Abstract
Thermal burns trigger complex systemic inflammatory and metabolic responses that may affect organs, including the pancreas, yet pancreatic pathology in burn-related deaths is poorly documented. This study evaluated gross and microscopic pancreatic changes in 63 medicolegal autopsies of fatal thermal burn cases and explored their relationship with total body surface area (TBSA) involvement and survival duration after injury. Gross examination commonly revealed congestion (42.9%) and necrosis (23.8%). On histopathology, congestion (52.8%) and necrosis (44.4%) were the predominant findings, along with varying degrees of edema, haemorrhage, fibrosis and inflammatory infiltrates. Pancreatic necrosis was minimal or absent in individuals who died within 72 hours of sustaining burns, but its frequency and severity increased with longer survival, particularly beyond 97 hours. Moderate to severe necrosis was more evident in patients surviving more than one week. Interestingly, higher grades of necrosis were observed more often in moderate burns (30–50% TBSA) than in extensive burns involving more than 50% TBSA. Statistical analysis did not demonstrate a significant association between pancreatic necrosis and either TBSA (
Introduction
Thermal burns are among the most devastating forms of trauma, associated with high morbidity and mortality. They result from the application of heat to the body surface, leading to tissue destruction. 1 Deaths in burn cases are commonly attributed to neurogenic shock, hypovolemic shock, sepsis, acute kidney injury, hypokalemia, and fat embolism. 2 Prognosis depends on several factors, including the extent of total body surface area (TBSA) affected, the severity of heat exposure, age, sex, and the presence of inhalational injuries, which significantly increase fatality risk. 1 Early hospitalization also plays a crucial role, as prompt fluid resuscitation reduces immediate mortality, although post-burn multiple organ dysfunction remains a major concern. 3
Although systemic complications of burns are well recognized, pancreatic involvement has been less studied. Burn injuries trigger inflammatory, oxidative, and immune responses that can impair pancreatic function, leading to congestion, edema, necrosis, or even pancreatitis. Such changes aggravate the hypermetabolic state, increase insulin resistance, and worsen outcomes. Notably, systemic complications occur in over 30% of severely burned patients, with metabolic disturbances like insulin resistance contributing significantly to morbidity and mortality. 4
Understanding the histopathological spectrum of pancreatic changes in burn victims is therefore important for clarifying underlying mechanisms and guiding management strategies. Identifying such changes may help in early detection, prevention, and treatment of metabolic and inflammatory complications.
The present study investigates gross and microscopic changes in the pancreas in deaths due to thermal burns, with particular emphasis on their association with TBSA involvement and survival duration.
Objectives
Primary Objective
To study the gross and histopathological changes in the pancreas in cases of death due to thermal burn injury.
Secondary Objectives
To correlate the pathological changes in the pancreas with the TBSA involved.
To correlate the pathological changes in the pancreas with the period of survival (duration between burn injury and death).
Materials and Methods
Study Design and Setting
A descriptive cross-sectional study was conducted over nine months following the Institutional Ethics Committee approval dated March 3, 2024 at the Department of Forensic Medicine and Toxicology in collaboration with the Department of Pathology.
Study Population and Sampling Technique
The study included 63 cases of burn-related deaths brought for medicolegal autopsy. Cases were included if there was a documented history of thermal burn injury. Exclusion criteria were decomposed (including evidence of early decomposition) or mutilated bodies, charred remains, unknown identity, pre-existing pancreatic disease, or lack of consent from the legal claimant. No randomization was applied; hence, the sampling method was consecutive purposive sampling.
Sample Size
In 2022, 330 of 3,400 medicolegal autopsies were burn-related (9.71%). Using this prevalence, a minimum sample size of 54 was calculated at a 95% confidence level with 8% absolute precision. Sixty-three cases fulfilling the inclusion criteria were ultimately included. Although this exceeds the minimum required sample, the study remains primarily exploratory and is powered to detect moderate, but not small, differences in necrosis grades across subgroups.
