Abstract
Over the years, rival companies have appropriately turned their attention towards effectively monitoring their supply chains. There is an emphasis on implementing supply chain management practices (SCMP) and promoting supply chain agility, which will lead to increased performance and competitive edge. The researchers covered SCM practices and competitive advantage under five sub-constructs while SCM agility and performance were represented by two sub-constructs. The study aimed to provide a questionnaire that is both reliable and valid, designed to measure the impact of supply chain management (SCM) practices and SCM agility on competitive advantage and firm performance within the Indian pharmaceutical industry. Data collection was done using a simple random sampling technique which gave 227 complete responses obtained from listed as well as unlisted pharmaceutical companies. The data was then statistically analysed using IBM SPSS 26 and SmartPLS 4. The resultant questionnaire instrument can be used by supply chain professionals in the pharmaceutical industry in order to conduct primary research and to derive insights that would help in crucial decision-making regarding pharmaceutical SCM with the aim of improving organizational performance.
Keywords
Introduction
Over the last 20 years, the global trade value of pharmaceutical products has increased more than sixfold, and supply chains have become more interconnected and complex globally (Sahoo et al., 2022). As a major player in the global pharmaceutical sector, India is passing through a compelling phase. The companies in the country are boldly moving forward on the growth path with the ambition to establish themselves as a hub of cost-effective manufacturing and research and development. In response to competition from both domestic and multinational counterparts, Indian pharmaceutical companies are taking a proactive approach to diversify and strengthen their portfolio. This includes taking decisive steps such as enhancing capabilities to produce more sophisticated and differentiated generics, investing in innovative ventures and expanding the capacity to manufacture biosimilars(Chattopadhyay & Bercovitz, 2020). The pharmaceutical industry is one of the most regulated sectors in the economy, with stringent compliance requirements throughout its entire lifecycle, from drug development to post-marketing surveillance(Chakraborty & Venkatraman, 2023).
India has the distinction of being the world’s third-largest producer of pharmaceutical products and has earned the nickname ‘Pharmacy for the World’, and to live up to this designation and expand its global reach, it is imperative to establish and maintain a strong supply chain management (SCM) (Jha & Sharma, 2020). The pharmaceutical supply chain serves as a vital medium through which medicines are made available to end consumers, ensuring efficient and reliable distribution of pharmaceutical products around the world (Panda & Satapathy, 2021). Therefore, there is an urgent need to streamline the supply chain and ensure its resilience, making it shock-proof or minimally affected during situations of uncertainties (Ciger et al., 2022). According to Shah (2004), time-to-market is the prime driver in the pharmaceutical supply chain. As per the existing literature, only a few of the recent studies have delved into the consideration of upstream as well as downstream aspects of the pharmaceutical supply chain. These studies collectively contribute to addressing various diverse and interesting aspects of SCM practices—customer relationship practices and organizational performance; effects of supplier-customer integration on performance; impact of customer–supplier integration on organization performance and the orientation of supply chain and SCM.
Effective SCM involves several activities that are collectively known as supply chain management practices (SCMP). These include strategic supplier partnerships (SSP), customer relationships (CR), information sharing levels (IS), information sharing quality (IQ), postponement (POS), goal congruence (GC) and just-in-time (Gandhi et al., 2017; Li et al., 2006). Supply chain agility (SCA) refers to the organization’s ability to respond rapidly to changing circumstances with minimal time delay (Gerwin, 2005). Several empirical studies have indicated that both SCMP and SCA have a positive effect on performance and competitive advantage (Abdallah et al., 2021; Chen, 2019; Manzoor et al., 2022; Thongrawd et al., 2020; Waqas et al., 2022). It is also reported in the literature that competitive advantage has a positive influence on the organization performance (Flynn et al., 1995; Li et al., 2006; Luthans & Youssef, 2004). Organization performance is defined as the degree to which an organization is able to achieve its pre-defined financial and market objectives (Li et al., 2006).
