Abstract
Objectives
To report changes in incidence of cervical tumours by disease stage, following the introduction of an organized cytology-based screening programme.
Methods
An intention-to-screen study of a cytology-based screening programme targeting 1,219,000 women aged 25–64 in northern Italy was carried out. Based on the previously reported trend in total incidence of cervical cancer, the study period 1995–2014 was divided into 1995–1996 (pre-screening, or reference, years), 1997–1998 (screening implementation phase), 1999–2006 (transition phase, when incidence decreased), and 2007–2014 (steady-state phase, when incidence stabilized again). Tumour stage was categorized as preinvasive (cervical intraepithelial neoplasia grade 3 (CIN3) and adenocarcinoma in situ), early (pT1a), advanced (pT1b or greater, ypT), and unknown (pT1 not otherwise specified, pTx, missing information). Average annual incidence rates observed in each phase were compared with the expected (reference) rates, using the incidence rate ratio, calculated with a Poisson regression model.
Results
In the steady-state phase, incidence rate ratios were: CIN3, 1.55 (95% confidence interval, 1.41–1.70); early-stage squamous carcinoma, 0.49 (0.36–0.67); advanced-stage squamous carcinoma, 0.44 (0.33–0.57); unknown-stage squamous carcinoma, 0.69 (0.48–0.99); adenocarcinoma in situ, 1.44 (0.72–2.88); early-stage adenocarcinoma, 2.65 (0.82–8.53); advanced-stage adenocarcinoma, 1.03 (0.56–1.91); and unknown-stage adenocarcinoma, 0.46 (0.23–0.92).
Conclusions
After stabilization, changes in incidence by tumour stage included a 55% increase for CIN3 and a 50–55% decrease both for early- and advanced-stage squamous carcinoma, but no significant changes for glandular tumours. These data will serve to quantify the incremental impact of the implementation of human papillomavirus-based screening, introduced in 2015.
Introduction
Many European countries have initiated the conversion of cytology-based screening for cervical cancer into the combined strategy of human papillomavirus (HPV) vaccination and HPV-based screening. 1 This radical change of paradigm necessitates coordination, monitoring, and evaluation of the two complementary anti-HPV services.2–4 To assess the effectiveness of this combined strategy, it will be necessary to estimate the incremental benefit of HPV-based screening, both over cytology-based screening and over HPV vaccination, 5 which will require the availability of data consistently defined and collected. These tasks are challenging. In particular, there are doubts that the incremental impact of HPV testing over cytology-based screening will be measurable, because the baseline level of effectiveness achieved with the Pap smear is ill-defined for many programmes. The European guidelines for cytology-based organized screening activities recommend that the impact on incidence be accurately evaluated.2,6,7 To our knowledge, this has been done in only a few countries and regional areas.8–11
Since 1997, women aged 25–64 living in the Emilia-Romagna Region (northern Italy) have been targeted by a cytology-based cervical screening programme. In 2008, a regional HPV vaccination campaign was launched, 12 and in 2015 the conversion to HPV-based screening of women aged 30–64 was undertaken. We aimed to evaluate the changes in incidence of cervical tumours that are attributable to the cytology-based screening programme relative to the pre-screening years, when only opportunistic testing was available. We have previously addressed total incidence. 13 The present study complements the previous one by examining the effects on the incidence of cervical tumours according to their stage.
Methods
Before the implementation of the Emilia-Romagna Region cervical screening programme, resident women were offered only opportunistic screening. In 1995, the opportunistic screening coverage of the regional female population aged 25–64 (defined as the proportion reporting at least one Pap smear in their lifetime) was 74.5%. 14 The programme started in 1997. 13 Several articles have reported on its organization, 15 quality assurance,16,17 and epidemiologic surveillance.13,18–20 Every three years, resident women in the target age range (n = 1,219,000 on 1 January 2018) were invited by a personal letter to attend district screening centres. Pap smears were taken by trained midwives. Abnormal screening results were notified to women by telephone. Colposcopy assessment was performed at selected clinics. In 2008 (previous data not available), the three-year coverage of the target population was 60.2% for the organized screening programme (last Pap smear in the organized screening setting), and 26.7% for opportunistic screening (last Pap smear in the opportunistic setting), giving a total of 87.0%. Between 1998 and 2014 (ending year of the study, see below), these two measures followed opposite trends, with the organized screening increasing, and opportunistic screening reducing. In 2014, these were respectively 67.1% and 23.5%, giving a total of 90.7% of the target population. These data were collected through telephone surveys conducted by the Italian National Institute of Health (Italian: Istituto Superiore di Sanità). 21 Routine performance indicators of all Italian regional cervical screening programmes are collected annually and published by the Italian National Centre for Screening Monitoring (Osservatorio Nazionale Screening).22,23 The Emilia-Romagna Region cytology screening programme has constantly performed at acceptable levels compared with the Italian national standards.
We have previously reported that before the programme started in the second half of 1997, the total annual incidence rate of cervical carcinoma in the target age range had been stable since 1988 (Figure 1). 13 The rate peaked in 1998, decreased between 1999 and 2007, and stabilized thereafter. The rates that would have been expected in 1998–2013 in the absence of screening were estimated, with virtually coincident results, using a log-linear model and an age-period model. Table 1 shows the estimate based on the latter. The expected rates in the absence of screening were stable. Between 2007 and 2013, the median ratio between each observed and expected annual rate varied between 0.60 (95% confidence interval (CI), 0.45–0.81) and 0.58 (95% CI, 0.34–0.97), depending on the method for estimating the expected rates. These changes were entirely accounted for by squamous carcinoma. The incidence of adenocarcinoma remained stable over the whole study period.
