Abstract
Objective
To investigate the real-world impact of organised mammography screening on breast cancer mortality in metropolitan areas with widespread opportunistic imaging.
Methods
We conducted a population-based incidence-density case–control study among women aged 50–74 years resident in Milan, Italy. We matched 900 breast cancer deaths (diagnosed 2008–2022; died 2012–2023) to 3600 controls by age (±3 months), postcode and citizenship. Individual screening histories were reconstructed by deterministic linkage across five administrative databases. Conditional logistic regression estimated odds ratios (ORs) for breast cancer death by invitation and participation. Opportunistic mammography was modelled, and self-selection bias was corrected using a factor of 1.28.
Results
Invitation was associated with lower mortality (adjusted OR 0.71, 95% CI 0.58–0.85). Participation in at least one programme screen yielded an adjusted OR of 0.44 (0.36–0.52), which attenuated to 0.64 (0.53–0.77) after self-selection correction. Adjustment for opportunistic mammography had minimal impact on programme estimates.
Conclusions
In a large metropolitan setting characterised by intensive opportunistic imaging, organised screening confers a significant mortality reduction, chiefly among participants. Policies discouraging opportunistic substitution and enhancing adherence may increase population benefit.
Keywords
Introduction
Since the 1990s, early diagnosis through organised mammography screening programmes has been one of the most effective strategies for reducing breast cancer mortality, thanks to the identification of tumours at earlier stages, which are more amenable to conservative treatment. Numerous European systematic reviews and observational studies confirm this benefit: among all women who are invited, mortality falls by 22% to 31%, whereas the estimate rises to 38%-48% when analysis is limited to women who attend. The difference between these two ranges therefore reflects the combined influence of biological efficacy and the level of participation: the higher the uptake, the greater the gain in lives saved. These findings, described in the pioneering studies of Shapiro and Tabár and subsequently confirmed by further research,1–6 indicate that the impact of screening varies according to organisational and behavioural factors. The magnitude of the effect depends on several parameters, including target age range, screening interval, invitation and participation coverage, and the overall quality of the diagnostic–therapeutic pathway.
In Italy, a nationwide network of screening programmes has been in place since the early 2000s; multicentre studies such as IMPATTO 4 have documented significant reductions in mortality and laid the methodological foundations for real-world evaluations. Assessing effectiveness in real-world settings is methodologically complex. Incidence-density case–control studies remain appropriate because they directly compare exposure histories in cases and controls while accounting for key confounders. 7 Observational and case–control studies carried out in other Italian regions and in Europe have confirmed the effectiveness of screening in public health practice.4,7,8 Local data are essential to verify whether the expected benefit is being achieved and to identify organisational factors that may amplify or attenuate it.
The city of Milan is a metropolis with very high population density, considerable socioeconomic heterogeneity, and heavy reliance on ‘opportunistic’ mammography outside the organised pathway. Despite extensive international literature, the actual benefit of organised screening in the Milanese population has never been quantified using local data. This knowledge gap motivates the present study, which aims to evaluate whether, and to what extent, the offer of and adherence to organised screening reduce breast cancer mortality in a large metropolitan area.
To address the complexity of this question, case–control studies represent an efficient design because they can measure the association between participation in screening and death from breast cancer, taking account of potential confounders.
In this study, using individual-level linkages across population, screening, cancer registry and mortality databases, we aimed to capture both programme and opportunistic mammography, and explicitly correct for self-selection. This design isolates the mortality benefit attributable to the organised programme and informs policy in similarly urbanised settings. The metropolitan setting, characterised by dense health-care infrastructure and substantial population mobility, provides a stringent real-world test of screening performance that complements evidence from smaller or rural populations. By integrating robust bias-adjustment methods with comprehensive, population-based data, our analysis was intended to isolate the net mortality benefit attributable to the organised programme, illuminating how organised screening can remain effective even when opportunistic services are widely available.
