Abstract
Objective
To assess the potential impact of a melanoma screening programme, compared with usual care, on direct costs and life expectancy in the era of targeted drugs and cancer immunotherapy.
Methods
Using a Whole Disease Model approach, a Markov simulation model with a time horizon of 25 years was devised to analyse the cost-effectiveness of a one-time, general practitioner-based melanoma screening strategy in the population aged over 20, compared with no screening. The study considered the most up-to-date drug therapy and was conducted from the perspective of the Veneto regional healthcare system within the Italian National Health Service. Only direct costs were considered. Sensitivity analyses, both one-way and probabilistic, were performed to identify the parameters with the greatest impact on cost-effectiveness, and to assess the robustness of our model.
Results
Over a 25-year time horizon, the screening intervention dominated usual care. The probabilistic sensitivity analyses confirmed the robustness of these findings. The key drivers of the model were the proportion of melanomas detected by the screening procedure and the adherence of the target population to the screening programme.
Conclusions
The screening programme proved to be a dominant option compared with usual care. These findings should prompt serious consideration of the design and implementation of a regional or national melanoma screening strategy within a National Health Service.
Introduction
The economic burden of cancer is increasing, due to rising incidence, improved survival rates and increasing patient care costs.1–3 The incidence rates of malignant cutaneous melanoma in Europe and the United States have risen steadily in recent decades,4,5 and in Italy, the incidence increased from 1970 to 2015 for both men (from 1.6/100,000 to 21/100,000) and women (from 2/100,000 to 17/100,000). 6 Malignant cutaneous melanoma is an important public health issue as well as an economic concern, and the financial pressure on healthcare systems requires policy-makers to focus more on the appropriate allocation of resources.
Melanoma survival has a strong negative correlation with the extent of tumour invasion at diagnosis. 7 Advanced melanoma entails much higher costs than early stages of the disease.8,9 The impact of early detection on extended survival and subsequent treatment costs has increased the interest in analysing the cost-effectiveness of melanoma screening strategies.10–14 An Australian analysis performed over 20 years ago found that melanoma screening could be cost-effective if conducted every five years, particularly for men, with the incremental cost-effectiveness ratio (ICER) ranging from A$5745 (€3541) to A$25,214 (€15,543) per life-year saved. 10 In the United States, a Markov simulation model was developed in order to compare three general population melanoma screening strategies (one-time screening vs. every two years vs. annually from age 50) with usual care, and calculated ICERs of US $10,100/quality adjusted life year (QALY) (€7370/QALY), US $80,700/QALY (€58,885/QALY) and US $586,800/QALY (€428,172/QALY), respectively. 13 A more recent study used a Markov model with a latent period of 20 years and a time horizon of 50 years to analyse the cost-effectiveness of two population-based skin cancer screening programmes in Belgium, compared with no screening. Assuming that the total Belgian population aged 18 or older was invited for the screening programme, the two screening strategies achieved a similar ICER of about US $36,500/QALY (€32,975/QALY) in men and US $20,500/QALY (€18,520/QALY) in women. The authors also predicted that the cost of treating stages III and IV melanoma will continue to rise, due to new treatments, making screening an increasingly cost-effective strategy. 14
Evidence of the cost-effectiveness of melanoma screening considering the new target therapy is lacking. We therefore developed a cost-effectiveness model to assess the potential impact of a one-time general practitioner (GP)-based melanoma screening programme, in terms of direct costs and life expectancy, over a 25-year time horizon and including the most up-to-date drug therapies. We used a Whole Disease Model to simulate two counterfactual scenarios (with the target population receiving or not receiving the screening programme), within the perspective of the Veneto regional healthcare system within the Italian National Health Service.
Methods
The Italian National Health Service is organized at national, regional, and local levels, and provides universal coverage free of charge at the point of service. Regional health departments are responsible for ensuring the delivery of the essential levels of healthcare (defined at national level) through a network of population-based health management organizations (local health authorities) and accredited public and private hospitals.
A Whole Disease Model was developed in a previous study, 8 based on the patient care pathway for malignant cutaneous melanoma that was implemented in the Veneto Region in Northeast Italy on the grounds of international evidence and guidelines.15–17 Whole Disease Modelling is a methodological framework for developing economic models of whole health systems, including disease and treatment pathways, to inform resource allocation decisions. 15 The model contained clinical and process probabilities. The clinical probabilities were estimated from the literature or, failing this, from a clinical database of patients followed up by the Veneto Institute of Oncology. The deterministic chance (100%) of a process was established when the clinical pathway indicated a procedure for all patients with a given clinical condition. 17 Where the clinical pathway left decisions regarding particular diagnostic or therapeutic procedures to the physician’s discretion, process probabilities were identified using other guidelines, such as those of the National Comprehensive Cancer Network. 16 If the international guidelines were not sufficiently explanatory, several national melanoma experts were asked to indicate (based on their professional experience) the probability of patients with a given condition undergoing certain procedures, using the Delphi technique to obtain a consensus. 18 The annual direct costs for each disease stage from this previous study were used as input cost data for the present study.
On this basis, we considered two alternative scenarios for the Veneto regional healthcare system: a GP-based melanoma screening strategy of the population over the age of 20 and a strategy without any melanoma screening (usual care or status quo).
The GP-based melanoma screening strategy was devised largely on the strength of the SCREEN project conducted in Northern Germany,19–23 adapting some aspects of the model to make it more compliant with the Veneto healthcare system. In this screening strategy, people aged >20 who were resident in the Veneto Region would receive a mailed invitation, as this is the recruitment scheme already in use for other regional cancer screening programmes. The invitation includes a letter and a leaflet with explanatory photos, to help people to identify a mole as suspicious and to self-refer for a screening visit. Participants could refer to their own GP (if they were taking part in the project) or to another available GP working in the same Integrated Primary Care Team. In the primary screening test, participants undergo a total body skin examination by a trained GP, who refers them to a dermatologist if any genuinely suspicious lesions are found. In the secondary screening test, dermatologists examine patients referred by GPs, and may take a biopsy to reach a tentative clinical diagnosis.
