Abstract
Objectives
To evaluate the effect of general practitioner endorsement accompanying the screening kit rather than with the invitation letter on participation in the NHS Bowel Cancer Screening Programme and on the socioeconomic gradient in participation in the Programme.
Methods
The NHS Bowel Cancer Screening Programme in England is delivered via five regional hubs. In early 2016, we carried out a cluster-randomised trial, with hub-day of invitation as the randomisation unit. We randomised 150 hub-days of invitation to the intervention group, GP endorsement on the letter accompanying the guaiac faecal occult blood testing kit (75 hub-days, 197,366 individuals) or control, usual letter (75 hub-days, 197,476 individuals). The endpoint was participation, defined as return of a valid kit within 18 weeks of initial invitation. Because of the cluster randomisation, data were analysed by a hierarchical logistic regression, allowing a random effect for date of invitation. Socioeconomic status was represented by the index of multiple deprivation.
Results
Participation was 59.4% in the intervention group and 58.7% in the control group, a significant difference (p = 0.04). There was no heterogeneity of the effect of intervention by index of multiple deprivation. We found that there was some confounding between date and screening episode order (first or subsequent screen). This in turn may have induced confounding with age and slightly diluted the result.
Conclusions
General practitioner endorsement induces a modest increase in participation in bowel cancer screening, but does not affect the socioeconomic gradient. When considering cluster randomisation as a research method, careful scrutiny of potential confounding is indicated in advance if possible and in analysis otherwise.
Introduction
In England, the NHS Bowel Cancer Screening Programme (BCSP) has been offering faecal occult blood testing (FOBt) since 2006. 1 The programme has the lowest participation of the three cancer screening programmes, at around 60%, compared to participation rates of around 70% in screening programmes for breast cancer and 80% for those for cervical cancer.2,3 Participation is especially low in deprived populations, decreasing to 35% in the most deprived quintile of the population. 2
Several interventions have been observed to improve screening participation, including general practitioner (GP) endorsement, varying reminder strategies and advance notification, although there is little published work showing a successful reduction in socioeconomic gradients in screening participation.
4
In a series of randomised trials, the ASCEND programme investigated four interventions aimed at reducing this socioeconomic gradient in bowel cancer screening uptake (randomisations occurred between 2012 and 2013):
a supplementary ‘Gist’ leaflet very briefly summarising the key information about FOBt screening; a supplementary narrative leaflet incorporating individual patients’ stories; an enhanced reminder letter for those not returning a completed FOBt kit within four weeks; GP endorsement on the initial invitation letter (which does not include the FOBt kit).
The investigators found that an enhanced reminder to those who had not yet returned a kit after four weeks increased overall participation by 0.7 percentage points (from 25.1% using the standard letter to 25.8%) and reduced the socioeconomic gradient, and that GP endorsement increased overall participation by 0.7% percentage points (from 57.5 to 58.2%), without significantly altering the socioeconomic gradient. 5
In view of the results of ASCEND, an additional trial was carried out – ASCEND2, to evaluate the effect of GP endorsement on the letter which accompanies the FOBt kit, this letter and kit being subsequent to the original invitation. At the time of the trial, the English BCSP used a guaiac faecal occult blood test which was replaced with an immunochemical faecal occult blood test in 2019.
The aim was to increase screening uptake and reduce differences in uptake by socioeconomic status. This paper reports on the results of ASCEND2. We also flag up a warning on the design of large-scale cluster randomised trials.
Methods
The NHS BCSP in England is run by five regional hubs. Each hub is responsible for invitation, testing and recall for further investigation following a positive test. The basic timeline for a screening episode is as follows:
Week 0: first, an invitation letter is sent making the offer of screening, giving notification that an FOBt kit will arrive in around a week’s time and enclosing information on the pros and cons of FOBt screening. Week 1: the FOBt kit is sent, with instructions for its use. Week 5: if the kit has not been returned within four weeks of its issue, a reminder letter is sent. Receipt of completed kit or week 17: screening episode closed.
As part of the original ASCEND trials, 5 a Primary Care Advisory Group developed text that could be easily incorporated into the standard BCSP invitation as there is space on the letters to mention the support of the individual’s GP practice. The group created a GP endorsement that was programmed to appear as a banner across the initial invitation letter, linking each individual’s letter to their registered GP practice.
The study design of ASCEND2 was identical to that of the previous ASCEND GP endorsement trial except that the intervention banner of the GP practice was now included on the letter with the screening kit rather than as part of the initial invitation letter, and ASCEND2 contained a larger number of subjects. An example letter is included as Supplemental Material (online only). The cost of including the banner on the letter with the kit was the same as including it on the invitation letter, so no additional costs were incurred relative to that of the GP endorsement arm of the ASCEND trial.
