Abstract
The personal identification of unknown individuals continues to pose a significant challenge in the aftermath of criminal activities and mass disasters. Saliva is among the most frequently encountered biological fluids, as the oral cavity readily comes into contact with various surfaces, making it a valuable source of forensic evidence. The present study aimed to assess sex differences and age-related variations in total salivary protein levels. This in vivo prospective study included a total of 120 participants. Approximately 5 mL of whole unstimulated saliva was collected from each subject and subsequently centrifuged at −20°C. The resulting supernatant was carefully aspirated, and the aliquots were stored at −20°C until the biochemical assay was performed. The salivary protein quantification was done by the Bradford assay. Statistical analysis was done using Stata version 17. A highly significant statistically difference in protein concentration was seen between age groups (p < .0001), with lower values reported in the geriatric population. A highly statistically significant difference in protein concentration was seen between males and females (p < .0001), with higher values reported in females. A statistically significant difference in protein concentration was seen among age groups (p = .026), with lower values reported in the post-dental eruption group. The total salivary protein concentration can predict gender with a specificity of 23% and a sensitivity of 65%. It can be predicted whether an individual would belong to the geriatric population or other age groups with a specificity of 83% and a sensitivity of 73%. This shows that salivary total protein has limited predictive strength and can be used as an adjunct to the existing forensic identification methods.
Introduction
Saliva is a complex biological fluid characterized by its viscoelastic properties and plays a crucial role in the maintenance of oral health. Its functional capacity is determined by its biochemical composition and rheological characteristics, which collectively contribute to essential processes such as lubrication and moistening of oral tissues, maintenance of microbial homeostasis, protection against pathogens, initiation of digestion, bolus formation and clearance, taste perception, buffering of oral pH, mineralization of dental structures, and facilitation of wound healing. 1
Saliva as an identification tool in the field of forensic sciences is gaining popularity due to its noninvasive nature and the ease of sample collection as compared to other body fluids. The role of saliva in serological as well as cellular analysis plays a major role in the identification of the individual in question. Saliva obtained from bitemarks can be subjected to genetic profiling and deoxyribonucleic acid (DNA) extraction to aid in the personal identification of the accused. 2 The assessment of salivary concentration of various analytes and comparison with the matched plasma samples is still a major arena of research. 3
The advent of molecular biology, genetic material isolation methods, biotechnology techniques, and serological techniques has paved the way for the development of salivary diagnostics. At present, salivary diagnostics is applied in the fields of medicine, dentistry, pharmacotherapy, narcotics, epidemiology, and forensic applications. In existing literature, there were not many studies done correlating the age and gender-related changes in salivary total protein. Research on the use of saliva for forensic identification remains at an early stage, and advancement in this area is constrained by the limited availability of efficient and cost-effective laboratory techniques. Consequently, there is a clear need to develop reliable methods for molecular identification using saliva and to establish standardized protocols for the evaluation and analysis of salivary samples.
In situations involving criminal incidents or mass disasters, establishing the identity of unknown individuals continues to be a significant challenge. In such instances, body fluids like saliva play an inevitable role. The modern progress in molecular biology has helped different aspects of forensic odontology, including salivary proteomics and genomics.
Saliva is composed predominantly of water, accounting for approximately 99% of its volume, along with a diverse array of electrolytes, including sodium, potassium, calcium, chloride, magnesium, bicarbonate, and phosphate. In addition, it contains a wide range of proteins such as enzymes, immunoglobulins, antimicrobial agents, mucosal glycoproteins, trace amounts of albumin, and peptides that are essential for maintaining oral health. 4 Saliva also includes organic constituents such as glucose and nitrogenous waste products, including urea and ammonia. Whole saliva represents a composite fluid derived from the secretions of both major and minor salivary glands, gingival crevicular fluid, and exfoliated oral epithelial cells, along with oral microorganisms. The salivary proteome is primarily composed of amylases, proline-rich proteins (acidic, basic, and glycosylated), statherins, histatins, peroxidases, cystatins, and mucins. The concentration of total protein in whole saliva typically ranges from approximately 2,000 to 4,000 µg/mL. 4 Salivary constituents often act as useful biomarkers, which in turn has an influence on personal identification. Saliva is one of the most common body fluids obtained in every instance where the oral cavity comes in direct contact with skin, clothes, cigarettes, and glass.