Regional authorities decide the number of GPs to involve in the screening process. GPs are invited to participate in the project and authorized to conduct the primary screening test after completing a one-day (8-h) mandatory training course. Twelve-hour shifts of out-of-hours medical service are planned to enable GPs to attend the course. The number of GPs participating is assumed so that each of them would spend, on average, about 30 min of each working day on screening the participating population over the course of one year (assuming 220 working days and that a total body skin examination takes 12 min on average). This equates to about 550 screens in a year (2.5 visits each day × 220 working days). Based on the figures in the study by Breitbart et al., 19 we set the percentage of the target population participating in the screening programme at 19% and estimated that one GP is needed for every 2895 citizens (calculated as 550/0.19). For the hypothetical target population of 100,000 citizens, 34.5 GPs would have to be involved. The number of courses organized is estimated as one for every 30 GPs receiving the training, resulting in 1.15 courses per 100,000 target people. To cover GP absences due to the training course, we considered the involvement of out-of-hours doctors, estimating that one out-of-hours shift would be required for every 4.3 GPs (as out-of-hours doctors and GPs take care of 6500 and 1500 people, on average, respectively 24 ).
Table 1 shows the screening-related variables, which were derived from the SCREEN pilot project.19–23
Screening-related variables.
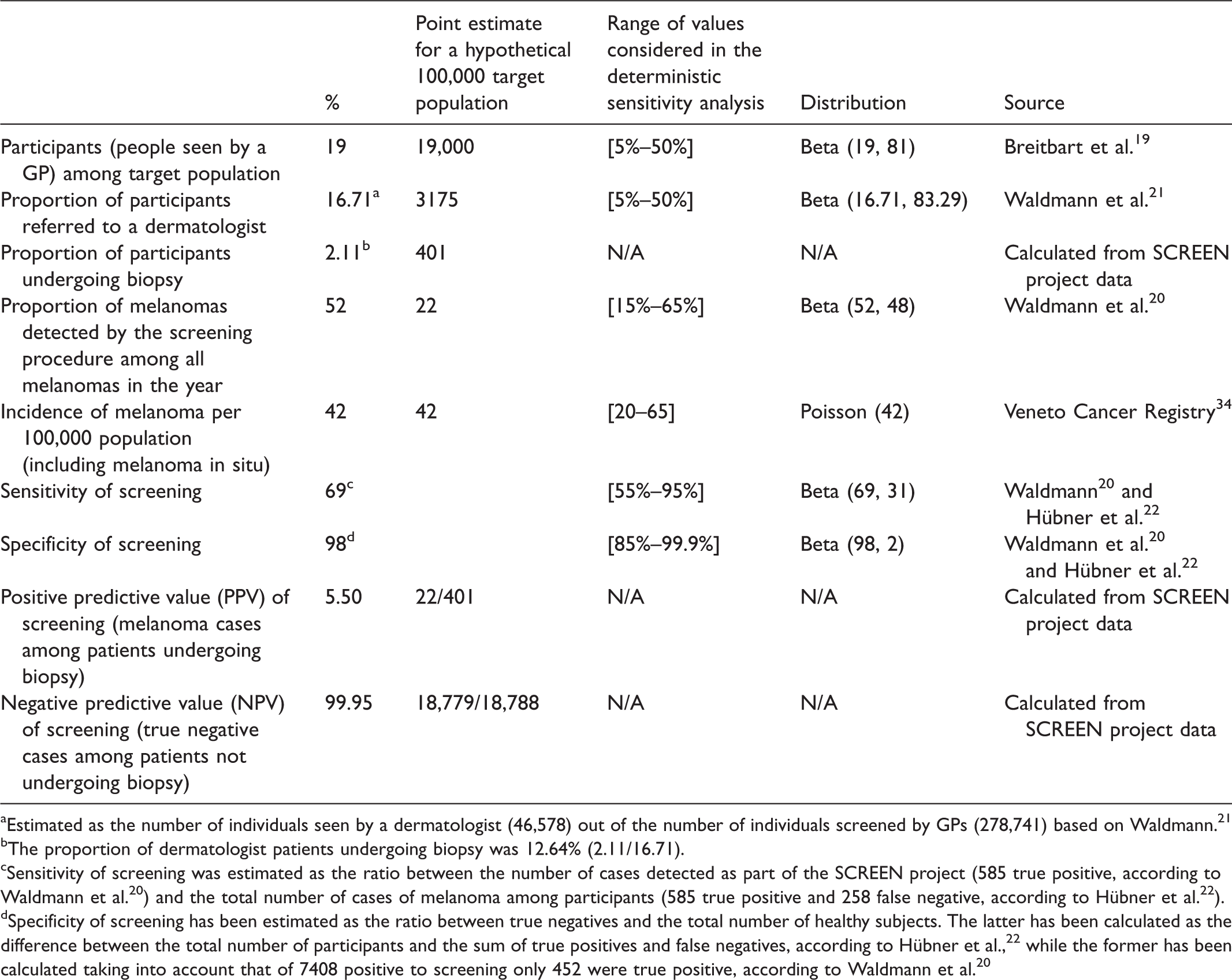
aEstimated as the number of individuals seen by a dermatologist (46,578) out of the number of individuals screened by GPs (278,741) based on Waldmann. 21
bThe proportion of dermatologist patients undergoing biopsy was 12.64% (2.11/16.71).