We randomised by clusters by day within hubs (hub-days) rather than for individuals, as the hubs anticipated administrative difficulties in individual randomisation. Although blinding of hubs was not possible, there was no direct contact with participants.
The primary aim of the study was to test the hypothesis that participation, in terms of percentage of invitees adequately screened, was the same in the GP endorsement arm as in the standard invitation arm, against the alternative that participation increases in the GP endorsement arm.
Study population
Consent had been sought from all general practices in England (N = 8142) in collaboration with a Primary Care Advisory Group and the Health & Social Care Information Centre (HSCIC, now NHS Digital), by sending invitations to be part of the ASCEND trials, followed by reminders at four and eight weeks. Permission to link the general practice address to the invitation was granted by 80% of practices (N = 6480). Eighty-one practices from three clinical commissioning groups in the Leeds area that had previously participated in ASCEND were excluded from the ASCEND2 trial to avoid conflicting with another bowel screening uptake trial.
Men and women aged 60–74 years from the general practices included who are routinely invited for FOBt screening in the BCSP were randomised in equal proportions (clustered by date of invitation, stratified by BCSP hub) to receive a GP endorsement letter with their FOBt kit on randomly selected days within a pre-specified time-period or to the control group who received the kit with the standard letter without the GP endorsement banner. Randomisation was carried out via the Bowel Cancer Screening System (BCSS) on which the necessary modifications had to be programmed to enable the system to generate the intervention letters for patients at participating practices. Within each hub, we randomised the working days between 1 February 2016 and 11 March 2016.
A consent form was not applicable in this study as the intervention took place as part of the subject’s usual communication from the BCSP. As there was no direct contact, individuals would have been unaware of participation unless they had been invited on a previous occasion and recalled the content of the invitation or if a household. member had received an invitation during the study period that contained different information. The activities of the BCSP are covered under an existing Confidentiality Advisory Group Section 251 approval with regard to the handling of patient-identifiable data (Ref: PIAG 1–08(a)/2003). Ethical approval for ASCEND2 was obtained from the East Midlands – Leicester Central Research Ethics Committee (15/EM/0561). The ASCEND2 trial is registered with the ISRCTN (http://www.isrctn.com/ISRCTN11660314).
Outcomes
We used the English Index of Multiple Deprivation (IMD) score to measure socioeconomic status because of its established representation of deprivation by income, education, housing, health, environment, employment and crime 6 and its demonstrated ability to explain area-level variation in bowel cancer screening uptake. 2 IMD is freely available and widely accepted and used, enabling direct comparison of our results with other studies. The postcode variable was substituted with an IMD score by HSCIC. IMD was applied using the geographic unit of Lower Super Output Area (LSOA) level. There are 32,844 LSOAs in England, each covering about 1500 individuals. Each individual’s postcode was linked to the relevant LSOA with the corresponding IMD scores grouped into quintiles of their national distributions.
The primary outcome was the proportion of people in each IMD quintile returning an adequate FOBt within 18 weeks of being sent their initial invitation letter; this time period coincides with when the BCSS closes an episode due to non-response. An adequate FOBt in this study was defined as reaching a definitive FOBt outcome of either a ‘Normal’ (no further clinical investigation required) or ‘Abnormal’ (referral for colonoscopy).
A pseudonymised dataset from the BCSS was provided by HSCIC. Age, sex and the type of screening episode (whether individuals were being invited for the first time, being sent a biennial invitation having previously not responded or being sent a biennial invitation having been screened before) were obtained from the BCSS database.
Sample size
To calculate the required study size, we needed to take into account: (1) the size of the effect/interaction we wish to detect; (2) the anticipated distribution of socioeconomic status and the expected participation by socioeconomic status in the control arm and by screening round; (3) the proportion of general practices agreeing to be included in the trial; (4) a variance inflation factor to account for the cluster (hub-day) randomisation; and (5) the size of the effect/interaction by screening round.
We assumed an interaction such that in the least deprived quintile, there was a 1% increase in participation with the intervention, in the second least deprived a 1.5% increase, and so on, with a 3% increase in the most deprived quintile. This would give an average 2% increase with the intervention, lower than the 3% observed in ASCEND. 5 The distribution of IMD quintiles from the GP endorsement on the initial invitation letter in ASCEND 5 by completion rates in the control group is shown in Table 1. We anticipated these rates in the control group of ASCEND2 and the intervention participation rates in the final column of Table 1.
