Abstract
The prevalence of mislabeled pharmaceutical preparations poses a significant threat to public health and presents considerable challenges in forensic narcotics investigations, particularly under the legal framework of the Narcotic Drugs and Psychotropic Substances (NDPS) Act. Such mislabeling not only endangers consumers due to potential adverse pharmacological effects but also complicates legal adjudication and regulatory enforcement. This case study details the forensic identification and analytical characterization of a suspected psychotropic drug sample submitted as alprazolam. Preliminary presumptive tests indicated inconsistencies with the declared identity, prompting comprehensive instrumental analyses. Thin-layer chromatography (TLC), ultra-violet (UV) visible spectrophotometry, and gas chromatography–mass spectrometry (GC–MS) were employed for confirmatory examination. The results conclusively demonstrated that the seized sample contained cyproheptadine, an antihistaminic compound, rather than the labeled alprazolam. The distinct chromatographic and spectral profiles provided unequivocal differentiation between the declared and actual constituents. These findings underscore the critical necessity for robust, multi-tiered analytical protocols in forensic narcotics laboratories and highlight the broader implications of drug mislabeling for public safety, law enforcement, and judicial processes.
Introduction
Mislabeled pharmaceutical and psychotropic products are an emerging concern in forensic chemistry, with significant legal, health, and regulatory implications. An increasing body of literature documents cases in which products marketed as one substance are adulterated or substituted with others, often leading to dangerous consequences. For instance, Brandt1–2 reported that a traditional herbal “Gambir” product was contaminated with toxic toad venom compounds, revealed through gas chromatography–mass spectrometry (GC–MS) analysis. Similarly, studies on dietary supplements have revealed the existence of undeclared phenethylamine derivatives, underscoring the public health risks associated with product misrepresentation. 3 These precedents demonstrate that even non-illicit substances can conceal harmful or foreign compounds, highlighting the need for demanding forensic examination. According to the recent study, 4 in which crushed tapentadol pills were injected intravenously and caused cutaneous adverse drug responses. Because injectable drug usage frequently results in such responses, clinical evaluation of injection sites is crucial. Strict regulations are necessary in light of the growing abuse of tapentadol in India. When used to treat moderate to severe pain, the relatively novel synthetic opioid analgesic tapentadol has proven to be more effective than conventional opioid analgesics. 5
Pharmaceutical mislabeling, the incorrect identification, or intentional substitution of drug contents, represents a significant public health concern and a critical challenge in forensic science. Even minor discrepancies in labeling can lead to severe therapeutic failures, unexpected adverse effects, and life-threatening toxicological consequences. Such risks are further amplified in the case of controlled substances, where pharmacological potency, metabolic behavior, and legal implications are tightly regulated.
GC–MS is considered the “gold standard” in the field of confiscated drug analysis due to its ability to offer unambiguous chemical identification by integrating mass spectrum information with retention time (RT) data. 6 As new psychoactive drugs proliferate quickly and frequently avoid detection by conventional immunoassay screening, forensic laboratories are increasingly using GC–MS and liquid chromatography mass spectrometry (LC–MS) methods to reliably identify a variety of chemical moieties. 7 These advanced methods, along with derivatization, automation using the Automated Mass Spectral Deconvolution and Identification System, and RT locking, enhance both sensitivity and specificity, enabling the detection of known and unexpected substances. 8 Maurer 9 demonstrated that automated spectral deconvolution significantly improves detection rates by identifying substances missed during manual review, further reinforcing the value of advanced data-processing pipelines in forensic laboratories. A strong and ethical forensic workflow is produced when thin-layer chromatography (TLC), UV, and GC–MS are used in tandem. TLC and UV are the quick screening methods for identifying initial sample composition, while GC–MS offers confirmatory characterization and accurate chemical structure identification.