Data Collection
Demographic details (age, sex, residence), comorbidities, and circumstances of injury were recorded from documents and accompanying relatives. Burn characteristics such as date of injury, time to hospital admission, and survival duration were documented. TBSA involvement was assessed using the Wallace Rule of Nines or the Lund and Browder chart, depending on age.
Autopsy and Tissue Sampling
During the autopsy, the pancreas was removed, examined for gross changes (edema, congestion, hemorrhage, necrosis, fibrosis), and preserved in 10% formal saline. For histopathological examination, three representative tissue blocks were routinely sampled—one each from the head, body, and tail regions of the pancreas. Tissue sections were processed into paraffin blocks and stained with hematoxylin and eosin. In this series, most autopsies were conducted within approximately 12 hours (none exceeded 24 hours), and pancreatic tissue was placed into fixative almost immediately following removal (typically within about 15 minutes), which helped minimize autolysis-related artefact. Microscopic features evaluated included congestion, edema, hemorrhage, fibrosis, inflammatory infiltrates, and necrosis. Changes were graded on a semi-quantitative scale (nil, 1+, 2+, 3+) to indicate the extent and severity of tissue involvement across the microscopic fields examined. Microscopic grading was based on evaluation of an average of 10 high-power fields (HPF) per case to ensure adequate field representation. A grade of nil denoted complete absence of the change; 1+ (mild) indicated focal or minimal changes involving <25% of the tissue section; 2+ (moderate) represented changes involving approximately 25%–50% of the tissue; and 3+ (severe) referred to diffuse or extensive changes affecting >50% of the pancreatic tissue. This grading system reflects both intensity and distribution of lesions across the histological slide. Histopathological grading was performed by a single pathologist (slides were coded, and the pathologist was blinded to survival period and TBSA); therefore, inter-rater reliability analysis was not applicable.
Data Analysis
Data were compiled in spreadsheets and expressed as numbers and percentages. Associations of pancreatic necrosis with survival duration and TBSA were analyzed using the chi-square test and Spearman’s rank correlation.
Results
The study included 63 burn-related deaths, with a female predominance (57 females, 6 males). No cases occurred below 10 years of age. The highest number of deaths was in the elderly: >70 years (28.6%), followed by 61–70 years (23.8%), and 51–60 years (19%). Younger age groups contributed fewer cases (4.8% in 11–20 years, 9.5% in 21–30 years).
Most victims were hospitalized promptly; 38.1% arrived within 1 hour, and 42.9% within 1–6 hours of injury. Only 19.1% presented after 6 hours.
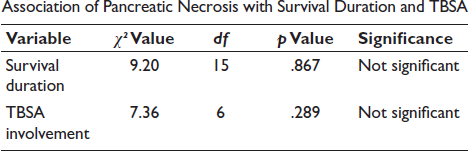
Regarding survival after burns, 38.1% lived beyond one week, 26.9% survived 97 hours to one week, and 11.1% survived 73–96 hours. Early deaths were less frequent: 14.3% within 24 hours, 4.8% each within 24–48 hours and 48–72 hours.
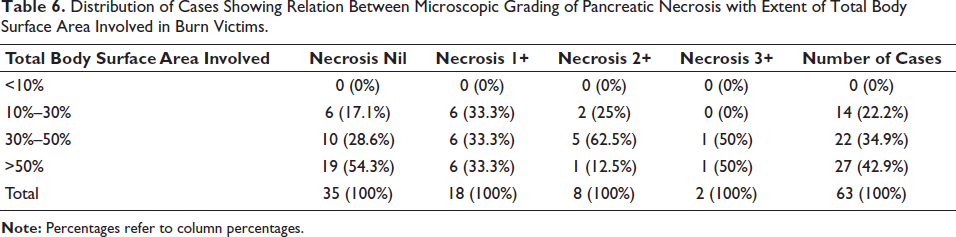
Fatal burns were generally extensive: 42.9% involved >50% TBSA and 34.9% involved 30%–50% TBSA. Moderate burns of 10%–30% accounted for 22.2%; none had <10% TBSA.