Observed socioeconomic status distribution and control group completion rates from ASCEND GP endorsement trial (n = 133,449) 5 and estimated completion rate in the intervention group of ASCEND2.
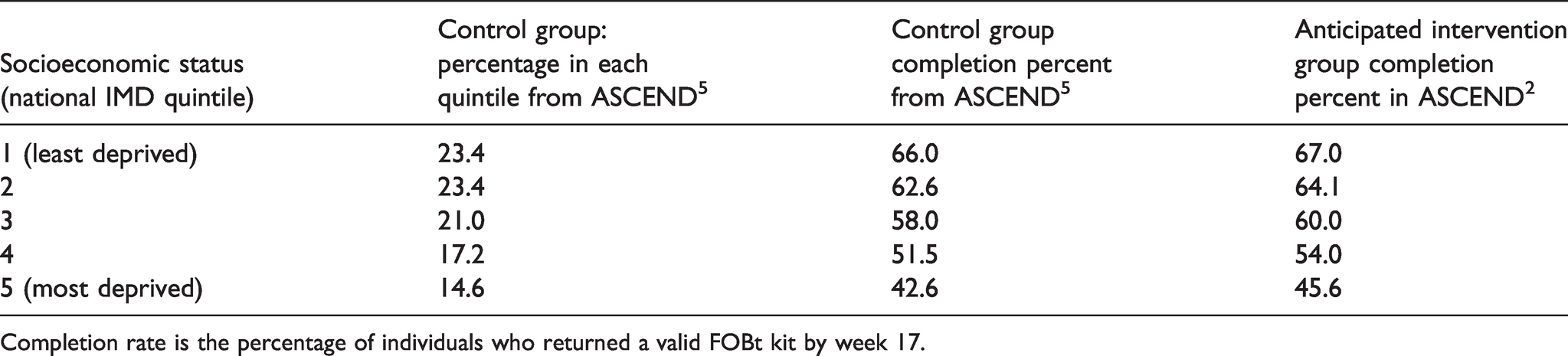
Completion rate is the percentage of individuals who returned a valid FOBt kit by week 17.
We calculated the required sample size using the method of Brentnall et al. 7 To obtain 90% power to detect the interaction as significant at 5% level with two-sided testing, in an individually randomised study, based on the control group IMD distribution and participation rates in Table 1, we would require 11,714 individuals in each arm. In the original ASCEND trial, 80% of the GPs approached agreed to participate in the GP endorsement trial. We therefore inflated the required size for the current trial to 14,643 (=11,714/0.8) subjects per trial arm. We also estimated a variance inflation factor for cluster randomisation of 1.7, but results post hoc suggest that a larger inflation would be more appropriate. We therefore used an inflation factor of 2.5 for the trial reported on here, giving a total study size of 36,608 per arm and a total number of 73,216 individuals randomised over all five hubs. Assuming approximately 3000 kits were sent per hub-day, this indicates that we should randomise 25 hub-days in total. To cope with any plausible failures of our assumptions, and to allow for subgroup analyses, we randomised 30 days in each hub, completing trial accrual from the five hubs in six working weeks with a target population of 450,000 subjects if our assumption about numbers invited per hub-day were correct.
Statistical analysis
Data were first analysed by a fixed effects logistic regression and a simple chi-squared test to compare participation rates between the two trial arms. Thereafter, a formal analysis, taking account of the cluster randomisation, was carried out using a hierarchical logistic regression model, with a random effect for date, and fixed effects for hub, trial arm IMD quintile, gender and screening round, with estimation by Markov Chain Monte Carlo. 8 The endpoint was participation, defined as returning a valid completed kit by week 17, that is, 18 weeks after the initiation invitation letter, as noted above. We used the same analysis to examine the secondary endpoints: time taken to return the kit, rate of spoilt kits, and rate of kits not delivered.
