Abstract
Objective
Cervical cancer mortality has halved in Australia since the national cervical screening program began in 1991, but elevated mortality rates persist for Aboriginal and Torres Strait Islander women (referred to as Aboriginal women in this report). We investigated differences by Aboriginal status in abnormality rates predicted by cervical cytology and confirmed by histological diagnoses among screened women.
Methods
Using record linkage between cervical screening registry and public hospital records in South Australia, we obtained Aboriginal status of women aged 20–69 for 1993–2016 (this was not recorded by the registry). Differences in cytological abnormalities were investigated by Aboriginal status, using relative risk ratios from mixed effect multinomial logistic regression modelling. Odds ratios were calculated for histological high grade results for Aboriginal compared with non-Aboriginal women.
Results
Of 1,676,141 linkable cytology tests, 5.8% were abnormal. Abnormal results were more common for women who were younger, never married, and living in a major city or socioeconomically disadvantaged area. After adjusting for these factors and numbers of screening episodes, the relative risk of a low grade cytological abnormality compared with a normal test was 14% (95% confidence interval 5–24%) higher, and the relative risk of a high grade cytological abnormality was 61% (95% confidence interval 44–79%) higher, for Aboriginal women. The adjusted odds ratio of a histological high grade was 76% (95% confidence interval 46–113%) higher.
Conclusions
Ensuring that screen-detected abnormalities are followed up in a timely way by culturally acceptable services is important for reducing differences in cervical cancer rates between Aboriginal and non-Aboriginal women.
Introduction
Since the Australian National Cervical Screening Program started in 1991, cervical cancer rates have halved.1–3 Until December 2017, when human papillomavirus (HPV)-based primary screening commenced, the Program targeted women aged 20–69 using biennial cytology screening. 4 Aboriginal and Torres Strait Islander women (analysed collectively as “Aboriginal women” due to small numbers) have a two-fold higher incidence of cervical cancer than non-Aboriginal women and a four-fold higher cervical cancer mortality rate.1,5,6 National data on screening outcomes are lacking by Aboriginal status, because data sources (laboratory cytology and histology reports) do not record Aboriginal status. 7
Aboriginal women have been found to be under-screened in the Northern Territory 8 and, using linked hospital records, in Queensland. 9 The linked Queensland data found that the odds for Aboriginal compared with other women were 40% higher for low grade, and 120% higher for high grade cytological abnormalities, and 100% higher for high grade histological abnormalities. 10 While Queensland data indicated similar rates of follow-up investigation by Aboriginal status within six months of a histologically confirmed high grade abnormality, a lower proportion of Aboriginal women had a follow-up clinical investigation within the two months specified in national guidelines.10,11
During 2013–2014 (24 months), the age-standardised biennial cervical screening participation among 20–69-year-olds was similar for South Australia and nationally (59% vs. 57%), 4 but screening participation by Aboriginal status was not known. While Aboriginal people comprise only 2% of the South Australian population, they are a priority for cervical screening due to higher cervical cancer rates and cultural needs.1,5,6 Culturally appropriate programs for increasing screening participation have been implemented, 12 but effects on screening participation among Aboriginal women have not been quantified. Without private hospital data, population-based screening participation cannot be determined. This study used public hospital data to investigate differences by Aboriginal status in abnormality rates predicted by cervical cytology and confirmed by histological diagnoses among screened women attending these hospitals.
Methods
This study used two data sources, the South Australian Cervix Screening Registry, and the South Australian inpatient statistics collection. The Registry included results of all cervical cytology screens and cervical histology, reported under legal mandate by laboratories. Women could “opt out” of the Registry, but <1% take this course, as reported for Victoria. 13 The inpatient statistics applied to all hospitals, public and private, and covered all items specified in the National Health Data Dictionary, 14 but names of hospital patients were collected for public hospitals only (60% of admissions). Hospital data were used to ascertain Aboriginal status and marital status, because this was not available through the Registry. Screening information on test dates and results was only available through the Registry. Both sources recorded place of residence and age, but Registry data were preferred as they were better aligned with dates of screening tests.
The Registry recorded 1,676,141 cytology screening tests for women aged 20–69, which were linked to public hospital inpatient data for 1993–2016 (50% of tests). Tests not linked were for women who had never attended a public hospital, including those who only attended private hospitals, and non-hospital attenders. Tests included in this study were for a large cohort of screened women who had both attended a public hospital and had a cervical screening episode recorded on the Registry during 1993–2016.