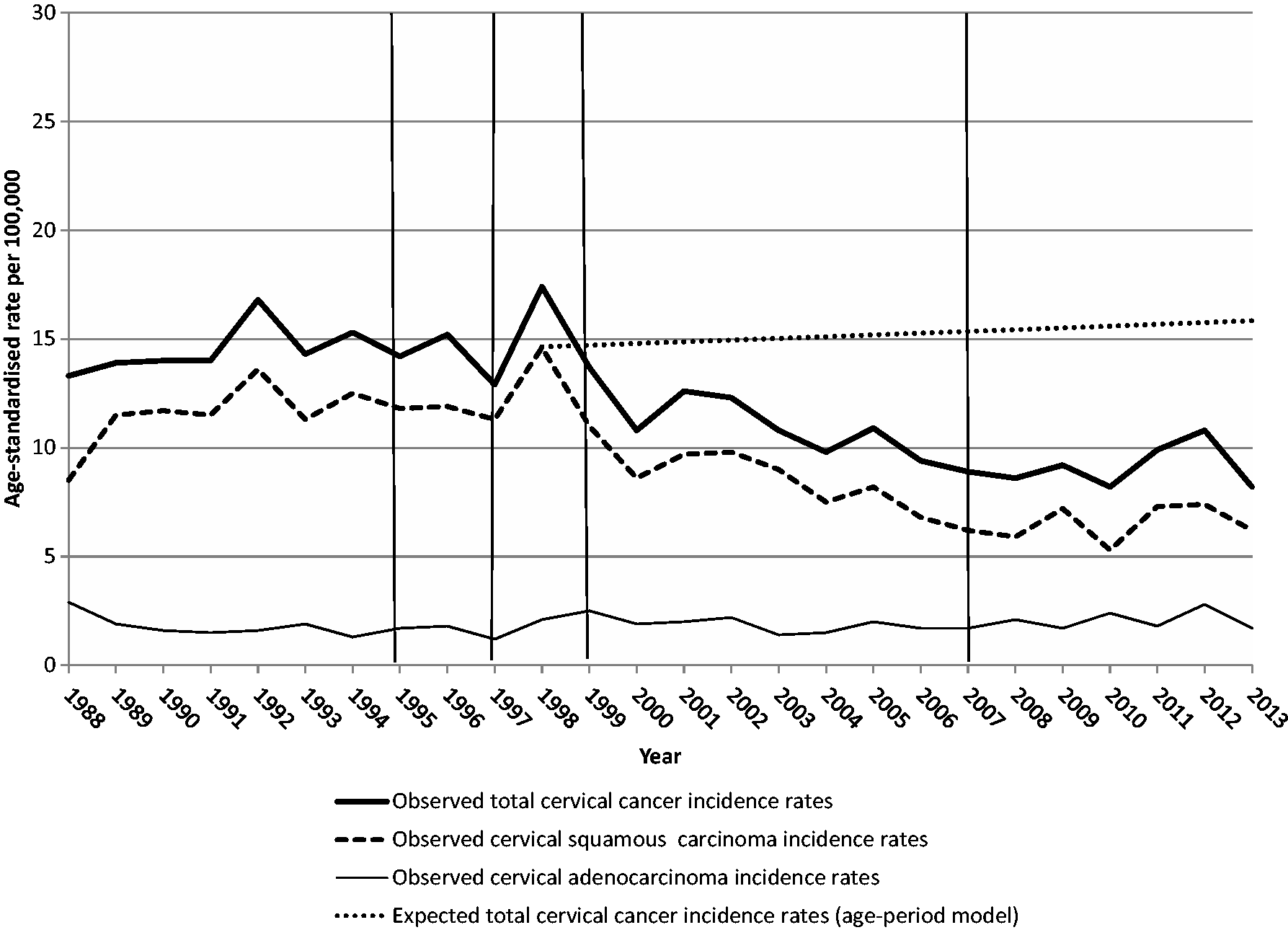
Curve of observed total annual incidence rates of cervical cancer per 100,000 women aged 25–64 in 1988–2013, curve of total annual rates that would have been expected in 1998–2013 in the absence of screening, derived from an age-period model analysing the rates in the period 1988–2013, and curves of observed annual incidence rates of cervical squamous carcinoma and cervical adenocarcinoma. All rates were age-standardized (2013 European standard population). The graphs, generated for a previous study, 13 were reproduced with permission. The time period covered by the present study was divided into 1995–1996 (pre-screening years), 1997–1998 (screening implementation phase), 1999–2006 (transition phase), and 2007–2014 (steady-state phase). Emilia-Romagna Region, Italy.
Ratio between the average annual age-standardized (2013 European standard population) incidence rate of cervical tumours per 100,000 women aged 25–64 in 1997–1998, 1999–2006, and 2007–2014 and the rate of pre-screening years (1995–1996) by type and stage of disease. Emilia-Romagna Region cancer registries and health care district cervical cancer screening programmes, Italy, 1995–2014.
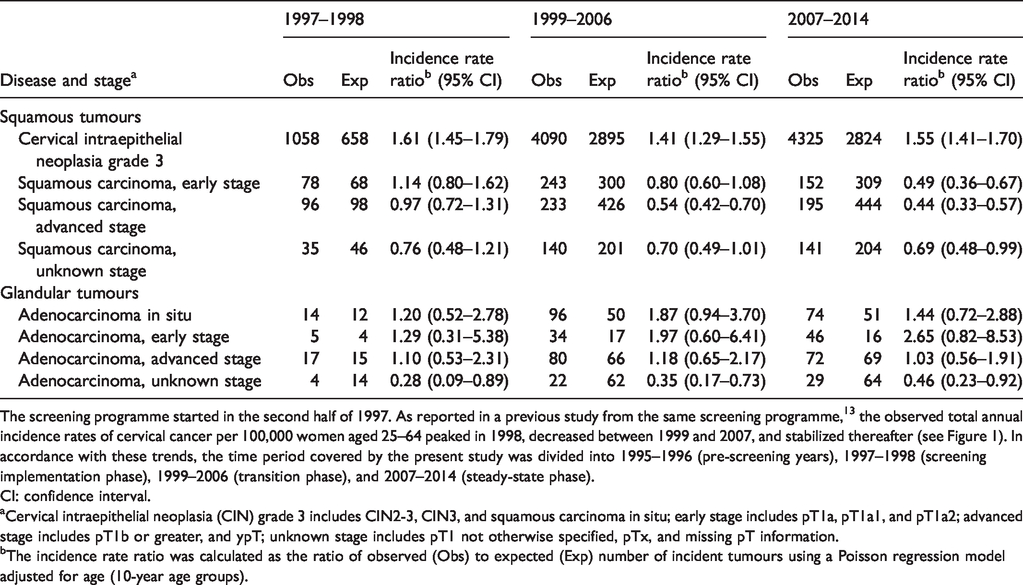
The screening programme started in the second half of 1997. As reported in a previous study from the same screening programme, 13 the observed total annual incidence rates of cervical cancer per 100,000 women aged 25–64 peaked in 1998, decreased between 1999 and 2007, and stabilized thereafter (see Figure 1). In accordance with these trends, the time period covered by the present study was divided into 1995–1996 (pre-screening years), 1997–1998 (screening implementation phase), 1999–2006 (transition phase), and 2007–2014 (steady-state phase).