Comorbidities were present in 65.1% of victims. Diabetes was most common (38.1%), followed by coronary artery disease (14.3%) and hypertension (6.3%); 34.9% had no known comorbidities.
Grossly, pancreatic congestion (42.9%) and necrosis (23.8%) were the most frequent findings, while hemorrhage and fibrosis were uncommon (Table 1).
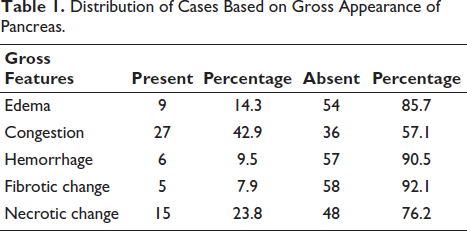
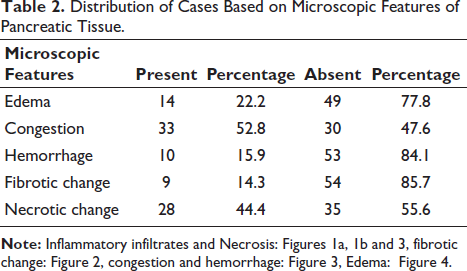
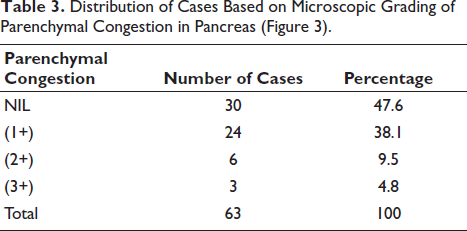
Microscopically, pancreatic congestion (52.8%) and necrosis (44.4%) were the most common changes, with fewer cases showing hemorrhage (15.9%) or fibrosis (14.3%) (Table 2). Nearly half of the cases (47.6%) showed no parenchymal congestion, whereas mild (1+) congestion was observed in 38.1% of cases (Table 3). Inflammatory infiltrates were absent in 58.7% of cases, while mild (1+) infiltration was noted in 19% and moderate to severe in 22.2% (Table 4).
Distribution of Cases Based on Gross Appearance of Pancreas.
Distribution of Cases Based on Microscopic Features of Pancreatic Tissue.
Distribution of Cases Based on Microscopic Grading of Parenchymal Congestion in Pancreas (Figure 3).
Distribution of Cases According to Microscopic Grading of Inflammatory Infiltrates in the Pancreas (Figures 1b and 4).
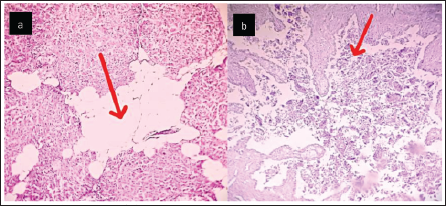
(a) Infiltrated Adipocytes in Pancreatic Tissue. (b) Necrosis and Inflammation of Pancreas (H&E Stain, ×200; Scale Bar = 50 µm).
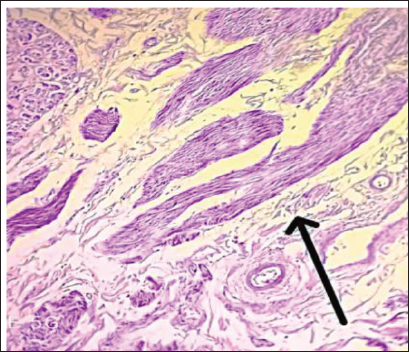
Pancreatic Fibrosis.
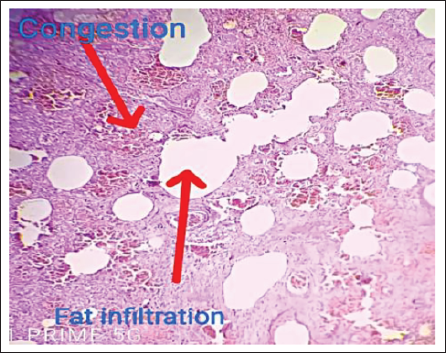
Congestion and Fat Infiltration of Pancreas (H&E stain, ×200 Magnification; Scale Bar = 50 µm).