Unique linkage keys were developed and used by the State Linkage Unit. Deterministic and probabilistic linkage were used, supplemented by clerical checks using name, sex, birth date, and street address for linking. The standard process of separation of identified and health data was followed to protect privacy. 15 The quality of the linkage was monitored closely and detailed in the Linkage Unit “hand-over” report. Data items included laboratory test date, birth date, marital status, residential Statistical Local Area, number of screening tests, and test results. Women were assumed to be Aboriginal if so recorded at any hospital admission. 7
Approval for the study was obtained from the South Australian Aboriginal Health Research Ethics Committee [04/14/580], the Human Research Ethics Committee of the South Australian Department of Health [HREC/14/SAH/118], the University of South Australia Human Research Ethics Committee [200454], and the Human Research Ethics Committee of Northern Territory Department of Health and Menzies School of Health Research [HOMER-2012-1737], with a waiver of consent.
Cytology and histology results were classified as normal or low or high grade, using Australian Institute of Health and Welfare criteria (Table 1). 4 Age at screening was categorised as 20–29, 30–39, 40–49, 50–59, or 60–69. Aboriginal status was summarised as a binary variable, there being insufficient numbers to analyse Torres Strait Islander women separately. Marital status was recorded as never married, married/de facto, or divorced/separated/widowed at first admission. Where multiple records existed, the most common and most proximal recording to the screening test were used in sensitivity analyses. Residential remoteness was classified as major city, inner regional, outer regional, remote, or very remote, using the 2006 Remoteness Area codes employed in the Australian Standard Geographical Classification. 16 Socioeconomic status was inferred from the Socio-Economic Indexes for Areas (SEIFA) Index of Relative Socioeconomic Disadvantage, and 2011 Statistical Local Area coding, and grouped by socioeconomic quartile (an ordinal scale), with the first quartile referring to the most disadvantaged areas. 17 Screening periods were categorised as 1993–1997, 1998–2002, 2003–2007, 2008–2012, and 2013–2016. Numbers of screening episodes were defined as 1, 2, 3, 4, or ≥5 for each participant for the 1993–2016 study period.
