Abstract
Precise estimation of time since a death has great importance while investigating suspicious foul play in deaths. Findings help to estimate time since death gradually diminishes along with the progression of time. In the case of decomposed bodies, entomological evidence plays a great role in finding out the probable time since death. In this case series, we observed and analysed the life cycle of flies and gave opinions about the time since death. For this purpose, we developed an artificially ventilated environment and did not change the feeding substance. We observed that when the body is in an advanced stage of decomposition; entomological evidence acts as an important tool to estimate the time since death.
Introduction
Entomological evidence plays a crucial role in forensic investigations, especially in estimating the time since death. Forensic entomologists analyse the insects found on a corpse, considering their developmental stage to determine the postmortem interval. This analysis provides crucial information for investigators to reconstruct events leading to death and serves as valuable support that can confirm or challenge other evidence in a case.1, 2 In addition to estimating the time since death, entomological evidence can also be used to determine the location of death and aid in toxicological evaluations when traditional biological samples are not available. This information can be crucial in criminal investigations and provide valuable insights into the circumstances surrounding a suspicious death.1, 3
The accurate estimation of the time since death is crucial in forensic investigations. It not only helps establish a timeline of events but also assists in determining the cause of death, identifying potential suspects and providing valuable evidence in the court of law. 4 Forensic entomology, specifically the analysis of insect evidence, has emerged as a reliable method for estimating the time since death in forensic investigations. 5 By studying the life cycle and behaviour of necrophagous insects, forensic entomologists can estimate the minimum time since death based on the developmental stage and species composition of the insects found on the body. This information, combined with other postmortem findings, can provide important insights into circumstances surrounding death. 1
Case Series
In the past six months, our department has handled three cases involving bodies in an advanced stage of decomposition and entomological evidence. In one instance (Case 1), the decomposed body of an unidentified female was found in bushes and transferred to our mortuary for examination. During the examination, we observed marbling and blackish discoloration on the skin, loose teeth inside the oral cavity, easily pluckable hair, loosened nails, absence of rigour mortis and postmortem staining. Internally, liquefaction of the brain and spleen was noted along with softening of abdominal tissues. Additionally, multiple live maggots were found in various orifices; these were carefully collected alongside muscle bits for further study in a controlled environment to complete their life cycle. Few live maggots were collected separately for gross and microscopic examination.
In Case 2, The body of an unknown middle-aged man was discovered in a forested area, showing early signs of decomposition. Upon examination, there was bluish discoloration on the abdomen and well-developed rigour mortis in the large and small joints. Postmortem staining was also visible on the back. Additionally, a stab wound was found on the left side of the chest. The soft tissues were missing from the left forearm at elbow level with a crescent-shaped margin suggesting postmortem animal scavenging. The body was carefully examined for entomological evidence, and a pale-yellow colour cluster of eggs was noted over the left elbow area & left side of the chest. These eggs were meticulously gathered alongside muscle fragments and relocated to a controlled environment with artificial ventilation to undergo their life cycle.
In Case 3, The body of a young adult male was found in a paddy field. Upon external examination, weak rigour mortis was present over the large joints of the lower limbs. Postmortem staining was not noticeable, and patchy discoloration ranging from blackish to greenish was observed on the abdomen, neck and upper chest.
Evidence of insect activity was apparent, with multiple live maggots discovered around body openings and surrounding areas. These maggots were carefully collected along with muscle bits and transferred to an artificially ventilated environment for their life cycle to complete.
We set up an artificially ventilated environment using a rectangular glass specimen jar. We placed around 500 grams of dry soil and several dry leaves inside, then added entomological samples and muscle bits for observation. To ensure proper ventilation, we covered the top of the jar with a white paper sheet perforated with multiple small holes. Finally, we positioned the entire setup in a shaded area. 6
The collected maggots and eggs were observed closely under controlled conditions, monitoring their growth and development. During the observation period, we documented the sequence of developmental phases of eggs & maggots and their developmental rates. 7 In all three cases, we noted the range of temperature during the observation period, which ranged from 26°C to 32°C. 1
To determine the developmental stages, a small number of live maggots from controlled environments were gathered. These maggots were preserved in rectified spirit for wet mounting to facilitate microscopic examination and assess their developmental stage. To achieve this, cross-sectioning of the posterior end was conducted and secured with DPx mounting solution for identification of posterior spiracles.
Discussion
Case 1
At the time of postmortem, collected maggots were examined grossly and their size was up to 12 mm with a hairy appearance. On microscopic examination, a segmented posterior spherical with incomplete three lobes & tubercles were noted. After 24 hours maggots were kept in the artificially ventilated environment, and a significant increase in maggot size was observed & gross their size was up to 15 mm. On microscopic examination, a cross-sectional view of the posterior segment shows distinct three lobes (Figure 1). In the next 24 hours, maggots migrate away from these remains, indicating their continued development and movement. Initially, they turn into reddish-brown colour soft shell-like structures and gradually turn into dark black colour hard shell-like structures, suggestive of the pre-pupae and pupae stage (Figure 2).
Larvae Length 12 mm with Hairy Appearance; on Microscopically Posterior Spiracle with Incomplete Three Lobe Followed by Distinct Segmented Posterior Spherical Three Lobe.
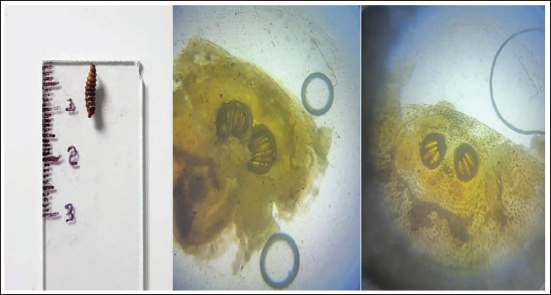
Reddish Colour Soft Shell-like Structure—Pre-pupae, Blackish Colour Hard Shell-Like Structure—Pupae.