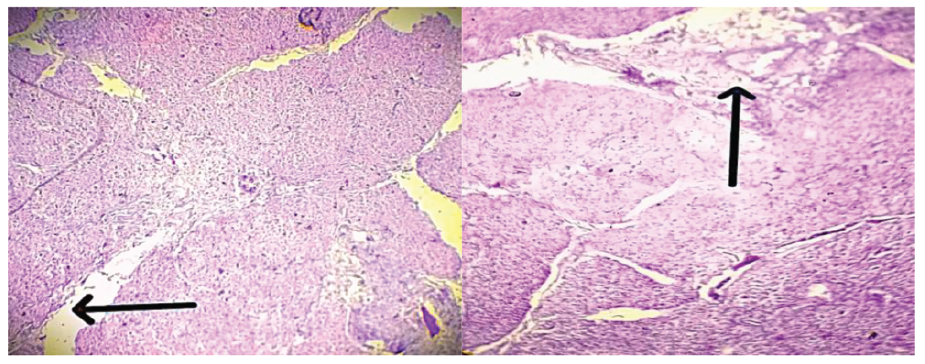
Edema and Inflammation of Pancreatic Tissue (H&E Stain, ×200 Magnification; Scale Bar = 50 µm).
Microscopically, pancreatic congestion (52.8%) and necrosis (44.4%) were the most common changes, with fewer cases showing hemorrhage (15.9%) or fibrosis (14.3%).
Pancreatic necrosis was almost absent in cases that died within the first 72 hours following burn injury, with most showing no necrotic change. As the survival period increased, especially beyond 97 hours, the frequency and severity of necrosis rose considerably. Among those who survived more than one week, nearly half of the cases demonstrated some degree of necrosis, including moderate (2+) and severe (3+) grades. This trend indicates that pancreatic necrosis is a time-dependent pathological change, developing more prominently with prolonged survival after burns. The chi-square test showed no significant association between survival duration and necrosis severity (
Distribution of Cases Showing Relation Between Microscopic Grading of Pancreatic Necrosis with Period of Survival of Burn Victims.
It was observed that more than half of the cases with extensive burns involving >50% TBSA showed no necrosis, whereas lower grades of necrosis (1+ and 2+) were more frequently associated with moderate burn areas (10%–50% TBSA). Notably, the highest grades of necrosis (2+ and 3+) were predominantly seen in the 30%–50% TBSA group rather than in those with very extensive burns. This indicates that the severity of pancreatic necrosis does not increase proportionately with burn extent; instead, it appears to be influenced by additional factors such as survival duration, systemic inflammatory response, and underlying comorbidities. The chi-square test again showed no significant association (
Distribution of Cases Showing Relation Between Microscopic Grading of Pancreatic Necrosis with Extent of Total Body Surface Area Involved in Burn Victims.
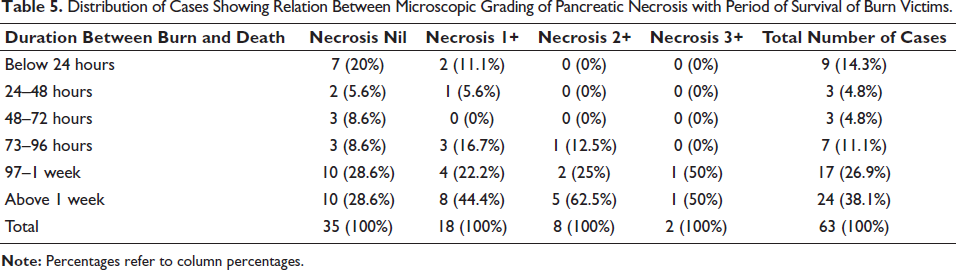
Association of Pancreatic Necrosis with Survival Duration and TBSA
In addition to chi-square, no parametric trend analysis was performed. Spearman’s rank correlation demonstrated a weak positive association between necrosis grade and survival duration (
Grossly, pancreatic congestion (42.9%) and necrosis (23.8%) were the most frequent findings, while hemorrhage and fibrosis were uncommon (Table 1).