CI: confidence interval.
aCervical intraepithelial neoplasia (CIN) grade 3 includes CIN2-3, CIN3, and squamous carcinoma in situ; early stage includes pT1a, pT1a1, and pT1a2; advanced stage includes pT1b or greater, and ypT; unknown stage includes pT1 not otherwise specified, pTx, and missing pT information.
bThe incidence rate ratio was calculated as the ratio of observed (Obs) to expected (Exp) number of incident tumours using a Poisson regression model adjusted for age (10-year age groups).
The present study was designed in accordance with the above trends in total rate of cervical carcinoma. Tumour stage-specific incidence data began to be available in 1995. We considered the years 1995–1996 to represent the pre-screening or reference years, when only opportunistic testing was available. We divided the screening years into 1997–1998 (screening implementation phase), 1999–2006 (transition phase) and 2007–2014 (steady-state phase). The latter was the phase in which a new dynamic equilibrium was reached between the inception of new preclinical diseases and the capability of the screening process to prevent their progression to invasive disease.
The results of the study were independent of the effects of HPV vaccination and HPV-based screening. In 1997–2014, the first cohorts undergoing HPV vaccination, those of 1996 and 1997, had not yet entered the target population of the screening programme. In that period, the target population was exposed exclusively to cytology-based screening, as HPV testing was introduced in 2015.
The rationale of the study was based on three assumptions: the impact of the screening programme on incidence may vary across tumour stages; the benefit may include not only a reduction in total incidence of cervical carcinoma, but also a shift toward lower tumour stage among those cases that are not prevented, i.e. a downstaging effect; and the expected shift from total invasive to preinvasive disease may be considered a different kind of downstaging, which needs to be assessed. The primary objectives of the study were: (1) to calculate the average annual incidence rates of preinvasive tumour, early-stage and advanced-stage invasive squamous carcinoma and adenocarcinoma in the above four time segments; (2) to compare the rates in the three screening phases with those expected, based on the pre-screening years; and (3) to compare the invasive to preinvasive tumour incidence rate ratio (IRR) and the advanced- to early-stage cervical carcinoma IRR in the three screening phases with the IRRs observed in the pre-screening years. A decrease in these ratios was considered to indicate a downstaging effect. A secondary objective of the study was to evaluate the changes in incidence of non-screen-detected cervical tumours.
All estimates were obtained independently for squamous and glandular tumours. Adenocarcinoma was included in the study on the basis of the hypothesis that a downstaging effect might underlie the reported stability of total incidence.13,20
Records of preinvasive and invasive cervical tumours (International Classification of Diseases-10th Revision code C53) cases for the years 1995–2014 were made available by the six general and specialized cervical cancer registries – all accredited by the International Agency for Research on Cancer – that cover the Emilia-Romagna Region. Details on the registration process can be found in our previous article. 13
Preinvasive diseases were divided into cervical intraepithelial neoplasia grade 3 (CIN3) (including CIN2-3, CIN3, and squamous carcinoma in situ) and adenocarcinoma in situ (AIS). The stage of invasive tumours was categorized as early (including pT1a, pT1a1, and pT1a2), advanced (including pT1b or greater, here referred to as pT1b+, and ypT), and unknown (including pT1 not otherwise specified, pTx and missing pT information).
All observed and expected average annual incidence rates in the age range 25–64 were age-standardized (five-year age groups) using the 2013 European standard population. Incidence rates of screen-detected and non-screen-detected tumours were calculated using the number of registered screen-detected and, respectively, non-screen-detected tumours as numerator, and the whole target population as denominator. Observed average annual incidence rates were compared with those expected by means of the IRR with 95% CI. The IRRs were calculated as observed to expected ratios using a Poisson regression model adjusted for age (10-year age groups). The invasive to preinvasive tumour IRRs and the advanced- to early-stage cervical carcinoma IRRs in the four time segments were calculated using the same methods.
The study protocol was approved by the Ethics Committee at the Romagna Cancer Institute (ID: IRST100.37).
