Abstract
Objectives
Georgia has a high prevalence of hepatitis C virus (HCV) infection. In 2015 a national HCV elimination program was launched providing free access to screening and treatment. To achieve elimination, innovative approaches to increase screening coverage and linkage to care are needed. This study estimates feasibility, acceptability, and outcomes of the door-to-door pilot HCV testing program in three cities.
Methods
Households were approached by system random sampling and all members were invited for study participation. Researchers used a detailed guide for conducting door-to-door testing and served as case navigators to link anti-HCV-positive individuals to care.
Results
Testing acceptance rate was high. In total 4804 individuals were tested and 48 (1.0%) were HCV positive. Among the entire sample of newly and previously tested individuals, overall HCV antibody prevalence was 3.6%. Through case navigation, of 48 newly identified and 26 previously identified anti-HCV-positive individuals, 42 (87.5%) and 17 (65.4%), respectively, were successfully linked to care.
Conclusions
Door-to-door HCV testing has potential to increase testing uptake. Such community-based approaches not only improve testing, but can also serve to increase linkage to care, which is important in achieving the goal of HCV elimination. The study provides a model for high prevalence countries aiming to eliminate hepatitis C.
Introduction
The country of Georgia has a high prevalence of hepatitis C virus (HCV) infection (estimated 5.4% RNA-positive adult population in 2015).1,2 A national HCV elimination program was launched in 2015 which provides free access to treatment using direct acting antivirals (DAAs).3–5 Despite wide-scale screening, a large proportion of HCV-infected individuals remain unaware of their HCV status and others are not willing to start treatment. After peaking in September 2016, the number of patients initiating treatment has declined. 6 The WHO recommends a community-based testing approach, including door-to-door outreach models, to expand the coverage and impact of hepatitis testing. 7
To achieve HCV elimination, innovative approaches to increase screening coverage and linkage to care are needed. This study estimates feasibility, acceptability, and outcomes of door-to-door pilot testing program for hepatitis C in Georgia.
Materials and methods
Three cities were selected to pilot the door-to-door HCV testing model: Tbilisi (capital), Zugdidi, and Batumi. Households were selected by systematic random sampling. Study teams consisting of two researchers were trained in completing data collection forms, pre- and post-test counseling, using national HCV program databases, performing HCV antibody rapid diagnostic tests (RDTs), and serving as case navigators to link HCV-positive individuals to care. Three study teams (a total of six researchers) conducted door-to-door visits in the evenings and on weekends. Study participation was voluntary. If a household agreed to participate, each member, regardless of age, was eligible for study enrollment; any household member not at home at the time of visit was marked “absent.” An informed consent form was required to be signed by willing study participants or parents/guardians of individuals <18 years old. A free hemoglobin (Hb) test was offered as an incentive. Pre- and post-test counseling were provided; demographic data and test results were recorded. Anti-HCV test results were entered into the national screening database.
Participants who tested anti-HCV positive during the household visit were provided with information on the importance of care and treatment, and a list of facilities where HCV viremia testing was available for free. Study teams served as case navigators, and two weeks after household visits contacted anti-HCV positive individuals to ensure that their viremia test had been performed, and, if positive, advised on next steps for diagnosis and treatment.
Household members refusing participation were registered and reasons for refusal and demographic data were recorded. If a household member refused due to previously being tested, this was verified in the national HCV screening database using the participant's unique national ID number. If this person was denoted as HCV antibody positive and not linked to care (has not received HCV viremia testing), reasons for not seeking care were recorded.
Collected data were entered in statistical software SPSS (Version 20) and a descriptive analysis was performed. The study protocol was reviewed and approved by the Institutional Review Board of the Health Research Union, Georgia.
Results
During March–October 2018, 4062 households were approached. Out of 2949 families who were at home during visits 23.1% refused participation. The total number of individuals at the selected households was 6925 and of these 20.8% were not present at home during visits (see Figure 1).