In this study, we employed GC–MS with stringent controls for artifacts and RT standards alongside TLC and UV methods to confirm the presence of an antihistamine in a mislabeled sample. This integrative approach addresses core analytical challenges documented in prior research. By presenting this case, our aim is to raise scientific awareness regarding mislabeled exhibits in forensic drug analysis and to encourage the adoption of modified protocols that ensure the detection and documentation of such anomalies with both scientific rigor and legal accountability. To validate the presence of an antihistamine in a mislabeled sample, we used TLC, UV in scientific conjunction with GC–MS, which has strict controls for analytical artifacts and RT standards. This integrated method holds the fundamental analytical issues that have been reported in earlier studies. Our aim in representing this example is to increase scientific awareness of mislabeled exhibits in forensic drug analysis and to promote the use of modified techniques that validate the identification and recording of such anomalies with both legal responsibility and scientific rigor.
Materials and Methods
All chemicals, reagents, and reference standards employed in this study were of analytical grade. The solvents used for chromatographic analysis were of high- performance liquid chromatography (HPLC) grade, and all chemicals were used as received without further purification; their purity conformed to the specifications provided by E. Merck and Sigma-Aldrich.
Thin-layer Chromatography and UV–visible Spectroscopy
Standard reference samples of the chemicals used were procured from certified pharmaceutical sources, accompanied by certificates of analysis. Analytical grade diethyl ether, acetone, and diethylamine were obtained from Merck India Ltd. For chromatographic analysis, silica gel 60 F254 pre-coated TLC plates (Merck, 20 × 20 cm, 0.25 mm thickness) were used. TLC was performed to validate the identity of the tablet labeled as containing alprazolam. The mobile phase consisted of diethyl ether (water saturated 1:1 v/v), acetone, and diethylamine (85:8:7, v/v). The test sample (tablet extract) was dissolved in methanol and spotted on the TLC plate alongside a standard alprazolam solution (1 mg/mL in methanol). Plates were developed in a pre-saturated glass TLC chamber to a height of 8 cm, air-dried, and visualized under UV light at 254 nm. 10 In the supportive study, we have analyzed the received sample on a UV–visible spectrophotometer (UV-1900, SHIMADZU, Japan) single tablet was dissolved in 5 mL of 0.1 N HCl solution, and the wavelength of the given sample was measured on the mentioned instrument.
GC–MS Analysis
All analyses were performed using a Thermo Fisher Scientific GC–MS system, model ISQ 7000, integrated with a Trace 1310 gas chromatograph and operated in electron ionization mode. The system utilizes a single quadrupole mass analyzer and is equipped with a multimode inlet and a Thermo Scientific™ single taper liner (4 mm inner diameter). The glass wool liner aided in minimizing contamination from non-volatile sample residues while maintaining high analyte sensitivity, especially in comparison to plain liner configurations. Sample injection was carried out in pulsed split (17:1) with purge flow 5.0 mL less mode at 30 psi for 0.35 minutes, ensuring efficient analyte transfer to the column while reducing band broadening. A 1 µL injection volume was used, and the injector temperature was held constant at 250 °C throughout the run. Chromatographic separation was achieved using a Thermo Scientific™ TraceGOLD™ TG-5-MS capillary column (30 m × 0.25 mm I.D. × 0.25 µm film thickness), composed of polyethylene glycol, a highly polar stationary phase optimized for acidic, polar, and thermally sensitive compounds. The GC oven was initially held at 100 °C, followed by a temperature increase to a final temperature of 300 °C at 15 °C/min. The carrier gas was ultrahigh-purity helium (99.999%), maintained at a constant flow of 1.2 mL/min. Ultrahigh-purity nitrogen was used as the collision gas, and ultrahigh-purity helium served as the quenching and carrier gas.