The current literature reveals that there are very few studies conducted to study salivary protein profiling and its applications in the field of forensic odontology.
The study aimed to estimate the gender and age-related changes in the total salivary protein level.
Methods
The study participants were selected from the outpatient section of the department of oral medicine and radiology. The study was conducted after approval of the Institutional Review Board (IRB reference no.: 4/IRB/2019 dated August 22, 2019). The study is an in vivo prospective clinical study. Written informed consent was obtained from all participants prior to inclusion in the study.
Patients satisfying the inclusion criteria were examined and findings were recorded.
The research study included healthy individuals aged five years and above belonging to both genders. The exclusion criteria were individuals with acute or chronic illnesses, pregnant individuals, individuals with autoimmune diseases like Sjogren’s syndrome, completely edentulous patients, smokers, and patients with severe periodontal conditions.
The participants visiting the outpatient department were allotted into different groups based on gender and age according to the fulfillment of the inclusion criteria, and were randomly selected for the study using the lottery method.
Group A consisted of males (n = 60) and group B consisted of females (n = 60) subdivided further into different age categories as 5–13 years (children, n = 15); 14–19 years (adolescents, n = 15); 20–64 years (adults, n = 15); and 65 years and above (geriatric, n = 15).
Saliva Collection, Storage, and Processing
In this study, the saliva samples were collected in the morning from 8 am to 11:00 am. Patients were advised to avoid consuming any food, drugs, or water one hour before the collection of the sample. The participant was made to sit in a comfortable, upright position, and around 5 mL of whole unstimulated saliva was collected by asking the patient to spit into 40 mL sterile wide-mouth plastic containers. Using disposable plastic Pasteur pipettes, the saliva sample was transferred to ethylenediaminetetraacetic acid (EDTA)-coated centrifuge tubes. The tubes were placed on ice packs during the collection of saliva and transport for centrifugation. The samples were transported in sealed vaccine storage boxes packed with ice packs to maintain the cold chain. The saliva processing and storage from the time of collection was limited to two hours. The saliva samples were centrifuged in a refrigerated centrifuge at −4°C, 2,600 g for 15 minutes. The clear supernatant was pipetted and aliquoted into sterile 2 mL Eppendorf tubes. The samples were stored at −20°C until analysis was performed. The salivary protein quantification was done by the Bradford assay. The samples and protein standards were measured using an ultraviolet (UV) spectrophotometer. The data obtained was subjected to statistical analysis.
Estimation of Total Protein Using Bradford Reagent
The protein concentration in the tissue culture samples was determined using the Bradford dye binding assay as described by Bradford (1976).5, 6 Coomassie brilliant blue G 250 was prepared by dissolving the dye in ethanol and orthophosphoric acid, followed by dilution with distilled water to a final volume of 1,000 mL. The absorbance of the reagent was measured at 595 nm using a spectrophotometer and adjusted to obtain an optical density of 1.18, with distilled water serving as the blank. The reagent was then filtered through Whatman No. 1 filter paper and stored in an amber-colored container at 4°C.
For the assay, 1 mL of the culture filtrate was mixed with 5 mL of Bradford reagent. The intensity of the resulting blue coloration was measured at 595 nm using a spectrophotometer. Protein concentration was quantified by comparison with a standard curve generated using Bovine Serum Albumin Fraction V (Sigma, USA) (Figure 1).
Estimation of Total Protein Concentration Using Bradford Assay.