The absence of a thorough understanding of SCM practices hampers the studies’ capacity to provide comprehensive and broadly applicable suggestions on SCM practices and further research. The present study focuses on instrument validation and reliability of the construct developed to study the influence of SCM practices and agility on the performance and competitive advantage of pharmaceutical firms. SCM practices have been represented by five sub-constructs that is, strategic supplier partnership (SSP), customer relationship (CR), quality of information sharing (IQ), level of information sharing (IS) and postponement (POS). Competitive advantage is also represented by five sub-constructs namely price, product quality (PQ), delivery dependability (DD), product innovation (PI) and time-to-market (TTM). Organization performance has two sub-constructs that is, financial performance (FP) and market performance (MP). SCM agility is represented by organizational agility (OA) and SCA. An instrument questionnaire was designed and developed using a Likert scale (five-pointer) to test the proposed framework and hypothesis. Responses were collected from pharmaceutical industry professionals involved in supply chain management. A total of 227 usable responses were obtained which were statistically analysed using IBM SPSS 26 and SmartPLS 4 to check for reliability, validity and to draw conclusions. The findings of the study will benefit pharmaceutical supply chain professionals by providing strategic insights and aid in decision-making. This article aims to provide supply chain professionals with a reliable and valid instrument questionnaire to measure SCMP, SCMA, CA and OP in pharmaceutical supply chains.
Literature Review
SCM Practices
SCMP refer to the set of initiatives and strategies that an organization adopts to promote and enhance the efficiency, effectiveness and overall performance of its supply chain processes (Li et al., 2006). SSP is a long-term association of the firm with its suppliers that is established with an intent to leverage the operational as well as strategic capabilities of both firms for overall benefit (Faruquee et al., 2021; Qrunfleh & Tarafdar, 2013). This type of collaboration goes beyond the traditional buyer-supplier relationship and involves a long-term, mutually beneficial collaboration between a firm and its suppliers, for example, such participation at early stages in the process of designing the product presents multiple options on effective cost-saving, product designs and technology(Alshurideh et al., 2023; Siagian et al., 2021). Establishing and nurturing lasting relationships with customers is a fundamental aspect of customer relationship management (CRM) (Aripin et al., 2024). Customer Relationship includes a set of practices that are used to build lasting customer relationships, manage customer queries and those that improve the overall customer satisfaction and customer loyalty (Gil-Gomez et al., 2020; Khan et al., 2022). The level of information sharing (IS) can be explained as the level to which an organization shares exclusive and critical data with its partners (Alzoubi & Yanamandra, 2020; Pan et al., 2023). The currency of information is often important, especially in rapidly changing environments (Dwivedi et al., 2020). Shared information is said to have high quality depending on the level of how timely, accurate, adequate, reliable and credible the information passed is (Htay et al., 2022; Moberg et al., 2002; Monczka et al., 1998). Postponement refers to the intentional delay of certain activities, operations or processes until a further juncture (Prataviera et al., 2020). This strategy adds flexibility and responsiveness to the entire supply chain (Beamon, 1998; Johnson & Davis, 1998; Rau et al., 2021; Van Hoek et al., 1999). Organizations that actively deploy supply chain management practices are reported to have a competitive edge as well as enhanced market, financial and overall organizational performance (Al-Madi et al., 2021; Cahyono et al., 2023; Jawabreh et al., 2023; Li et al., 2006).
Competitive Advantage
Competitive advantage refers to the distinct superiority and competencies which equip an organization to outnumber its competition and attain impeccable results through differentiation and critical decisions (Lakhal, 2009; Nu’man et al., 2020). The effectiveness and efficiency of a supply chain can impact various aspects of a business, including cost, speed, flexibility and customer satisfaction and drive competitive advantage for the firms (Asamoah et al., 2021; Davis & Spekman, 2004; Maaz & Ahmad, 2022; Madhani, 2020). Organizations are able to achieve competitive advantage by focusing their efforts on SCM which acts as a quintessential factor (Afraz et al., 2021; Schneller & Smeltzer, 2006). Competitive advantage can be measured by indicators that reveal past performance as well as potential competitiveness(Sachitra, 2017). Supply chain personnel can also create a competitive advantage by means of the capabilities and competencies they bring into the team (Salgado et al., 2022). Permitting the involvement as well as accepting and acting on the inputs of suppliers and other stakeholders brings in key insights and hence an edge over its competition (Wang & Yang, 2021). Organizations that have competitive advantage have a substantial positive impact on the overall financial as well as market performance (Azeem et al., 2021; Chatterjee et al., 2022; Ferreira et al., 2020; Lestari et al., 2020; Nu’man et al., 2020).