cSensitivity of screening was estimated as the ratio between the number of cases detected as part of the SCREEN project (585 true positive, according to Waldmann et al. 20 ) and the total number of cases of melanoma among participants (585 true positive and 258 false negative, according to Hübner et al. 22 ).
dSpecificity of screening has been estimated as the ratio between true negatives and the total number of healthy subjects. The latter has been calculated as the difference between the total number of participants and the sum of true positives and false negatives, according to Hübner et al., 22 while the former has been calculated taking into account that of 7408 positive to screening only 452 were true positive, according to Waldmann et al. 20
We developed a Markov model using TreeAge Pro 2011 over a time horizon of 25 years, to compare costs and outcomes with and without (usual care) screening (Figure 1(a) to (c)). In this study, we focused only on cases of melanoma detectable during the one-time screening campaign, and followed them over a time horizon of 25 years. Thus, future cases of melanoma have not been considered in our analyses, assuming that these cases will have equal costs and life expectancy in both scenarios.
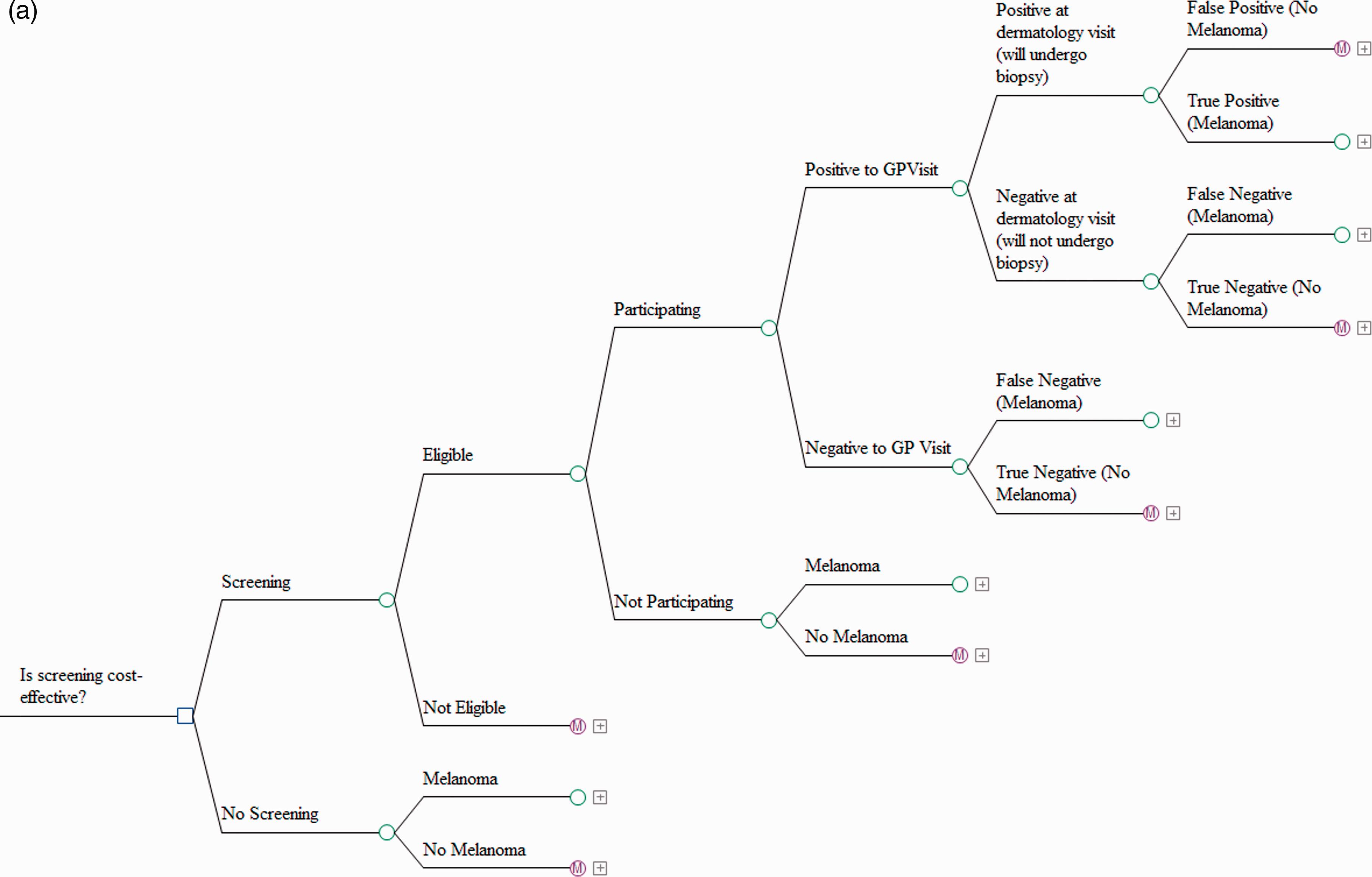
(a) Structure of the model – screening and no screening branches. (b) Structure of the model – Markov models. (c) Structure of the model – Markov models.
The model shows four mutually exclusive health states for patients with melanoma in stages I, II, III and in regression: stable, locoregional relapse, distant relapse and death (melanoma-related or due to natural causes). It shows only two health states – stable or natural death – for the melanoma-free and stage 0 (melanoma in situ) population. The model for stage IV melanoma patients presents three mutually exclusive health states: stable, relapse and death. The clinical probabilities governing the transition through each state of the Markov model25–27 are reported in online Appendix Tables 1 to 4. To separately obtain the probabilities of locoregional and distant relapse, the above likelihoods must be multiplied by those given in online Appendix Table 2.