Results
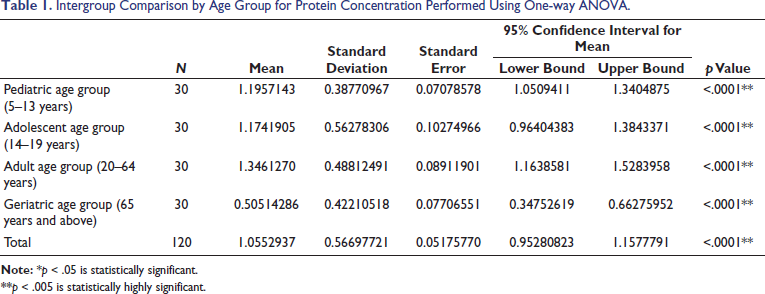
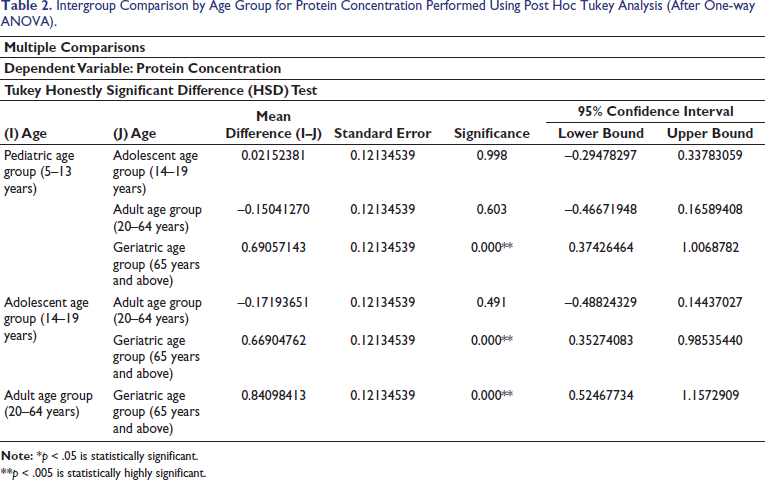
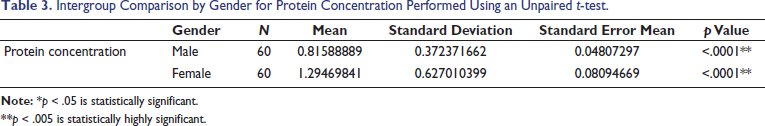
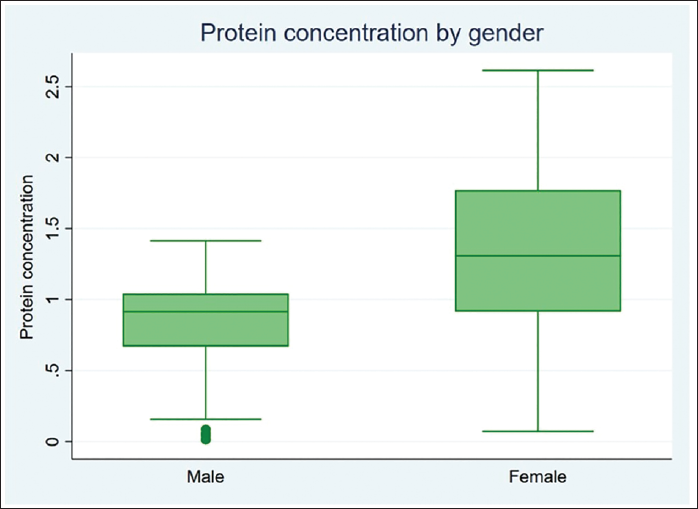
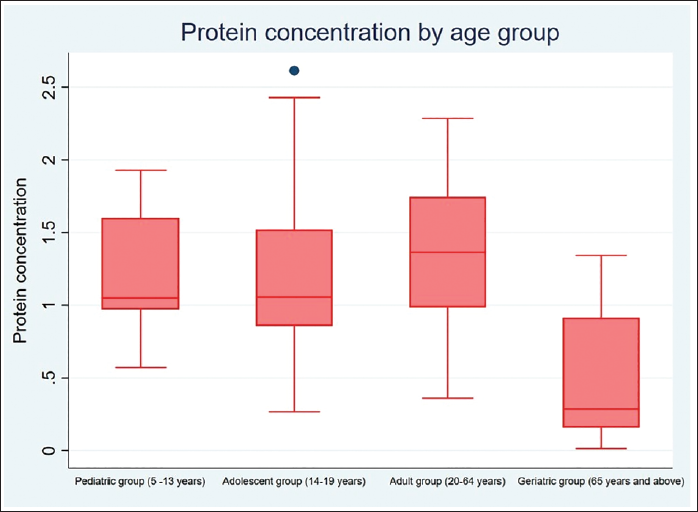
The statistical analysis was done using Stata version 17, standard edition (SE) 17 software package (StataCorp, College Station, Texas, USA). A total of 120 subjects were included in the study, 60 males and 60 females, divided into four specific age categories (5–13 years, 14–19 years, 20–64 years, and 65 years and above). Descriptive statistics were computed (Tables 1 and 3). Intergroup comparison by age group for protein concentration was performed using one-way analysis of variance (ANOVA). Intergroup comparison by age group for protein concentration was performed using post hoc Tukey analysis after one-way ANOVA. Intergroup comparison by gender for protein concentration was performed using an unpaired t-test. Intergroup comparison by dentition status for protein concentration was performed using Welch ANOVA. Median, interquartile range (IQR), minimum, and maximum values for protein concentration with respect to the categorical variables (age group and gender) are represented using box and whisker plots (Figures 2 and 3). The median (p50) is represented using the thick line in the vertical box, and the first quartile (p25) and third quartile (p75) are represented by the upper and lower limits of the box. Maximum and minimum values are presented using upper and lower limits of the whiskers and the outliers as dots above and below the whiskers. A highly statistically significant difference in protein concentration was observed between the age groups (p < .0001) with lower values reported in the geriatric population (0.505 ± 0.422) (Table 1). Post hoc Tukey analysis revealed all other age groups demonstrated a statistically highly significant difference when compared with the geriatric population (p < .0001) (Table 2). The geriatric population refers to patients aged ≥65 years. Other paired comparisons did not demonstrate a statistically significant difference (Table 2). A statistically highly significant difference in protein concentration was observed between males and females (p < .0001), with higher values reported in females (1.295 ± .627) (Table 3). A statistically significant difference in protein concentration was observed between the age groups (p = .026), with lower values reported in the post-dental eruption group (0.882 ± 0.627). Post hoc Tukey analysis revealed permanent dentition until the third molar eruption group demonstrated a statistically significant difference when compared with the post-dental eruption group (p = .030). The post-dental eruption group refers to individuals whose permanent teeth have all erupted in the oral cavity, including the third molars. Other paired comparisons did not demonstrate a statistically significant difference.