These insights have direct relevance for policy makers in similarly urbanised regions who must weigh the coexistence of public programmes and private imaging against the goal of equitable, high-impact breast cancer control. Our aim therefore was to produce indications that can be transferred both to the other areas governed by the Agency for Health Protection of the Metropolitan City of Milan – covering Milan and 191 additional municipalities – and to other highly urbanised settings characterised by intense diagnostic opportunism.
Methods
Organised screening programme setting
The organised mammography screening programme in Lombardy reached full regional operation in 2007, but in the city of Milan it had already been active since 1999, with complete implementation in 2000 and coverage exceeding 60% from 2007 onwards. Biennial invitations were initially addressed to women aged 50–69 years, a threshold later extended to 45–74 years in accordance with the National Screening Programme and the National Prevention Plan. From 2016 onward, following Regional Law 23/2015, coordination and monitoring of the programme were assigned to the Agency for Health Protection (ATS) of the Metropolitan City of Milan, while provision was delegated to the local health trusts (ASSTs), which inherited information flows from three former local health authorities. As a pilot study, the investigation was conducted in the municipality of Milan, chosen because of the long-standing continuity of the screening programme, the availability of complete and centralised historical series of invitations and attendance information to develop screening evaluations, and the presence of a local Cancer Registry accredited by the International Agency for Research on Cancer (IARC) active since 1999. Conducting the study in a favourable setting will also make it possible, in the coming years, to test methodological approaches that can subsequently be extended to the entire ATS population and to other population-based screening programmes.
Data sources
Data were obtained through a deterministic linkage of the Cancer Registry, the Mortality Registry, the screening programme database, outpatient ambulatory procedure flows (SSP/28-SAN) related to opportunistic mammograms (those performed outside the organised programme), and the Population Registry (NAR). The linkage was performed using individual tax code with consistency check on date of birth and sex. Extracted information included tumour incidence, cause of death, invitation and attendance history, opportunistic mammography, and sociodemographic covariates. Clinical cancer characteristics were not systematically available across data sources and were therefore not included in the analysis. The resulting datasets were validated with internal and external quality control procedures.
Study design
A retrospective incidence-density case–control design was adopted, a methodology commonly used to evaluate the effectiveness of population-based screening programmes in the absence of randomisation. The study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines for case–control studies. Cases were defined as breast cancer deaths occurring between 2012 and 2023 among women aged 50–74 resident in the municipality of Milan and eligible for invitation to the organised screening programme. This age range corresponds to the core target population of the programme, characterised by biennial invitation protocols. Women aged 45–49, who were introduced more recently into the programme and are invited according to an annual screening protocol, were excluded to avoid heterogeneity in screening exposure and limited follow-up for mortality outcomes. Inclusion of deaths from 2012 was chosen to allow a latency period between screening exposure and the mortality outcome, and to ensure that women had screening opportunities before outcome assessment, given that individual organised screening history started from the 2006–2007 screening round. For each case, four controls were selected from the dynamic cohort of resident women identified through the Population Registry. Controls were eligible for screening between 2006 and 2023, alive and cancer-free at the case's diagnosis date and were matched on age (±3 months), postcode and citizenship. Controls were assigned the diagnosis date of the corresponding case (pseudo-diagnosis date) 4 9–12.
Women (cases and controls) who had not maintained continuous residence in the municipality of Milan for at least 2 years were excluded. Sensitivity analyses restricted to women with ≥4 years of continuous residence were performed to reduce exposure misclassification in the ‘not invited’ group.
The study is consistent with the IMPATTO protocol published by Puliti in 2008 4 and estimates programme effectiveness by comparing the screening histories of women who died from breast cancer with those of controls drawn from the same population. Because woman may undergo mammography outside the organised programme independently of invitation or participation status, opportunistic mammography was distinguished from programme screening and accounted as a potential confounder. The rationale is that, if screening is effective in reducing breast cancer mortality, participation in organised screening will be less frequent among cases than among controls, allowing the association to be quantified by odds ratios (ORs).