Results
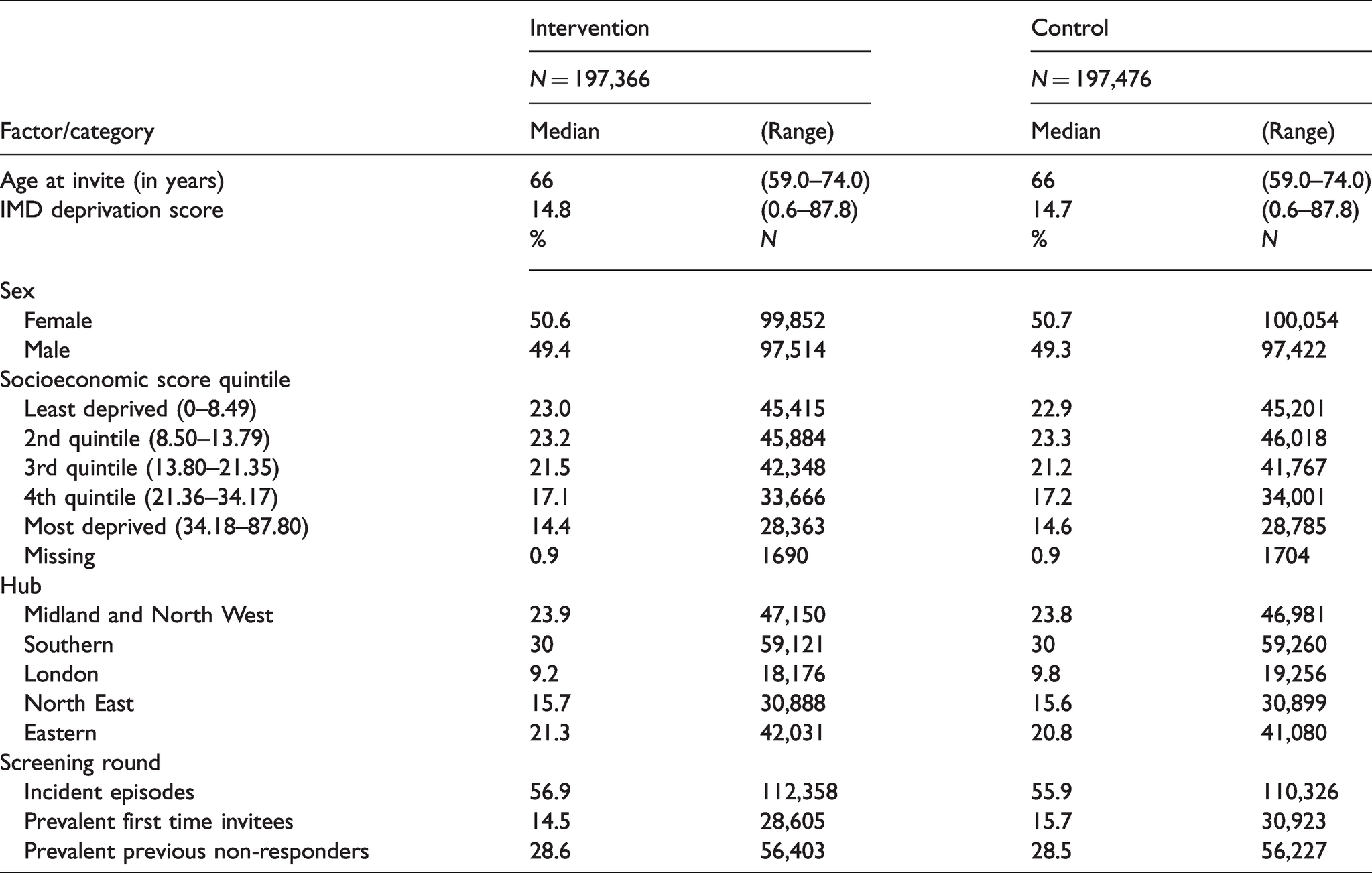
Figure 1 shows the flow of participants through the trial. After exclusion of subjects for which GP consent to endorsement was not in place by the appropriate date, there were 395,014 subjects randomised, and after exclusion of the 0.04% of subjects erroneously included from non-participating practices, there were 394,842 subjects, 197,366 randomised to the intervention and 197,476 to the control arm. These represented 150 randomisation units (hub-days), 75 intervention and 75 control. Table 2 shows the basic characteristics of the study population in the intervention and control arms; these were evenly balanced between the groups except for a slightly lower percentage of first-time invitees in the intervention arm (14.5% versus 15.7% in the control arm).