Organizational Performance
The improvement in organizational performance (OP) is attributed to the financial and market-related initiatives implemented by the organization (Akintokunbo, 2018; Kiu & Chan, 2024). Simply put, OP arises from the combined impact of both non-financial and financial factors that assess the achievement of results relative to predetermined targets in line with objectives and organizational goals (O’Boyle & Hassan, 2013; Raval et al., 2020). Indicators such as investment returns (ROI), profitability percentage, overall share of the market, sales and ROI growth, increased market share and overall position of the firm are employed to measure organizational performance (Bilan et al., 2020; Lee et al., 2022; Singh et al., 2016; Utomo et al., 2023). Effective SCM enhances financial performance and, as a result, generates value for shareholders by leveraging control over key performance indicators such as working capital, revenues and cost of capital (Del Giudice et al., 2021; Ellinger et al., 2011; Ivanov, 2021; Khanfar et al., 2021). Organizational effectiveness, as a broader concept, extends beyond financial indicators and includes assessment of factors such as degree of customer satisfaction, operational efficiency and corporate social responsibility (Richard et al., 2009; Zhang et al., 2020). Studies have found that SCM practices have shown to exert a positive influence on organizational performance (Kumar et al., 2020; Lee et al., 2022; Saragih et al., 2020; Shahzad et al., 2020; Younis & Sundarakani, 2020).
Supply Chain Management Agility
SCA relates to a firm’s ability to rapidly modify its approach to address rapidly evolving supply chain demands, particularly in purchasing, inventory management and distribution (Shukor et al., 2021). Organizations with a high level of SCA can efficiently navigate changes, keep pace with changing market conditions and take advantage of emerging business opportunities (Miceli et al., 2021; Patel, 2023). The extent of SCM agility is closely linked to the effectiveness of the overall SCM (Abdallah et al., 2021). SCA is an integral function that acts as a mediator between the demand and supply side and operational performance (van Hoek et al., 2022). Agility in a supply chain can be ensured by incorporating factors like quickness, competency, responsibility, flexibility and cost efficacy (Al-Zabidi et al., 2021; Patel, 2023; Shukor et al., 2021). Continuous changes that occur in market situations have created a need for supply chains to become more responsive by leveraging new-age technologies and operation strategies (Aitken et al., 2002). The capacity of organizations to maintain innovativeness is influenced by the agility throughout its supply chain (Al-Zabidi et al., 2021; Chen, 2019). SCA boosts financial, non-financial, operational and overall organizational performance (Al-Zabidi et al., 2021; Chan et al., 2017; Chen, 2019; Gligor et al., 2015; Miceli et al., 2021; Shukor et al., 2021; Swafford et al., 2006; Yang, 2014).
Research Framework of the Study
Research Framework.
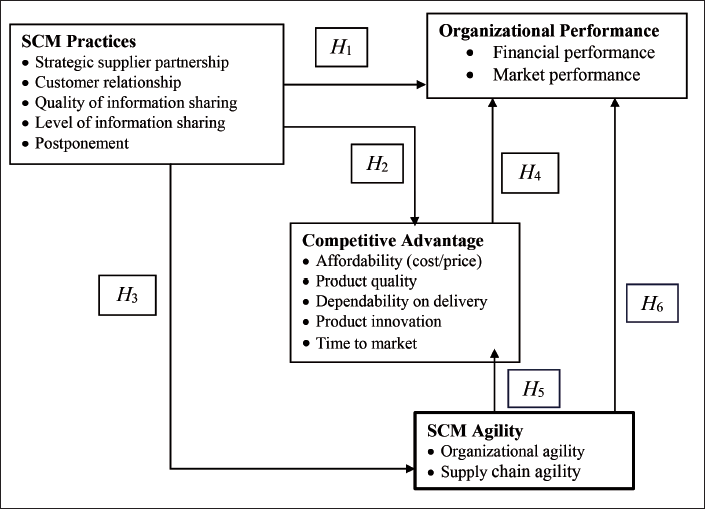
Research Methodology
Conducting a thorough literature review served as the basis for developing an instrument questionnaire, conceptualized on a Likert scale (five-point) with values ranging from five (strongly agree) to one (strongly disagree). The questionnaire covers four key domains: supply chain management practices (SCMP), supply chain management agility (SCMA), organizational performance (OP) and competitive advantage (CA) (Figure 1).