Opportunistic mammography
Opportunistic mammography was defined as examinations performed outside the organised programme, in the absence of known clinical symptoms, carried out on the woman's initiative or following referral by a general practitioner/specialist, not performed in response to an active programme invitation and outside or in addition to the programme-defined screening interval. Identification relied on outpatient administrative flows (SSP/28-SAN, years 2006–2023). Because outpatient administrative information does not contain direct information about symptoms, we applied a temporal exclusion (wash-out) strategy to minimise inclusion of symptom-driven exams. In the primary analyses, opportunistic mammograms performed within 12 months before the index date, as well as those temporally associated with other investigations (MRI, biopsy or histology) in the same interval, were excluded. Sensitivity analyses were conducted using alternative wash-out windows of 6 and 18 months. For adjustment, opportunistic exposure was coded as 0, 1, ≥ 2 exams before the index date, restricted to age 50–74 years and censored after diagnosis/pseudo-diagnosis. This approach distinguishes routine opportunistic use from symptom-driven clinical imaging near diagnosis, thereby reducing distortion in the estimated programme effect.
Exposure definition
Exposure was described along two complementary axes. The first concerned whether at least one formal programme invitation had been received, distinguishing ‘invited’ from ‘not invited’ women. The second considered whether at least one programme mammogram had been executed in response to the invitation, thus identifying ‘screened’ compared with ‘unscreened’ women. For each subject, the screening history was reconstructed starting from the 2006–2007 round. All invitations up to the 2022–2023 cycle were included and only examinations performed after the corresponding invitation were defined as screening mammography.
For exposure classification, invitations issued after the index date were ignored (censored) but retained in the database for quality checks. Women who received only one invitation that the Screening Data Warehouse flagged as ‘Excluded before invitation’ – without any documented reason – were likewise placed in the ‘not invited’ category, because they should ordinarily have received several invitations over time.
For women with more than one recorded invitation, entries labelled ‘Excluded before invitation’ were not counted as valid invitations. Women with only such entries were considered ‘not invited prior to the index date’. As a result, the ‘Invited’ group in Table 1 comprises only those invitations that were delivered and not subsequently invalidated, while all others fall into the ‘Not invited’ group.
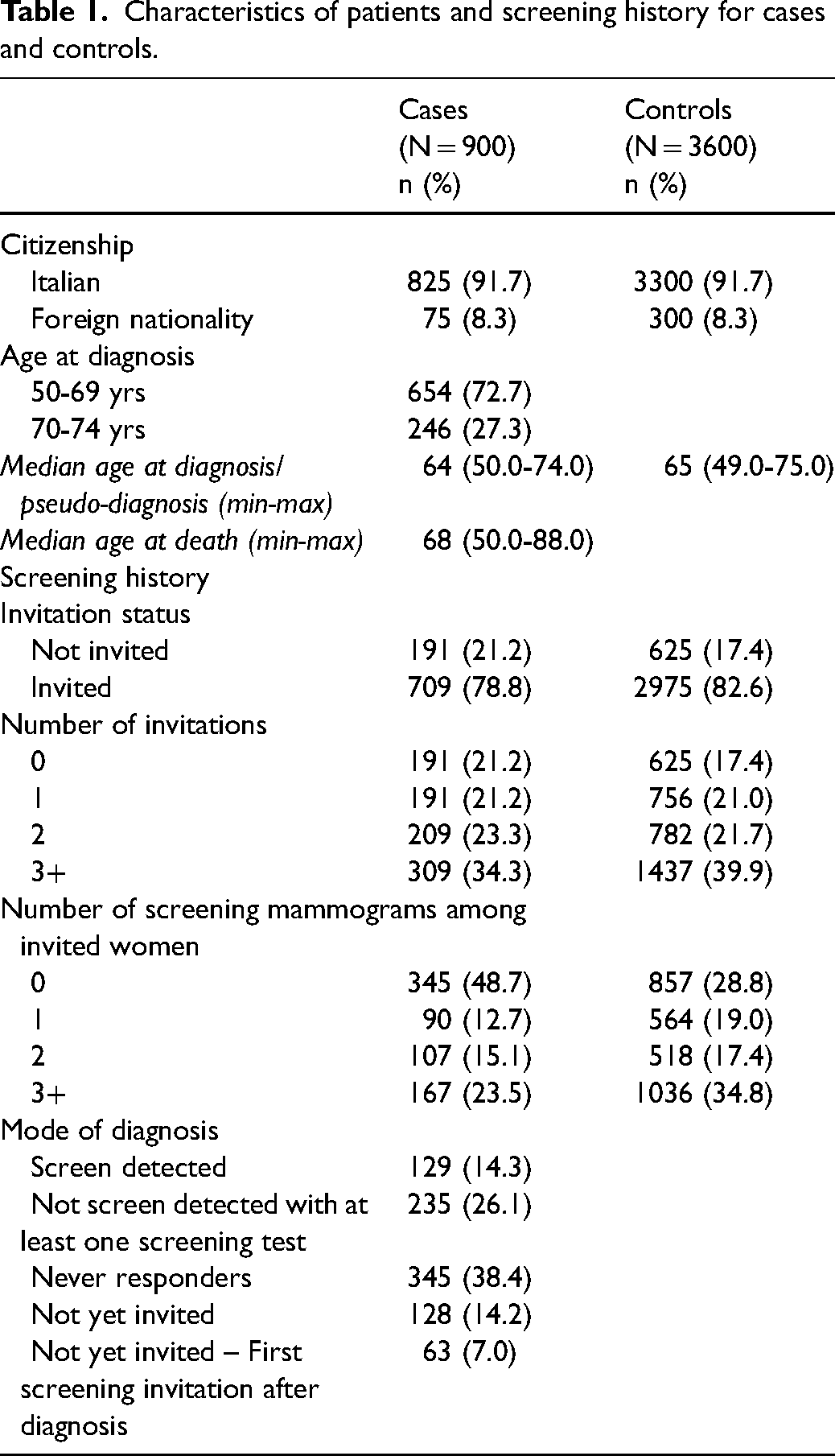
Characteristics of patients and screening history for cases and controls.
