Abstract
Objective
A better understanding of factors associated with cervical cancer screening can inform strategies for cervical cancer prevention. This study examined the relationship between age at human papillomavirus (HPV) vaccination and participation in cervical cancer screening among a nationally representative sample of women in the United States.
Methods
We utilized data from the National Survey of Family Growth for the years 2015–2019 focusing on women aged 18–24 vaccinated against HPV. Age at first HPV immunization was analyzed as both a dichotomous (vaccinated at 9–12 vs. 13–23 years) and a continuous variable. The outcome measured was ever having a Pap smear. Multivariable logistic regression that accounted for complex survey design was employed to estimate adjusted prevalence ratios and differences from average marginal predictions.
Results
The study comprised 981 individuals, representing 6.05 million women. Over half of the study population had a Pap test (57.4%). Women vaccinated at ages 9–12 were less likely to participate in screening compared to those vaccinated at ages 13–23 [risk difference: −9.1, 95% confidence interval (CI) −16.7 to −1.5)] which translates into 120,260 fewer women nationwide getting cervical cancer screening. Each 1-year increase in age at first vaccination was associated with a 1.1% (95% CI, −0.1 to 2.4%) higher probability of having a Pap test, but this linear trend was not statistically significant.
Conclusions
Our study underscores the importance of promoting cervical cancer screening not only among unvaccinated women but also among those who received the HPV vaccine at the recommended ages of 9–12.
Introduction
Strategies for cervical cancer prevention in the United States (US) encompass both human papillomavirus (HPV) vaccination and regular screening. Although evidence suggests that HPV vaccination significantly reduces the risk of invasive cervical cancer (88% lower rate if vaccinated before the age of 17, 95% CI 0–66%),1,2 routine screening in women over the age of 21–25 remains essential for cervical cancer prevention regardless of immunization status. 3 However, a concerning decrease in cervical cancer screening utilization was observed between 2005 and 2019, with the primary self-reported barrier being a lack of knowledge of screening. 4 These observations raise questions about the impact of increased HPV vaccination uptake and changes in screening behavior during this period.
The timing of vaccination can be a contributing factor to screening participation. Screening should begin at age 21, as per US Preventive Services Task Force 3 and the American College of Obstetricians and Gynecologists, 5 and at age 25, according to the American Cancer Society's guidelines. 6 Women approaching the age of screening eligibility may be more likely to undergo opportunistic screening or vaccination when attending a clinic for either service. Conversely, those vaccinated at the recommended age (9–12) may be less likely to initiate screening compared to those vaccinated later in life due to a variety of behavioral factors, including feeling protected against cervical cancer or having different knowledge, risk perception, or healthcare access than their parents or guardians who were likely responsible for their children's vaccination at an early age.7,8
Only a few studies have addressed the association between screening participation and age at first HPV vaccination.9–13 These studies lack screening data on women vaccinated at recommended age (9–12), and they lack generalizability to the broad US population.
This study aims to overcome the limitations of previous research by using a nationally representative dataset with data available up to 2019 encompassing screening behavior of those vaccinated at recommended age and including health and sexual behavior variables. The primary goal is to examine the association between age at first HPV vaccination and participation in cervical cancer screening, focusing on individuals vaccinated at recommended age (9–12) compared to those vaccinated at later ages. Enhancing our understanding of screening patterns by age at HPV vaccination will inform more effective, targeted cervical cancer preventive strategies.
Methods
Study population and measures
For this cross-sectional analysis, we combined the National Survey of Family Growth (NSFG) data from 2015–2017 and 2017–2019. The NSFG is a nationally representative survey conducted by the Centers for Disease Control and Prevention (CDC). Through a multistage probability clustering and stratification design, NSFG provides a nationally representative sample of the non-institutionalized civilian population aged 15–49 in the United States. Information is collected through in-person interviews and includes demographic characteristics, sexual activity, healthcare utilization, HPV vaccination history, and cervical cancer screening. 14 The response rates among female participants were 66.7% and 65.2% in 2015–2017 and 2017–2019, respectively. 15 Our study population included women who did not report prior hysterectomy, cervical cancer, or cervical dysplasia; were aged 18–24 at the time of assessment, and were aged 12 or younger in 2006 when HPV vaccines were first introduced. The last criterion was applied to ensure that all women in the study population had a theoretical possibility of getting vaccinated at recommended age (9–12). The study population excluded women over age 24 because they were not asked about HPV vaccination in the survey.