Cervical cytology classification code; Australian Institute of Health and Welfare.4

Cytology and histology test results were compared by Aboriginal status, as repeated tests (level 1) at different time points nested within participants (level 2). For each participant, screening results could vary, as could covariates at different tests. Two-level mixed-effect multinomial logistic regression models were used to determine differences in screening results by Aboriginal status, using normal results as the reference category.
Complete case analyses were used in multivariable analyses. Covariates in the fixed part of the model comprised age at test, marital status, residential remoteness, SEIFA socioeconomic quartile, testing period, and number of tests. When analysing cytology data, these characteristics were included in the model as categorical variables, with the first category treated as the reference (see Table 2). Relative risk ratios (RRR) and 95% confidence intervals (95% CI) were obtained using Stata 13 (Stata Corp, College Station, Texas, USA). A parallel analysis was undertaken for histology results, using binomial logistic regression to estimate the adjusted odds ratio (95% CI) of a high grade abnormality compared with a negative screen.
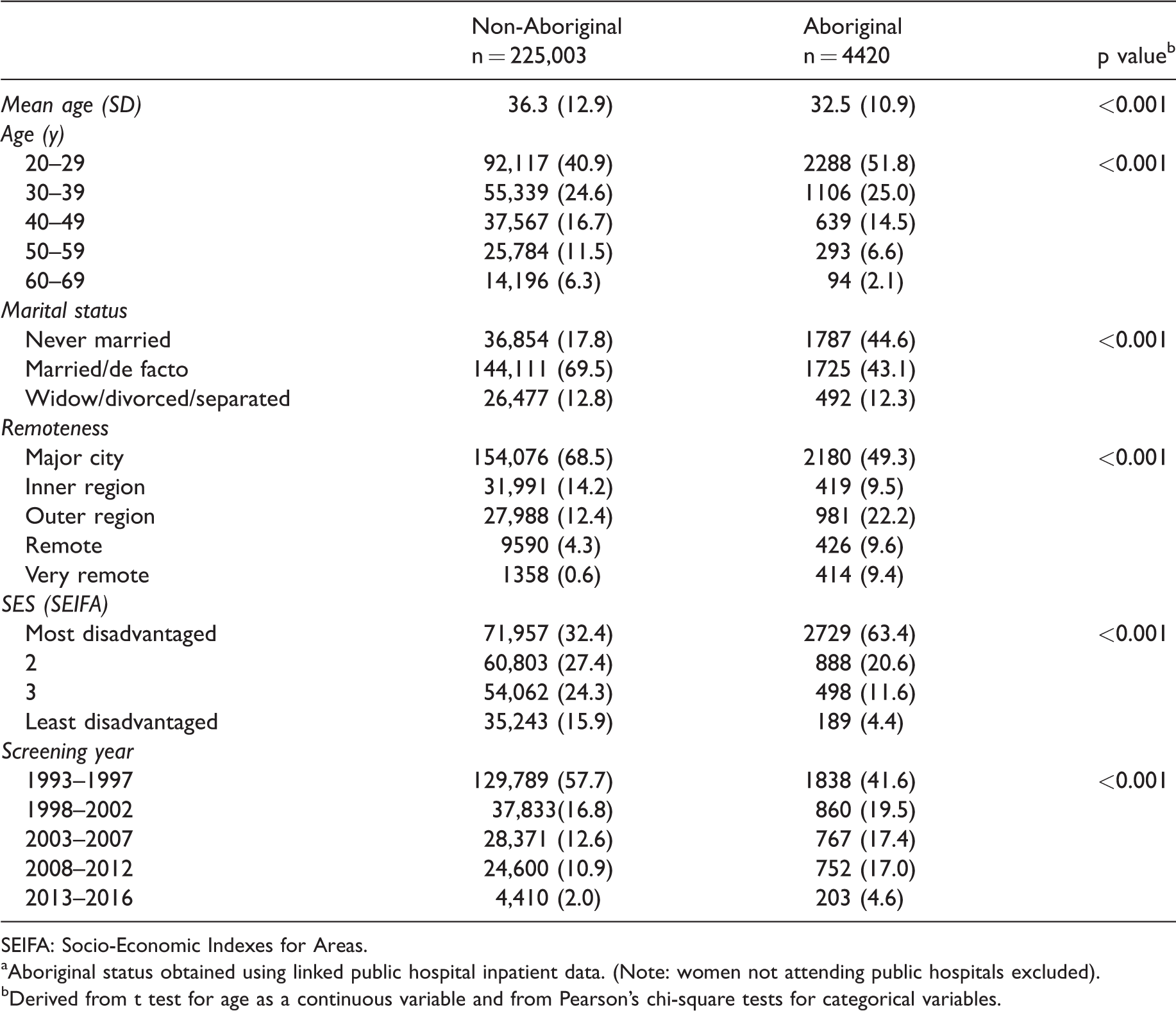
Characteristics (means or numbers and percentages) at first cytology screening test by Aboriginal status; South Australian women, 1993–2016. a
SEIFA: Socio-Economic Indexes for Areas.
Aboriginal status obtained using linked public hospital inpatient data. (Note: women not attending public hospitals excluded).
Derived from t test for age as a continuous variable and from Pearson’s chi-square tests for categorical variables.
Results
The mean age at first recorded screen for all screened women was 36.2 (41.2% aged under 30). Other study characteristics are presented in Table 2. Linked data indicated that 4420 (1.9%) of screened women were Aboriginal. They were younger than non-Aboriginal women at first screening test in the study period (mean age: 32.5 vs. 36.3). The proportions aged 20–29 were 51.8% and 40.9%, respectively. More Aboriginal than non-Aboriginal women had never married at time of