The aim of the research was to include participants who should have extensive knowledge about the functioning of supply chains within their organizations only managers with higher designations were considered to fill out the questionnaire. The instrument containing the questions and a cover letter that mentioned the importance and purpose of this study were sent via email to 750 pharmaceutical companies in India. The data of listed pharmaceutical companies in India was obtained through the official websites of the Bombay Stock Exchange and the National Stock Exchange.
The questionnaire consisted of a total of 63 items that were subjected to pre-testing to ensure content validity (Forza, 2002). The constructs and sub-constructs thereof for SCMP, OP and CA were taken from (Li et al., 2006) while for that for SCMA were given by the author’s understanding on the subject through the literature review. The instrument was analysed for its content validation through a panel of four academic professionals and as per their suggestions, the necessary changes were incorporated. A pilot survey was then conducted with 10 responses collected from pharmaceutical companies in Goa by means of personal visits, to test the appropriateness of the instrument (Green, 2020). Owing to limited access to supply chain professionals at managerial levels, a simple random sampling method was used for data collection (Amaya et al., 2015). A total of 227 complete useful responses were received and used for the analysis. The rate of response was found to be 30.26%. This low rate of response is because the respondents considered in this study at managerial levels have time as well as availability constraints. Rigorous statistical analysis was conducted by using the software IBM SPSS 26 and SmartPLS 4 to determine the reliability as well as validation of the instrument questionnaire.
Constructs and the Sub-constructs Included in the Study.
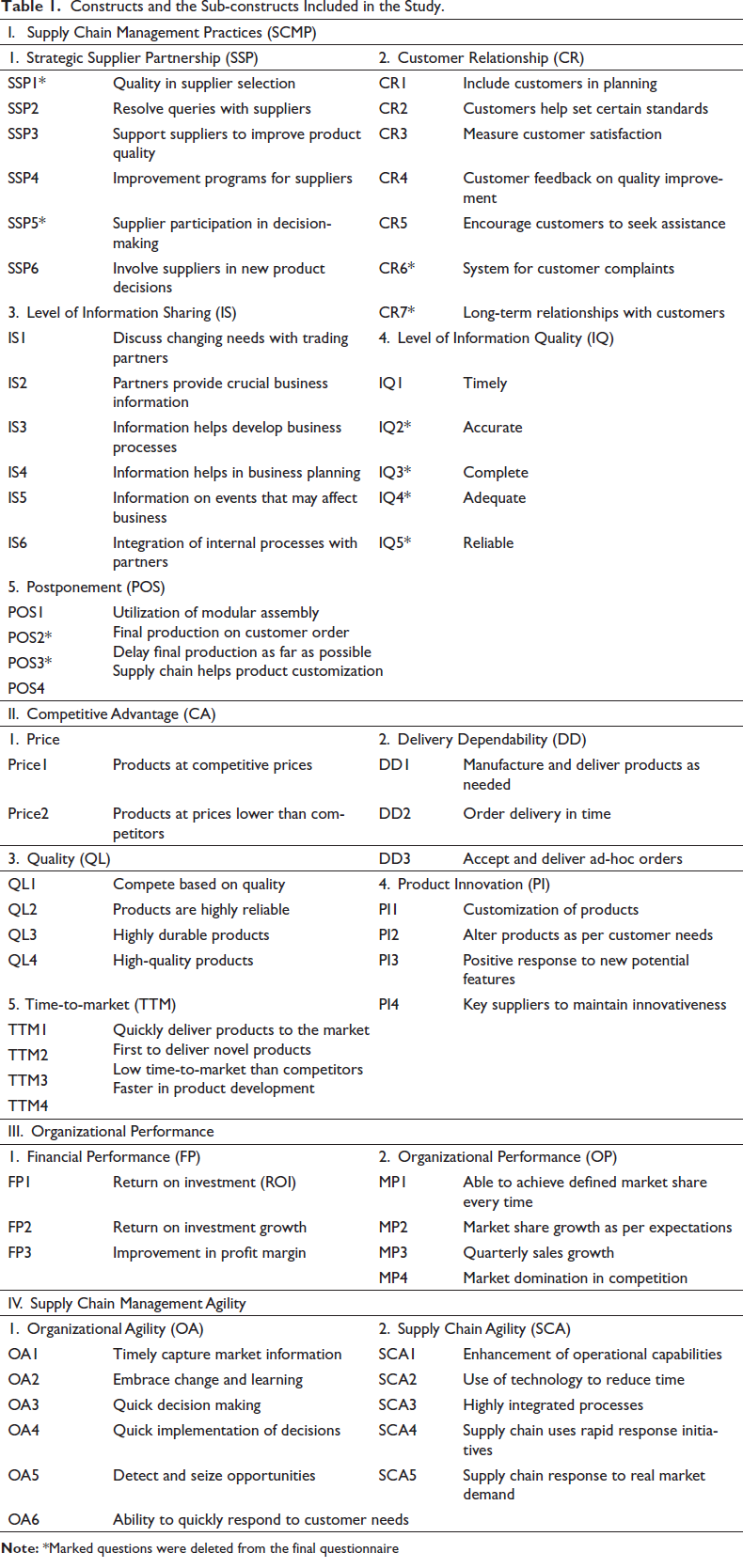