Results
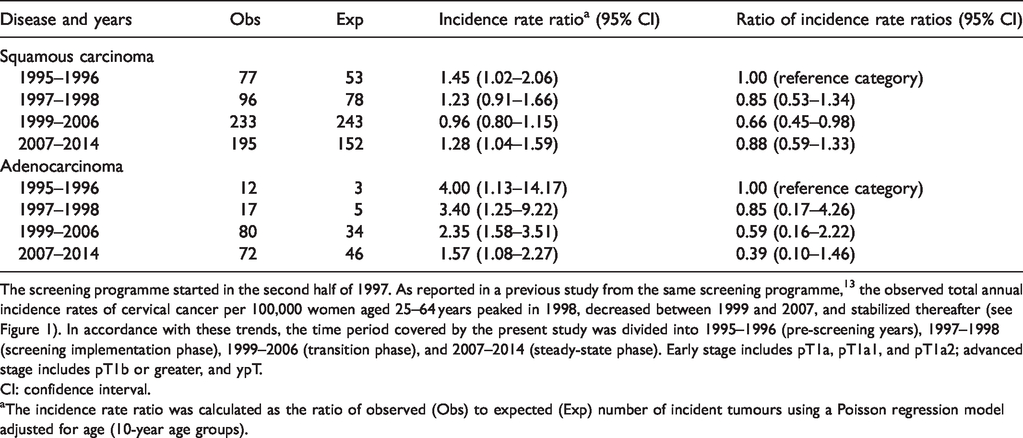
Figure 2 shows the curves of annual incidence rates of squamous tumours. The rates of CIN3 and the much lower ones of invasive carcinomas are plotted on two different vertical axes for presentational purposes. The increase in the rate of CIN3, characterized by large fluctuations, was paralleled by a drop in the incidence of both early- and advanced-stage squamous carcinoma. The upper section of Table 1 shows a formal analysis of these changes. In the implementation phase (1997–1998), the rate of CIN3 increased by approximately 60% compared with the pre-screening years. Subsequently, the increase was slightly smaller, varying between 40% and 55% in the steady-state phase. For invasive carcinoma, the annual rate of advanced-stage lesions decreased earlier than that of early-stage lesions, with an IRR in the transition phase of 0.54 versus a weakly significant 0.80. In the steady-state phase, however, the magnitude of the incidence decrease was similar for both tumour stage categories at about 50–55%. The upper section of Table 2 shows that the different trends of CIN3 and invasive squamous carcinoma resulted in a constant decrease in the invasive to preinvasive squamous tumour IRR. In the steady-state phase, the incidence of invasive lesions was almost ten times lower than that of CIN3. The upper section of Table 3 confirms that a significant decrease in the advanced- to early-stage squamous carcinoma IRR was observed only in the transition phase (1999–2006), when the rate of advanced-stage lesions decreased more rapidly, and the ratio to pre-screening rate was 0.66 (95% CI, 0.45–0.98). In the steady-state phase, the rate of early-stage squamous carcinoma decreased as well and, consequently, the IRR was no longer significantly different from that of the pre-screening years.
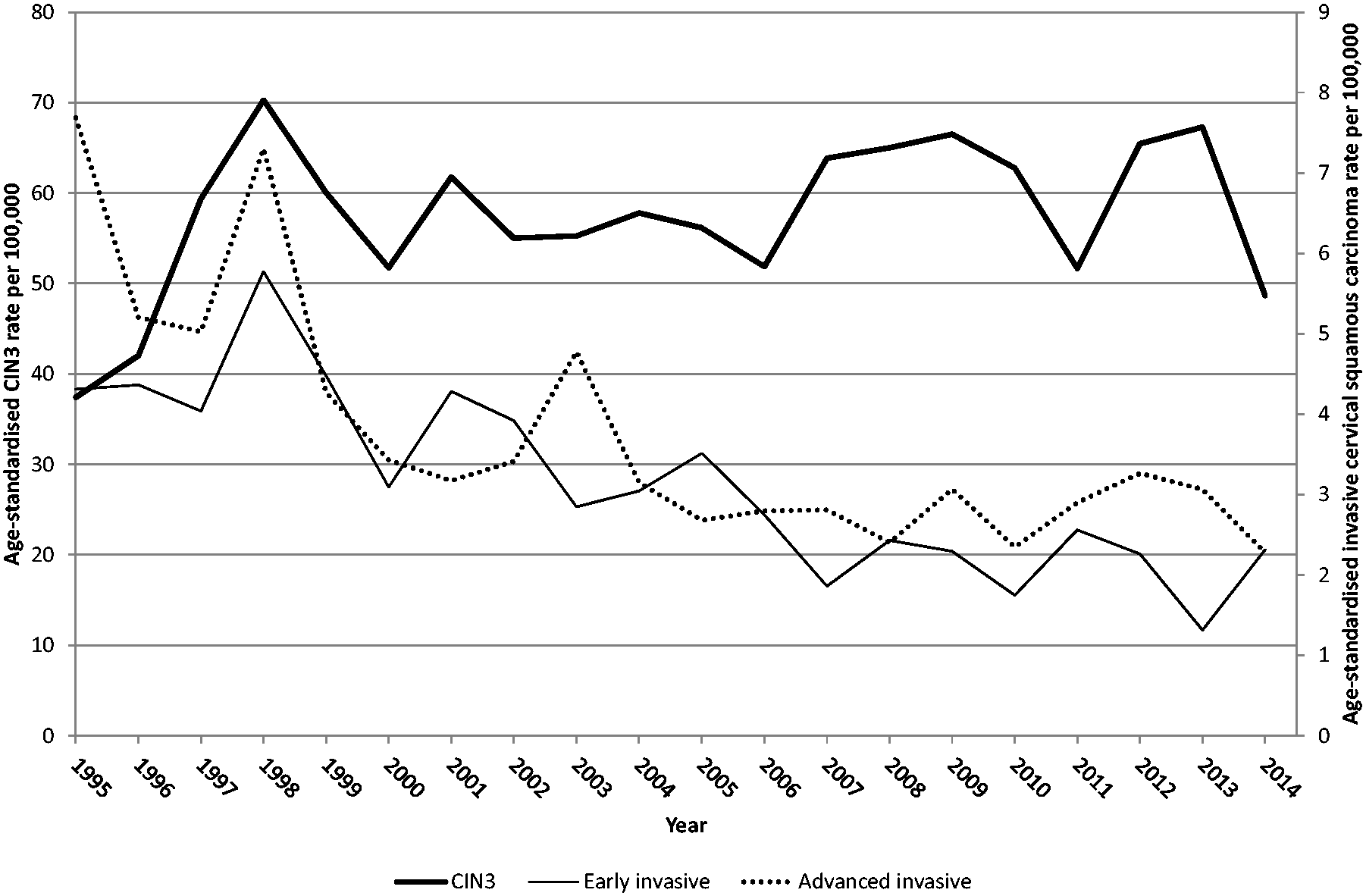
Curve of observed annual incidence rates of cervical intraepithelial neoplasia grade 3 (CIN3), early-stage cervical squamous carcinoma, and advanced-stage cervical squamous carcinoma per 100,000 women aged 25–64 in 1995–2014. CIN3 includes CIN2–3, CIN3, and squamous carcinoma in situ; early stage includes pT1a, pT1a1, and pT1a2; advanced stage includes pT1b or greater, and ypT. All rates were age-standardized (2013 European standard population). Emilia-Romagna Region, Italy.