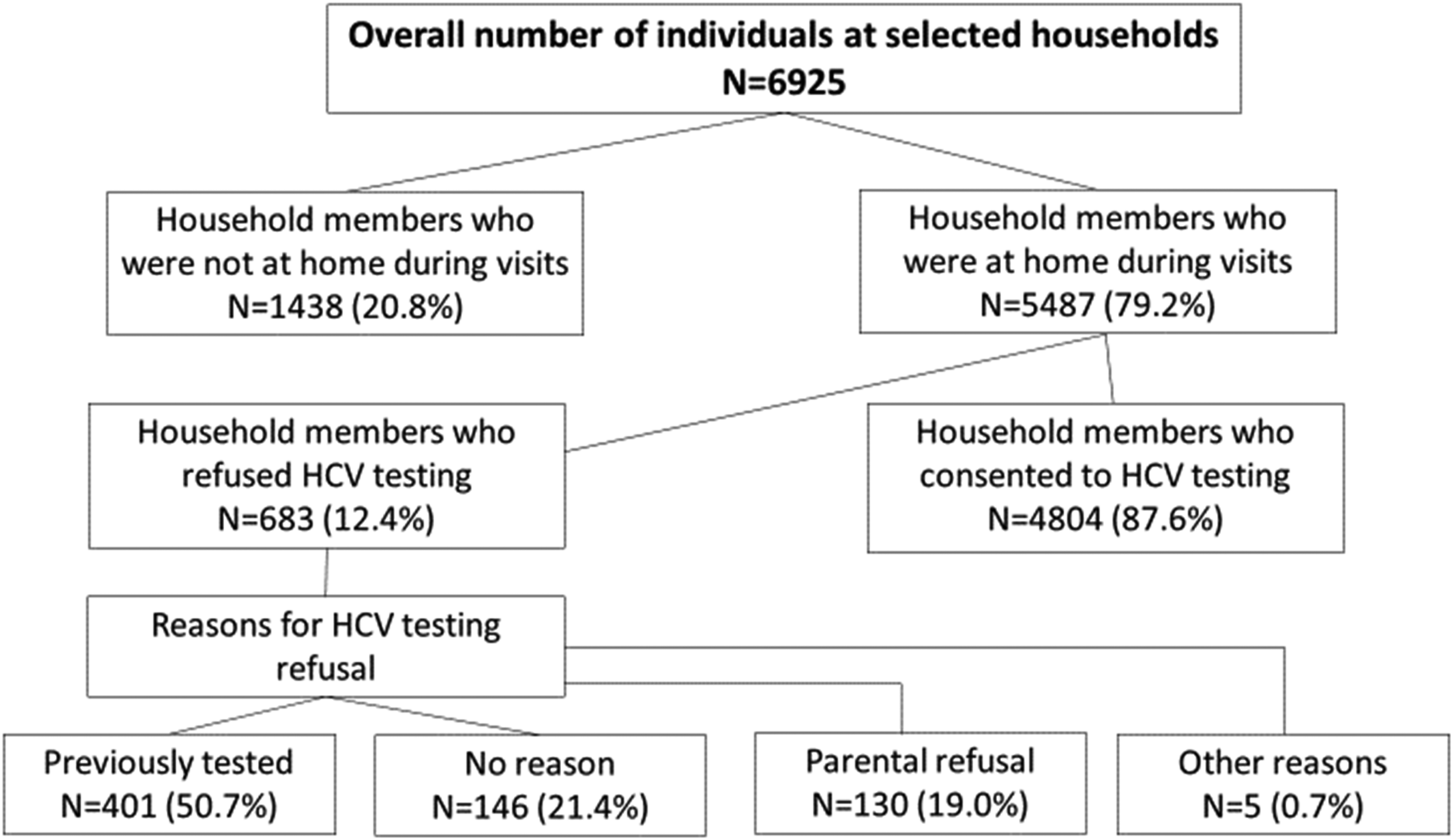
Households reached by door-to-door HCV screening, participation rates and reasons for refusal to get tested for HCV.