Long-term survival was estimated by interpolating the probability of death in the second year with survival in the 10th year, as described by Balch et al., 25 and plotting Kaplan–Meier curves for stage IV, imposing the condition assumption that all stage IV patients must be dead within 10 years. For patients in stages II and III undergoing adjuvant interferon therapy, we applied a 17% relative reduction in the probability of occurrence of a first relapse, and a 9% relative reduction in the probability of death in the first year, as described in the meta-analysis by Mocellin et al. 28 Because our Whole Disease Model considered the most up-to-date drug therapies for the advanced stages (namely III inoperable and IV), in online Appendix Tables 3 and 4, the melanoma-related death risk was updated in line with the results of trials whenever data were available,29–32 and then the likelihoods were gradually aligned, over a period of 10 years, with those of the period before the latest drugs were introduced. 25 A proportion of patients was assumed to have permanently discontinued their therapy due to treatment-related adverse events during their second-line treatment. This likelihood was set at 11% during the first year and at 15% lifelong.29–32 An annual risk of natural death (ranging from 0.9% in year 1 to 1.6% in year 25) was also applied to all individuals in the model, based on the life tables for the Veneto Region. 33
The incidence rate of melanoma for adults over age 20 in the Veneto Region for 2016 was calculated using the incidence rate in the same region in 2013, and the compound annual growth rate (3.6% in men, 2.4% in women) calculated from 1987 to 2013. 34 The same overall incidence was set for both the screening and the no-screening branches of the model. This choice was based on the assumption that the incidence of melanoma is not influenced by the screening strategy during a steady-state period. In the study by Breitbart et al., 19 despite an initial increase during the implementation of the screening project, after a period of three years, the incidence of melanomas returns to the levels that existed before the screening campaign.
Online Appendix Table 5 shows the distribution, by stage, of melanoma at diagnosis in the Veneto Region. These stage frequencies at diagnosis corresponded to the current incidence of melanoma, and they were assigned both to participants in the non-screening and screening branch who failed to attend or were false-negative cases. Data were obtained from the Veneto Cancer Register (personal communication). To obtain the distribution by melanoma stage at diagnosis of the true positive screened patients, we applied the percentage of relative variation observed before and after implementing the SCREEN project in Northern Germany to the distribution described above. 23 We chose to adopt this approach, as the population of the Veneto Region and that of Germany have a similar pre-screening stage at diagnosis distribution, and so similar variations are expected after screening.
The health effect of screening was taken as the variation in life expectancy as a result of the melanoma stage shift. A score of 0 (death) or 1 (alive) was assigned to each person in the model for each year and was discounted at a 3% annual rate.
The study was conducted from the perspective of the Veneto regional healthcare system within the Italian National Health Service, and considered only the direct costs borne by a regional government. Direct and indirect patient costs were excluded. Costs (in Euros) are drawn from official reimbursement tariffs in effect in 2017. Costs (and outcomes) after the first year were discounted at a 3% annual rate. The costs to a healthcare system of implementing the screening campaign, per 100,000 target population, are presented in online Appendix Table 6.
The annual direct costs for each stage of disease reported in online Appendix Table 7 are drawn from a previous study. 8 The same stage-specific clinical pathways were modelled after diagnosis in both scenarios. We assumed that each transition from one Markov state to another happens in the sixth month. In the year when a transition towards locoregional or distant relapse occurred, we stopped the follow-up costs after six months, and applied a cost that included re-staging and surgical/medical treatments for the remaining six months (see online Appendix Table 7). In the case of death due to natural causes, we stopped the follow-up and any possible medical treatments at six months, while in the case of melanoma-related death, we stopped the costs of medical therapy six months beforehand (no treatment costs were applied because the patients were expected to die by the end of the sixth month), and we applied the cost of three months of palliative care. 35 The costs associated with the occurrence of a locoregional lesion include surgery and the introduction of medical therapy, amounting to €10,560. For every melanoma stage, the cost associated with the occurrence of distant relapse was €40,929, taking into account the weighted probability of different metastasis sites, and the probability of surgical therapy for each site, as well as the related costs. 8 The cost associated with palliative care was €2199 and included hospitalization, hospice and outpatient care. 35
The National Institute for Health and Clinical Excellence established a willingness-to-pay threshold between £20,000 and £30,000 per QALY. 36 We considered a willingness-to-pay threshold of €25,000 per life-year gained to ascertain the cost-effectiveness of one-time screening compared with the usual care option. This threshold should be appropriate even if we considered life-years gained instead of quality of life, as any adjustment would lower this threshold.
We conducted a series of sensitivity analyses, varying some parameters to assess the robustness of our model and to identify the parameters with the greatest impact on cost-effectiveness. We focused particularly on screening-related variables, such as the participation rate and sensitivity/specificity of the screening procedure (Table 1), as well as on the discount rate (using a range from 0% to 5%).
A probabilistic sensitivity analysis was performed to determine the impact of the uncertainty surrounding the model input parameters. Beta distributions were assigned to the screening-related probabilities, such as the participation rate and the proportion of participants referred to a dermatologist (Table 1). In addition to these variables, we chose to assign a Dirichlet distribution to the stage-at-diagnosis probabilities shown in online Appendix Table 5, to embed the uncertainty of these percentages in our analysis. As costs were assigned on the basis of guidelines, and defined according to official reimbursement tariffs, these were not included in our sensitivity analyses. A random value from the corresponding distribution was selected, from which we generated an estimate of the mean cost and outcomes associated with each scenario. This was repeated 10,000 times, and the results for each simulation were noted. An estimate of the average costs and outcomes can thus be obtained, along with the 95% credibility intervals. A sensitivity analysis was conducted for the tornado diagram, by holding the target variable at its lowest and highest values and running a 10,000 probabilistic sensitivity analysis simulation.