Table 1 depicts the constructs along with its sub-constructs that have been included in the study.
Data Analysis and Interpretation
Assessing the Measurement Model
Content Validity
Content validity relates to the suitability of measurement items employed to assess the domains covered in a study through a data collection instrument (Lynn, 1986). Extensive examination of the literature, along with feedback from academic experts and industry professionals in relevant fields, constitutes the most commonly employed approach to ensuring the content validity of the instrument (Polit & Beck, 2006). Content validation of the instrument questionnaire was conducted through a thorough review of the literature on SCMP, SCMA, OP and CA in the supply chain. Content validation of the instrument was ensured by a panel of four academic experts whose suggestions were incorporated. A pilot survey was carried out over 10 respondents to ensure content validity in the questionnaire. The industry professionals working in supply chain management and allied areas who responded to the questionnaire did not face any practical difficulties in understanding and responding to the questions nor did they suggest any major changes. Thus, content validity was established in the instrument questionnaire.
Uni-dimensionality
It refers to a condition wherein an entire set of items/sub-constructs are explained by one and only one major construct (Hair et al., 2014). It ensures that every item has a significant association with its own construct and that is related to only a single construct (Chen & Paulraj, 2004). Exploratory Factor Analysis was performed to check for uni-dimensionality. According to (Hair et al., 2017) the threshold value of factor loadings for significant consideration is 0.5 and those items that either demonstrate values below 0.5 or show cross-loadings with more than one construct are deemed fit for deletion.
Factor Analysis
Factor Analysis for SCMP, SCMA, OP and CA.