Opportunistic mammography, although recorded in the SSP/28-SAN flows from 2006 to 2023, was analysed separately and did not contribute to classify every woman as invited or not invited and screened or not screened. Women could experience mixed patterns of mammogram examination (e.g. programme exams combined with ordinary/programmed or direct non-Emergency Dept access) over time. In such cases, for exposure classification, opportunistic mammograms were included, to reflect screening activity outside exclusive adherence to the organised programme. Non-programme examinations classified as ‘urgent’ were not considered as opportunistic screening and therefore excluded. For controls matched to screen-detected cases, the pseudo-diagnosis date was postponed by 1 month (primary) to correct for lead-time bias, ensuring a comparable duration of the preclinical detectable phase between cases and controls. 13 Opportunistic mammograms performed outside the 50–74 age range or executed after diagnosis in cases and after pseudo-diagnosis in controls were excluded from analysis to avoid overestimating extra-programme exposure.
Statistical analysis
Conditional logistic regression models were used to estimate the association between screening and mortality, accounting for individual-level matching of case and controls by birth cohort, area of residence and citizenship. The first model compared risk between women already invited and women not yet invited, thus assessing the impact of programme offer. The second model estimated the effect of actual participation by comparing women who had undergone at least one programme mammography before the index date with those who had never done so, including among the latter both non-responders and not-yet-invited women. The third model, limited to invited women only, contrasted screened and unscreened women to measure the effectiveness of participation within the population reached by the invitation. This last comparison is subject to self-selection bias because the decision to attend is not random but influenced by individual characteristics that may affect the risk of death; adjustment for socioeconomic or educational variables does not necessarily eliminate this bias, as shown by Duffy and subsequently by Puliti. To correct this distortion the method proposed by Duffy was applied; it uses a multiplicative factor based on the mortality ratio between non-responders and non-invited women. The original estimate proposed by Duffy 14 is 1.36, derived from trials with about 80% adherence. Using relative risks of 1.17, 1.11 and 0.84, respectively, Gabe, Puliti and Paap calculated a correction factor specific to their study areas, considering participation of about 60%–65% for Puliti and Gabe and 82% for Paap.4, 7, 15 Self-selection was corrected using Duffy's method with participation rate of 70% and the mortality ratio R between non-attenders and non-invited estimated from our data (R = 1.28). We propagated uncertainty to obtain corrected ORs.