In addition, we investigated the impact of the screening intervention on expected costs and health outcomes in the Veneto Region via cost–consequence analysis. 37 The total budgetary impact of the screening programme and the overall improvement in terms of survival over the 25-year time horizon were estimated.
Results
The incremental discounted average life expectancy and costs in the two alternative scenarios (screening strategy vs. usual care) over a 25-year period are shown in Table 2. Over 25 years, the average cost per citizen (over age 20) to diagnose and treat melanoma was estimated to be €70.06 (95% credibility interval: €65.60–74.84) for the screening strategy and €72.65 (95% credibility interval: €67.87–77.94) for the usual care option (Table 2). Considering only the patients with melanoma, we observed an average cost of €156,045 and €170,943 with the screening campaign and the status quo, respectively. Compared with treatment as usual, the screening strategy would result in an average increase of four months of life for each patient (from 13.27 to 13.60 years), resulting in an overall gain of 13.90 years of life in the 100,000 target population.
Average costs and life-years estimated with 10,000 simulations.
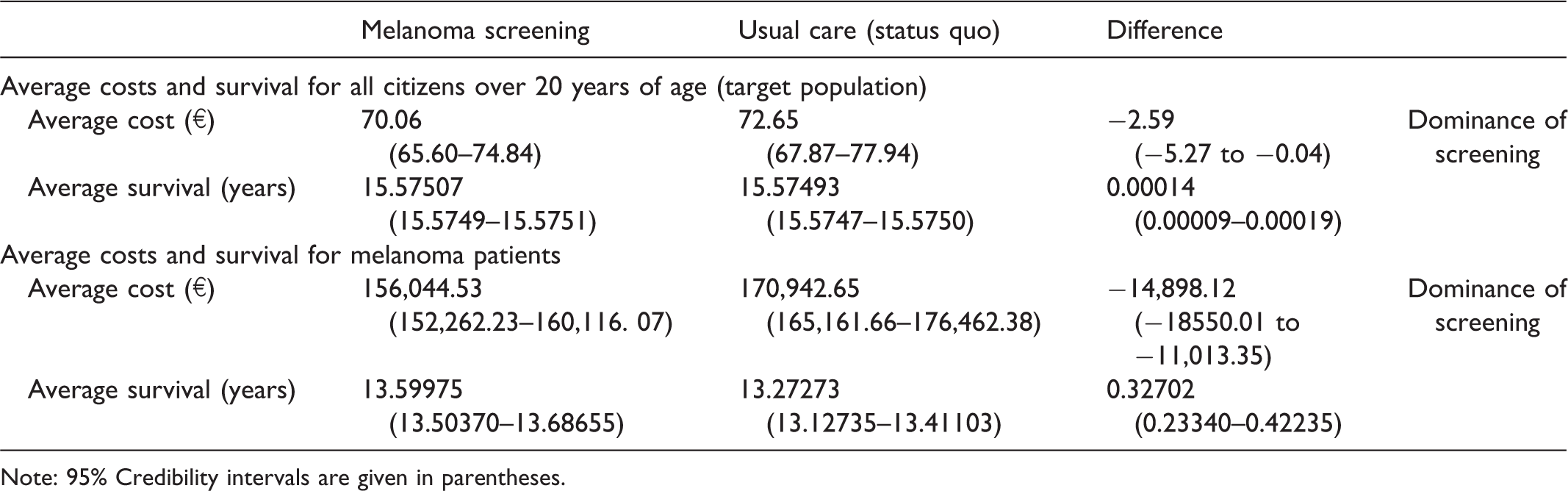
Note: 95% Credibility intervals are given in parentheses.
As the screening strategy was associated with lower mean costs and higher survival rates than usual care, the screening intervention was therefore dominant. The screening strategy led to savings in both money and life-years, namely, −€18,551 per year of life gained across the entire target population.
The one-way sensitivity analyses show that the proportion of melanomas detected as part of the screening programme and the screening adherence rate are the variables that contribute most to the variability of the ICER (Figure 2).