Invasive to preinvasive squamous and glandular tumour incidence rate ratio, by time period, and ratio of ratios comparing 1997–1998, 1999–2006, and 2007–2014 with the pre-screening years (1995–1996). Emilia-Romagna Region cancer registries and health care district cervical cancer screening programmes, Italy, 1995–2014.
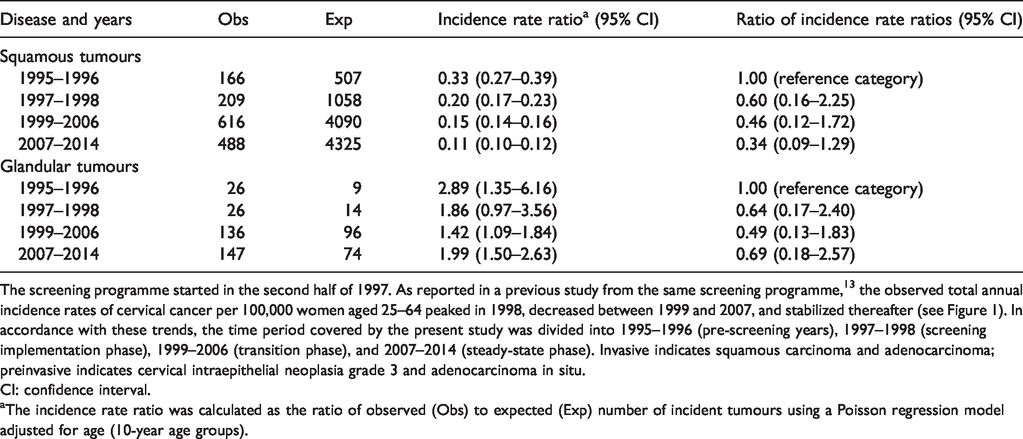
The screening programme started in the second half of 1997. As reported in a previous study from the same screening programme, 13 the observed total annual incidence rates of cervical cancer per 100,000 women aged 25–64 peaked in 1998, decreased between 1999 and 2007, and stabilized thereafter (see Figure 1). In accordance with these trends, the time period covered by the present study was divided into 1995–1996 (pre-screening years), 1997–1998 (screening implementation phase), 1999–2006 (transition phase), and 2007–2014 (steady-state phase). Invasive indicates squamous carcinoma and adenocarcinoma; preinvasive indicates cervical intraepithelial neoplasia grade 3 and adenocarcinoma in situ.
CI: confidence interval.
aThe incidence rate ratio was calculated as the ratio of observed (Obs) to expected (Exp) number of incident tumours using a Poisson regression model adjusted for age (10-year age groups).
Advanced- to early-stage squamous carcinoma and adenocarcinoma incidence rate ratio, by time period, and ratio of ratios comparing 1997–1998, 1999–2006, and 2007–2014 with the pre-screening years (1995–1996). Emilia-Romagna Region cancer registries and health care district cervical cancer screening programmes, Italy, 1995–2014.
The screening programme started in the second half of 1997. As reported in a previous study from the same screening programme, 13 the observed total annual incidence rates of cervical cancer per 100,000 women aged 25–64 years peaked in 1998, decreased between 1999 and 2007, and stabilized thereafter (see Figure 1). In accordance with these trends, the time period covered by the present study was divided into 1995–1996 (pre-screening years), 1997–1998 (screening implementation phase), 1999–2006 (transition phase), and 2007–2014 (steady-state phase). Early stage includes pT1a, pT1a1, and pT1a2; advanced stage includes pT1b or greater, and ypT.
CI: confidence interval.
aThe incidence rate ratio was calculated as the ratio of observed (Obs) to expected (Exp) number of incident tumours using a Poisson regression model adjusted for age (10-year age groups).
Figure 3 shows the curves of annual incidence rates of glandular tumours. The IRRs are presented in the bottom section of Table 1. After the implementation phase, the incidence of AIS and of the rare early-stage adenocarcinoma rose irregularly and non-significantly. For advanced-stage adenocarcinoma, the incidence changes were generally modest, and totally absent in the steady-state phase. These trends must be viewed taking into account the fact that the introduction of the screening programme was accompanied by a marked drop in the rate of unknown-stage adenocarcinoma. The bottom section of Table 2 shows that the invasive to preinvasive glandular tumour IRR decreased non-significantly after the introduction of the screening programme. The bottom section of Table 3 shows that the advanced- to early-stage adenocarcinoma IRR also decreased permanently after the introduction of the screening programme, but not significantly.
