Abstract
Objective
To exploit the prevalence of HPV genotypes 52/58 in a Chinese population, we evaluated algorithms that the use the Cervista Assay A9 group for primary cervical cancer screening.
Methods
The SHENCCAST II trial database was re-analyzed, focussing on the A9 pool of the Cervista HR-HPV Assay. Results for the detection CIN2+ and CIN3+ were correlated with a genotyping assay (MALDI-TOF) and cervical cytology to explore various screening algorithms.
Results
This analysis included 8,556 women with a mean age of 38.9. CIN 2+ rates were 2.7% (233/8556); CIN 3+ rates were 1.7% (141/8556). Overall HPV infection rates were 11.1% (950/8556) for Cervista, in which A5/A6, A7 and A9 groups were 26.5% (227/950), 22.9% (218/950) and 67.8% (644/950), respectively. The HPV A9 group is highly predictive of high-grade cervical lesions (CIN2+ OR = 103.61, CIN3+ OR = 128.059). Sensitivity and specificity for Cervista A9 group for CIN 2+ was 85.4% and 94.7%, and for CIN 3+ 89.4% and 93.8% respectively. Cervista A9 Assay followed by triage cytology for non-A9 positives has sensitivity and specificity for CIN2+ of 91.5% of 93.5%, and for CIN 3+ 94.3% and 92.6%.
Conclusion
Using the Cervista A9 as the primary screen instead of the full Cervista assay, the percentage referred to colposcopy would decrease from 11.1% to 8.8% and percentage requiring cytology would decrease from 11.1% to 3.6%. Sensitivity of detecting CIN 2+(91.5%), CIN3+(94.3%) would remain similar to the complete Cervista HR-HPV assay for CIN 2+(93.1%), CIN3+(95.0%).
Introduction
It is well accepted that testing women for the presence of high-risk types of the human papillomavirus (HPV) is the most sensitive primary screening method for the detection and prevention of cervical cancer.1,2 As the majority of women testing positive will have non-neoplastic HPV infections that will spontaneously resolve or not progress, the search for assays with higher specificity to use in a triage role, or possibly even for primary screening is an important objective in contemporary HPV research. 3 Potential secondary biomarkers include HPV genotyping (for HPV16 or HPV16/18),4,5 HPV mRNA testing, 6 and/or detection of other non-HPV biomarkers (eg. p16INK4A). 7 All have aimed to improve specificity, but population based data regarding the test performance of these biomarkers are limited.
From April 2009 to April 2010 we conducted the Shenzhen Cervical Cancer Screening Trial II (SHENCCAST II) in Shenzhen, China and surrounding rural communities. 8 Among the various new technologies studied in this population based screening trial, we tested the now Food and Drug Administration (FDA) approved Cervista High-Risk HPV Assay (Hologic Inc., Bedford, Mass, USA) and the polymerase chain reaction (PCR)-based MassARRAY (Sequenom, San Diego, CA) matrix-assisted laser desorption/ionization time-of-flight mass spectrometry system (MALDI-TOF). 9 This trial of 10,000-paired specimens demonstrated a non-significant difference to detect CIN3+ between the new Cervista assay and HC2 (Hybrid Capture II, Qiagen, USA). These results were confirmed by others.10,11 We also demonstrated at the established cut-points for the two assays a significant difference (favouring Cervista) in specificity. 12 We believed at that time that the Cervista assay had some characteristics that could be exploited, which could result in a decrease in the number of women referred for positive management. We hypothesized that as HPV types 52/58 are highly prevalent in a Chinese population, a more targeted screening protocol using the Cervista Assay A9 pool might result in a more efficient screening model for cervical cancer in China. To test this hypothesis we re-analyzed the SHENCCAST II data.
Methods
The study population consisted of the 8,556 women (of 10,000 enrolled) from SHENCCAST II who had all prescribed screening and diagnostic procedures, and had complete data. 8 This project and all associated studies have been approved by the human subject review boards of the Cleveland Clinic (CCF, 08-457, 7/11/2008) and the Peking University Shenzhen Hospital (2/10/2009). The protocol, previously detailed, 8 referred women for colposcopy based on three direct (physician obtained) HPV assays, two self-collected HPV assays, and cytology. All patients seen for colposcopy had a minimum of five cervical biopsies, as per the Preventive Oncology International micro-biopsy protocol of directed and random biopsies. 13
All histology slides were interpreted by a gynaecologic pathologist from Peking University Shenzhen Hospital as Normal, CIN 1, CIN 2, CIN 3, AIS, or cancer, with review by a pathologist from the Cleveland Clinic (Author BY).