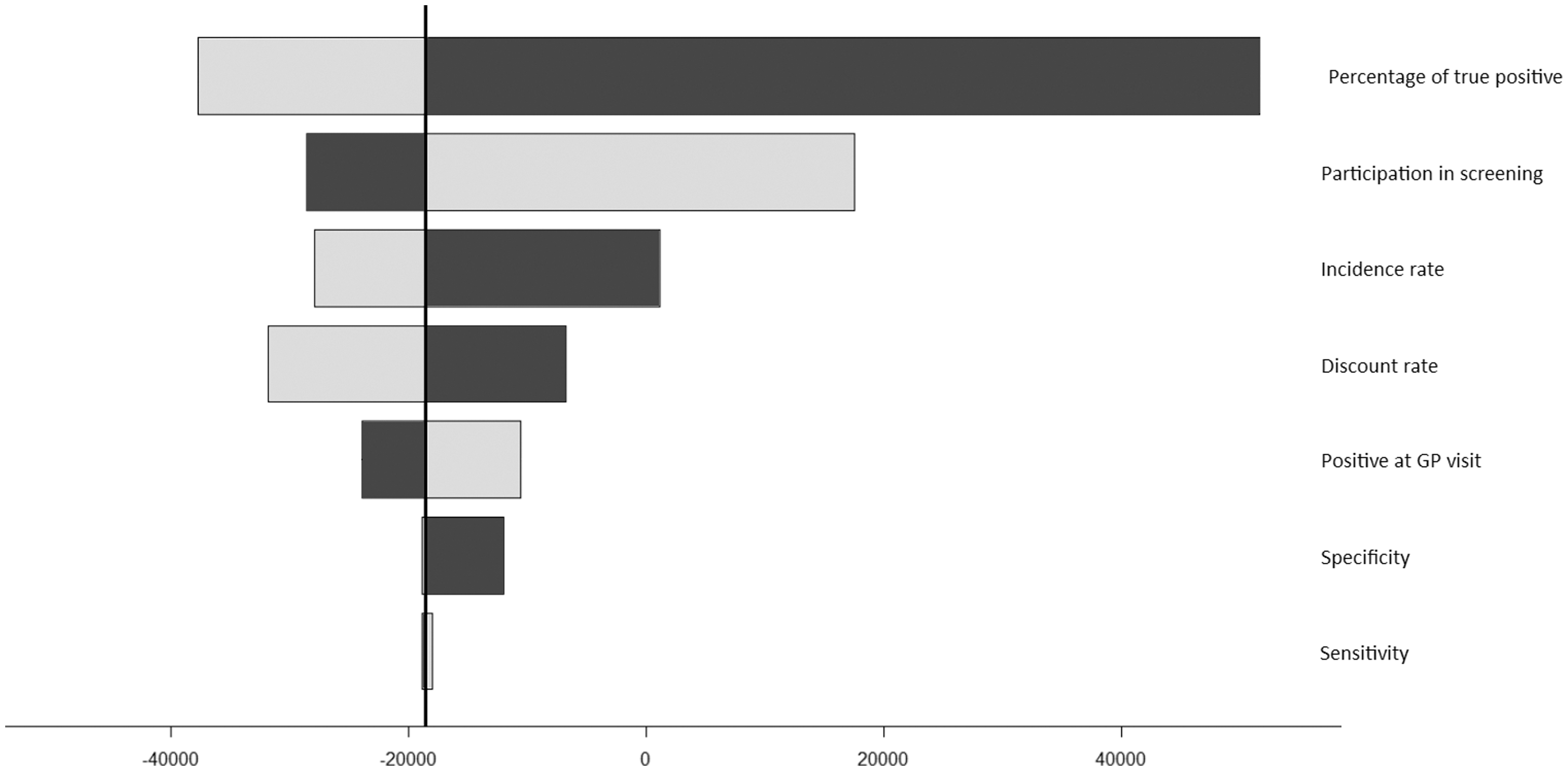
Tornado diagram showing the most influential screening variables for ICER. White bars show the maximum values of the variable while black bars show the minimum value. For instance, lower values for the percentage of melanomas detected by screening procedures lead to higher ICER.
The results of the probabilistic sensitivity analysis show that almost all of the 10,000 simulations are cost-effective, if not cost saving (Figure 3), and that the screening option has a 97.38% probability of being dominant.
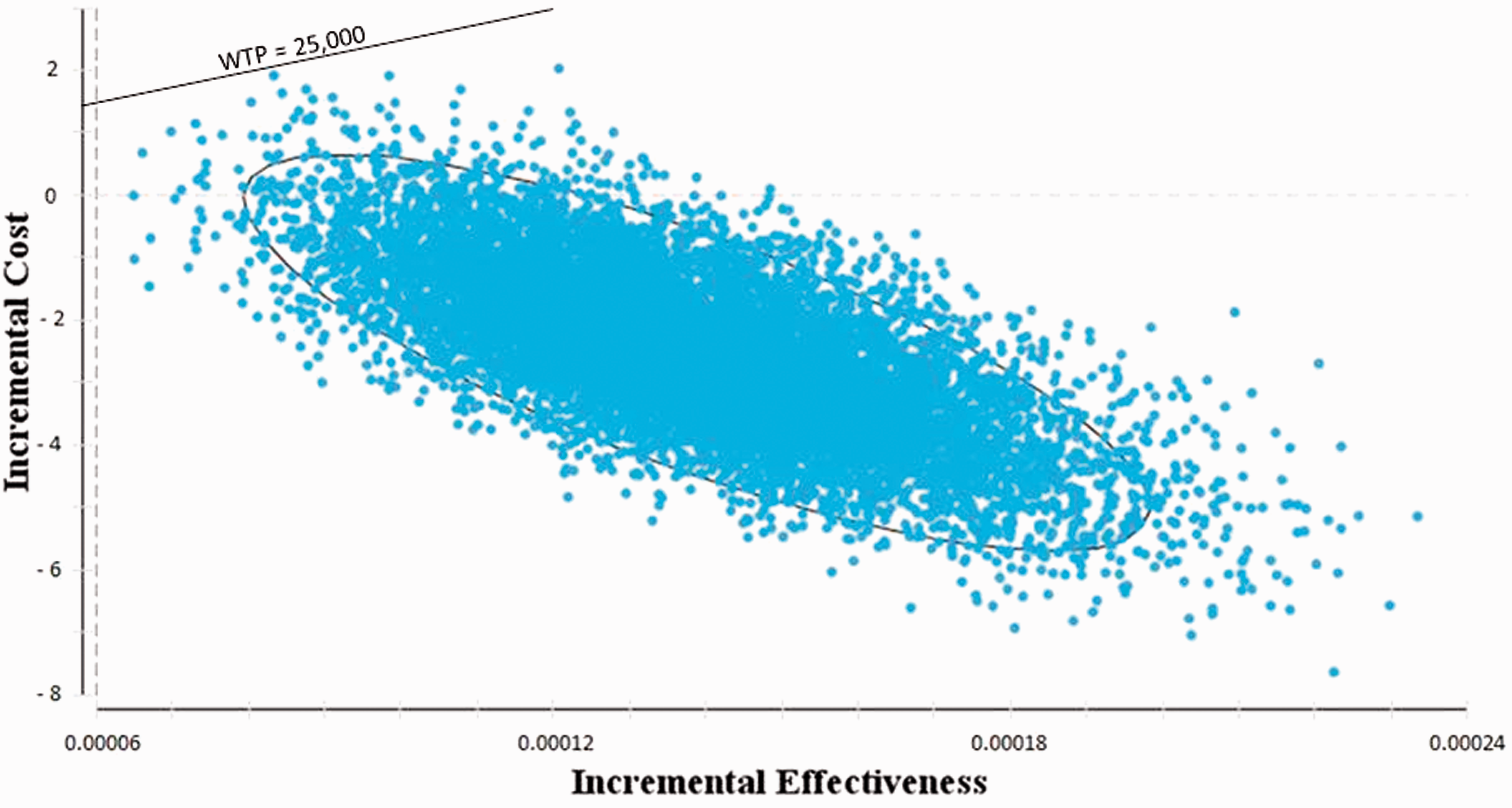
Results of probabilistic sensitivity analysis with 10,000 simulations. The ellipse represents the 95% confidence interval.