first screening test (44.6% vs. 17.7%). The percentage of Aboriginal women in the most socioeconomically disadvantaged category was twice that for non-Aboriginal women (63.4% vs. 32.4%). More Aboriginal than non-Aboriginal women were residents of very remote areas (9.7% vs. 0.6%). The proportion whose first tests were undertaken in 2003–2016 was higher for Aboriginal than non-Aboriginal women (39.0% vs. 25.5%) (Table 2).
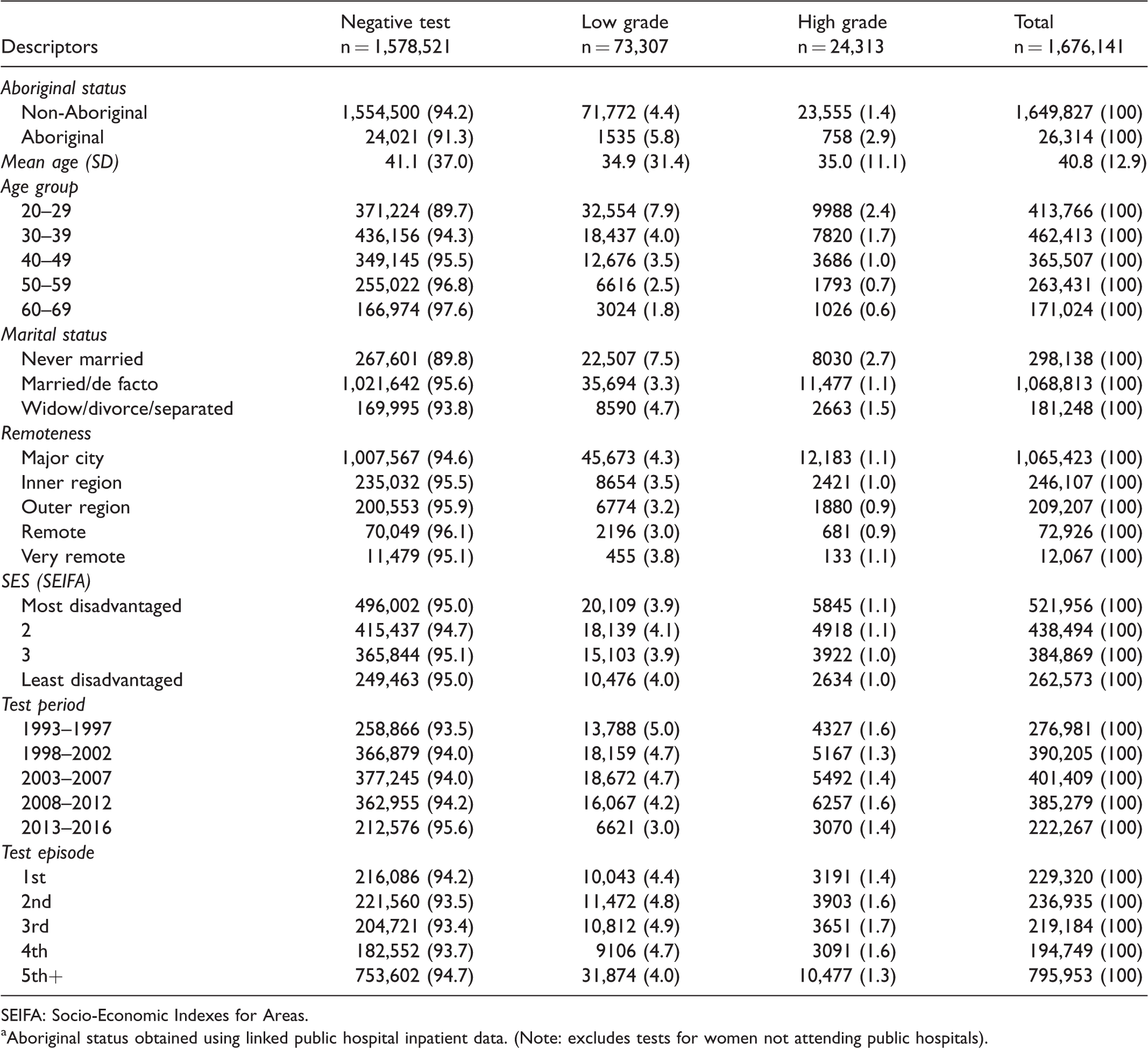
Of the 1,676,141 cytology tests in the linked dataset, 97,620 (5.8%) were classified as abnormal (Table 3). Of these, 24,313 (24.9%) were high grade. Crude rates of low grade and high grade abnormal results were higher for Aboriginal (5.8% and 2.9%, respectively) than non-Aboriginal women (4.4% and 1.4%, respectively). Crude rates of abnormal test results were also higher for women who were younger, never married/de facto, resident of a major city area, or resident of an area classified in the two most socioeconomically disadvantaged quartiles (Table 3).
Characteristics (numbers and percentages) by outcomes of cervical screening tests; South Australian cytology tests, 1993–2016. a
SEIFA: Socio-Economic Indexes for Areas.
Aboriginal status obtained using linked public hospital inpatient data. (Note: excludes tests for women not attending public hospitals).
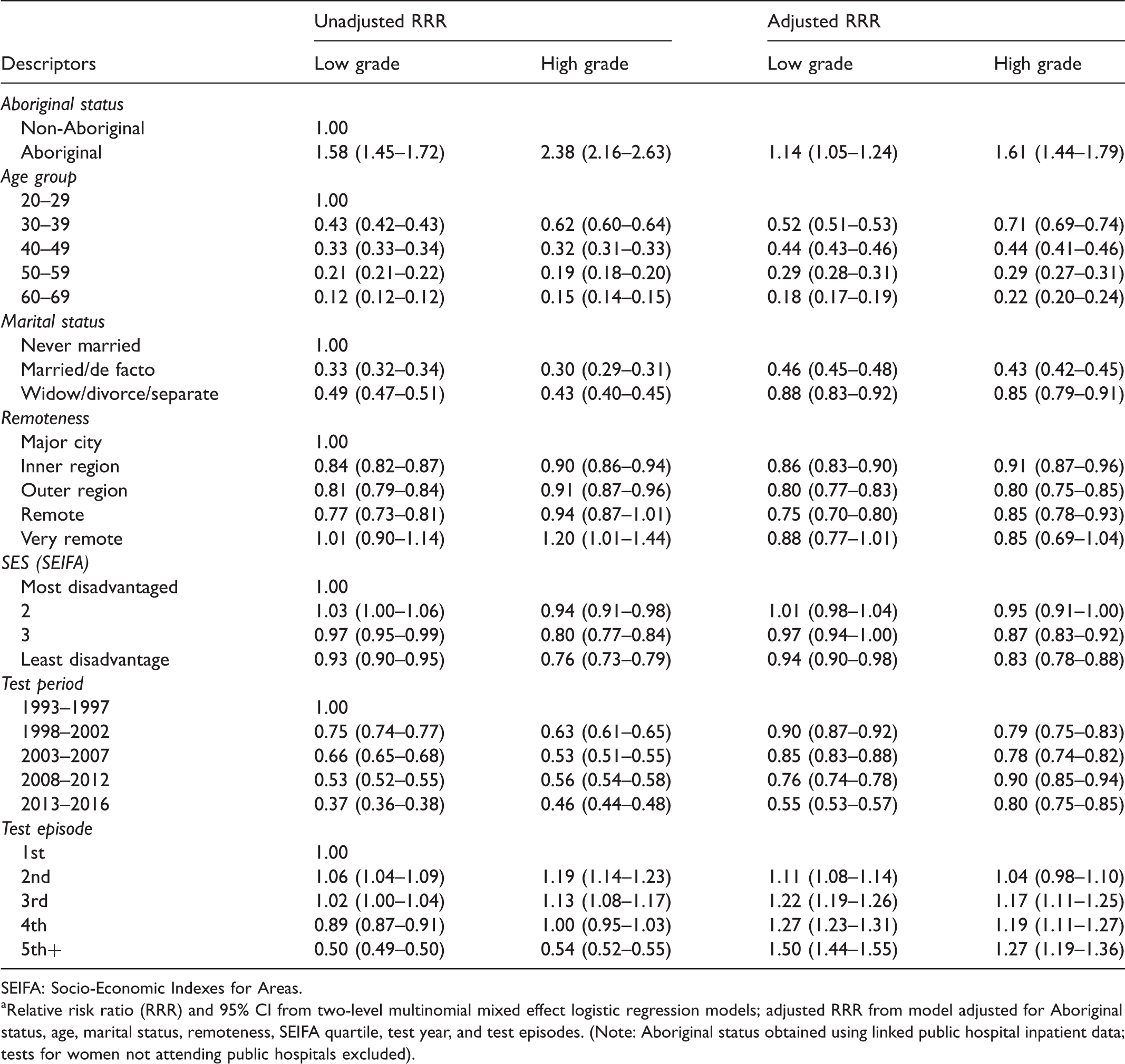
Using negative test results as the reference, multinomial logistic regression indicated a lower adjusted relative risk of low grade test results with older age, compared with the age 20–29 reference category, reaching a low for ages 60–69 (RRR 0.18, 95% CI 0.17–0.19) (Table 4). Again, using negative test results as the reference, tests for married/de facto women had an adjusted relative risk of a low grade result about 54% lower than for never married women (RRR 0.46, 95% CI 0.45–0.48) and the relative risk of low grade was about 12% lower for divorced/widowed/separated women than for those never married (RRR 0.88, 95% CI 0.83–0.92). Compared with tests for women living in a major city area, the adjusted relative risk for low grade test outcomes was lower for women living in inner regional areas (RRR 0.86, 95% CI 0.83–0.90), outer regional areas (RRR 0.80, 95% CI 0.77–0.83), and a remote area (RRR 0.75, 95% CI 0.70–0.80). While the adjusted