Abstract
Objective
To test whether reduced-frequency risk-stratified breast screening would be perceived more favourably by transposing the order of information on benefits and risks.
Methods
After reading vignettes describing non-stratified three-yearly screening and a risk-stratified alternative with five-yearly invitations for women at low risk, 698 women completed an online survey. Participants were allocated at random to information on screening benefits followed by risks, or vice versa, and asked to state preferences for either screening system. Participants also rated perceived magnitude of screening benefits and risks, and breast cancer susceptibility.
Results
Binomial logistic regression did not find order effects on preferences (p = 0.533) or perceived benefits of screening (p = 0.780). Perceived screening risks were greater when risks were presented first (p < 0.0005). Greater perceived susceptibility was associated with lower proportions preferring risk-stratified screening (15% vs. 39% in highest and lowest groups; p = 0.002), as were greater perceived screening benefits (e.g. 13% vs. 45% in highest and lowest groups; p < 0.0005).
Conclusions
No information order effect on preferences was observed. Information order did affect screening risk perceptions. Efforts to improve perceptions may need to be more intensive than those tested. Women perceiving themselves as high risk or perceiving greater benefits of screening may be particularly averse to less frequent screening.
Introduction
Risk-stratified breast screening, tailoring screening interventions according to individuals’ risk status (e.g. based on genetic factors), 1 has been mooted to improve existing programmes. Women at low risk could be screened less intensively, minimizing opportunity costs and harms due to test inconvenience and overdiagnosis, but acceptability to the target population is necessary to ensure high levels of uptake. 2 Research shows that prospective participants may have preconceptions that more intensive screening is superior to less intensive screening. In particular, women are relatively accepting of those at high risk being invited for mammography more frequently than the current triennial default in England, but less willing to accept longer intervals for those at lower risk. 3
A potentially important determinant of perceived acceptability of screening programmes is the written information provided to invitees before participation, 4 which aims to convey knowledge about screening benefits and risks, and enable informed choice about participation. 5 Written information is generally the main or only way that invitees learn about screening. Written information is generally the main or only way that invitees learn about screening in organised programmes.6,7 However, there may be opportunities to increase perceived acceptability of reduced-frequency risk-stratified breast screening, using subtle methods of designing information that still respect these requirements.
Previous research has found that preferences and perceptions can be influenced using a wide range of psychological methods, and in diverse healthcare contexts and clinical groups. 8 Authors of screening information materials necessarily decide whether to summarize benefits of screening before risks, or vice versa, 6 and this order may have psychological effects. 8 A study of genetic testing for breast cancer risk found that participants had more positive attitudes and intentions (but greater risk perceptions and fewer disadvantages) when advantages were presented before disadvantages. 9 We hypothesized that similar order effects (so-called “primacy” and “recency” effects) may also affect the acceptability of longer screening intervals for women at low risk. For example, if women are more inclined to have genetic testing for breast cancer when advantages are presented first, this order may result in a larger proportion favouring risk-stratified breast screening, in which genetic testing is part of the process.
In a pilot survey testing four manipulations (in addition to the information order manipulation, these were a reminder that participants would be personally invited for screening vs. no reminder, information about the current triennial screening interval vs. no information on the interval and social norm information on the proportion of women who accept an invitation to the Breast Screening Programme in England vs. no information), the provisional effect size estimate was largest for the information order manipulation. In a sample of 205 participants, 36% stated a preference for risk-stratified screening with longer intervals when allocated at random to read information on screening benefits before those of risks vs. 19% with this preference who read screening risks first (Χ(1) = 7.25; p = 0.007). We conducted a larger experimental survey as a confirmatory test of this order effect. Secondary aims were to test (i) whether information order affected perceptions of screening risks and benefits and (ii) associations between preferences and other factors (e.g. perceived susceptibility to breast cancer).