Figure 4 shows the different ICER values over a 5-, 10-, 15-, 20- and 25-year period. Again, these estimates and their credibility intervals were obtained using a probabilistic sensitivity analysis, with 10,000 simulations.
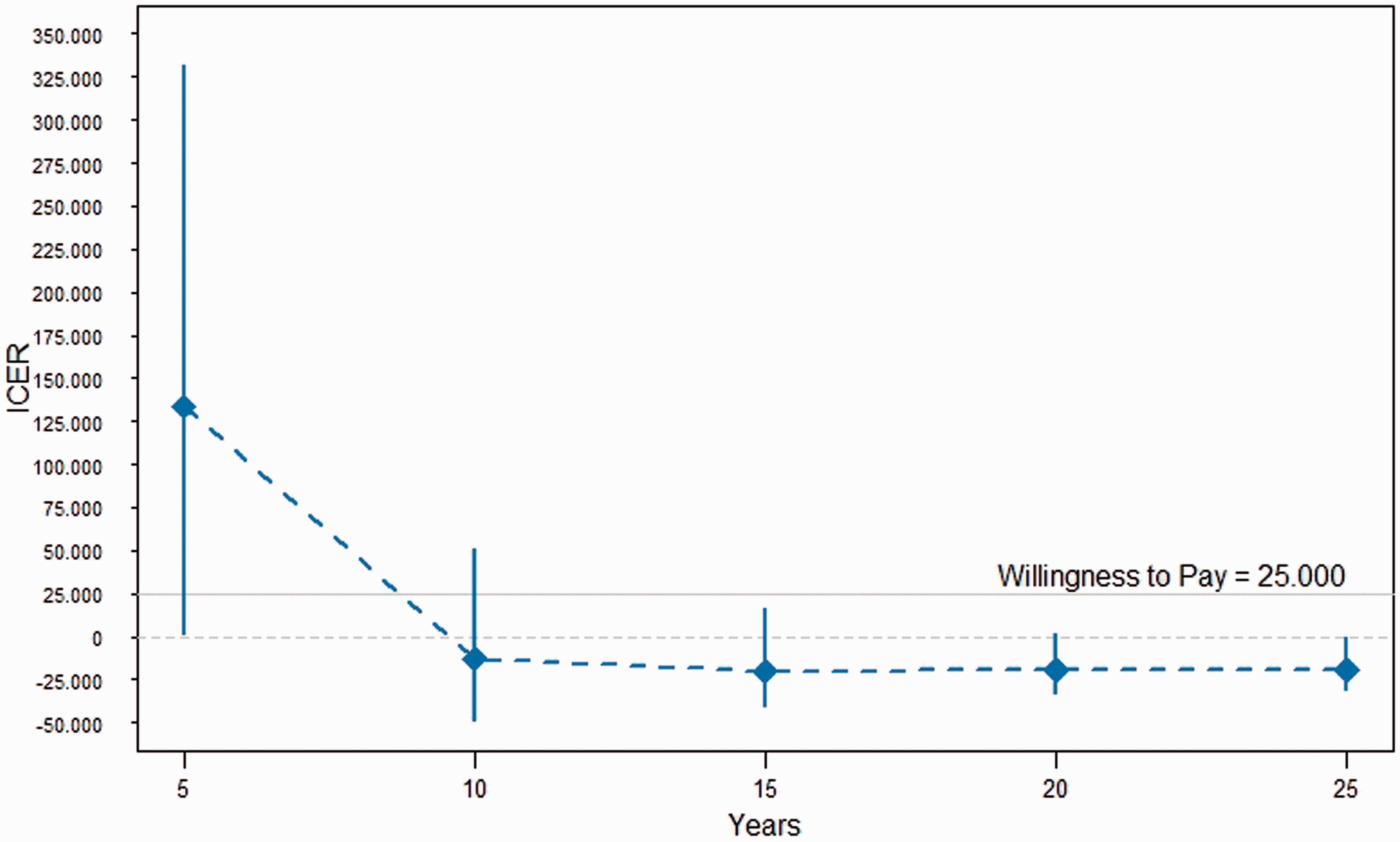
ICER by years, with 95% credibility intervals.
According to our Whole Disease Model, from the perspective of the Veneto regional healthcare system (with a target population of 4,004,266 citizens aged over 20), a one-time screening programme could save 557 life-years and reduce the economic burden of melanoma by €10.2 million (€280.4 million in the screening scenario vs. €290.6 million Euros in the usual care scenario).