We reported both the total effect (without adjustment for opportunistic mammography) and the direct effect (adjusted), given that opportunistic imaging may act as a mediator of the invitation–mortality pathway. All analyses were performed using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA) and RStudio (Posit Software, Boston, MA, USA).
Results
Between 2012 and 2023, 900 deaths from breast cancer diagnosed between 2008 and 2022 were recorded among women aged 50–74 and resident in Milan; for each case four controls were selected, yielding a total of 3600 controls, and for all 4500 women the screening history was reconstructed from the 2006–2007 round to the 2022–2023 cycle. Table 1 shows that 91.7% of subjects were Italian citizens, the median age at diagnosis was 64 years and the median age at death among cases was 68 years; 78.8% of cases and 82.6% of controls had received at least one invitation, whereas 21.2% of cases and 17.4% of controls had not yet been invited.
Among invited women, 48.7% of cases and 28.8% of controls had never attended, while 23.5% of cases and 34.8% of controls showed high attendance (more than three programme mammographies). Table 1 also shows that 14.3% of deaths corresponded to screen-detected tumours, while 26.1% were not screen-detected cases but occurred among women who had undergone at least one programme mammography.
Table 2 documents that 85.6% of cases and 78.1% of controls had not undergone opportunistic mammography, and that among invited women 12.0% of cases compared with 19.9% of controls had in fact had opportunistic mammographies. Among non-invited women the frequency of at least one opportunistic test was slightly higher in cases (8.9%) than in controls (7.4%). Conversely, controls have higher opportunistic use among both screened and unscreened women.
Opportunistic mammography among invited/non-invited and screened/non-screened women.

The results of conditional logistic-regression models reported in Table 3 show that an offer is protective: the risk of death is lower in invited than in non-invited women with a crude OR of 0.76 (95% CI 0.63-0.92) which decreased to 0.71 (0.58-0.85) after adjustment for opportunistic mammography. By contrast, analysis based on screening status indicates OR 0.50 (0.43–0.56) in the crude model and OR 0.48 (0.41–0.54) after adjustment for opportunistic mammography, corresponding to 50–52% lower odds of death. Limiting the comparison to invited women only, participation is associated with a 55% reduction in the crude model, OR 0.45 (0.38-0.53), which slightly increased to 56% after adjustment, OR 0.44 (0.36-0.52). Applying the correction factor of 1.28 for self-selection bias, the benefit is attenuated but remains evident with OR 0.66 (0.56-0.78) in the crude model and OR 0.64 (0.53-0.77) in the adjusted model, corresponding to reductions of 34% and 36% in the adjusted model (Table S1, see online Supplemental Material). When evaluated without temporal restrictions, opportunistic mammography appeared positively associated with breast cancer mortality (OR 1.7, p < 0.05), suggesting contamination by exams performed for symptoms close to diagnosis (Table S2, see online Supplemental Material). After applying progressive wash-out windows (6, 12 and 18 months before the index date, excluding exams associated with MRI, biopsy or histology), the association progressively attenuated. At 6 months, the OR was 0.95 (not significant), while at 12 and 18 months it fell to 0.6 and 0.5, respectively (both p < 0.05). The same pattern was observed across invited, not invited, screened and non-screened subgroups (Table S2). These findings support the interpretation that the apparent increased risk associated with opportunism reflects diagnostic mammography for symptomatic women, whereas ‘routine’ opportunistic use outside the programme does not confer a clear protective effect.