Methods
We recruited approximately 700 women aged 40–70 and not previously diagnosed with breast cancer to an online survey (Appendix 1 (available as online Supplemental materials)). Participants read a vignette outlining the current Breast Screening Programme in England, and a risk-stratified alternative: screening every three years for women at average genetic risk of breast cancer and every five years for women at low genetic risk. No specific information was provided on a screening strategy for women at high genetic risk. To test for an order effect, the vignette ended with information on screening benefits followed by information on screening risks or vice versa (i.e. two groups, in which the order was determined at random). To minimize cognitive load for participants and maintain their attention, information on screening risks and benefits was brief, and no quantitative estimates of magnitude were provided.
Participants were asked about their preference for either “breast cancer screening offered every 3 years to all women aged 50 to 70 years” or “breast cancer screening offered every 3 years to women aged 50 to 70 years if they are at average genetic risk and breast cancer screening offered every 5 years to women aged 50 to 70 years if they are at low genetic risk”. After reading the information, participants were asked a multiple choice question testing their comprehension of part of it (the proportion of breast cancers diagnosed in women aged over 50). If they did not respond to this correctly, they were asked the question again (with the option to return to the previous page) until they gave the correct answer, after which they were asked the next question in the survey. Perceived benefits and risks of screening were assessed using a scale with response options from 0 to 100, denoting how positively (for benefits) or negatively (for risks) participants rated these. We hypothesized that if preferences were related to the order of risk and benefit information, the magnitude of perceived risks and benefits would also differ between manipulations. The main analysis used binomial logistic regression to test whether women’s stated preference for status quo screening was associated with the experimental manipulation, after controlling for covariates (e.g. perceived susceptibility to breast cancer, perceived benefits and risks (as quintiles) and age). Perceived benefits and risks of screening were compared between the two levels of the experimental manipulation using Mann-Whitney U tests.
Institutional ethical approval was obtained (reference: 2951/005).
Results
Of 733 eligible participants, 698 completed the survey (five provided inconsistent data on the comprehension question and were excluded). Mean age was 53.4 years (standard deviation: 7.9), 285 (41%) were aged 40–46 (currently ineligible for breast cancer screening) and 97% spoke English as a first language. The majority stated a preference for status quo screening in both benefits-first and risks-first manipulations (78% vs. 74%; 76% overall). Results of binomial logistic regression (n = 587) did not show an effect of the experimental manipulation on preferences, although three covariates were associated: those rating themselves at higher perceived susceptibility to breast cancer, those who answered the comprehension question incorrectly on the first attempt and those with higher scores of perceived benefits were more likely to prefer status quo screening (Table 1).