Results
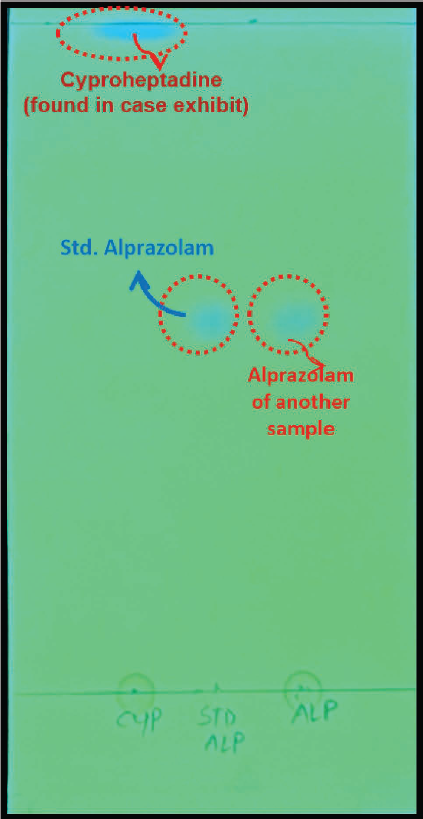
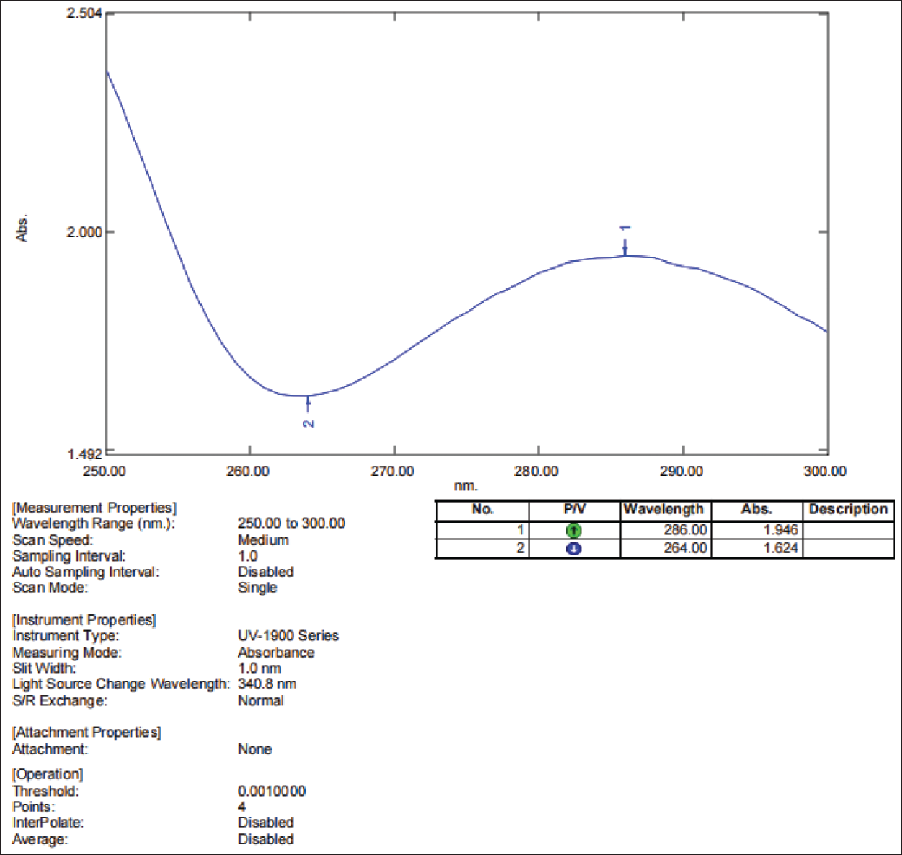
In the case sample received for drug analysis, contradictions were observed in the results between the labeled drug and the compound detected. Although the seized pharmaceutical tablet was labeled as containing alprazolam, preliminary screening was done using colorimetric detection and TLC in Figure 1, which suggested the presence of a compound with spectral and chromatographic characteristics inconsistent with alprazolam due to the different retention factor (Rf) values of the standard compound with the spot found near the mobile phase. Similarly, Figure 2 shows that the wavelength at 286 nm does not correspond to alprazolam (260 nm), and the data literate the plausible probability of cyproheptadine in the received sample.11–12 The UV–visible spectrophotometric analysis of this significant spectral shift validated the presence of different compounds. To confirm these findings, the sample was subjected to GC–MS analysis. Under the selected solvent system, the chromatographic profile of the alprazolam standard differed from that of the test sample, whereas the test sample produced a spot identical in position to that of the cyproheptadine. This observation indicated that the tablet, although labeled as containing alprazolam, contained another chemical moiety as the active ingredient (Figures 3 and 4).