The outcome of interest in this study was participation in cervical cancer screening. This was determined by whether participants reported ever being screened for cervical cancer, using a Papanicolaou (Pap) test. The primary exposure of interest was the reported age at first HPV vaccination. We examined this exposure in two ways: (1) dichotomized into having been vaccinated at age 12 or younger (the recommended age) or at age 13 or older, and (2) as a continuous variable to assess a potential for linear trend. The secondary exposure was ever having been vaccinated.
Based on previous research and available data, the following factors were considered for analysis as potential covariates: age at the time of assessment (18–24), race and ethnicity (Hispanic, non-Hispanic Black, non-Hispanic of another race or multi-racial, non-Hispanic White), area of residence (metropolitan or non-metropolitan area), poverty level income (above poverty, at or below poverty level), primary language (English or other), birth country (US or other), insurance (yes/no), healthcare utilization in the past 12 months (yes/no), smoking history (smoked 100 or more cigarettes in a lifetime), and number of lifetime sexual partners (0–10+).
This study was exempted from the institutional review board of the University of California, San Francisco since it only used publicly accessible de-identified data.
Statistical analysis
The relationship between age at the first HPV vaccination and cervical cancer screening participation was analyzed using logistic regression models. Since NSFG is a multi-stage probability-based, nationally representative sample of the household population aged 15–49, we accounted for the complex survey design and applied sample weights following the NSFG documentation to obtain accurate estimates. We used adjusted predicted marginal models 16 to estimate adjusted marginal prevalence ratios (aPR) and adjusted marginal prevalence differences (aPD) with associated 95% confidence intervals (CI).
We used step Akaike Information Criterion 17 to assess model fit and select the covariates from the prespecified list for inclusion in the regression models. Both age at assessment and age at first HPV vaccination (when examining age on a continuous scale) were included in the model as linear terms due to no evidence of non-linearity.
We additionally examined the socio-demographic characteristics of those who were vaccinated at 9–12 years and did not participate in cervical cancer screening later in life and conducted a sensitivity analysis excluding women who were over-screened, i.e., received a Pap test before the age of 21, and were under age 22 at the time of assessment. For our main analysis, we chose not to exclude over-screened women for several reasons: (1) to enhance statistical power, and (2) to reflect real-world practices, as a significant proportion (44%) of screened women underwent Pap testing between ages 18 and 21. The sensitivity analysis among over-screened women aimed to determine whether the findings aligned with those of the main analysis.
Unless noted otherwise, the percentages and estimates in the text below are reported for the weighted dataset that reflects the US population represented by the participants of the NSFG survey.
STATA (version 16.1) was used for all data analyses. 18 All statistical tests were two-sided with a pre-specified type I error rate of 0.05.
Results
After excluding individuals who did not report getting vaccinated (n = 895) or had missing data on age at first vaccination (n = 47), the study population comprised 981 individuals representing 6.05 million US women. No women reported being vaccinated at 24 years, making 23 years the upper limit of vaccination age. There were some differences in baseline characteristics between women vaccinated against HPV at the recommended age (9–12) and women vaccinated later in life (13–23) (Table 1). In particular, compared to those vaccinated at 13–23, women vaccinated at the recommended age were more likely to be Hispanic (23.3 vs. 20.7%) or non-Hispanic of other or multiple races (12.5 vs. 11.0%), have been born in the US (95.2 vs. 90.7%) and have English as their primary language (95.5 vs. 92.2%), be insured (92.5 vs. 87.6%), have had 2–4 sexual partners (37.3 vs. 23.8%), and to not have been over-screened (65.6 vs. 58.5%) (Table 1).