Odds ratios for breast cancer mortality risk according to different modes of screening exposure.
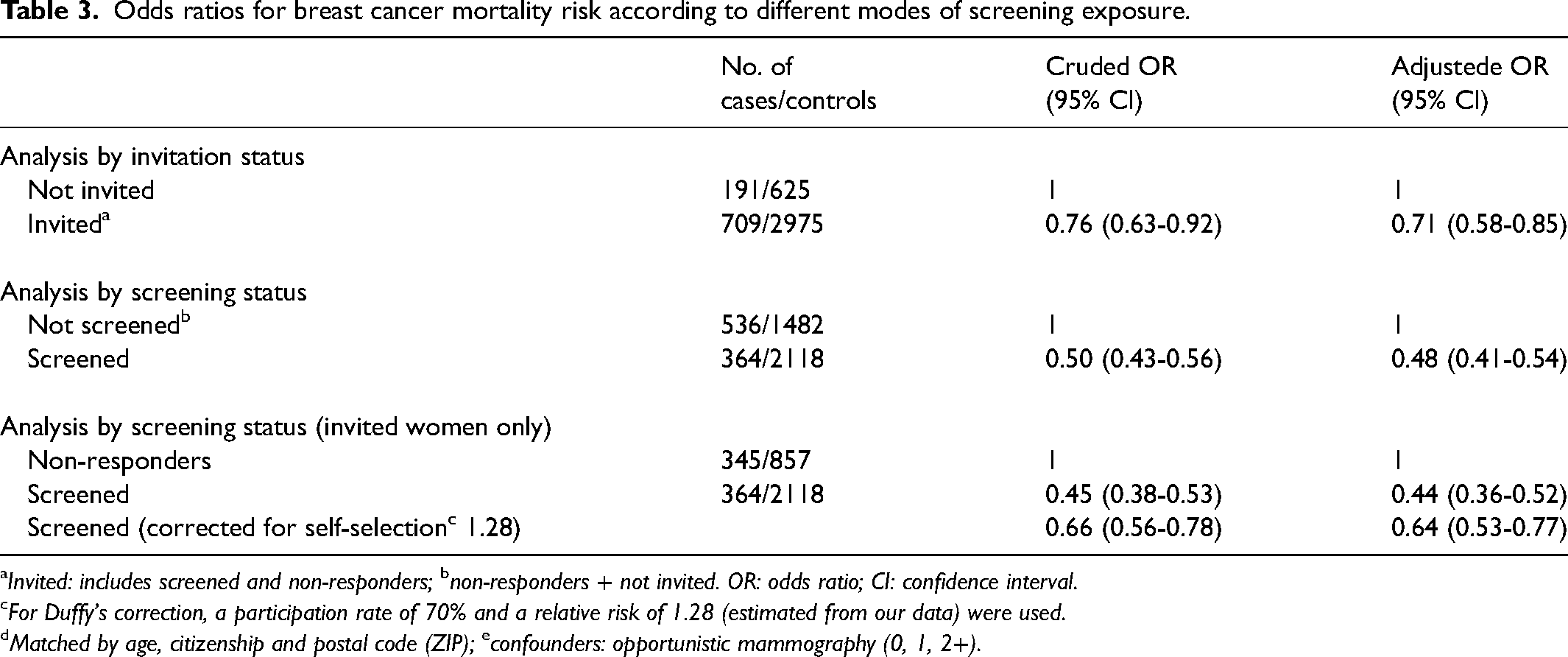
Invited: includes screened and non-responders; bnon-responders + not invited. OR: odds ratio; CI: confidence interval.
For Duffy's correction, a participation rate of 70% and a relative risk of 1.28 (estimated from our data) were used.
Matched by age, citizenship and postal code (ZIP); econfounders: opportunistic mammography (0, 1, 2+).
Overall, the data confirm that invitation alone has an important impact in reducing mortality; regular participation in the programme offers a significant advantage, although partially attenuated by self-selection correction. The adjustment for opportunistic mammography had minimal impact on programme estimates.