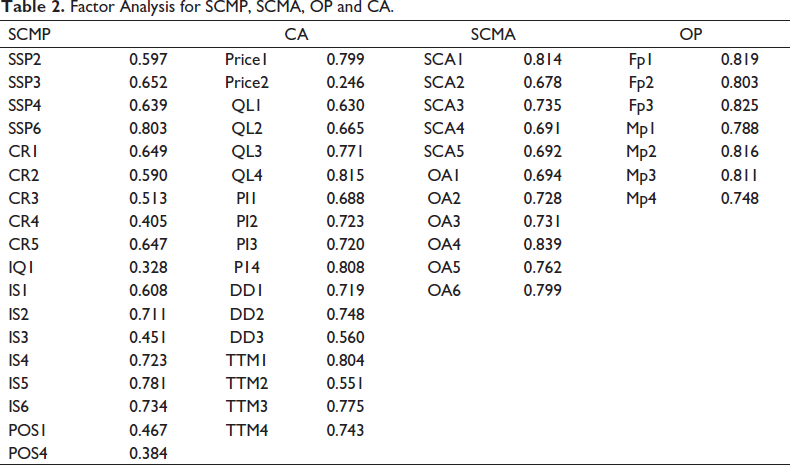
Table 2 is related to analysis of the sub-constructs belonging to SCMP, SCMA, OP and CA and the output is shown.
SCMP construct consisting of 28 items intended to measure five dimensions were factor analysed. It was observed that SSP1, SSP5, CR6, CR7, IQ2, IQ3, IQ4, IQ5, POS2 and POS3 either exhibited cross-loadings or values below 0.50 and hence had to be excluded. The CA construct contained 17 items intended to measure five dimensions while OP and SCMA had 7 and 11 items, respectively to be measured over two dimensions. Considering the literature, the importance of the items for measurement and their loadings well above the threshold, no items were deleted from CA, OP and SCMA. Though IQ1 and Price2 failed to meet the criteria, they were not deleted since literature suggested that information quality and price play an important role in competitive advantage (Baqleh & Alateeq, 2023). CR4 is an important measure as customer feedback is essential in order to understand and cater to the existing market demand and in quality improvement (Alzoubi & Yanamandra, 2020). IS4 was not deleted as certain proprietary information helps in crucial business planning and decision-making (Mc Loughlin et al., 2023). POS1 and POS4 were not deleted as they had the highest loading among the postponement sub-constructs and to understand the relevance of modular assembly and product customization in the Indian pharmaceutical sector. All the other values were found to be well above the threshold. Hence, according to Wang et al. (2006) convergent validity and uni-dimensionality is confirmed.
Multicollinearity Statistics (VIF) for Indicators
Variance inflation factor (VIF) helps recognize the extent of collinearity issues in the dataset. A dataset is said to not have any multicollinearity problems when the VIF values are above 0.1 and below 10 (García et al., 2015). As evident from Table 3, all the VIF values are below 10 hence confirming the absence of multicollinearity issues.
Variance Inflation Factor (VIF).

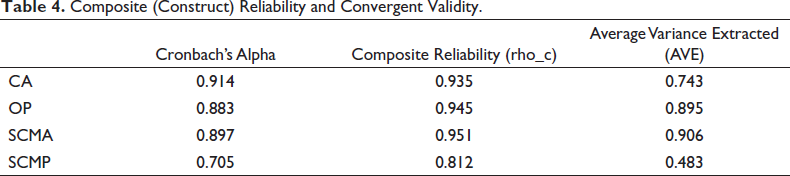
Construct Reliability and Convergent Validity
Reliability is important because it measures the quality of research. Reliability analysis, specifically internal consistency, was conducted using Cronbach’s alpha on each dimension—SCMP, CA, OP and SCMA (Cronbach, 1951).
According to (Cronbach, 1951), reliability values of 0.60 and above lie in the acceptable range. As evident in Table 4, the alpha value for every construct is above 0.70, which is considered acceptable. Since Cronbach’s alpha explains the internal consistency, it can be inferred that the level of internal consistency in the questionnaire instrument is acceptable. Construct validity can also be confirmed using Average Variance Extracted, wherein outcomes exceeding 0.50 exhibit above 50% of variance (Shahbaz et al., 2019). Therefore, it can be concluded from Table 4 that construct reliability and convergent validity exist in the questionnaire instrument.
Composite (Construct) Reliability and Convergent Validity.
