approach to drug discovery is testing whether existing antiviral drugs are effective in treating related viral infections (Wang et al., 2020b).
Chloroquine
An expert consensus was published on 20 February. It was based on in vitro evidence and clinical experience that has not yet been published. Chloroquine phosphate tablets at a dose of 500 mg twice daily for 10 days can be used for mild, moderate and severe pneumonia SARS-CoV2 without contraindications to the drug (Multicentre collaboration group of Department of Science and Technology of Guangdong Province and Health Commission of Guangdong Province for Chloroquine in the treatment of novel coronavirus pneumonia, 2020). According to the US CDC and US FDA, the optimal dose and duration of therapy are unknown (Coronavirus Disease—2019, 2020; US FDA, 2020). In the use of Chloroquine for malaria therapy, there is a small risk of macular retinopathy, which depends on cumulative dose (Bernstein, 1991), and the presence of several reports of cardiomyopathy as a severe side effect (ITS, 2020). Other side effects of Chloroquine Phosphate are dizziness, headaches, nausea, vomiting, diarrhoea and various types of skin rashes. The most severe adverse reaction is a heart attack. An electrocardiogram needs to be checked before taking medication. Medication must be prohibited for arrhythmia patients (e.g., conduction blocks), retinal disease, or hearing loss (ITS, 2020).
Hydroxychloroquine
Based on the Physiologically Based Pharmacokinetic Models (PBPK), a loading dose of 400 mg twice daily Hydroxychloroquine Sulphate given orally, followed by a maintenance dose of 200 mg given twice daily for 4 days is recommended for SARS-CoV2 infection. It reaches three times potency of Chloroquine Phosphate 500 mg twice for 5 days (Yao et al., 2020). According to US CDC and US FDA, the optimal dose and duration of therapy are unknown (Coronavirus Disease-2019, 2020). Hydroxychloroquine is an analogue of chloroquine which has less concern about drug interactions (Biot et al., 2006). Hydroxychloroquine side effects are GI intolerance, cytopenia, QT prolongation, headache, dizziness (Multicentre collaboration group of Department of Science and Technology of Guangdong Province and Health Commission of Guangdong Province for chloroquine in the treatment of novel coronavirus pneumonia, 2020). Hydroxychloroquine is preferred over Chloroquine due to its lower ocular toxicity (Lim et al., 2009).
Lopinavir/Ritonavir
The use of Lopinavir (400 mg)/Ritonavir (100 mg) is two tabs orally q12h. Based on reports, it can be used up to 14 days. Most patients showed side effect symptoms that require early withdrawal (Tian et al., 2020; Young et al., 2020). Lopinavir/Ritonavir is metabolised through the CYP3A enzyme in the liver. It needs to monitor potential drug interactions. This has been proven by 51.9% incidence of abnormal liver function which has received combination of Arbidol and Lopinavir/Ritonavir. Therefore, unnecessary drug combinations must be reduced. Side effects of Lopinavir/Ritonavir and Darunavir/Cobicistat are diarrhoea, nausea, vomiting, increased serum aminotransferase, jaundice, dyslipidaemia and increased lactic acid. These symptoms will recover after stopping the drug. This medicine may be used for pregnant women (ITS, 2020).
Oseltamivir
Chen et al. (2020b) reported that the dosage of Oseltamivir in the case series of 99 patients was 75 mg orally every 12 hours. Oseltamivir is contraindicated for children and adolescents, 10–19 years, in principle since March 2007, due to concerns that it may cause abnormal behaviours (Maxwell, 2007). Nausea and vomiting were demonstrated as adverse reactions to Oseltamivir in treatment trials involving both adults and children and are the established adverse reactions to Oseltamivir. Dose-dependent increase of headache was demonstrated only in prophylaxis trials because common symptoms of influenza including headache were not considered adverse event in the treatment trials (Jefferson et al., 2014).