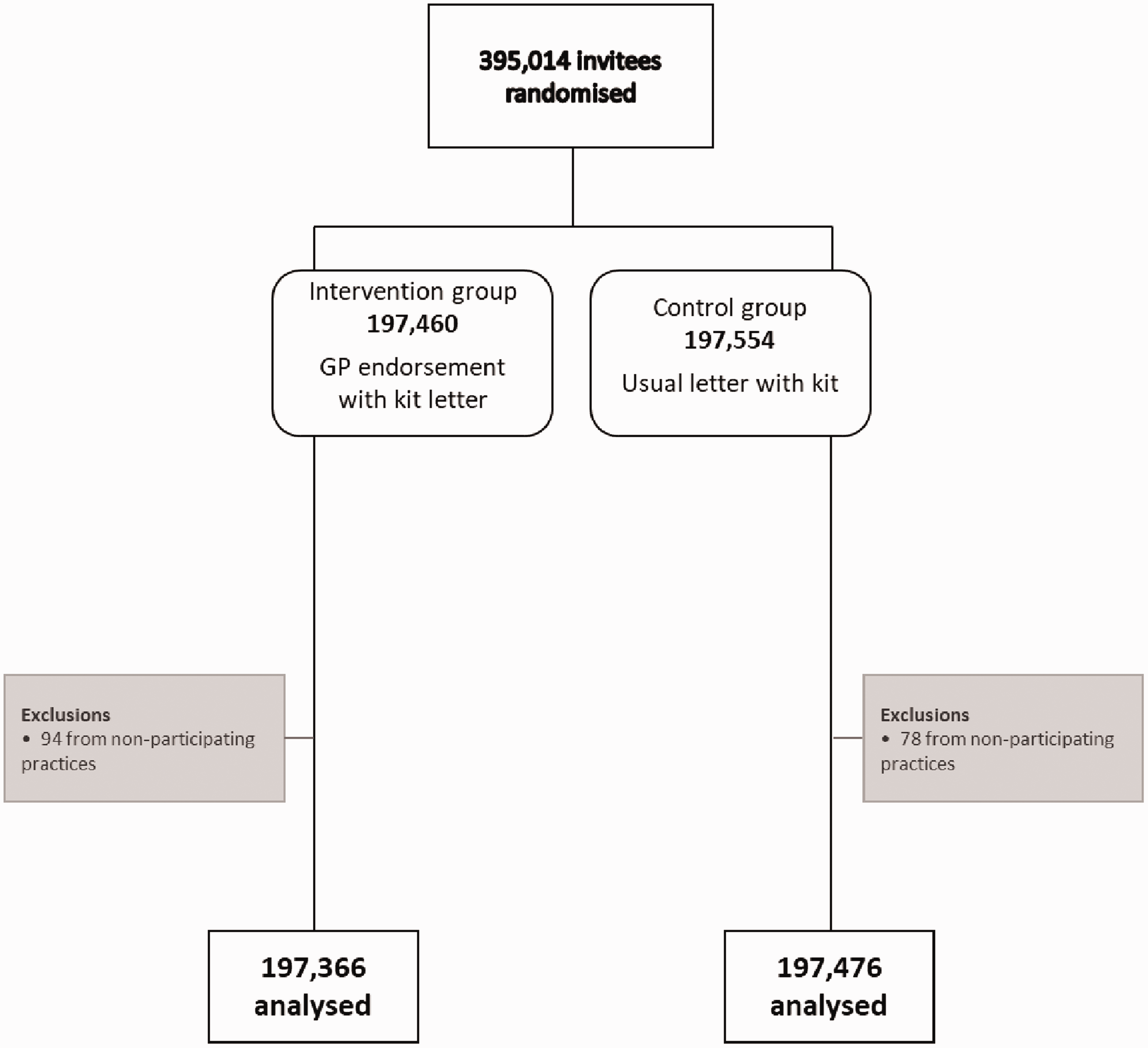
The flow of participants through the trial.
Basic description of the study population.
The effect of intervention on being adequately screened within 18 weeks of initial invitation is shown in Table 3. Participation was slightly higher in the intervention arm at 59.4% compared to 58.7% in the control arm (0.7 percentage point increase). With a conventional analysis at individual level, this would be highly significant (OR = 1.03, 95% CI 1.01–1.05, p < 0.0001). However, when we took account of the cluster randomisation in a hierarchical logistic regression model, the statistical significance was considerably attenuated, at p = 0.04, and with an odds ratio of 1.02 (95% CI 1.00–1.04).
Effect of the intervention on screening participation, as measured by percentage of invitees adequately screened.