Discussion
This autopsy-based study evaluated pancreatic gross and microscopic alterations in thermal burn deaths and explored their relationship with survival duration and TBSA involvement. The marked female predominance reflects regional epidemiological patterns of flame burns in domestic settings, and therefore, external generalizability may be limited.
Pancreatic Necrosis and Survival Duration
Pancreatic necrosis was minimal in early deaths (<72 hours) but progressively increased in both frequency and severity after 97 hours, with the highest grades among > one-week survivors. This supports the concept that necrosis is a delayed downstream effect of a sustained systemic inflammatory surge, catecholamine excess, and prolonged metabolic stress rather than an immediate direct thermal effect. This temporal trend parallels the observations of Abhishek Das et al.,
5
who noted hemorrhage/necrosis appearing after 2–4 days. Although this trend was biologically consistent, chi-square did not reach statistical significance (
Pancreatic Necrosis and TBSA
Higher necrosis grades were more frequently seen in moderate TBSA (30%–50%) rather than extreme TBSA (>50%), possibly because extensive burns cause earlier mortality, limiting time for necrotic evolution. Thus, TBSA alone does not proportionally determine necrosis severity. This partly aligns with Gauglitz et al.,
6
who described intense metabolic dysregulation beyond ~40% TBSA. The association remained statistically non-significant here (
Mechanistic Context
Burn physiology is characterized by hypermetabolism, insulin resistance, and systemic inflammatory cascades,7–10 which can explain the observed congestion, edema, and necrosis. Differences in hemorrhage prevalence compared to Das et al. 5 may reflect variable post-burn survival windows and histological thresholds. Additionally, the pancreas is among the most autolysis-prone organs postmortem, and although autopsies were done early with prompt fixation in this series, subtle autolysis-related exaggeration cannot be entirely excluded.11, 12
Overall, these findings suggest pancreatic involvement is an evolving downstream pathophysiological phenomenon in burns, more influenced by time-course than TBSA magnitude alone. Larger multicentric prospective clinico-pathological studies integrating metabolic markers and real-time pancreatic injury indexing may clarify its prognostic role.
Conclusion
This study highlights that pancreatic congestion and necrosis are not rare in deaths due to thermal burns, and that necrosis tends to be more frequently encountered in individuals who survive beyond the early acute phase. Although the statistical associations with TBSA and survival duration were modest, the observed trend with prolonged survival suggests that pancreatic involvement may form part of the evolving systemic inflammatory and metabolic response in burn injuries. Given the rapid PMI and prompt fixation, the microscopic findings in this series reflect meaningful pathological patterns rather than mere postmortem artifact.
These results help to broaden the understanding of organ-level changes in burn fatalities and provide a useful baseline dataset for future work in this direction. The observations made here create groundwork for more detailed clinicopathological studies integrating biochemical, metabolic, and imaging parameters in living patients, which may eventually clarify how pancreatic injury interacts with burn physiology and influences overall outcomes.
Limitations of the Study
While this study provides meaningful descriptive insights, some constraints warrant mention. It is a single-center series with a predominantly female cohort, reflecting local epidemiology but limiting generalizability. Histopathological grading was performed by one pathologist, and inter-observer comparison was not feasible. Although the postmortem interval was short and fixation prompt, subtle early autolysis in pancreatic tissue cannot be entirely excluded. Since stratified analysis for diabetes could not be reliably performed, diabetic status may have confounded the observed association between burn-related changes and necrosis.
These limitations do not negate the relevance of the observations; rather, they define areas for future work. Larger multicentric studies incorporating multivariable analysis and ante-mortem metabolic markers may further clarify the contribution of pancreatic pathology to systemic responses in burn injury.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Approval was obtained from the Institutional Ethics Committee (NRSMC/IEC/22/2024). Informed consent was taken from the legal claimant of the deceased, and confidentiality was maintained.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