These findings suggest that the maggots collected from the decomposed body were undergoing successive developmental stages, progressing from larvae to pre-pupae and pupae. It remained in the same condition for two to three days & turned into a green metallic colour adult fly, identified as a green bottle fly (Figure 3). The complete life cycle of the maggots collected from the decomposed body was successfully observed and documented, with the maggots progressing through various developmental stages from third instar larvae, pre-pupae, pupae, and finally emerging as adults. The total observation period was approximately five to six days. The maggots present at the time of postmortem belong to the third instar phase which finally developed into green bottle flies.
Green Metallic Colour Fly—Green Bottle Fly.

The findings from the observation of the collected maggots and their developmental stages suggest that the time since the death of the decomposed body in Case 1 was approximately three to five days. But, if we consider the decomposition features to estimate the time since death, it will be between 5 and 10 days. 8
In Case 2, collected eggs hatched on the very next day after being placed in the artificially ventilated environment & larvae appeared. Grossly their size was up to 5 mm and microscopically unsegmented posterior spherical noted (Figure 4), suggestive of the first instar phase. 9 Within the following 24 hours, the larvae experienced growth, reaching a size of about 10 mm. Under microscopic examination, they exhibited a segmented posterior sphere with two lobes, indicating the 2nd instar phase (Figure 5). 9 In the next 24 hours, maggot size increased to approximately 15 mm with a hairy appearance, and a distinct three-lobed cross-sectional view of the posterior segment (microscopic view), indicating the progression to the third instar phase. 9 This stage lasts for two days. By the 6th day, some maggots moved away from the feeding site to selectively pupate. Subsequently, a reddish-brown soft shell-like structure was observed, gradually transforming into a blackish, hard shell-like structure identified as pupae. This pupal stage lasts for two to three days before finally developing into green bottle flies. Considering the postmortem features, the estimated time since death was 12–24 hours. However, when considering entomological evidence, the time frame was approximately within 24 hours, which lacks specificity.
Larvae Length 5 mm. On Microscopically Unsegmented Posterior Spiracle Noted—1st Instar Stage.

Larvae Length 10 mm. On Microscopically Segmented Posterior Spiracle With Two Lobes Noted—2nd Instar Stage.
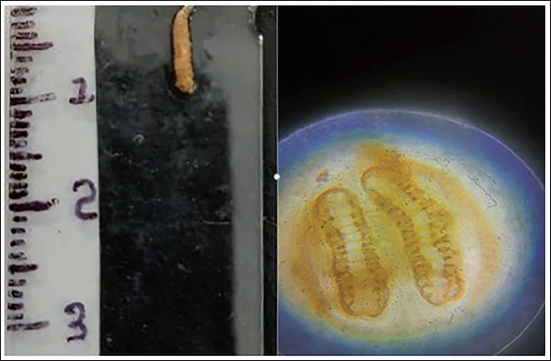
In Case 3, during the initial examination of the maggots, their size was noted to be up to 5 mm. Microscopically, an unsegmented posterior sphere was observed, indicating the first instar phase. Subsequently, within the next 24 hours, these maggots reached a size of around 10 mm, displaying a segmented posterior sphere with two lobes, signifying progression to the second instar phase. In the subsequent 24 hours, their size increased to approximately 15 mm, and they developed a hairy appearance. Additionally, a distinct three-lobed cross-sectional view of the posterior segment was observed, indicating the third instar phase lasts for up to 48 hours.
In this stage, the maggots showed active feeding behaviour. Starting from the 6th day, the maggots moved away from their feeding site for selective pupation. They underwent a gradual transformation, first adopting a reddish-brown soft cell-like structure and later developing into a dark black hard cell-like structure, indicative of the pre-pupae and pupae stage. The pupal stage lasted for two to three days before the emergence of green bottle flies. For Case 3, the estimated time since death derived from entomological evidence was approximately 24–48 hours. However, considering postmortem changes, the time since death is more accurately narrowed down to within 24–36 hours, rendering it more precise than the entomological evidence.
Green bottle flies deposit eggs in relatively fresh corpses within the first 24 hours of death. These eggs hatch within a day, giving rise to first instar larvae that commence feeding on decomposing tissues. As the larvae progress through their instar stages, they enter the 2nd instar phase typically within the subsequent 24 hours, followed by the development of the 3rd instar phase within an additional 24 hours. During the 2nd and 3rd instar phases, maggot sizes reach up to 10 mm and 15 mm, respectively. Microscopically, the posterior segments exhibit a segmented appearance with two lobes during the 2nd instar phase and three lobes during the 3rd instar phase.2, 10
Conclusion
The study of green bottle fly maggots and their life cycle is crucial in forensic entomology for estimating the time since death in decomposed bodies. Careful observation of the different instar phases, from egg hatching to adult fly emergence, provides vital evidence and helps narrow down the time since death. 11 The combination of entomological evidence and postmortem changes is crucial for accurate estimations, as demonstrated in observed cases. It is important to note that identifying fly species based on adult characteristics alone can be challenging. Examining eggs and maggots at different stages of development, along with observing postmortem changes, provides a more comprehensive and reliable estimate of the time since death in decomposed bodies. This case series emphasises the importance of using species-specific developmental data sets and considering environmental conditions when estimating the time since death in decomposed bodies using entomological evidence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
Ethical approval taken from Institutional Ethics Committee and informed consent taken from legal guardian.