Among the 5487 individuals who were approached, 401 were ineligible because they had been previously tested for HCV antibodies and 4804 agreed to participate in the study (refusal rate 5.5%) (see online Appendix for the socio-demographic characteristics of the study participants). Out of the 4804 tested participants, 48 (1.0%) were positive. Among the 401 previously tested individuals, 34.4% were recorded in the national HCV screening database. Among those who tested HCV positive, 81.4% had already been linked to care. Among the entire sample of newly and previously tested individuals, overall HCV antibody prevalence was 3.6% with the highest rate in Zugdidi (6.0%), followed by Batumi (3.9%) and Tbilisi (2.0%).
Among previously tested HCV antibody-positive individuals, 26 (6.4%) were not tested for viremia. More than half of HCV-positive persons were informed about further diagnostic investigations, but they did not get the viremia test due to financial barriers (n = 15) or thinking there was no need of treatment because of absence of symptoms (n = 11).
Through case navigation, out of 48 newly identified and 26 previously identified HCV antibody-positive individuals, 42 (87.5%) and 17 (65.4%) respectively were successfully linked to care within two weeks of the home visits by our study teams. The main reason newly and previously identified anti-HCV-positive individuals turned down being linked to care was the fear of treatment side effects.
Discussion
Although door-to-door HIV screening is utilized in some countries, it is a novel approach for hepatitis C. A study conducted in high HIV prevalence area in Africa revealed that door-to-door HIV testing affords higher rates of testing for underserved communities by overcoming barriers to care. 8 Our study shows the effectiveness of this approach in Georgia, to identify HCV-infected individuals and link them to care.
Testing acceptance rate was high; nearly 90% of those who were at home during visits and 70% of overall individuals living in visited households were tested. Among those who refused to get tested, more than half had been tested previously. Some of those who were not already linked to care named financial barriers to viremia testing. At the start of the program patients were required to pay for diagnostic tests. Recognizing testing costs as a barrier to HCV elimination, the government of Georgia reduced the number of tests required for each patient and, from March 2018, provided HCV RNA or HCVcAg testing free-of-charge. 5 Currently all diagnostic tests related to HCV treatment and monitoring are free in Georgia, but people are unaware of that. The door-to-door testing approach will increase awareness which in turn will improve linkage to care of HCV-positive individuals.
A study limitation was that households were approached only once. Although evening hours and weekends were used for visits, 27% of households were not available. Total household refusal rate was 23%. Overall, door-to-door HCV testing seemed acceptable in our pilot study and has the potential to increase testing uptake.
All individuals who tested positive previously or during the study team visit (n = 74) received counseling, and 80% of them were enrolled in the HCV elimination program within two weeks of the home visit. Such community-based approaches not only improve testing, but can also serve to increase linkage to care,9,10 which is important to achieve the goal of HCV elimination. This study can serve as a model and may be of utility in high prevalence countries aiming to eliminate hepatitis C.
Supplemental Material
sj-docx-1-msc-10.1177_09691413221086497 - Supplemental material for Door-to-door hepatitis C screening in Georgia: An innovative model to increase testing and linkage to care
Supplemental material, sj-docx-1-msc-10.1177_09691413221086497 for Door-to-door hepatitis C screening in Georgia: An innovative model to increase testing and linkage to care by Maia Butsashvili, Tamar Zurashvili, George Kamkamidze, Maia Kajaia, Lasha Gulbiani, Ana Gamezardashvili, Lia Gvinjilia, Tinatin Kuchuloria, Amiran Gamkrelidze, Shaun Shadaker, Muazzam Nasrullah, Francisco Averhoff and Paige A. Armstrong in Journal of Medical Screening
Footnotes
Acknowledgements
This work is supported by Liver Institute and Foundation for Education and Research (LIFER) grant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online. <Pre-editor: Please check the supplementary files on the server. This standard statement is not required if the supplementary file is a reviewers response or letter etc.>
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