Discussion
Although our study showed that the melanoma screening strategy was dominant compared with usual care (status quo), our results differ to some degree from those of previous studies. While comparisons are difficult to make because of differences in the screening methods, the composition of the screening team, the model designs and epidemiological factors related to the involved populations, all previous cost-effectiveness analyses show that a screening option would be more effective than usual care but also more expensive.10–14 Although our results are dissimilar, they are not surprising, considering that the cost of treating stages III and IV melanoma will continue to increase as a result of new, more expensive treatments, making screening an increasingly more cost-effective strategy. 14 In addition, a screening programme is more effective and more cost-effective when the prevalence of the undiagnosed disease is high enough to justify the effort and costs of screening. For example, in 1992, in high-incidence countries such as Australia, economic evaluations suggested that the potential cost-effectiveness of melanoma screening programmes, with discounted ICER, ranged from €8,483 to €25,843 per years of life gained, based on different assumptions (i.e. in many cases below the current National Institute for Health and Clinical Excellence threshold of acceptability for cancer care). 10 Although the incidence of melanoma in Italy and in the Veneto Region has yet to reach the levels seen in Australia in 1992, it has been rapidly and constantly increasing in recent decades, 34 and is moving towards levels that would justify a screening campaign.
The results of the sensitivity analysis in our model highlight the crucial impact of the adherence rate on life expectancy and the ICER. A high participation rate is desirable, as it increases the proportion of melanomas diagnosed in the early stages and, consequently, the years of life saved by screening and early treatment. However, a high participation rate also reduces the cost-effectiveness of the screening, because larger numbers of visits and biopsies add to the costs.
Importantly, the sensitivity and specificity of the screening did not result in marked changes in the baseline ICER value.
Several studies have highlighted the importance of implementing screening that enrols only higher risk individuals. The study of a simulated screening programme by Wilson et al. found that a campaign targeting people at higher risk of developing melanoma was more cost-effective than population-wide screening; 38 however, the model was built on the basis of the 2010 guidelines, which do not include the newer, expensive treatments for advanced stages of the disease. As Hübner et al. point out, focusing on individuals at high risk for melanoma may also improve the benefit-to-harm balance of melanoma screening programmes. 39 The screening programme in our study, in accordance with the SCREEN project conducted in Northern Germany, includes a recruitment scheme based on explanatory images that should help people identify a suspect lesion, and so the invitation for screening tends to recruit potentially higher risk individuals.
There are some limitations to this study that should be addressed in future work. First, melanoma screening programmes are likely to increase the number of other skin cancers that would be detected during skin examinations. Our model did not consider the costs and health outcomes related to this surplus diagnosis of keratin cell cancers that might otherwise go undetected without screening. We therefore cannot tell whether the costs of detecting a greater proportion of melanomas in the early stage of the disease will outweigh the unavoidable costs of also finding and treating more keratinocyte cancers. Moreover, we did not consider the number of melanomas that would never develop clinical signs (overdiagnoses) but which, once diagnosed, inevitably expose patients to psychological harms 40 and treatments, and which increase the real costs of screening. Second, some other costs and measures of effectiveness were omitted, such as indirect costs (known to be high for this kind of tumour, especially in its advanced stages). 41 Any toxicity-related healthcare costs were also not considered, because evidence regarding the toxicity of the new therapies is still too recent and often controversial. Third, quality of life was not considered, although melanoma, particularly in the advanced stages, may well affect this. Had it been taken into account, this would have presumably made the screening branch of the model even more cost-effective, because screening prompted a shift towards less-advanced cancers, which should have a smaller effect on indirect costs and quality of life. Reducing the number of advanced melanomas, and consequently using less medical therapy, should also mean less toxicity and lower toxicity-related healthcare costs. However, the only costs that were not considered that might lower the cost-effectiveness of the screening were the indirect costs of the screening programme, and particularly its intangible costs due to worries over unnecessary visits and biopsies. Finally, we did not take into account the uncertainty in the survival probabilities, which may be a further source of variation in the cost-effectiveness of the screening strategy. In the case of the curves drawn from Balch et al., 25 however, the standard errors are relatively small because they are based on a large dataset, and would therefore not be significant.
Conclusion
Using a Whole Disease Model approach, we have shown that a one-time GP-based melanoma screening programme is dominant to usual care. Although some assumptions made in calculating the cost-effectiveness estimates may not be precise, and may be subject to variability, sensitivity analysis confirmed the robustness of our screening strategy. These findings could prompt policymakers to consider the design and implementation of a melanoma screening strategy within public healthcare systems. This study focused on a one-time screening intervention because we simulated a new scenario based on the SCREEN project conducted in Northern Germany. Further studies are warranted to demonstrate the cost-effectiveness of repeated melanoma screening programmes. This study will assist healthcare commissioners in melanoma cancer control, taking into account the latest evidence on epidemiological changes and health technology assessment evaluation.
Supplemental Material
MSC885998 Supplementary material - Supplemental material for Cost-effectiveness of a melanoma screening programme using whole disease modelling
Supplemental material, MSC885998 Supplementary material for Cost-effectiveness of a melanoma screening programme using whole disease modelling by Alessandra Buja, Michele Rivera, Giovanni Girardi, Antonella Vecchiato, Vincenzo Rebba, Elena Pizzo, Vanna Chiarion Sileni, Angelo Claudio Palozzo, Maria Montesco, Manuel Zorzi, Gino Sartor, Manuela Scioni, Mario Bolzan, Paolo Del Fiore, Maria Giuseppina Bonavina, Massimo Rugge, Vincenzo Baldo and Carlo Riccardo Rossi in Journal of Medical Screening
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. Pizzo was supported by the National Institute for Health Research (NIHR) Collaboration for Leadership in Applied Health Research and Care (CLAHRC) North Thames at Bart’s Health NHS Trust. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by BRISTOL-MYERS SQUIBB Srl. The company had no role in the design of the study, the collection, analysis or interpretation of the data, the writing of the manuscript or the decision to submit the paper for publication.
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