Results of the binomial logistic regression model testing variables associated with preferences for either status quo or risk-stratified screening.
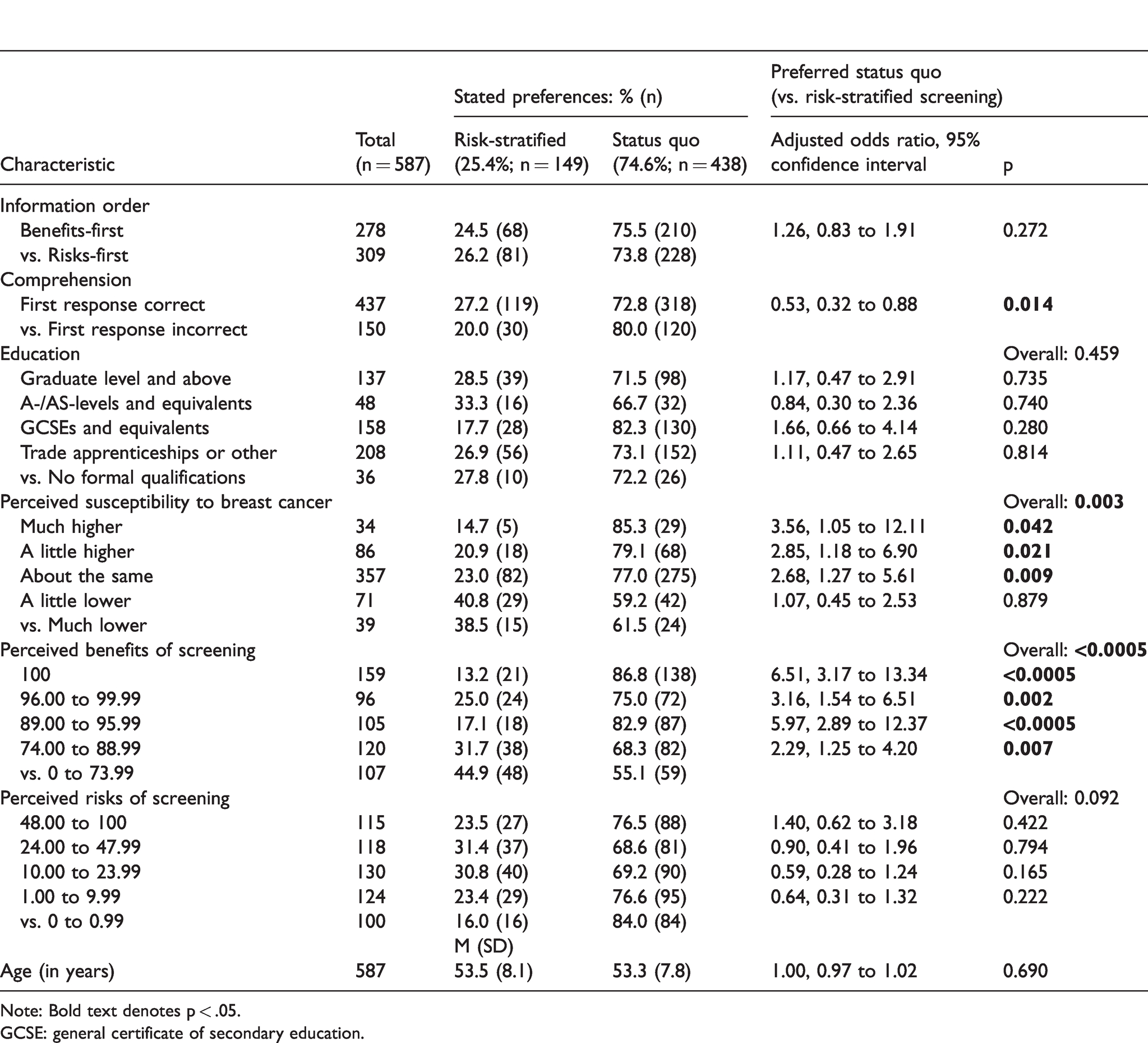
Note: Bold text denotes p < .05.GCSE: general certificate of secondary education.
Mann-Whitney U Tests for differences between experimental conditions in perceived screening benefits found only weak evidence against the null hypothesis when analysing (i) all participants (medians and interquartile ranges: 93.0, 20 vs. 92.0, 21 for benefits-first vs. risks-first; U = 60,036, z = –0.280, p = 0.780) and (ii) only participants who answered the comprehension question correctly on the first attempt (94.0, 20 vs. 93.0, 19; U = 34,921, z = 0.018, p = 0.986). However, there was strong evidence against the null hypothesis for comparisons of perceived screening risks. Median scores were lower for participants who read benefits-first vs. risks-first among all participants (12.0, 28 vs. 22.0, 40, U = 72,043, z = 4.25, p < 0.0005) and in the sensitivity analysis (12.0, 26 vs. 20.0, 39; U = 40,354, z = 3.12, p = 0.002).
Discussion
This study did not find an effect of information order on preferences; however, there were several key findings of relevance to future implementation of risk-stratified breast screening. First, we found an order effect for perceived risks of screening, with risks rated lower when presented after benefits. This supports the current design of the NHS breast screening leaflet, 4 which summarizes risks and benefits in this order.
Second, a notable majority (76%) of this large sample of women stated a preference for status quo screening over risk-stratified screening. Although the sample was not selected to be fully representative of the general population, these results reinforce similar findings from previous studies3,10,11 and suggest that transitioning to risk-stratified breast screening would be met with resistance if some women are invited less frequently. A recent quantitative survey found that only 57% of women surveyed in five European countries stated that they would consider less cancer screening if they were at lower than average genetic risk, 12 and qualitative research from Australia found that women feel a strong emotional connection to current methods of breast screening. 13 A relevant caveat to this finding is that a greater number of screening risks were presented compared with benefits. This is consistent with the existing NHS screening information leaflet, 4 but may have increased preferences for reduced-frequency (i.e. risk-minimizing) breast screening, in accordance with a processing difficulty effect. 14 However, participants’ ratings for the one benefit stated (saving lives from breast cancer) were clustered near the maximum allowed by the scale, whereas ratings of perceived screening risks were distributed more widely, suggesting that the nature of risks and benefits was more important than the number.