Characteristics of women vaccinated against HPV, NSFG 2015–2019.
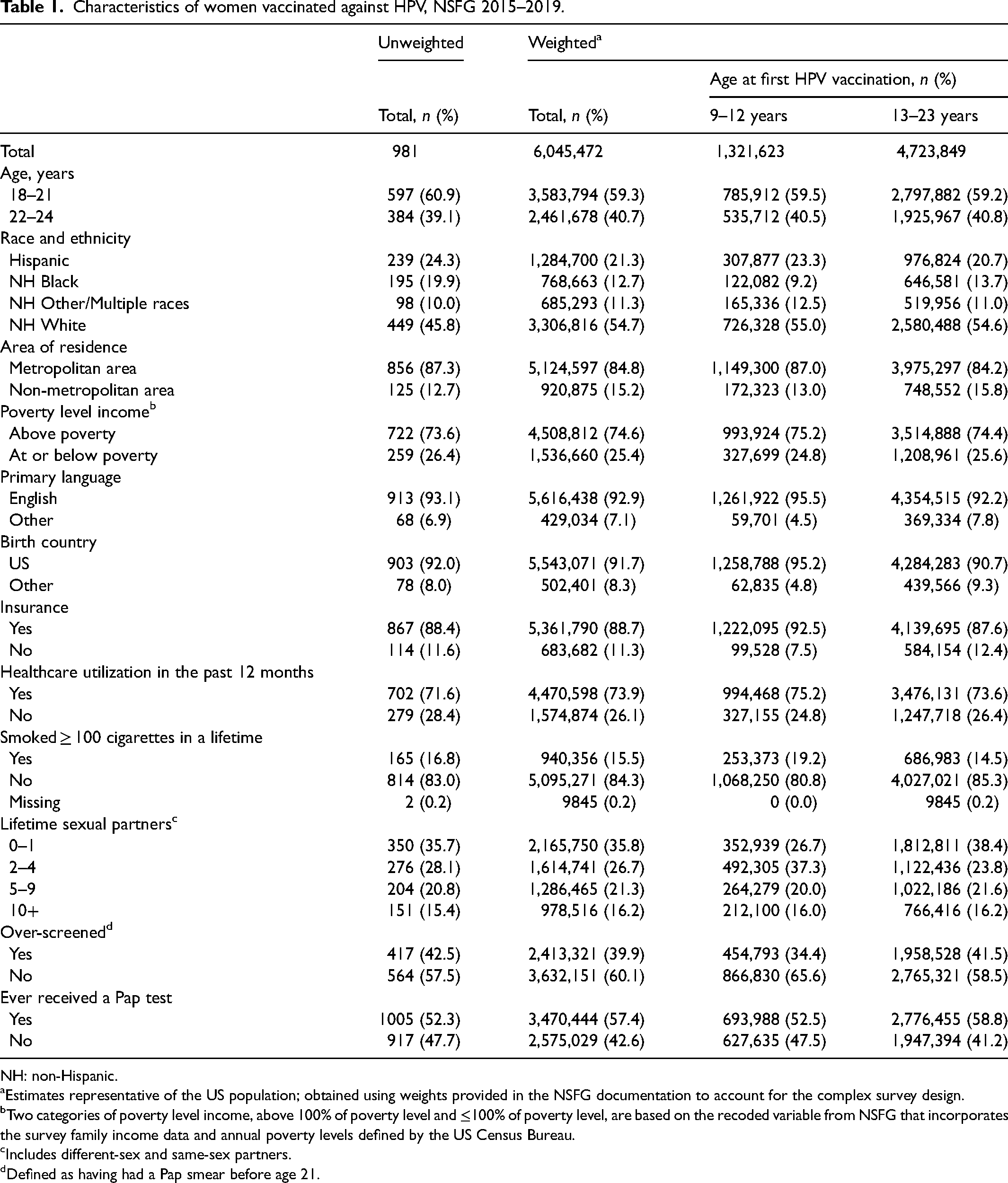
NH: non-Hispanic.