Discussion
The results of this study confirm that regular participation in the organised mammography screening programme of the city of Milan is associated with a reduction in breast cancer mortality, amounting to 55% in the crude model and 34% after correction for self-selection, rising to 36% when diagnostic opportunism is also controlled. Invitation alone, with a 29% reduction mortality risk, appears to exert significant protection.
The benefit observed in this case series, adjusted and uncorrected OR = 0.44 and corrected OR= 0.64, lies in the average of the European range: in the London case–control study the mortality ratio between participants and non-participants is 0.61, 10 while in the Dutch cohort of de Munck the hazard ratio is 0.76 after lead-time correction 16 and in the cohort of Otto the hazard ratio is 0.51 after self-selection correction. 17 The result is nevertheless consistent with the meta-analysis of Dibden and the overview by Broeders which, applying correction factors that vary with adherence, places Italy among the countries with reductions of about 20–30%. It appears lower than the model-based estimates of Monticciolo, 18 which hypothesise scenarios of optimal adherence yielding reductions above 40%. Compared with the IMPATTO study by Puliti, where invitation was associated with an OR of 0.75, our estimate (OR = 0.71) is consistent. In Lombardy, however, invitations now reach more than 80% of the target population, leaving a relatively small and possibly higher risk group of non-invited women.
The observed median ages at diagnosis and death reflect the inclusion criteria adopted to ensure a reliable assessment of women's exposure to organised screening. Restricting the analysis to women diagnosed between 2008 and 2022 and who died between 2012 and 2023 allowed that at least one screening round could be documented for each woman, while maintaining comparability between cases and controls. This selection may have led to the exclusion of some cases who died outside the chosen interval; however, this does not affect the internal validity of comparisons between cases and controls, as the same temporal criteria were consistently applied to both groups.
Correction for self-selection is the most delicate methodological issue. Applying Duffy's factor of 1.28 in our scenario, with a participation rate for the city of Milan at 70%, reduces the estimated benefit only slightly. Other correction factors proposed in the literature change the extent of the advantage without reversing its direction, suggesting that protection exists even if its magnitude is sensitive to the choice of correction (Table S1).
Residual healthy-screened effects could bias estimates away from the null (i.e. exaggerate apparent protection among participants), despite self-selection correction. Women who regularly attend preventive programmes generally have healthier lifestyles, greater propensity to undergo check-ups and better management of comorbidities. These protective behaviours affect all-cause mortality and overall mortality, making it difficult to distinguish the benefit due to screening from the effect of selection.
Lead-time was managed by delaying by 12 months the pseudo-diagnosis of controls of screen-detected cases, a compromise supported by literature that estimates an average sojourn time of 4 years; future refinements could parameterise the postponement by age and tumour biology as already done in the Netherlands. The main empirical limitation remains the non-negligible prevalence of opportunistic mammography (∼20% overall; higher among controls), which may attenuate exposure contrast.
The extent of opportunistic screening observed in Milan appears to be consistent with figures reported in previous studies, as in Italy, similarly to most European countries, opportunistic and organised screening routes coexist. 19 The apparent excess risk observed for opportunistic mammography in the crude analysis (OR 1.7) is plausibly explained by inclusion of examinations performed for symptoms or diagnostic suspicion close to cancer diagnosis. To reduce misclassification, we applied wash-out windows excluding opportunistic mammograms performed within 6–18 months before diagnosis and those temporally associated with MRI, biopsy or histology. Although this strategy mitigates contamination by diagnostic exams, residual misclassification cannot be ruled out, and some degree of bias in the estimated effect of opportunistic mammography may persist.
Once a temporal wash-out was applied, the association attenuated to null or slightly protective values, with estimates consistently below 1 across invitation and participation strata, although not always statistically significant. These results indicate that routine opportunistic use outside the programme does not generate the same structured mortality benefit observed for organised screening, but rather reflects heterogeneous behaviours driven by individual or clinical factors. From a methodological perspective, the analyses highlight the importance of applying exclusion windows to reduce contamination by diagnostic imaging.