relative risk of low grade was 12% lower for women living in very remote areas compared with a major city area, this may have been due to chance (RRR 0.88, 95% CI 0.77–1.01). Compared with tests for women from the most disadvantaged areas, the lowest adjusted relative risk of a low grade test was for the least disadvantaged areas (RRR 0.94, 95% CI 0.90–0.98) (Table 4). The adjusted relative risk of a low grade test decreased in the later test periods to a low for 2013–2016 compared with the 1993–1997 reference (RRR 0.55, 95% CI 0.53–0.57). A trend was also evident for an increased adjusted relative risk of low grade compared with a negative result for subsequent tests, reaching a maximum for the fifth or subsequent compared with the initial test (RRR 1.50, 95% CI 1.44–1.55).
Relative risk ratios (RRR) (95% CI) of abnormal cervical screening test outcomes relative to normal results by Aboriginal status; South Australian cytology tests, 1993–2016. a
SEIFA: Socio-Economic Indexes for Areas.
Relative risk ratio (RRR) and 95% CI from two-level multinomial mixed effect logistic regression models; adjusted RRR from model adjusted for Aboriginal status, age, marital status, remoteness, SEIFA quartile, test year, and test episodes. (Note: Aboriginal status obtained using linked public hospital inpatient data; tests for women not attending public hospitals excluded).
The pattern of adjusted RRRs for a high grade result, compared with no abnormality, approximated that for low grade results by age, marital status, and remoteness of residential area. The reduction in adjusted relative risk of a high grade abnormality with deceasing socioeconomic disadvantage was more pronounced than for low grade abnormalities (Table 4). The adjusted relative risks for a high grade test result were lower for 1998–2016 than for the 1993–1997 reference, and higher for the third and subsequent test episode than first episode (Table 4).
Aboriginal compared with non-Aboriginal women had a higher adjusted relative risk of low grade (RRR 1.14, 95% CI 1.05–1.24) and high grade cytology test results (RRR 1.61, 95% CI 1.44–1.79) compared with a negative test (Table 4). When low grade cytology test results were used as the reference, the adjusted relative risk of high grade was 50.0% (95% CI 34–68) higher for Aboriginal women.