Estimates representative of the US population; obtained using weights provided in the NSFG documentation to account for the complex survey design.
Two categories of poverty level income, above 100% of poverty level and ≤100% of poverty level, are based on the recoded variable from NSFG that incorporates the survey family income data and annual poverty levels defined by the US Census Bureau.
Includes different-sex and same-sex partners.
Defined as having had a Pap smear before age 21.
Over half of the study population had received a Pap test (57.4%, Table 1). In the crude analysis (not shown), the predicted prevalence of a Pap test did not significantly differ by age at HPV vaccination, with 6.3% (95% CI, −0.3 to 1.6%) lower probability of having a Pap test among women vaccinated at the recommended age in comparison to women vaccinated later in life.
In the analysis adjusting for age at time of assessment (as a linear term), race and ethnicity, insurance, birth country, primary language, number of lifetime sexual partners, and healthcare utilization, women vaccinated at the recommended age had a statistically significant 9.1% (95% CI, 1.5–16.7%) lower probability of having a Pap test than women vaccinated later in life (Table 2). This 9.1% difference translates into 120,260 (95% CI, 19,823–220,697) fewer women nationwide getting screened for cervical cancer than could be if those vaccinated at the age of 9–12 had the same screening probability as those vaccinated at 13–23 years.
Weighted adjusted prevalence of Pap test among women vaccinated against HPV, NSFG 2015–2019 (unweighted, n = 981; weighted, n = 6,045,472).
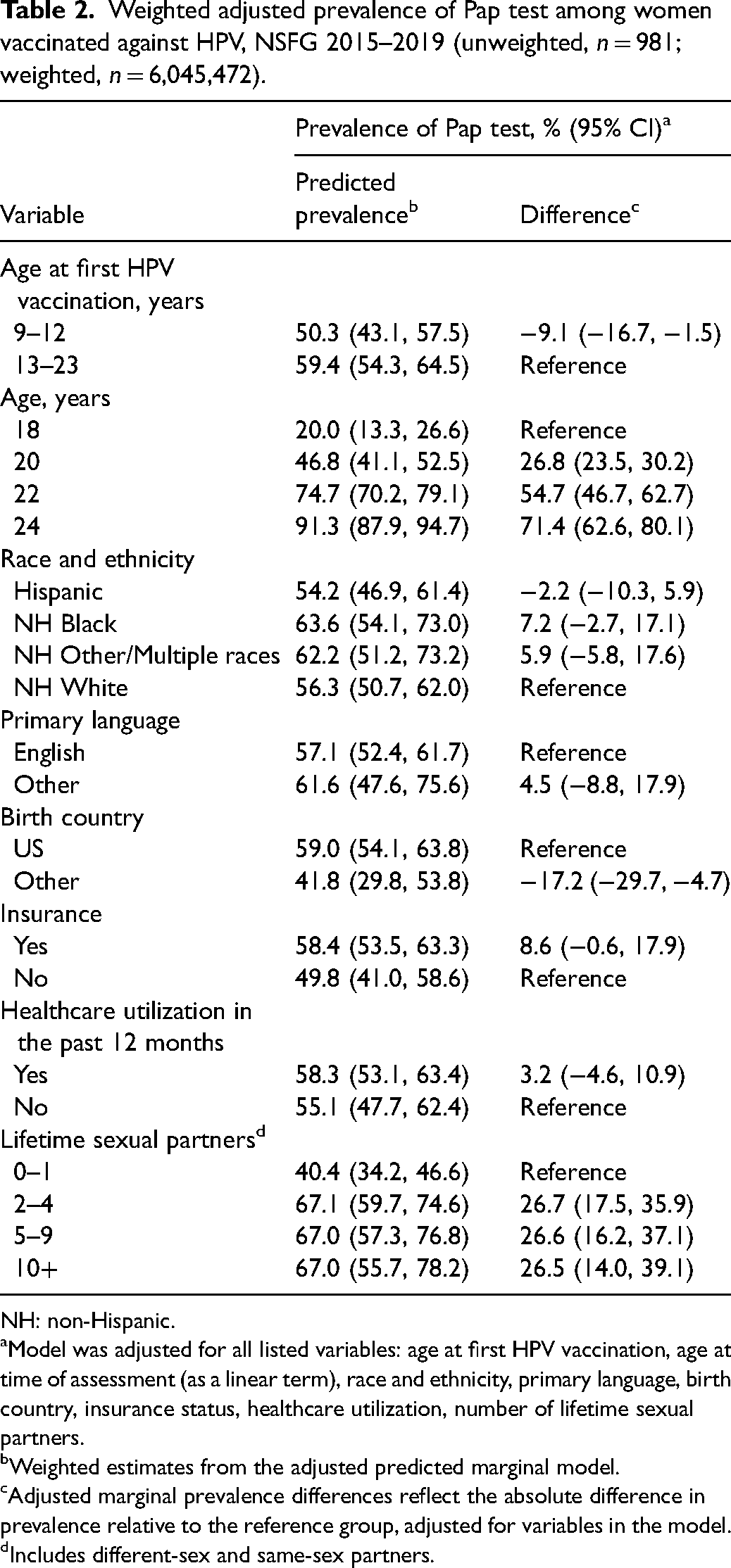
NH: non-Hispanic.
Model was adjusted for all listed variables: age at first HPV vaccination, age at time of assessment (as a linear term), race and ethnicity, primary language, birth country, insurance status, healthcare utilization, number of lifetime sexual partners.
Weighted estimates from the adjusted predicted marginal model.