Remdesivir
Until now, the optimum dose and duration of remdesivir therapy are unknown. Based on phase 3 clinical trial protocols in severe COVID-19 patients, remdesivir is used: 200 mg IV on day 1, then 100 mg IV daily on days 2–5 (arm 1) or 200 mg IV on day 1, then 100 mg IV daily on days 2–10 (arm 2) (Clinical Trials, 2020); 200 mg IV on day 1, then 100 mg IV daily on days 2–10 (Clinical Trials, 2020). Based on the compassionate use access protocol, remdesivir was used: 200 mg IV on day 1, then 100 mg IV on days 2–10 (Grein et al., 2020). Transaminase increases are presently the only adverse effect that appears clearly linked to remdesivir’s use. They need special attention to elevations of transaminases, hypersensitivity reactions, renal events, pregnancy and unexpected adverse reactions in the periodic safety reports and the expedited summary safety reports (European Medicines, 2020). Another article states that no adverse events were observed (Holshue et al., 2020).
Favipiravir
Chen et al. (2020a) state that a FPV dosage of 1,600 mg twice daily on day 1, then 600 mg twice daily thereafter for 7–10 days was used in one open-label COVID-19 study. Another guideline states that FPV’s starting dose was of 1,600 mg followed by 600 mg three times a day (tid) (ITS, 2020). In a study conducted by Cai et al. (2020), the total number of adverse reactions in the FPV study group was four (11.43%), which was significantly less than 25 adverse reactions (55.56%) in the control group (P <.001). Two patients had diarrhoea, one had a liver injury, and one had a poor diet in the FPV group. Meanwhile, there were five patients with diarrhoea, five with vomiting, six with nausea, four with rashes, three with liver injuries and two with chest tightness and palpitations in the control group. FPV is prohibited for use in pregnant women (ITS, 2020).
The success of COVID-19 therapy is also influenced by the patient’s condition and disease severity. Older patients and those who have comorbidities, such as cardiovascular disease and diabetes mellitus, have an increased risk of severe illness and death. They may present with mild symptoms but have a high risk of damage and must be admitted to an appropriate unit for close monitoring (WHO, 2020). A median number of days from the first symptom to death is shorter among people aged 65 years or older (Wang et al., 2020c; Yang et al., 2020). Similar to H7N9 (Gao et al., 2013), elderly male patients with comorbidities and ARDS show a higher risk of death.
The success of therapy is also influenced by complications experienced by patients. Wang et al. (2020a) report that among 138 hospitalised patients with COVID-19, 7.2% had an acute heart injury and 16.7% had arrhythmias. In a smaller group of 41 patients, 12.1% had an acute heart injury (Huang et al., 2020). In a cohort study of more than 416 patients, cardiac injuries occurred in 19.7% of patients during treatment in hospital (Grein et al., 2020). In another cohort of 191 patients with COVID-19, 17.2% of patients had a heart injury (Zhou et al., 2020). A meta-analysis of six studies published from China, including 1,527 patients with COVID-19, reported that 8% of patients had a heart injury (Li et al., 2020).
COVID-19 is a viral infection, therefore antibiotics are not recommended to prevent bacterial infections in mild or ordinary patients; it must be used with caution in severe patients based on their condition. Some COVID-19 patients have risk of secondary fungal infections because of weakened cellular immunity due to viral infections, use of glucocorticoids and/or broad-spectrum antibiotics. It is necessary to carry out microbiological detection of respiratory secretions such as smear and cultivation for critically ill patients; and provide D-Glucose (G-test) and galactomannan (GM-test) blood or bronchoalveolar lavage fluid for suspected patients (ITS, 2020).
Conclusion
Six antivirals have been used in the treatment of infections caused by RNA viruses. They are also potent for the treatment of COVID-19 disease caused by the SARS-CoV2 virus which is also an RNA virus. They also have safety profile. Because of pandemic situation and no specific antivirus found, the six antiviruses can be of use. There are differences in the use of antivirals cause by patient severity. The difference in severity also causes additional necessary support medications. The use of antivirus is influenced by its availability in hospitals and its presence or absence of adverse effects.
Suggestion
The use of six antiviruses must be supported by comprehensive observation of the effectiveness of therapy, side effects, drug interactions and others. Therefore, it needs collaboration between doctor, pharmacist and nurse to obtain the optimum effect.
Footnotes
Acknowledgement
We thank Mr Hendrianto Trisnowibowo, Ms Yuslely Usman, Ms Yuyun Yuniar and Mr Herianto Lahing who have been a discussion partner.
Authors’ Contribution
Lukman Prayitno is the main contributor in making this article. Other authors help to prepare data. They are member contributors.
Declaration of Conflicting Interests
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