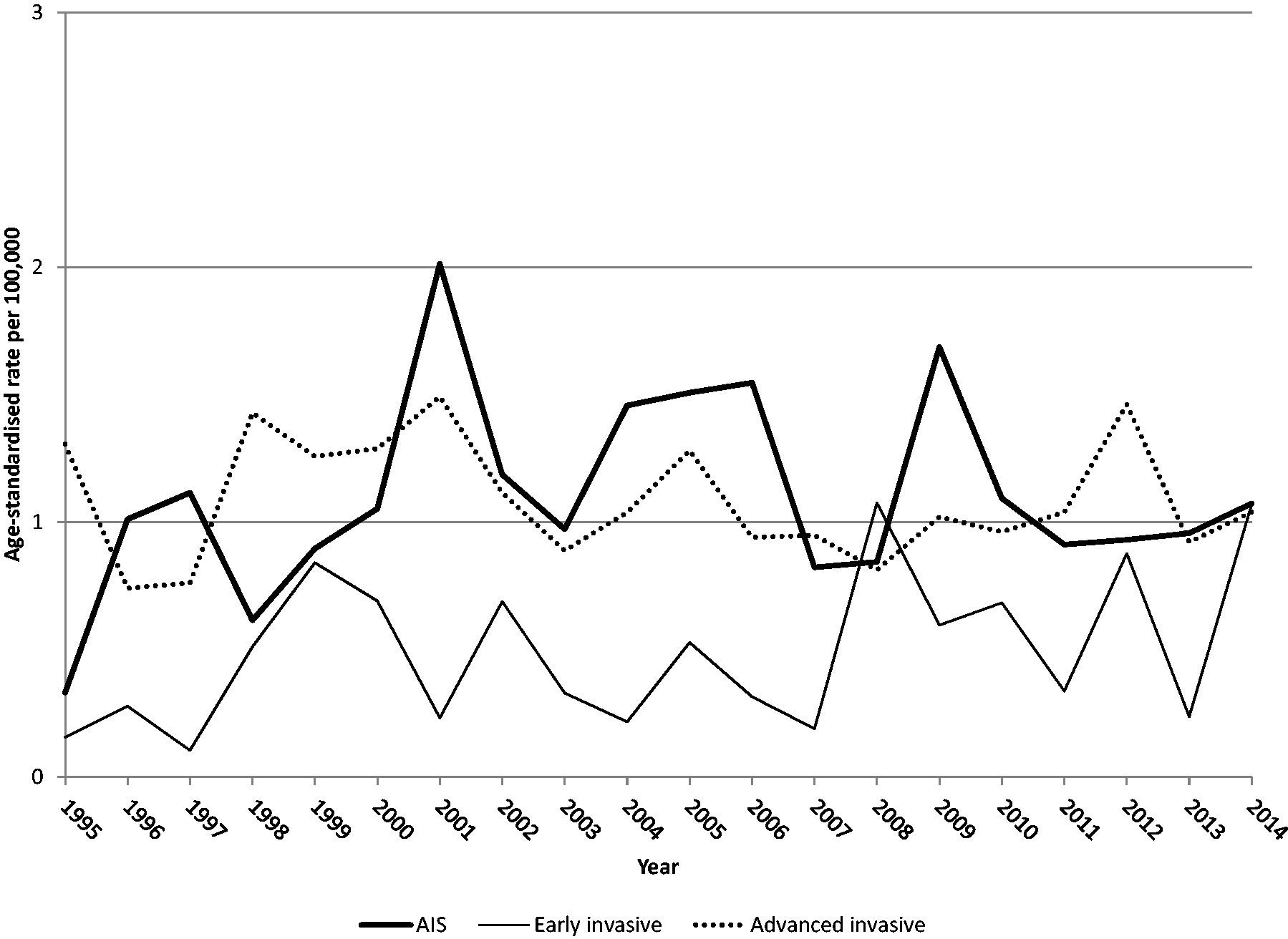
Curve of observed annual incidence rates of cervical adenocarcinoma in situ (AIS), early-stage cervical adenocarcinoma, and advanced-stage cervical adenocarcinoma per 100,000 women aged 25–64 in 1995–2014. Early stage includes pT1a, pT1a1, and pT1a2; advanced stage includes pT1b or greater, and ypT. All rates were age-standardized (2013 European standard population). Emilia-Romagna Region, Italy.
After the implementation phase, as shown in Figure 4, the rate of screen-detected CIN3 appeared to be roughly comparable with the pre-screening rate. With the introduction of the screening programme, the component of incidence generated by the opportunistic screening practice decreased rapidly, with an IRR of 0.35 (95% CI, 0.31–0.39) in the steady-state phase. This was expected. The change in the incidence of squamous carcinoma diagnosed on an opportunistic basis was of comparable magnitude (IRR ratio, 0.27; 95% CI, 0.22–0.33). Another important fact that can be drawn from Figure 4 is that the fluctuations of total incidence of CIN3 seen in Figure 2 depended, for the greater part, on the instability of the rate of screen-detected CIN3. In the steady-state phase, there was also a significant decrease in the incidence of glandular tumours diagnosed in the opportunistic setting, with an IRR of 0.28 (95% CI, 0.12–0.64) for AIS and 0.50 (95% CI, 0.32–0.78) for invasive adenocarcinoma (data not shown).