Intergroup Comparison by Age Group for Protein Concentration Performed Using One-way ANOVA.
**p < .005 is statistically highly significant.
Intergroup Comparison by Age Group for Protein Concentration Performed Using Post Hoc Tukey Analysis (After One-way ANOVA).
**p < .005 is statistically highly significant.
Intergroup Comparison by Gender for Protein Concentration Performed Using an Unpaired t-test.
**p < .005 is statistically highly significant.
Box and Whisker Plot for Protein Concentration by Gender.
Box and Whisker Plot for Protein Concentration by Age Group.
The empirical cut point (protein concentration) for gender was estimated to be 0.836 with a sensitivity of 0.65, specificity of 0.23, and area under the curve (AUC) of 0.44. A value below the estimated empirical cut point signifies that the individual would be male, and a value above that would be female. The empirical cut point (protein concentration) for age groups was estimated to be 0.936 with a sensitivity of 0.73, specificity of 0.83, and AUC of 0.78. A value below the estimated empirical cut point signifies that the individual would belong to the geriatric population, and a value above would belong to other age groups. The empirical cut point (protein concentration) for dentition status was estimated to be 0.993 with a sensitivity of 0.64, a specificity of 0.60, and an AUC of 0.62. A value below the estimated empirical cut point signifies that the individual would have post-dental eruption status, and a value above would be other groups.
Discussion
Forensic odontology, or forensic dentistry, is a specialized field of study covering a wide variety of topics like individual identification, mass identification, and bitemark analysis. 7 Application of salivary protein markers has emerged as a technology-driven science focusing on the analysis of the salivary proteome, including their expression of proteins as well as their chemical structures, interactions, and post-translational modifications.8–13 The origin of salivary proteins is from the salivary glands, oral tissues, blood, gingival crevicular fluid, and microorganisms.14–16 In the near future, human saliva has the potential to be a relevant diagnostic fluid for clinical diagnosis and prognosis of a variety of conditions.