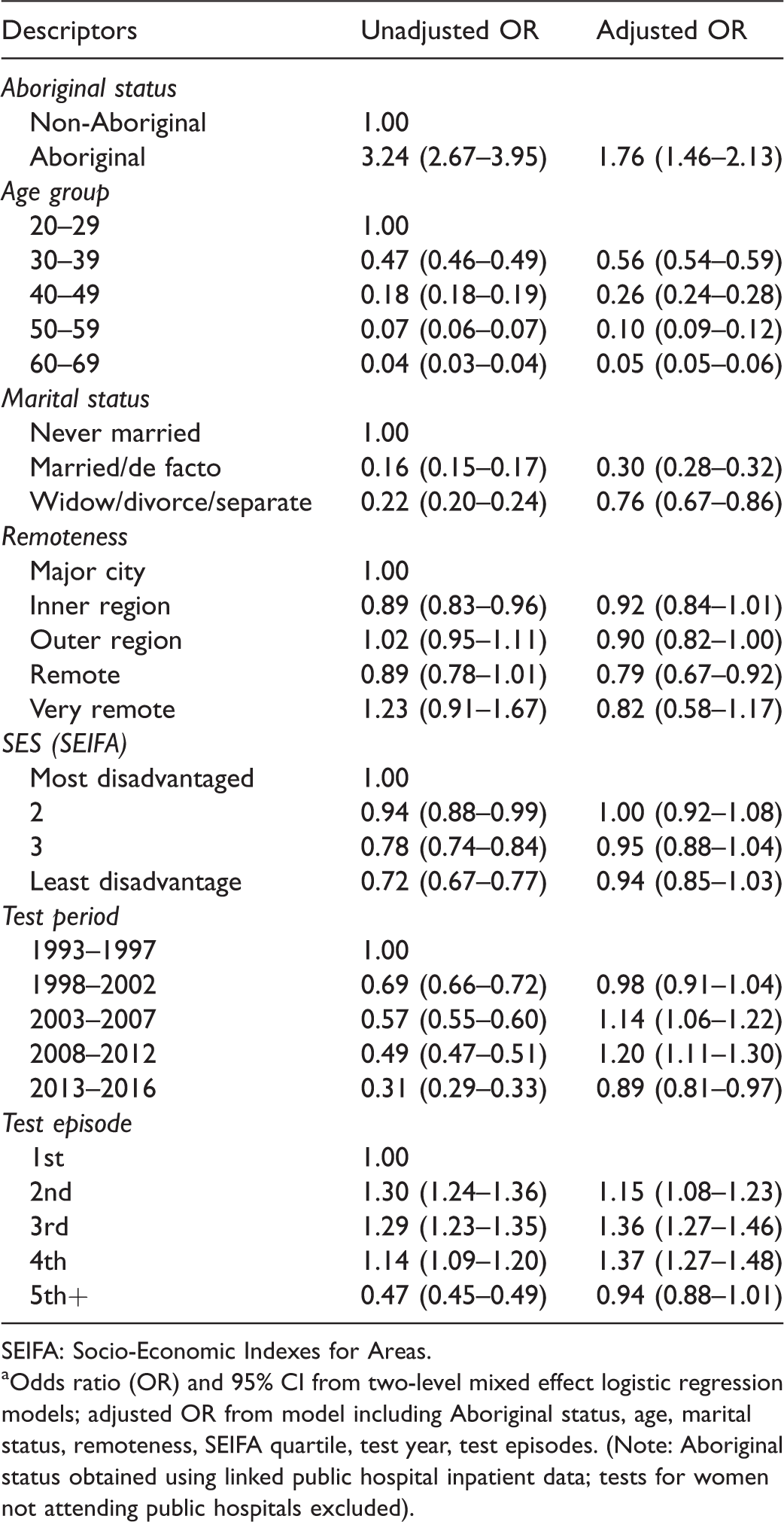
The percentage of high grade cytology results confirmed by high grade histology within six months was similar for Aboriginal and non-Aboriginal women, at 68% and 65%, respectively. Using a negative screen as the reference, the odds ratio for a high grade histology test result for Aboriginal compared with non-Aboriginal women was 1.76, 95% CI (1.46–2.13), after adjusting for age, marital status, socioeconomic status and remoteness of residential area, testing period, and test sequence (Table 5). Including interaction terms for Aboriginal status by testing period and sociodemographic variables did not indicate heterogeneity for this odds ratio.
Odds ratios (95% CI) of high grade histology outcomes compared with normal screening results by Aboriginal status; South Australia, 1993–2016. a
SEIFA: Socio-Economic Indexes for Areas.
Odds ratio (OR) and 95% CI from two-level mixed effect logistic regression models; adjusted OR from model including Aboriginal status, age, marital status, remoteness, SEIFA quartile, test year, test episodes. (Note: Aboriginal status obtained using linked public hospital inpatient data; tests for women not attending public hospitals excluded).