The Cervista assay uses proprietary Invader technology (Hologic, Inc., Madison, WI), a signal amplification method for the detection of specific nucleic acid sequences. 14 Based on the correlation among 14 types of high-risk HPV DNA gene sequences, three different oligonucleotide mixtures, A5/A6, A7 and A9, are designed for the assay to test the 14 HPV DNA types in groups. An A5/A6 oligonucleotide probe mixture (Oligo Mixes) is used for detecting HPV types 51, 56 and 66 (the A5/A6 group of HPV), an A7 oligonucleotide probe mixture is used for HPV types 18, 39, 45, 59 and 68 (the A7 group of HPV), and an A9 oligonucleotide mixture is used for HPV types of 16, 31, 33, 52 and 58 (the A9 group of HPV). The Cervista assay and Cervista HPV 16/18 genotyping test (Hologic, Inc.; Marlborough, MA) were approved by the US FDA and recommended for clinical use in published guidelines. 15
The PCR-based MALDI-TOF genotyping assay is a mass spectrometry method that uses a multiplex primary PCR. 16 MALDI-TOF can accurately identify 14 HR HPVs, including HPV types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68, which include the Cervista A9 group. Neither the Cervista HPV assay using the A9 group or the MALDI-TOF assay have received approval by the SFDA (in China) or the FDA in the USA.
The performance characteristics of the screening tests were evaluated by calculating the sensitivity and specificity for detecting high-grade lesions (CIN 2+ and CIN 3+) as the endpoint. McNemar’s test was used to compare the sensitivities and specificities of the tests, and P-values of less than 0.05 were considered significant. The expected values and 95% confidence intervals for sensitivity, specificity were calculated. The percentages of the study population referred for colposcopy or cytology were calculated based on the number of HR-HPV of positive or cytology of ≥ASC-US in the screening population. All confidence intervals are exact binomial confidence intervals. Data analyses were performed using STATA 10.1 (StataCorp LP, College Station, Tx).
Specifically for this analysis we used the data for Cervista HPV and MALDI-TOF on the direct samples of the study population, to evaluate the subsets of the Cervista HPV assay for positive triage. We analyzed the sensitivity and specificity of each of the Cervista HPV groups for the detection of CIN 2+ and CIN 3+. In addition we compared the sensitivity and specificity with MALDI-TOF by using a MALDI-TOF A9 Group plus HPV 18 (MALDI-TOF A9 +18) and MALDI-TOF A9 Group minus 52/58 (MALDI-TOF A9 -52/58).
Results
The mean age of the 8,556 women was 38.9 years. Cytological abnormalities of ≥ASCUS were found in 12.1% (1031/8556), ≥LGSIL in 4.8% (413/8556) and ≥HGSIL in 1.4% (120/8556) of the study population. Pathology results show that CIN 2+ and CIN 3+ were found in 2.7% (233/8556) and 1.7% (141/8556) respectively.
The HPV positive rate for endocervical (direct) specimens was 11.1% (950/8556) for the Cervista assay. Among the Cervista HPV positives, the A5/A6, A7 and A9 groups represented 23.89% (227/950), 22.95% (218/950) and 67.79% (644/950), respectively.
Odds ratios of Cervista HPVA5/A6, A7 and A9 group for CIN 2+/ CIN 3+ (risk estimate).