TLC Analysis Presenting that the Received Drug Sample Did Not Contain Alprazolam; Instead, the Spots Correspond to Cyproheptadine.
UV–visible Spectra of Detected Compound (Cyproheptadine) Depicted Distinct Absorption Traits from Alprazolam.
GC–MS Chromatogram Comparing RT of Detected Compound with that of Alprazolam Standard.
Mass Spectral Data Showing Distinct Fragmentation Patterns and Characteristic m/z Values of the Detected Compound (Cyproheptadine), Which Differ Significantly from Those of the Alprazolam Standard, Thereby Supporting Compound Identification.
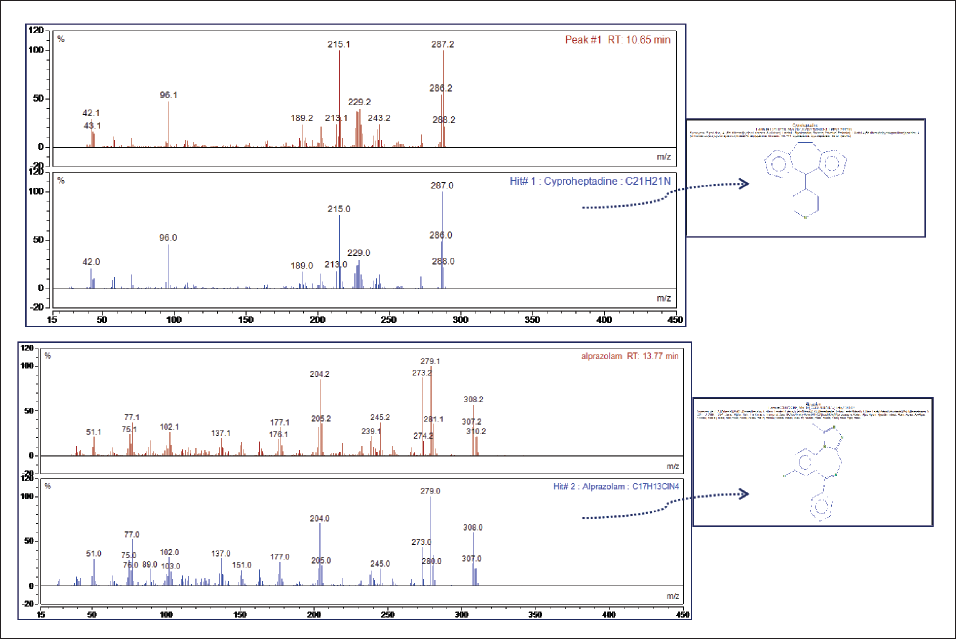
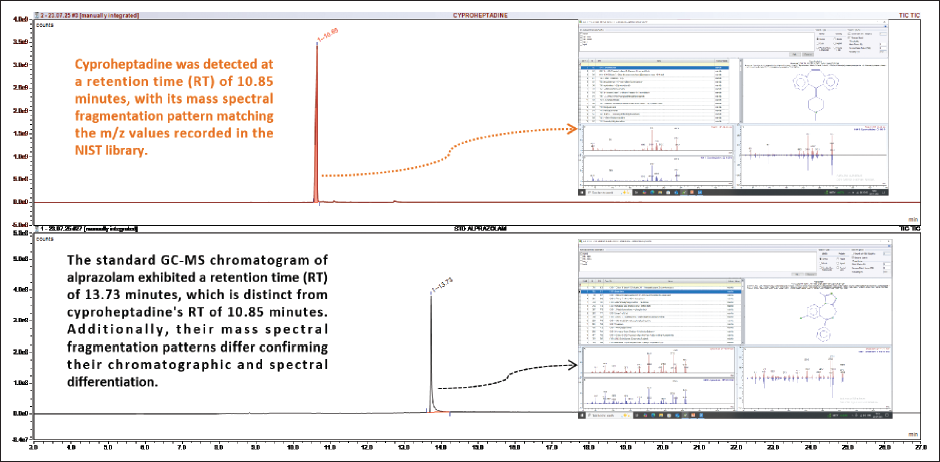
To further validate the chromatographic findings, GC–MS analysis was conducted on both the suspect tablet and the certified reference standards. The mass spectral data were processed using the National Institute of Standards and Technology (NIST) Mass Spectral Library, 13 enabling compound identification based on RT and spectral match probability. Figure 3 shows that the test sample showed a single prominent peak at an RT of 10.65 minutes, which corresponded to cyproheptadine, with a high match score against the NIST database. In contrast, the certified alprazolam standard exhibited a peak at RT 13.73 minutes, with fragmentation patterns consistent with known spectra for alprazolam, confirming its absence in the test sample.