Discussion
Aboriginal women had higher relative risks of low and high grade results than non-Aboriginal women. The likelihood of high grade abnormalities compared with a negative screen was higher for Aboriginal women in both cytology and histology testing. Our results are consistent with Queensland evidence. 10
The elevation in high grade histology is consistent with the elevated risk of cervical cancer in the Aboriginal population. 1 Finding high grade lesions through screening can be interpreted favourably, however, in that curative care can follow for women who receive timely care.
Reported elevations in cervical cancer rates in Aboriginal women1,5,6 are consistent with a lack of early detection of precursor abnormalities and early treatment. Studies in Queensland and the Northern Territory have shown screening participation to be lower for Aboriginal than non-Aboriginal women.8,9 While studies in Australia have advocated measures to close the screening gap,18–21 larger shortfalls in screening coverage are apparent for Aboriginal Australians. 9
After adjusting for screening behaviour, Aboriginal women were still found to be at higher risk of cervical abnormalities.9,18–21 This could be due to increased exposure to HPV and co-factors in HPV progression (e.g. tobacco use).18–21 Low grade abnormalities reflect acute HPV infections that often regress spontaneously. By comparison, infections with oncogenic HPV types are more likely to persist with integration of HPV DNA into the host genome, leading to high grade cytological abnormalities and oncogenic effects.22,23 Prior to the introduction of HPV vaccination, the two most oncogenic HPV types (16 and 18) occurred with similar frequency in Aboriginal and non-Aboriginal women, whereas other oncogenic HPV types appeared to be more prevalent in Aboriginal women for the mid-adult age. 24
Since the introduction of the National HPV Vaccination Program in 2007, which offered the quadrivalent vaccine to all girls aged 12–13 (with an initial catch up to age 26 and vaccination of males from 2013), the numbers of high grade cytological abnormalities in young women aged up to 25 has reduced, with declines now starting to be observed in ages ≤34.25,26 While the impact of HPV vaccination seems similar in young women irrespective of Aboriginal status, 27 vaccination coverage and completion rates were found to be lower for Aboriginal than non-Aboriginal adolescent girls in Queensland and the Northern Territory. 28
“Renewal” of the national cervical screening program, which commenced in December 2017 with an entry age of 25 and an exit age between 70 and 74, 29 is using primary HPV testing with partial genotyping for HPV16/18 every five years. 29 It is hoped that the renewed program will facilitate broader screening participation from direct invitations to screen, and the added option of in-clinic self-collection of vaginal samples for HPV testing for under-screened women. A pilot project in Victoria found that self-sampling was acceptable for Aboriginal women when offered in a culturally appropriate environment. 30
An important finding of the present study was the reduced elevation in relative risks of abnormal screening results for Aboriginal compared with non-Aboriginal women after adjusting for covariates (i.e. from 1.58 to 1.14 for low grade cytology tests, and 2.38 to 1.61 for high grade cytology tests), and with a corresponding reduction in odds ratio from 3.24 to 1.76 for high grade histology tests after covariate adjustment. Opportunities to reduce risk by addressing modifiable risks associated with these covariates should be considered. They may include social and environmental factors, and not maintaining screening.31,32 Culturally appropriate health care and support services are important, as well as addressing issues relating to stigma and shame associated with cervical cancer and abnormalities. Qualitative research is needed, in addition to quantitative research, to explore the social and environmental factors potentially involved, including those which may affect participation in the HPV vaccination and cervical screening programs.
This study had several limitations. Aboriginal status was ascertained by linking with hospital inpatient records, as was undertaken in Queensland. 9 As only public hospital data could be linked, results may not be representative of the general population, however, about 98% of Aboriginal people’s hospital admissions in South Australia occur in public hospitals, and 91% of Aboriginal women are hospitalised in public rather than private hospitals.33,34 We therefore consider that the present study data are probably reasonably representative for Aboriginal women. By comparison, closer to 60% of admissions for non-Aboriginal women occur in public hospitals. Because private hospitals include a higher proportion than public hospitals of residents from upper socioeconomic areas, and because the risk of cervical cancer is lower in upper socioeconomic areas, 1 we consider that differentials in cervical abnormality rates between Aboriginal and non-Aboriginal women at a population level would be greater than estimated in our public hospital cohort.
The accuracy of the data linkage process was reported to be high in the Linkage Unit “handover report”, and applied in a standard manner across the public hospital cohort. However, there were differences in demographic profiles between linked public hospital participants and the non-linked (including women using private hospitals) with the former having a younger profile (e.g. aged <40: 52% vs. 43%), a lower proportion from major city areas (67% vs. 81%), and (c) a lower proportion from least disadvantaged areas (19% vs. 31%). We do not consider that these differences necessarily biased comparisons of test results by Aboriginal status within the public hospital cohort, particularly as adjustments were made for sociodemographic differences in the regression analyses.