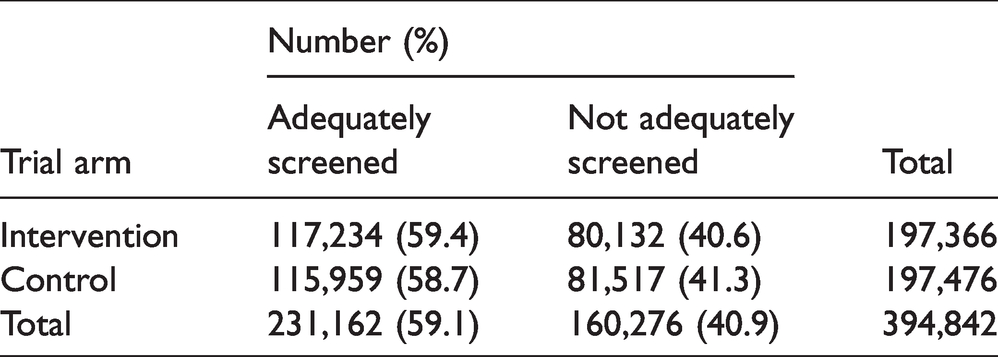
Table 4 shows the results by IMD quintile of invitee and by hub. There was no significant relationship with the size of the effect of intervention by IMD quintile, nor did the effect vary significantly by age or sex (data not shown). However, there was a noticeable difference between hubs with respect to effect size. In the Midlands and North Western Hub, there was an effect in the opposite direction to the other hubs, with individuals in the control group more likely to participate than those in the intervention group. In the hierarchical model, there was a significant heterogeneity of the effect of the intervention by hub (p < 0.001), which lost its significance when adjusted for screening round (p = 0.2).
Effect on participation stratified by IMD and by hub, as measured by percentage of invitees adequately screened.
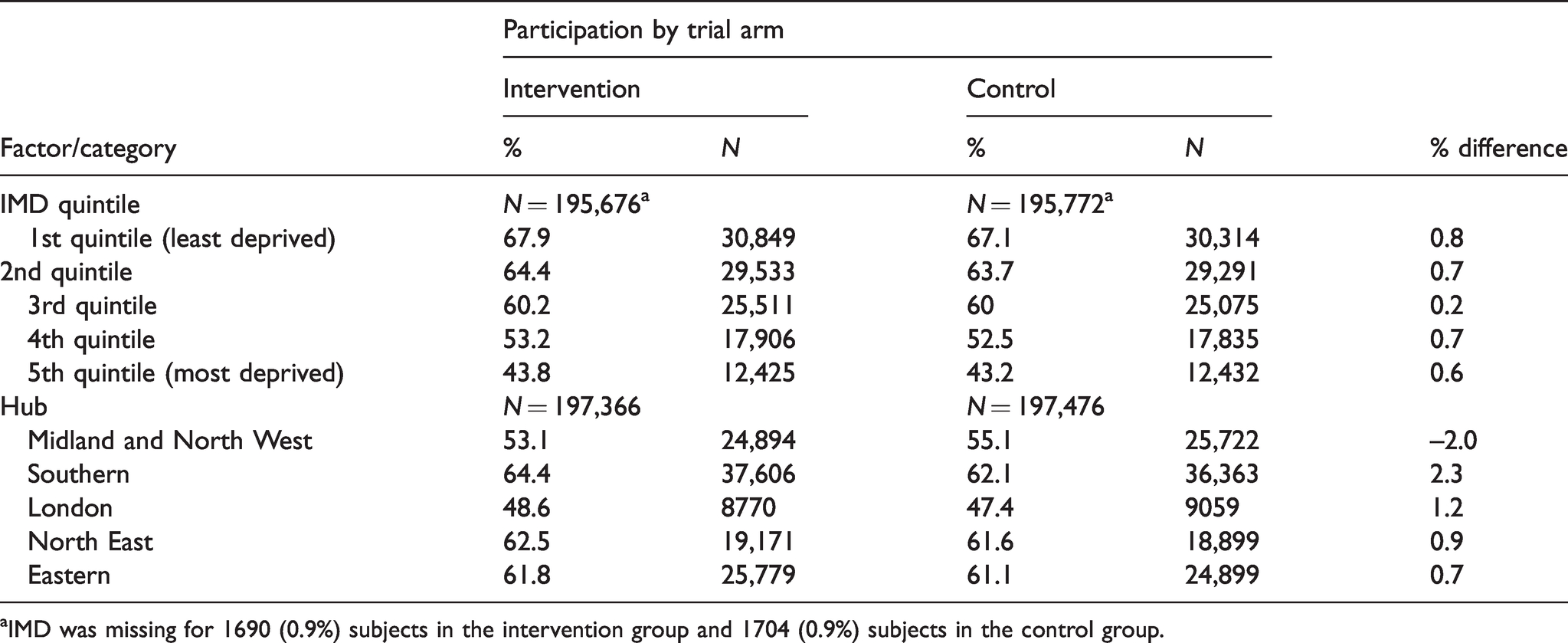
aIMD was missing for 1690 (0.9%) subjects in the intervention group and 1704 (0.9%) subjects in the control group.