Third, perceived benefits of screening were positively associated with favouring status quo screening, consistent with this being a key determinant of preferences.
Fourth, greater perceived susceptibility to breast cancer was associated with preferences for status quo screening. This finding may explain results of previous studies showing that women who have previously experienced a (false) positive screening result may be more resistant to reduced screening 15 (i.e. they may perceive themselves to be more susceptible). These findings are also broadly consistent with the PROCAS (Predicting Risk of Breast Cancer Screening) project, where the overwhelming majority of women who were informed that they were at high risk of breast cancer took up their next mammography invitation (99%). 1 However, an appreciable proportion of women continue to overestimate their risk after feedback, 16 and may require specific reassurance if they are offered less frequent screening.
Finally, null findings for the manipulation tested suggest that preferences for status quo screening may not be easily changed using subtle information modifications. However, a limitation of this study was that after excluding three alternatives that were ineffective in piloting, only one manipulation was tested. Other manipulations may have had more success. For example, there is some evidence that simple manipulations such as loss-framing (e.g. “not going for screening means you miss an opportunity to avoid dying from breast cancer”) increases perceptions of screening effectiveness vs. gain-framing (e.g. “going for screening means you have an opportunity to avoid dying from breast cancer”), 17 and that personalized risk feedback increases knowledge of screening and may increase uptake. 18 Further relevant psychological heuristics and biases are described elsewhere. 8
Another limitation was that information was not part of a full screening invitation. Responses may have differed if additional information had been provided, outside of a hypothetical context. In particular, we assumed that participants would infer the rationale for why screening intervals might be longer for women at low genetic risk than those at average genetic risk (i.e. because screening invitations were stated to be based on risk), but this was not stated explicitly and may have reduced the perceived acceptability of risk-stratified screening. In addition, the information referred to the possibility of overdiagnosis, but not the corollary risk of overtreatment, which may have led participants to rate screening risks less negatively and increased the proportion stating a preference for status quo screening. We also did not provide quantitative estimates of the magnitude of screening risks (e.g. chances of overdiagnosis) or benefits (i.e. chances of preventing breast cancer death), as the primary aim of the study was to test for the presence of an effect of information order on preferences, not to estimate population perceptions of acceptability, specifically. This may have affected participants’ evaluation of risks and benefits. However, a trial assessing knowledge of overdiagnosis in breast cancer screening reported far higher mean scores for knowledge of the concept than associated statistics, suggesting invitees focus less on numerical information. 19 Finally, we did not assess preferences for other forms of risk-stratified screening, e.g. more frequent screening for women at higher risk. This could be tested in future studies.
Conclusion
We found little evidence that preferences for risk-stratified screening could be increased by amending the order of risk and benefit information; however, perceived risks of screening were lower when screening risk information was read after screening benefits. As preferences were associated with perceived screening benefits and susceptibility to breast cancer, women at low objective risk but high perceived susceptibility may be particularly concerned if offered less frequent screening. Efforts to increase perceived acceptability may need to be more intensive than those tested.
Footnotes
Authors’ note
Sandro T Stoffel is also affiliated with Business School, University of Aberdeen, Aberdeen, UK.
Author contribution
AG, JW, STS, IV, and CVW conceived and designed the study. AG analysed the data. AG, JW, STS, IV, and CVW participated in the interpretation of results. AG, JW, STS, IV, and CVW drafted the manuscript, participated in critical revision, and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a programme grant from Cancer Research UK awarded to Jane Wardle (C1418/A14134). Jo Waller is supported by a Career Development Fellowship from Cancer Research UK (C7492/A17219). Cancer Research UK was not involved in the design of this study; the collection, analysis, or interpretation of the results; in the writing of the article; or in the decision to submit for publication.
Supplemental material
Supplemental material is available for this article online.