This approach may have excluded some truly asymptomatic opportunistic examinations, potentially resulting in an underestimation of the opportunistic screening. Nevertheless, the long follow-up ensured that women undergoing multiple opportunistic examination over time were still classified as having opportunistic screening despite the wash-out procedure. From a policy perspective, the findings reinforce the role of organised programmes in delivering systematic quality control, timely recalls and equity of access, while opportunistic activity remains fragmented and less predictable in terms of population impact.
Finally, the incidence-density design with controls alive at the date of death of the case assumes correct classification of cause of death. Italian studies indicate an accuracy of 80–90% for ISTAT certificates for breast cancer, although systematic errors (e.g. coding bone metastases as ‘unknown primary’) could dilute the screening effect.
Among the strengths of this study, it is worth emphasising the deterministic linkage between five databases, the 16-year observation window, and the case–control design with strict matching for age, area of residence and citizenship. The evidence produced indicates that, in a metropolis with significant use of private diagnostics, organised screening retains a protective effect, but its magnitude is attenuated by out-of-programme mammography. To increase the collective health gain, it will be necessary to reduce opportunism, for example by integrating tariffs, implementing a centralised recall system and systematically recording diagnostic modality in oncology flows. Monitoring should also be extended to the new 45–74 age band provided for by the National Prevention Plan, periodically assessing impact with methods that incorporate pathway quality indicators, costs and outcomes beyond mortality, such as the disease-free interval recently proposed by de Munck 16 and the cost-effectiveness modelling suggested by Maroni. 11
Conclusions
Overall, organised mammography screening in Milan is associated with a reduction in breast cancer mortality, consistent with evidence from other European settings. After rigorous correction for self-selection and lead-time biases, the effect remains robust, although its magnitude is more moderate than in contexts less saturated with opportunistic mammography. The findings suggest that in metropolitan areas where opportunistic activity is extensive, the overall population-level benefit of organised screening may therefore be partly attenuated, yet remains evident and clinically relevant. Strengthening information systems, improving data quality, limiting opportunistic substitution and promoting regular participation represent key levers to enhance the impact of organised screening. Continuous monitoring of participation and accurate distinction between exposure types are essential to assess programme performance and ensure equitable access to early detection.
While opportunistic mammography represents a non-negligible component of breast imaging in metropolitan settings, its impact on mortality reduction appears limited and inconsistent. Organised screening, with its systematic invitations, quality standards and equitable coverage, remains the primary driver of population-level benefit.
Supplemental Material
sj-docx-1-msc-10.1177_09691413261433740 - Supplemental material for Real-world impact of organised mammography screening on breast cancer mortality: A population-based case–control study
Supplemental material, sj-docx-1-msc-10.1177_09691413261433740 for Real-world impact of organised mammography screening on breast cancer mortality: A population-based case–control study by Brunella Frammartino, Carlotta Buzzoni, Diego Iemmi, Danilo Cereda, Marco Zappa and Antonio Giampiero Russo in Journal of Medical Screening
Footnotes
Ethical considerations
The assessment of organised breast cancer screening programmes falls within the statutory public health duties assigned to the Lombardy Health Protection Agencies by Regional Law No. 33/2009 and by the annually issued Regole di Sistema, which oblige ATS to evaluate the impact of population-based preventive interventions. Because the present analysis was undertaken in fulfilment of a legal mandate, additional approval from an ethics committee was not required.
Authors’ contributions
AGR and BF conceived and designed the study protocol and defined the methodological approach. BF performed data management, data linkage and statistical analyses. AGR supervised the study and provided critical feedback to ensure consistency with epidemiological and public health perspectives. BF and AGR drafted the manuscript, incorporating input and revisions from all authors. CB, MZ, DC and DI critically revised the manuscript.
All authors read and approved the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data supporting this study cannot be shared due to restrictions imposed by the data provider.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