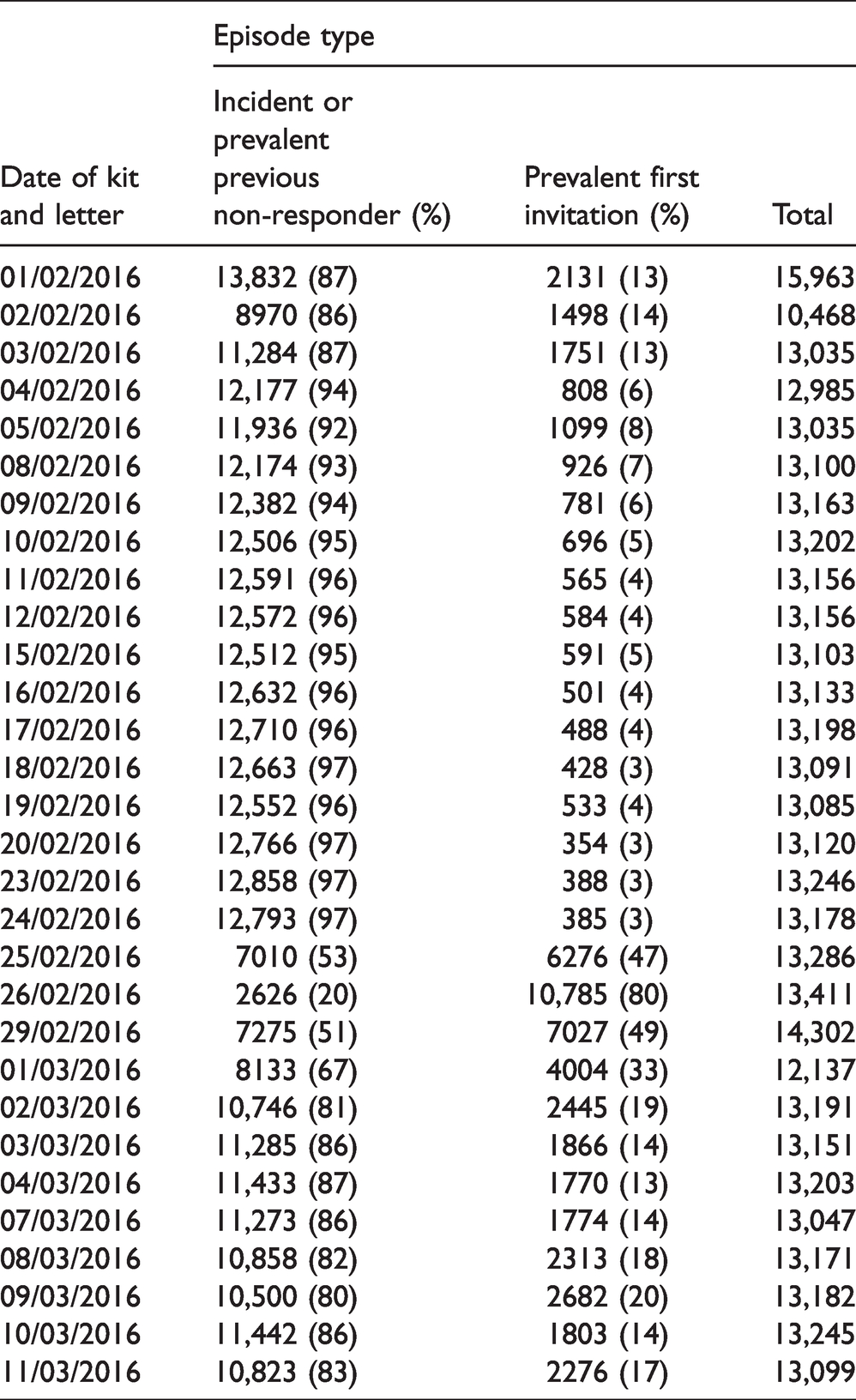
There was a systematic difference between dates with respect to screen type (first/subsequent) and therefore also with respect to age. Table 5 shows the screen type by date. It can be seen that generally less than 20% of episodes were first-time invitations. However, for 25 February, 47% were first-time invitations, for the 26 February, 80% were first time, for 29 February, 49%, and for 1 March, 67%. The same pattern was seen for all five hubs, and this had implications for the age of the invitees: first-time invitees were predominantly aged 59–64 years around the end of February and the beginning of March. Since first screens are associated with lower participation, the randomisation unit may have had some prior confounding with the outcome.
Episode type by date of kit and letter.