Adjusted marginal prevalence differences reflect the absolute difference in prevalence relative to the reference group, adjusted for variables in the model.
Includes different-sex and same-sex partners.
In a sensitivity analysis excluding women who were over-screened and those aged <22 at the time of assessment, the prevalence of having received Pap test was 71.8% (not shown), and the adjusted difference in screening probability between those vaccinated at the recommended age in comparison to those vaccinated later in life was 20.1% (95% CI was not possible to obtain) (Supplementary Table 1). This translates into 62,415 fewer screening-eligible women nationwide getting screened for cervical cancer than could be if those vaccinated at the age of 9–12 had the same screening probability as those vaccinated at 13–23 years.
In the analysis, there were 17 women (weighted, 95,982) aged 22–24 at the time of assessment who were vaccinated against HPV at 9–12 years and had not had a Pap test, i.e., women who followed the vaccination initiation but not the screening guidelines. Some differences were present in sociodemographic and behavioral characteristics in this subgroup in comparison to the general study population (Supplementary Table 2). In particular, they were more likely to be Non-Hispanic White women (75.5 vs. 54.7%), born in the US (98.4 vs. 91.7%), to have English as their primary language (98.4 vs. 92.9%), to not have insurance (29.2 vs. 11.3%), to not have used healthcare in the past 12 months (33.4 vs. 26.1%), and to have had over 10 lifetime sexual partners (26.0 vs. 16.2%).
In the model with age at the first HPV vaccine administration as a continuous variable, the adjusted prevalence of Pap test increased linearly with age. A 1-year increase in age at first vaccination was associated with a 1.1% (95% CI, −0.1 to 2.4%) higher probability of having had a Pap test, but this linear trend was not statistically significant (Figure 1). No evidence of a non-linear relationship between age at HPV vaccination and participation in cervical cancer screening was found.