The MS chromatogram (Figure 4) of the case sample revealed a prominent peak at an RT of 10.65 minutes, corresponding to cyproheptadine, as identified by spectral matching with the NIST library. The mass spectrum showed characteristic fragment ions at m/z 287, 229, 215, 189, and 96, consistent with reference spectra of cyproheptadine. In contrast, a standard alprazolam sample showed an RT of 13.73 minutes, with major m/z fragments at 308, 281, 279, 245, 204, 177, and 77.
The clear data distinction in both RT and fragmentation patterns confirms that the compound present in the case sample was cyproheptadine, not alprazolam, as labeled. This indicates a case of pharmaceutical mislabeling that requires further scientific investigation. Such analytical contradictions are not only scientifically significant but also carry important clinical and forensic implications. Misidentification of benzodiazepines by visual or packaging cues alone has previously been reported as a cause of diagnostic and therapeutic errors. 14 The reliability of TLC as a rapid screening method, when confirmed by spectrometric techniques such as GC–MS, provides a scientific foundation for detecting falsified or substituted medications. The findings emphasize the need for precise post-market surveillance, analytical verification, and adherence to Schedule H and the Narcotic Drugs and Psychotropic Substances (NDPS) Act compliance, such as psychoactive medications with high abuse potential.
Discussion
An article entitled “A Mislabeling Event with Batched Drugs: The Unintended Consequences of Practice Changes” by Grissinger 15 reported a critical medication error caused by mislabeling during the batching of anesthesia drugs. Grissinger 15 discussed a case where syringes containing neuromuscular blockers were mislabeled as lidocaine, leading to nearly undetectable mixings due to identical batch conditions and delayed labeling. Both incidents underscore the need for stringent, immediate labeling protocols and error-proofing strategies, especially when handling high-risk medications. A recent case report by Plæhn 16 published the adverse effects experienced by a patient who self-administered a pain-relieving counterfeit medication, believed to contain tramadol, but containing the opioid tapentadol and the muscle-relaxant carisoprodol. Mislabeling of psychoactive drugs, particularly the replacement of benzodiazepines with first-generation antihistamines, can lead to unpredictable pharmacodynamic interactions and a cascade of adverse effects owing to the distinct metabolic pathways and therapeutic actions of the compounds involved.17, 18 The inadvertent or intentional mislabeling of pharmaceuticals presents a significant challenge to both clinical practice and forensic toxicology. In this context, the substitution of alprazolam, a short-acting benzodiazepine, with cyproheptadine, a first-generation antihistamine possessing anticholinergic and serotonin antagonist properties, poses serious implications at the levels of pharmacodynamics, metabolism, and patient safety.