The average protein concentration was found to decrease progressively from the pediatric group to adults, then to adolescents, and finally to the geriatric age group. The lowered protein concentration in the geriatric age group was in line with Yeh et al. (1998), where it was found that there was a significant decrease in unstimulated saliva protein output with increasing age (p < .05). 17 An individual’s age can be predicted using genomic analysis in body fluids like saliva. 18
A highly significant statistical difference in protein concentration was seen between males and females (p < .0001), with higher values reported in females (1.295 ± .627). This was in agreement with the studies by Bhuptani et al. (2018) and by Dodds et al. (1997), where higher salivary total protein was found in females than in males. 19
The observed sex-related variations in salivary protein concentration may be attributed to underlying physiological differences, particularly the modulatory effects of sex hormones on salivary gland function. Female individuals are reported to have comparatively smaller salivary glands, which may partially account for differences in salivary biochemical composition.20, 21 In addition, studies have demonstrated distinct gene expression patterns in the salivary gland tissues of males and females, including differential expression of genes involved in saliva production and secretion. These variations are thought to be influenced by estrogen-mediated activity at the estrogen receptors of the salivary glands.22, 23 Salivary gland sexual dimorphism and divergent gene-expression profiles could explain the differences seen in salivary protein composition.
Using cutoff analysis, the empirical cut point (protein concentration) for gender was estimated to be 0.836. A value below the estimated empirical cut point signifies that the individual would be male and a value above that would be female. Similarly, the empirical cut point (protein concentration) for age groups was estimated to be 0.936. A value below the estimated empirical cut point signifies that the individual would belong to the geriatric population, and a value above would belong to other age groups.
Salivary total protein concentration performs weakly as a marker for gender discrimination (AUC = 0.44). It is a reasonably good marker for distinguishing geriatric from non-geriatric individuals (AUC = 0.78). The present study suggests that the AUC findings for age discrimination (AUC ~0.78) fit reasonably well into a broader narrative that protein levels change with age, while the AUC for gender discrimination (AUC ~0.44) aligns with most research studies that there are only modest differences in total protein by gender.
Limitations
The total salivary protein concentration was assessed without specific protein profiling, limiting biological interpretability. The single-center, limited-size sample restricts generalizability across populations. Uncontrolled pre-analytical factors (for example, collection method, diet, hydration, confounders like medications, or oral health) and a cross-sectional design preclude causal or longitudinal insights. Diagnostic performance was weak for gender (AUC = 0.44) and moderate for age (AUC = 0.78), with cutoffs derived from the same dataset lacking external validation or comparison to other biomarkers.
Conclusion
From this study, it can be concluded that there was a statistically significant difference observed between males and females for salivary total protein concentration, with higher values reported in females. A highly significant difference was observed between age groups for protein concentration, with lower values reported in the geriatric population. Although salivary total protein concentration demonstrates statistically significant differences between age groups and genders, the diagnostic performance and predictive value are low, especially for gender estimation.
This study is one of the few research studies that have been done to correlate the application of salivary total protein concentration in the forensic identification process. This study establishes age and gender-related changes in human salivary total protein levels, which will be an adjunctive aid in the identification of individuals in forensic cases.
Future Prospects
Salivary proteomics and its application in forensic identification are an upcoming and promising area of investigation within the field of forensic odontology. Further studies and an extended research spectrum with comparison of different populations, increased sample size, different modes of saliva collection, and different age groups can be used to validate the research findings in our study.
Footnotes
Authors’ Contribution
Archana Muralidharan: Conception, design of the work; acquisition, analysis, interpretation of data; substantively revised it, approved the submitted version, and preparation of tables and figures.
Jayachandran Sadaksharam: Conception, design of the work; acquisition, drafted the work, interpretation of data; preparation of tables and figures, substantively revised it, and approved the submitted version.
Availability of Data and Materials
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethics approval and consent to participate: Approval for the study was taken before conducting the study from the Institutional Review Board, Tamil Nadu Government Dental College and Hospital (Reference no 4/IRB/2019).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from all participants prior to inclusion in the study.