A further limitation of this study was the likelihood of recording error for Aboriginal status, which is supposed to be obtained through self-identification. Despite attempts to promote self-identification, it is likely that hospital staff would sometimes infer Aboriginal status from appearance, name, or other alternative means. As a result, under-ascertainment and mis-recording of Aboriginal status is likely to exist. An Australian Institute of Health and Welfare follow-up interview study indicated the proportion of Aboriginal people correctly identified by hospital records in South Australia to approximate 91%, suggesting minimal bias. 35 It is also probable that under-ascertainment would have had little effect on differences in test results, as the proportion of cases classified as non-Aboriginal who were Aboriginal is likely to have been very small, given the low proportion of Aboriginal women in the South Australian population.
Another study limitation was the varying use of hospital inpatient services by individual women, which meant that timely data on marital status were not always available by time of cervical testing. We do not believe this would have materially affected differences in test results by Aboriginal status, however, as sensitivity analyses (e.g. by using the first recorded marital status, the most commonly reported, or the most proximally recorded marital status to the screening test) gave similar results.
Despite design limitations, we consider the results of this study to be plausible in showing higher rates of screen-detected abnormalities in tests for younger women, never married/de-facto women, and those from lower socioeconomic and major city areas, and in earlier rather than later testing periods. Screening abnormality rates in this study during 2007–2014 compared with corresponding Australia-wide rates for women aged 20–69 (both age-standardised to the Australian 2001 reference population) were lower, at 4.9% compared with 5.7%. 36 The percentage of high grade as opposed to low grade abnormalities were 24.5% and 24.3%, respectively. 36
Conclusion
To our knowledge, this is the first report comparing Aboriginal and non-Aboriginal screening outcomes using South Australian cervical screening registry data. Abnormal cytology test results, including high grade abnormalities, are more common for Aboriginal than non-Aboriginal women, as too are high grade histology test results. Record linkage enables Aboriginal status to be determined for women attending public hospitals in South Australia, filling an important evidence gap. Differences observed in test outcomes by Aboriginal status may be more conservative in this study than would have applied had data for women attending private hospitals also been available. In addition to increasing screening participation, ensuring that screen-detected abnormalities are followed up in a timely way by culturally acceptable services will be important for reducing differences in cervical cancer rates between Aboriginal and non-Aboriginal women. To the extent feasible, services should be located for optimal access. Factors underlying Aboriginal disparity should be investigated further, focusing on educational status, income level, employment status, and behavioural risk factors, and using data from qualitative as well as quantitative research designs.
Footnotes
Acknowledgements
We thank the Indigenous Reference Group for guidance throughout the project; Sharon Inawantji Clarke, Department of Health and Wellbeing, Prevention and Population Health, Aboriginal Well Women’s Screening Program, South Australia, for advice on respectful conduct of research related to Indigenous women’s business; and Data Linkage Unit staff and data custodians, including Karen Winch and Genevieve Chappell of the South Australian Cervical Screening Register, for data extraction and linkage. We also acknowledge the ongoing support of the Lowitja Institute, Australia’s National Institute for Aboriginal and Torres Strait Islander Health Research.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Karen Canfell is co-PI of an investigator-initiated trial of cytology and primary HPV screening in Australia (“Compass”), which is conducted and funded by the Victorian Cytology Service (VCS), a government-funded health promotion charity. The VCS has received equipment and a funding contribution for the Compass trial from Roche Molecular Systems and Ventana Inc USA. However, neither the authors nor Cancer Council NSW receive direct funding from industry for this trial or any other project. The other authors declare no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Indigenous Cervical Screening Project (NICSP) was funded by a National Health and Medical Research Council (NHMRC) Project Grant [#1045591]. This project was conducted under the auspice of the Centre of Research Excellence in Discovering Indigenous Strategies to improve Cancer Outcomes via Engagement, Research Translation & Training (DISCOVER-TT CRE) funded by NHMRC [#1041111] and the Strategic Research Partnership to Improve Cancer Control for Indigenous Australians (STREP Ca-CIndA), funded through Cancer Council NSW [#SRP 13-01] with supplementary funding from Cancer Council WA. We also acknowledge the ongoing support of the Lowitja Institute, Australia’s National Institute for Aboriginal and Torres Strait Islander Health Research. Gail Garvey was supported by an NHMRC Early Career Fellowship [# 1105399] and Lisa Whop by an NHMRC Early Career Fellowship [#1142085]. Joan Cunningham was supported by an NHMRC Research Fellowship [#1058244]. Abbey Diaz was supported by an NHMRC Training Scholarship for Indigenous Australian Health Research [#1055587] and a Menzies Enhanced Living Top-up scholarship funded by the DISCOVER-TTCRE. Patricia C Valery was supported by an NHMRC Career Development Fellowship [#1083090]. Karen Canfell was supported by an NHMRC Career Development Fellowship [#1007994]. The views expressed in this publication are those of the authors and do not necessarily reflect the views of the funding agencies.