The pattern of median time in days to return of kit against IMD quintile was exactly the same in each treatment arm – 14 days in the four lowest quintiles of deprivation and 13 in the highest. The rate of spoilt kits rose from 0.6% in the least deprived quintile to 1.3% in the most, and the rate of non-delivered kits rose from 32.2 to 55.8%. Bayesian multilevel modelling similar to that carried out for participation showed that intervention had no significant effect on either rate nor did the intervention effect vary significantly by IMD quintile.
Discussion
In this cluster-randomised trial with almost 400,000 subjects, we found at best a modest effect on participation of GP endorsement with the FOBt kit, with 59.4% participation in the intervention group and 58.7% in the control group. This was consistent with the result of the first ASCEND trial, of GP endorsement on the initial invitation letter, which showed a participation proportion of 58.2% compared to 57.5% in the control group. 9 We had hypothesised that the increased uptake due to the intervention might have a different effect by socioeconomic status. However, there was no significant heterogeneity of the effect by IMD quintile.
We observed a systematic difference between days with respect to the invited population. Around the end of February and the beginning of March 2016, a higher proportion of younger and first-time potential screenees were invited. Given the lower participation at first round observed both here and elsewhere, 10 this constitutes a confounding risk.
While the cluster-randomisation option is attractive in very large-scale population trials in prevention and screening, the results here give a salutary warning that it is not without hidden costs. The use of a mixed-model analysis allows for additional variation to pure sampling error among clusters, but does not adequately adjust for the systematic difference observed here. Before choosing such a design, careful advance checking of assumptions with respect to both random and systematic differences among clusters is essential. Heterogeneities among randomisation units may inform further stratification, exclusion policy or indeed a decision not to use the cluster randomisation design or to define randomisation units differently.
We observed a difference between hubs with respect to effect size. In the Midlands and North Western Hub, there was an effect in the opposite direction to overall, with the control group having higher participation than the intervention group. This is likely due to the fact that in the Midlands and North Western Hub, the three days dominated by the first invitation round and the 59–64-year age group were all randomised to the intervention arm, conferring a lower participation in the intervention group. The result from all four other hubs (all of whom happened to have the crucial dates shared between intervention and control) combined gave a 1.6% increase in participation with intervention (61.4% versus 59.8%). This suggests that the true effect may be slightly larger than the primary result reported here, but even taking into account the potential confounding issue of date of invitation, the effect of the intervention on participation remained modest, and rather smaller than the effect of primary care endorsement seen in other contexts.4,5,11 As noted above, however, it was consistent with the results of the original ASCEND trial of GP endorsement. 9
Thus, in conclusion, two observations can be made. The first is that the effect of GP endorsement on the letter accompanying the bowel screening kit is modest (although from above, it may be slightly greater than observed overall in this study) and shows no sign of variation with socioeconomic status. Neither does placing the GP endorsement in a different letter closer to when people are asked to actually participate make a substantial difference. Second, cluster randomisation should only be used after careful checking of assumptions with respect to differences among clusters.
Finally, notwithstanding the small effects observed in GP endorsement trials, the BCSP now routinely places the GP banner on the invitation and test kit letters. This banner now accompanies a more user-friendly test kit which only requires a single sample and is associated with an improvement in uptake. 12 It is possible that small modifications like a GP endorsement will have a stronger impact after having removed an important practical obstacle (the need to take samples from three consecutive bowel motions).
Supplemental Material
sj-pdf-1-msc-10.1177_0969141321997480 - Supplemental material for Including a general practice endorsement letter with the testing kit in the Bowel Cancer Screening Programme: Results of a cluster randomised trial
Supplemental material, sj-pdf-1-msc-10.1177_0969141321997480 for Including a general practice endorsement letter with the testing kit in the Bowel Cancer Screening Programme: Results of a cluster randomised trial by Amanda J Cross, Jonathan Myles, Paul Greliak, Allan Hackshaw, Stephen Halloran, Sally C Benton, Caroline Addison, Caroline Chapman, Natasha Djedovic, Stephen Smith, Christian von Wagner, Stephen W Duffy and Rosalind Raine in Journal of Medical Screening
Footnotes
Acknowledgements
ASCEND2 was originally conceived by our dear colleagues Professor Wendy Atkin, who died in 2018, and Professor Jane Wardle, who died in 2015. The study was led by Professor Atkin until her death. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by Cancer Research UK awards (references C8171/A16894; C53889/A25004; and C1418/A14134). Professor Stephen Duffy and Dr Jonathan Myles participated in this study funded by the National Institute for Health Research (NIHR) Policy Research Programme, conducted through the Policy Research Unit in Cancer Awareness, Screening and Early Diagnosis, 106/0001. Professor Rosalind Raine is partly funded by NIHR ARC North Thames.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