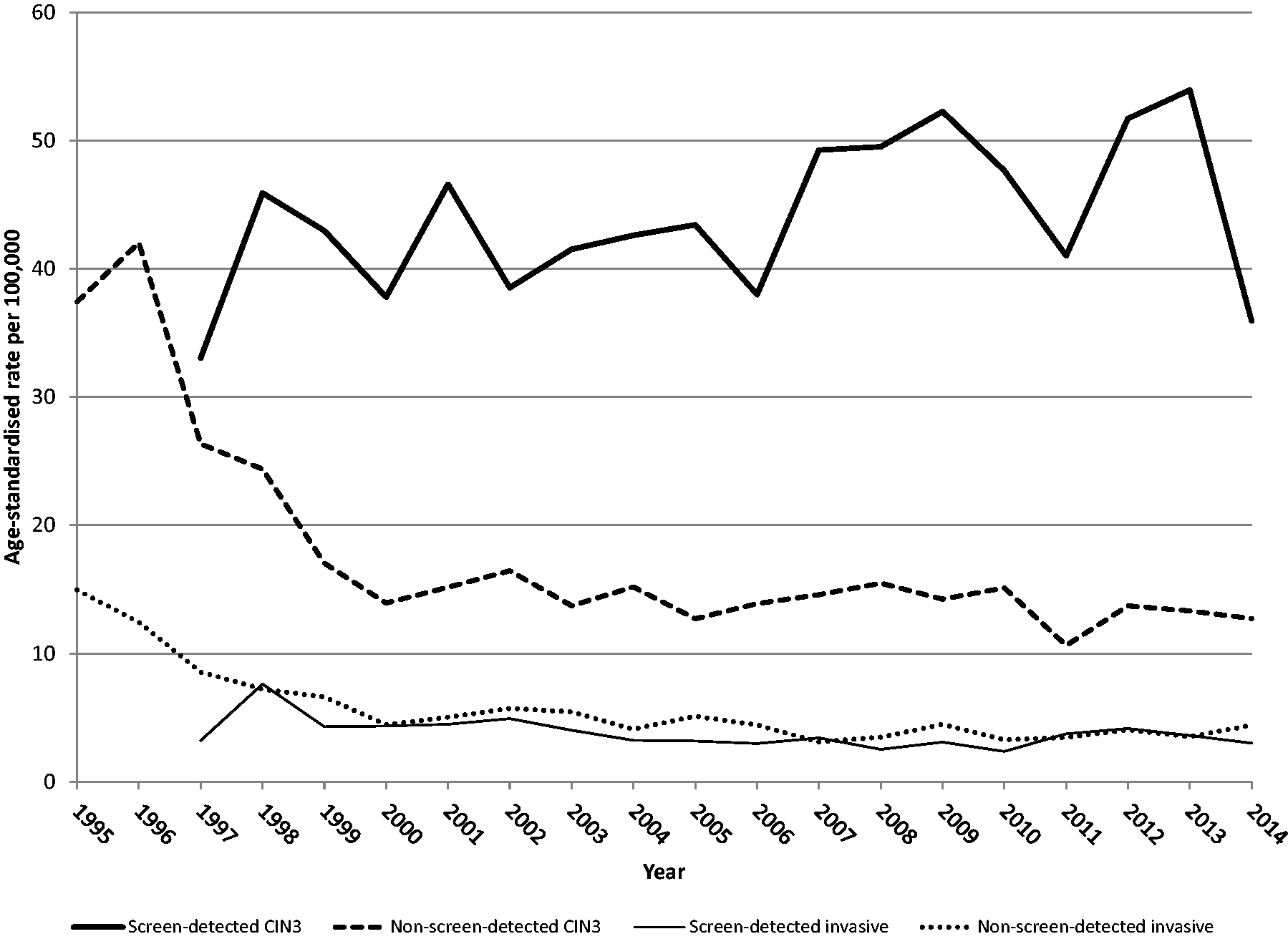
Curve of observed annual incidence rates of screen-detected and non-screen-detected cervical intraepithelial neoplasia grade 3 (CIN3), and screen-detected and non-screen-detected cervical squamous carcinoma per 100,000 women aged 25–64 in 1995–2014. CIN3 includes CIN2-3, CIN3, and squamous carcinoma in situ. All rates were age-standardized (2013 European standard population). Emilia-Romagna Region, Italy.
Discussion
There are four main results of this study. First, the increase in total incidence of CIN3 was limited to 55% due to the fact that cases detected outside the screening programme dropped by two-thirds. This finding is in line with Italian national data.21,24 The fluctuations of the rate were mainly due to the instability of the incidence of screen-detected CIN3, which suggests that the volume of invitations and/or the uptake were not constant. However, this instability is expected to attenuate with time. Second, we found that the rates of early- and advanced-stage squamous carcinoma decreased to the same extent. In the transition phase, however, the rate of pT1b+ carcinoma decreased earlier than that of pT1a carcinoma, because the average lead time of a prevented case of pT1b+ carcinoma is shorter. Unfortunately, the significant decrease in the advanced- to early-stage squamous carcinoma IRR observed in the transition phase was a temporary phenomenon. The third significant finding of the study is that the incidence decrease in the steady-state phase was so uniform that the incidence ratio between advanced- and early-stage squamous carcinoma was left unchanged. Our working hypothesis that the reduction in total incidence of cervical carcinoma could be associated with a downstaging effect was not confirmed. The only kind of downstaging seen in our data was the expected increase in the incidence of CIN3 coupled with the decrease of invasive squamous carcinoma. In the steady-state phase, the rate of invasive squamous carcinoma was almost ten times lower than the rate of CIN3. The fourth interesting finding is that the stability of incidence of adenocarcinoma, already analysed in our previous study, 13 involved all stages of disease. The incidence of AIS and early-stage adenocarcinoma increased non-significantly, and the rate of advanced-stage adenocarcinoma was perfectly stable. The inability of cervical cytology to prevent glandular lesions suggests that this subset of diseases is an area where the benefit of moving to HPV-based screening may become more rapidly apparent. The detection of HPV 18 and 45 infections allows earlier detection of glandular lesions 25 both through earlier referral for colposcopy and, if colposcopic assessment is negative, through more targeted surveillance. The impact on glandular lesions needs to be specifically investigated when assessing the effectiveness of HPV-based screening.