Discriminant Validity
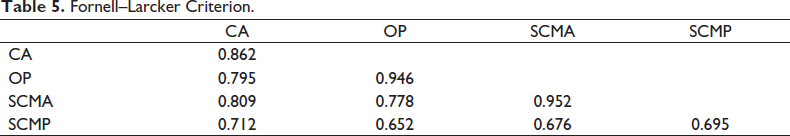
Fornell–Larcker Criterion
As per the criterion, the diagonal elements should have the highest values within its own construct (Fornell & Larcker, 1981). In the Table 5, the diagonal values are above 0.5 and are the highest among its own construct.
Fornell–Larcker Criterion.
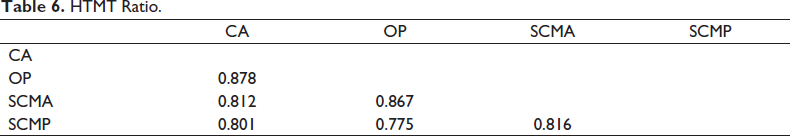
Heterotrait–Monotrait (HTMT) Ratio
All the HTMT values in Table 6 were found to be below or near to 0.85 which is acceptable (Yusoff et al., 2020). We can hence conclude the absence of discriminant validity issues.
HTMT Ratio.
Measurement Model.
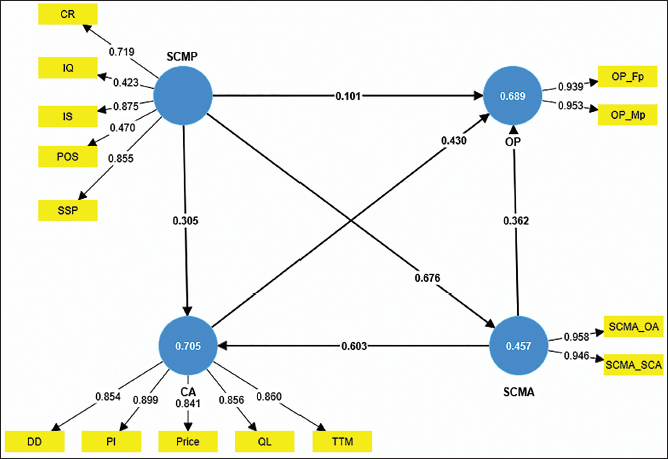
There exists a good degree of interactions and inter-relations between the constructs in the second-order model, thus confirming the validity, reliability and effectiveness of the questionnaire, its responses as well as the data analysis.
Assessing the Structural Model
Covariance-based structural equation modelling was conducted on SmartPLS 4 for determining the model fit.
Structural Model.
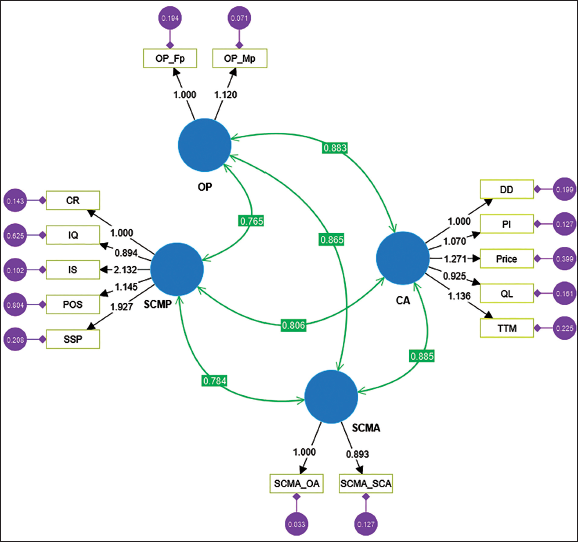
The results exhibit significant loadings that correspond to its construct (second-order). As can be confirmed from Table 7, the overall model has shown a satisfactory fit (Hair et al., 2017).
Results of the Structural Model

Figure 2 is the output related to SEM analysis using SmartPLS4 explaining the impact of independent constructs on the dependent constructs.
Figure 3 is the result of SEM analysis which explains structural model of covariance-based structural equation modelling analysis (CB-SEM).