Alprazolam acts primarily on the central nervous system (CNS) by enhancing the activity of gamma-aminobutyric acid (GABA) at GABA-A receptors, resulting in anxiolytic, sedative, and muscle-relaxant effects. It is metabolized in the liver primarily via cytochrome-P450-3A4 into α-hydroxyalprazolam and 4-hydroxyalprazolam metabolites with reduced pharmacological activity. Therapeutically, alprazolam is used in the management of anxiety and panic disorders, and abrupt therapeutic failure may lead to withdrawal symptoms, including agitation, insomnia, tremors, and, in severe cases, seizures and psychosis in dependent individuals. 17 Cyproheptadine undergoes hepatic metabolism to norcyproheptadine and related inactive metabolites. It exerts its effects through H1-histamine receptor antagonism as well as serotonin receptor antagonism (specifically 5-hydroxytryptamine2 (5-HT2)), in addition to pronounced anticholinergic activity. 19 Although generally regarded as safe at therapeutic doses, overdoses or inappropriate administration can lead to adverse outcomes such as central anticholinergic syndrome, delirium, mydriasis, dry mucous membranes, tachycardia, urinary retention, and, in severe cases, hallucinations and seizures. 18
The metabolic implications of such a substitution are profound. While both drugs are processed hepatically, their metabolites, plasma half-lives, and enzyme affinities differ, potentially leading to unexpected pharmacokinetic interactions when used concurrently with other medications. Moreover, patients expecting anxiolytic relief from alprazolam may instead experience paradoxical effects such as increased irritability or confusion due to the anticholinergic and anti-serotonergic profile of cyproheptadine. From a forensic and toxicological standpoint, such mislabeling can complicate the interpretation of drug levels in biological specimens, especially in the context of therapeutic failure, intoxication, or unexplained death. Analytical toxicology may detect only cyproheptadine, leading to potential misattribution of clinical symptoms or mortality, especially if the patient had a known prescription for alprazolam. This misrepresentation may raise medico-legal implications, particularly in scenarios involving custody deaths, impaired driving, or alleged medical negligence.14, 20–22 Furthermore, this incident underscores the critical need for stringent quality control, pharmaceutical authentication, and routine toxicological screening, particularly when managing CNS-acting medications with a high potential for dependence or misuse. It also emphasizes the role of clinical vigilance, as early recognition of anticholinergic toxicity or treatment failure may prevent serious outcomes. The above-explained biochemical ambiguity in this mislabeling causes many discrepancies in the normal functions of the consumer’s body. When pharmacological drugs have negative effects on a particular organ or organ system, it is referred to as drug-induced organ toxicity. It is crucial to correctly recognize and comprehend these toxicities to guarantee proper medical action and the mitigation of additional injury. 23 Nandhini et al., 2024 24 reported a study about the consequences of drug poisoning; this study depicts that sedative-hypnotics such as benzodiazepines and analgesics such as paracetamol (acetaminophen) were the most commonly used drugs. This data enlightens the crucial depiction of the prevalence of poisoning in India, but it does not find or demonstrate a general trend scientifically. The Poisons Information Center has a responsible duty to inform doctors about commonly used, abused medications so that the unaware public is aware of these before prescribing them.
From a legal point of view, such mislabeling also constitutes a serious violation of the NDPS Act, 1985, as alprazolam is classified as a psychotropic substance under the Act’s schedule. Possession, distribution, and administration of alprazolam without valid medical authorization or outside regulatory protocols are punishable by law. If a product labeled as cyproheptadine, an over-the-counter or Schedule H1 antihistamine in India, actually contains alprazolam, it can result in unauthorized and unintentional exposure to a controlled psychotropic drug, raising concerns about illegal possession, diversion, and trafficking under Sections 8, 21, and 22 of the NDPS Act. This mislabeling not only endangers public health but also circumvents legal controls meant to prevent the misuse and abuse of psychotropic substances. It highlights the critical need for forensic and pharmaceutical vigilance to prevent criminal misuse under the guise of therapeutic administration.
Conclusion
The present case underscores the critical implications of pharmaceutical mislabeling in forensic narcotics analysis. The substantial metabolic, pharmacological, and legal differences between alprazolam and cyproheptadine render their substitution highly unsafe, with even a single instance posing significant risks, particularly to public health. Analytical cross-verification using TLC, UV, and GC–MS conclusively demonstrated the absence of alprazolam and the presence of cyproheptadine, a compound with a distinct pharmacodynamic profile and regulatory status. This misrepresentation highlights the necessity for stringent quality control measures, robust forensic verification protocols, and a comprehensive understanding of drug metabolism in ensuring accurate identification. Ultimately, the findings emphasize the indispensable role of meticulous analytical scrutiny in safeguarding public health, supporting judicial processes, and upholding the integrity of narcotics law enforcement.
Footnotes
Acknowledgment
The team of authors acknowledges the analytical support of the Regional Forensic Sciences Laboratory, Hisar (Haryana), for providing the Laboratory facilities and legal context for this scientific study.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