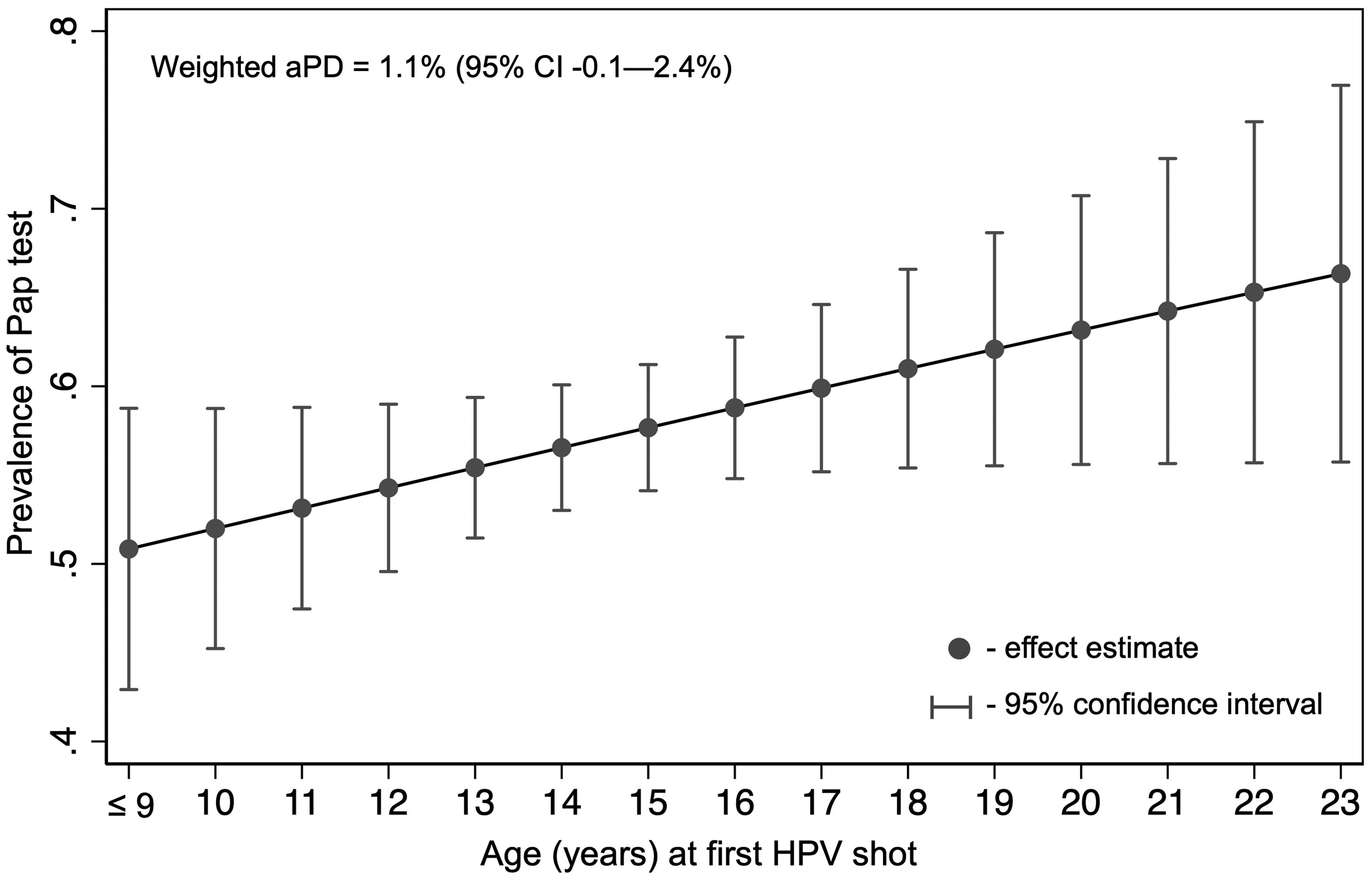
Association between age at first HPV vaccination and prevalence of Pap test, NSFG 2015–2019 (unweighted, n = 981; weighted, n = 6,045,472). Based on the linear model adjusted for age at time of assessment (as a linear term), race and ethnicity, primary language, birth country, insurance, healthcare utilization, and number of lifetime sexual partners. aPD: adjusted prevalence difference.