We are aware of only one previous study with a design closely resembling ours. After the reorganization of screening activities in South West Hampshire (United Kingdom) at the end of the 1980s, Herbert et al. observed a declining incidence of screen-detected T1b+ carcinoma (our own definition of advanced-stage carcinoma), but failed to demonstrate a decrease in the rates of earlier invasive tumours. 26
Several important methodological issues of our study need to be addressed. First, we based the design on the previous observation that changes in total incidence of cervical carcinoma in the population targeted by the screening programme followed a clear-cut temporal pattern, with three distinct phases. 13 We have already commented on this sequence. The transition phase, or the range of time over which the incidence of cervical carcinoma decreases after the initial peak, is an approximate measure of the lead time of cases averted by the detection and treatment of CIN. Incidentally, the stabilization of total incidence of cervical carcinoma in the steady-state phase confirms that cytology-based screening has reached the limit of its effectiveness, thus providing further justification for the conversion into HPV testing. Secondly, evaluating the impact of a screening programme on the whole invited population conforms to the intention-to-screen principle. This approach underestimates the magnitude of the effect on participants, but represents a pragmatic test of whether the programme is effective when established in the real world, in particular at the level of participation observed. In 2014, despite a constant increase, the three-year coverage for the organized screening programme was still slightly below the level of 70% considered acceptable by the European guidelines. 6 It must be noted, however, that total screening coverage obtained combining organized and opportunistic Pap smears reached the desirable level of 90%. 6 Thirdly, our definition of early and advanced tumour stage was based on literature data 26 and prognostic and therapeutic considerations. 27 Both pT1a1 and pT1a2 carcinoma are potentially treatable by conization, although patient age and parity are factors in clinical decision-making for the subset of pT1a2 tumours, and lymphadenectomy is recommended for all patients with pT1a2 carcinoma and for those with pT1a1 carcinoma and lymphovascular invasion. The final methodological issue is that the small number of pre-screening years of registration of tumour stage data did not allow us to estimate the expected stage-specific rates with an age-period-cohort modelling analysis. We used pre-screening rates as a reference, assuming that they would have been stable in the absence of screening. However, the curve of expected total incidence rates, previously estimated with robust methods, was flat, 13 and there is no obvious reason to believe that stage-specific incidence rates would have followed a different trend.
A temporal correlation like the one seen in our data (including our previous data) 13 is an ecological correlation, that cannot formally demonstrate a causal relationship between two events. 28 Considering this, our study has two key strengths. One is the timing of introduction of the screening programme, which was rapid and simultaneous across the whole target population. This supports a causal link to subsequent incidence changes,13,29,30 which began soon after, and clarifies the temporal correlation. The other strength is that only an increase in the rate of CIN3 due to Pap smear screening can be associated with a decreasing trend of invasive squamous carcinoma and a stable trend of both AIS and invasive adenocarcinoma. No natural change in exposure to risk factors could have such different effects.
Conclusions
This study provides further details of the events underlying the previously reported 40% decrease in total incidence of cervical carcinoma, due to the introduction of an organized screening programme, in comparison with the years when only opportunistic testing was available. In the steady-state phase, the rate of CIN3 increased by an average 55% in a stable manner. Early- and advanced-stage squamous carcinoma decreased almost equally by 50–55%. There were non-significant increases for AIS and early-stage adenocarcinoma, and a perfect stability for advanced-stage adenocarcinoma. Given that primary HPV screening is being implemented in different ways, and starting from different baselines in different countries, each country must devise its own monitoring matrix using robust data sources. Both our present and previous studies show that, using simple data readily available from cancer registries, it is possible to demonstrate the effectiveness of a cervical screening programme. Multiple incidence studies from different populations will allow meta-analyses to document the benefit of HPV-based screening over cytology-based screening, and the benefit of HPV vaccination when added to these secondary prevention measures.
Emilia-Romagna Region Workgroup for Cervical Screening Evaluation
The membership of the Emilia-Romagna Region Workgroup for Cervical Screening Evaluation is as follows: Nicoletta Bertozzi, Enza Di Felice, Alba Carola Finarelli, Carlo Naldoni, Priscilla Sassoli de Bianchi (Department of Health, Emilia-Romagna Region, Bologna, Italy); Flavia Baldacchini, Lauro Bucchi, Orietta Giuliani, Silvia Mancini, Alessandra Ravaioli, Rosa Vattiato (Romagna Cancer Institute, IRST, IRCCS, Meldola, Forlì, Italy); Fabio Falcini (Local Health Authority, Forlì, Italy, and Romagna Cancer Institute, IRST, IRCCS, Meldola, Forlì, Italy); Elisabetta Borciani, Pier Giorgio Dataro, Giorgio Gatti, Alessandro Lauri (Local Health Authority, Piacenza, Italy); Luigi Lombardozzi (Local Health Authority, Parma, Italy); Paolo Giorgi Rossi (Epidemiology Unit, Azienda Unità Sanitaria Locale – IRCCS di Reggio Emilia, 42122 Reggio Emilia, Italy); Cinzia Campari, Luisa Paterlini (Cancer Screening Unit, Azienda Unità Sanitaria Locale – IRCCS di Reggio Emilia, 42122 Reggio Emilia, Italy); Teresa Rubino (Cervical/Vaginal Cytology Unit, Azienda Unità Sanitaria Locale – IRCCS di Reggio Emilia, 42122 Reggio Emilia, Italy); Raffaella Andresini, Giuliano Carrozzi, Rossella Corradini, Carlo Alberto Goldoni, Agar Venturelli, Maria Dirce Vezzani (Local Health Authority, Modena, Italy); Carmen Bazzani, Patrizia Biavati, Paolo Cristiani, Marilena Manfredi, Francesca Mezzetti, Adriana Pasquini (Local Health Authority, Bologna, Italy); Licia Caprara, Margherita De Lillo, Roberto Nannini (Local Health Authority, Imola, Italy); Maria Cristina Carpanelli, Aldo De Togni, Paola Garutti, Caterina Palmonari (Local Health Authority, Ferrara, Italy); Stefano Ferretti (University of Ferrara and Local Health Authority, Ferrara, Italy); Nori Morini, Patrizia Schincaglia, Monica Serafini, Benedetta Vitali (Local Health Authority, Ravenna, Italy); Antonella Brunelli, Massimo Farneti, Claudia Imolesi, Mauro Palazzi (Local Health Authority, Cesena, Italy); Debora Canuti, Franco Desiderio, Giancarla Monticelli (Local Health Authority, Rimini, Italy).
Footnotes
Acknowledgements
The authors thank the referees of this article for their valuable comments, and Grainne Tierney (Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori, IRST, IRCCS, Meldola, Forlì, Italy) for the linguistic assistance in the preparation of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