Managerial Implications
The study provides a reliable and valid questionnaire instrument to test the impact of supply chain management practices and supply chain management agility on competitive advantage and organizational performance of Indian pharmaceutical firms. Professionals working in the field of supply chain can make use of this questionnaire to conduct primary market research in order to evaluate the impact of the constructs and its sub-constructs on CA, OP and SCMA. The insights gained in terms of identifying positive and negative points will help in making informed decisions, thereby increasing the effectiveness of the pharmaceutical supply chain.
Conclusions
The purpose of this article was to conduct validity and reliability tests of the instrument questionnaire that measures SCMP, SCMA, CA and OP among Indian pharmaceutical firms. The scale development was carried out as per the standards set by (King & Sethi, 1999; Rao et al., 1999). The study has managed to develop an authentic questionnaire instrument that can be used empirically by supply chain managers and professionals linked to logistics in order to evaluate the impact of SCMP, SCMA on CA and OP. The resultant questionnaire is reliable, valid and unidimensional in nature. The scale has been tested through the measurement model assessment as well as the structural model assessment and has been found to be satisfactory (Hair et al., 2017). The extension of this scale can be utilized to measure the indirect and direct effects of SCMP and SCMA on CA and OP. Future studies can incorporate other dimensions of SCMP, SCMA, CA and OP for the development and validation of a similar construct. The scale developed through this study can be also subjected to similar studies in different geographical locations.
Appendix A. Questionnaire Instrument for SCMP, SCMA, CA and OP.
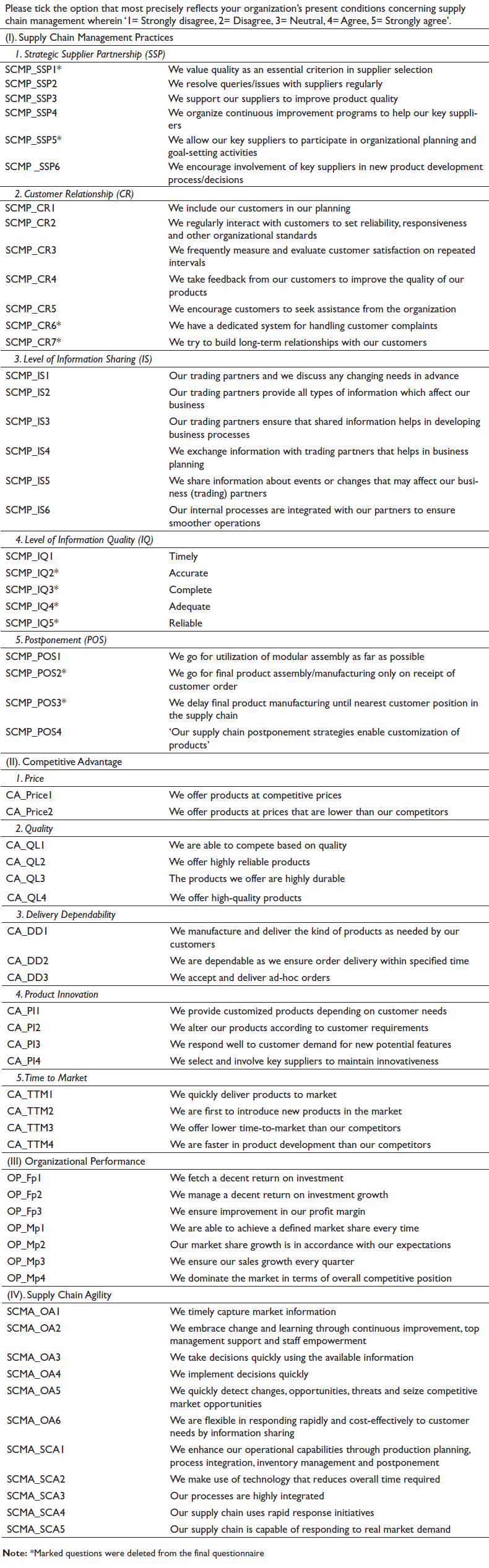
Appendix A is related to questionnaire that was used to collect responses from supply chain professionals from the Indian pharmaceutical industry.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