Among 1921 women in the study who were eligible for HPV vaccination (weighted, 11.8 million), 53.9% had received at least one HPV vaccination. Those who had received at least one HPV vaccination were 5.3% (95% CI, 0.1–10.5%) more likely to have had a Pap test than those who had not initiated HPV vaccination (P = 0.05) (Supplementary Table 3).
Discussion
In this cross-sectional analysis of a nationally representative sample of women in the US aged 18–24 and vaccinated against HPV, we found that although HPV vaccination was generally associated with a higher probability of having received cervical cancer screening, initiating vaccination at the CDC-recommended ages of 9–12 was associated with a statistically significant lower probability of cervical cancer screening than initiating HPV vaccination at 13–23 years. When accounting for confounding sociodemographic and behavioral risk factors, we found that if screening-eligible women vaccinated against HPV at the recommended age (i.e., 9–12) had the same probability of having had a Pap test as those vaccinated above that age, this would add 62,415 women nationwide getting proper cervical cancer prevention. In light of this evidence, there is a high potential for clinical interventions, such as sending reminders to providers and patients who were vaccinated against HPV early in life to return for cervical cancer screening as they become age-eligible (age 21 or 25, depending on the source of recommendations3,5,6).
In agreement with previous studies conducted in the US, Europe, and Israel, our analysis (Supplementary Table 3) has demonstrated that women vaccinated against HPV are more likely to get screened for cervical cancer.7,10–13,19–25 Some researchers attribute this association to the increased knowledge of cervical cancer screening practices among vaccinated women that they may have received from their healthcare provider at the time of vaccination.7,11,19,20 Another possible explanation is related to the idea that individuals practicing healthy behaviors are more likely to engage in other healthy behaviors. 9 Overall, this offers strong reassurance that vaccinated women are better protected against cervical cancer compared to those who are unvaccinated. However, women vaccinated against HPV are not a homogeneous group of people, and the context of HPV vaccination, such as age or year of vaccination or the number of received doses of the vaccine, could play an important role in cervical cancer screening participation.11,12,20,25
Only a few studies to date have examined the association between age at HPV vaccination and cervical cancer screening participation.9–13 With the exception of one Danish study that showed no evidence of association, 13 most found that younger age at HPV vaccination is associated with lower odds of cervical cancer screening participation. In particular, in a study utilizing the electronic health records of a safety net healthcare system in Missouri, US by Paynter et al., women vaccinated closer to age 21 were more likely to participate in cervical cancer screening than women vaccinated at age 14 (adjusted OR = 1.71, 95% CI 1.45–2.00). 12 Similarly, using insurance claims data, Hirth et al. found that women aged 22–26 at first HPV vaccination were more likely to get screened than women aged 19–21 (adjusted OR = 1.97, 95% CI 1.84–2.12). 9 In a study of catch-up HPV vaccination in Israel by Chodick et al., the rate of screening at 10 years of follow-up was higher among women vaccinated after the age of 25 than before that age (about 70 vs. 30%). 10 The findings from our study are in line with the previous research demonstrating a significantly higher probability of screening in women who initiated HPV vaccination later in life, at the age of 13–23, in comparison to the recommended vaccination age of 9–12 (aPD = 9.1%, 95% CI 1.5–16.7%).
A potential explanation for the pattern observed in the study, such that women vaccinated against HPV at ≥13 years have a higher probability of having had a Pap test, could be opportunistic screening. Health providers may recommend their patients get screened for cervical cancer as secondary prevention when they come for a catch-up HPV vaccination around the age of 21 and above. This finding can be examined in future studies with more detailed data on the dates of provider visits, HPV vaccination, and Pap tests.
Information on the timing of vaccination and screening is also crucial to verify the causal pathway since the relationship between these two events could be reversed, such that women who come for cervical cancer screening around the age of 21 and above get a provider's recommendation for a catch-up HPV vaccination. Future studies could benefit from exploring this association in more temporal detail to inform more effective clinical interventions.
