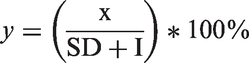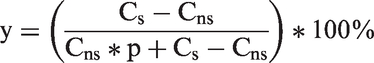Abstract
It is widely accepted that overdiagnosis is a major harm of screening, but its extent is still topic of controversy. This is partly the result of incomparable overdiagnosis estimates in scientific literature, as a variety of denominators are used to calculate the percentage of overdiagnosis in cancer screening. We propose to use the following denominator to calculate the percentage of overdiagnosis: ‘all cancers detected during the screening period, both interval and screen-detected, in participants of a screening programme’. This denominator is more appropriate than existing denominators because it presents overdiagnosis as a real percentage, is unaffected by attendance percentages, is applicable to all observational study designs, and can be easily recalculated to absolute numbers. This denominator can be widely applied and increases comparability between overdiagnosis estimates, which is needed to correctly present the balance between the benefits and harms of screening.
Keywords
It is widely accepted that overdiagnosis is a major harm of cancer screening. However, the discussion continues as to whether the benefit of screening (ie. cancer mortality reduction) outweighs the harm of overdiagnosis at population level, because there is so much uncertainty about the extent of overdiagnosis. One important factor influencing this uncertainty is the incomparability of overdiagnosis estimates: some studies express overdiagnosis as a percentage of cancers detected in absence of screening1,2, while other studies use the cancers detected in presence of screening as denominator. 3 In order to increase the comparability of overdiagnosis estimates in the scientific literature, a single denominator is needed, to express the percentage of overdiagnosis. This denominator should present overdiagnosis as a real percentage, be unaffected by attendance percentages, be applicable to all study designs, and be easily translated to relevant risks and numbers. We here propose a new denominator that fulfils all these requirements, and is therefore a strong candidate for replacing existing denominators.
A crucial first step, that is often forgotten, is the need to establish an agreed definition of the percentage of overdiagnosis in cancer screening. We define the percentage of overdiagnosis as: ‘the percentage of cancers detected during the screening period that would not present symptomatically during one’s lifetime, in the absence of screening’. The numerator is the absolute number of overdiagnosed cases, ie. ‘the detection of cancers that, in the absence of screening, would not present symptomatically during one’s lifetime’. The corresponding denominator is ‘all cancers detected during the screening period, both interval and screen-detected, in participants of a screening programme’. The formula (1) to calculate overdiagnosis is therefore
The numerator can be calculated by a number of study designs, including randomized trials, modelling, and observational studies. Most observational studies rely on the cumulative incidence approach 1 , which uses the difference in the cumulative cancer incidence in the presence (Cs) and absence (Cns) of screening to calculate the absolute number of overdiagnosed cases: x = Cs – Cns. In all study designs apart from randomized trials, Cns is unknown, therefore, most studies estimate Cns from pre-screening trends 4 , control regions 5 , non-attenders 6 , or trends adjusted for screening. 7
The denominator comprises all breast cancers detected in participants of the screening programme, and is therefore a strong candidate for replacing existing denominators.
Firstly, this denominator and definition express overdiagnosis as a real percentage, which is not the case for all denominators. For example, Biesheuvel et al. 1 proposed dividing the number of overdiagnosed breast cancer cases by the number of breast cancer cases in absence of screening. This is not a percentage. The definition of a percentage is ‘a part of a whole’. As overdiagnosed cases are not detected in absence of screening, they form no part of Biesheuvel’s denominator, and so Biesheuvel’s denominator does not result in a real percentage. In contrast, our proposed definition uses the breast cancers in presence of screening as denominator, and therefore forms a true percentage.
Secondly, the proposed denominator can be used to compare overdiagnosis between countries with different attendance percentages. Attendance percentages influence both the number of overdiagnosed cases and the total number of breast cancer cases. The numerator (ie. overdiagnosed cases) depends on the number of women attending screening. The denominator needs to be corrected for attendance. This only occurs if the cancers in women participating in screening are used instead of cancers in women invited to screening, as was, for example, proposed by the UK panel. 3
Thirdly, our proposed denominator can calculate overdiagnosis for all existing study designs, thereby increasing the comparability of overdiagnosis estimates between studies. In some studies in countries with a population-based screening programme4,6,7, it is unknown which breast cancers are detected in screening participants (ie. screen-detected) and which are detected in an interval, and in non-participants, so, the proposed denominator and denominators using only screen-detected cancers would be unknown. The percentage of overdiagnosis, however, can be calculated according to the proposed definition using an adapted formula (formula 2):
Derivation of formula 2.

Lastly, the percentage of overdiagnosis obtained by the proposed denominator is relevant to some constituencies and can easily be transformed to other relevant risks and numbers for other constituencies. The most appropriate way to express overdiagnosis depends on who is interested in the percentage of overdiagnosis. Persons detected with cancer during screening and cancer scientists are most likely to be interested in the chance that screen-detected cancer is overdiagnosed. In contrast, a person invited to screening is most likely to be interested in the absolute risk of an overdiagnosed cancer using the number of participants as denominator. The absolute risk of overdiagnosis can be obtained by multiplying the percentage of overdiagnosis with the cumulative risk of screen-detected and interval cancers, or by dividing the absolute number of overdiagnosed cases by the population at risk of overdiagnosis. Some constituencies (ie. governments) may be interested in the absolute number of overdiagnosed cases to calculate the health care costs of overdiagnosis among others. The absolute number of overdiagnosed cancers (x) can be recalculated from the percentage of overdiagnosis using the reverse of formula 1, or by the reverse of formula 2, ie.:
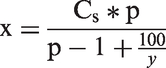
This formula was derived from formula 2 by replacing the numerator for x and Cns in the denominator for x-Cs. Then, the function was divided by x and rewritten as a function of x. The only data required for this formula is the percentage of overdiagnosis (y), total number of cancers in a screened population (Cs) and the attendance proportion (p).
To balance the benefits and harms of screening, agreement must be reached about the percentage of overdiagnosis. A crucial step towards achieving agreement is the standardization of the percentage of overdiagnosis. Our proposed definition for the percentage overdiagnosis, which expresses overdiagnosis as a real percentage, is not dependent on attendance, and is applicable to all study designs. Furthermore, the percentage overdiagnosis can easily be recalculated to other relevant numbers and risk.
Footnotes
Acknowledgments
We thank Roger Staats for his writing assistance.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.