An important aspect of our study is that comparison groups may differ substantially in terms of the decision-making process to initiate HPV vaccination, which may explain the observed screening pattern. The decision to vaccinate at 9–12 years was most likely made by parents or caregivers rather than individuals themselves as studies show that parental involvement has an impact on the vaccination decision-making process,26,27 and parental involvement likely dissipates with age. Consequently, early vaccination probably does not translate into other health-related behavior, such as cancer screening, unlike vaccination that is done at a later, more independent, age. Some work, including qualitative studies, has been done to elucidate the reasoning behind HPV vaccination decision and the role of HPV vaccination status in screening behavior,28–31 but there is a lack of studies that have explored the timing of HPV vaccination in this context. However, as more individuals get vaccinated against HPV at a young age, it becomes crucial to better understand this population to promote adequate cervical cancer screening more effectively.
Our study has some strengths and limitations. The main strengths of this study that distinguish it from previous research include a nationally representative dataset, the availability of important sociodemographic and behavioral covariates, and data availability until 2019 that offers an opportunity to study women who were both age-eligible for HPV vaccination (including being most age-appropriate, aged 9–12) when it was just introduced and reached the cervical cancer screening age by the time of the survey.
A few limitations of the study should be noted. Given the cross-sectional nature of the data, it is challenging to distinguish what came first, vaccination or screening, specifically for those who reported being vaccinated at the age when screening is also recommended. Another major limitation is that the data are self-reported. However, the potential measurement error due to self-reported HPV vaccination and cervical cancer screening status is likely non-differential as both practices tend to be overreported. Consequently, the observed association may be even stronger in real life than reported in this analysis.
Our study is limited to women aged 18–24 at the time of assessment because women aged above 24 were not asked about HPV vaccination in the survey. Although prior to 2020, the American Cancer Society's guidelines recommended that cervical cancer screening start at age 21, current guidelines increased the starting age to 25. Due to the age limit in our study, it does not provide evidence of an association between the age of HPV vaccination and cervical cancer screening beyond age 24.
Future studies may benefit from including a wider population and leveraging more detailed data on the timing of vaccination and cervical cancer screening. Electronic health records data supplemented by surveys to include behavioral factors would be an appropriate setting for future research in this area.
Cervical cancer prevention remains an important issue in the US, especially considering the recent upward trend in distant-stage cervical cancer incidence and mortality observed in low-income counties. 32 In conclusion, to improve cervical cancer prevention among women in the US, our results suggest that healthcare providers should consider reaching out with reminders to their patients vaccinated against HPV to inform them about the importance of this secondary cervical cancer prevention strategy through screening.
Supplemental Material
sj-docx-1-msc-10.1177_09691413251315879 - Supplemental material for The impact of age at initial HPV vaccination on cervical cancer screening participation in a nationally representative cohort of women in the United States
Supplemental material, sj-docx-1-msc-10.1177_09691413251315879 for The impact of age at initial HPV vaccination on cervical cancer screening participation in a nationally representative cohort of women in the United States by Ekaterina Chirikova, Vanessa Dorismond, Alyssa M. Cortella, Mindy C. DeRouen and George F. Sawaya in Journal of Medical Screening
Footnotes
Author contributions
Ekaterina Chirikova: conceptualization, data curation, formal analysis, methodology, project administration, writing—original draft preparation, writing—review and editing. Vanessa Dorismond: writing—review and editing. Alyssa M Cortella: formal analysis, writing—review and editing. Mindy C DeRouen: supervision, writing—review and editing. George F Sawaya: conceptualization, methodology, supervision, writing—review and editing.
Consent to participate
Not applicable
Consent for publication
Not applicable
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ekaterina Chirikova was supported by T32MD015070 from the National Institute on Minority Health and Health Disparities of the National Institutes of Health.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