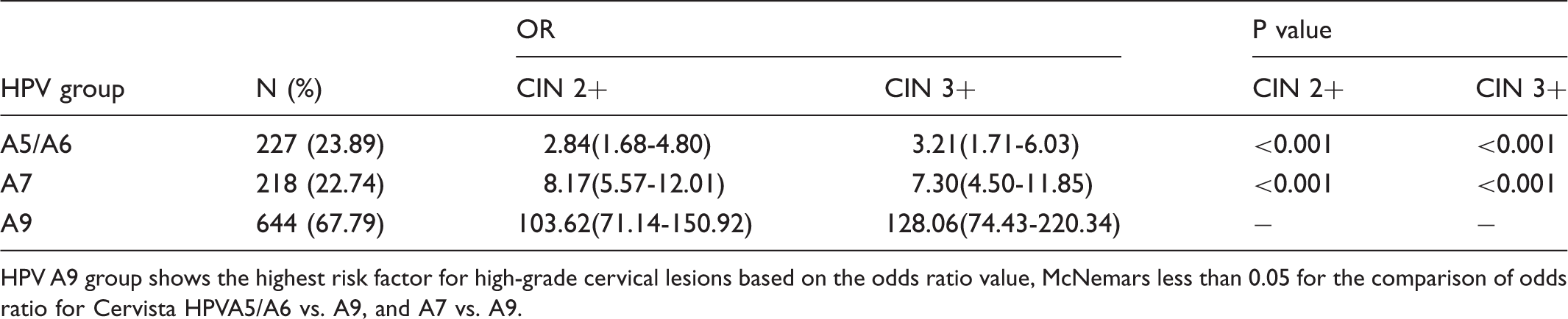
HPV A9 group shows the highest risk factor for high-grade cervical lesions based on the odds ratio value, McNemars less than 0.05 for the comparison of odds ratio for Cervista HPVA5/A6 vs. A9, and A7 vs. A9.
The sensitivity, specificity of three Cervista HPV groups for CIN 2+/ CIN 3+.
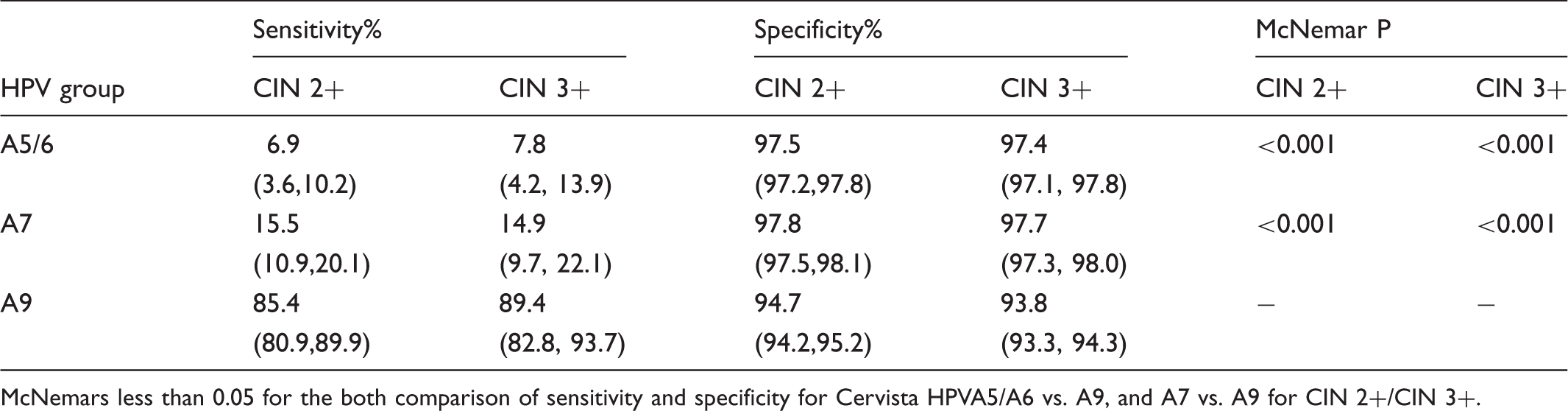
McNemars less than 0.05 for the both comparison of sensitivity and specificity for Cervista HPVA5/A6 vs. A9, and A7 vs. A9 for CIN 2+/CIN 3+.
The positive rates of cytology, Cervista, Cervista A9, Cervista A9 followed by cytology, MALDI-TOF A9+ type 18, MALDI-TOF A9 minus 52/58 and their sensitivity and specificity for CIN 2+/ CIN 3+.
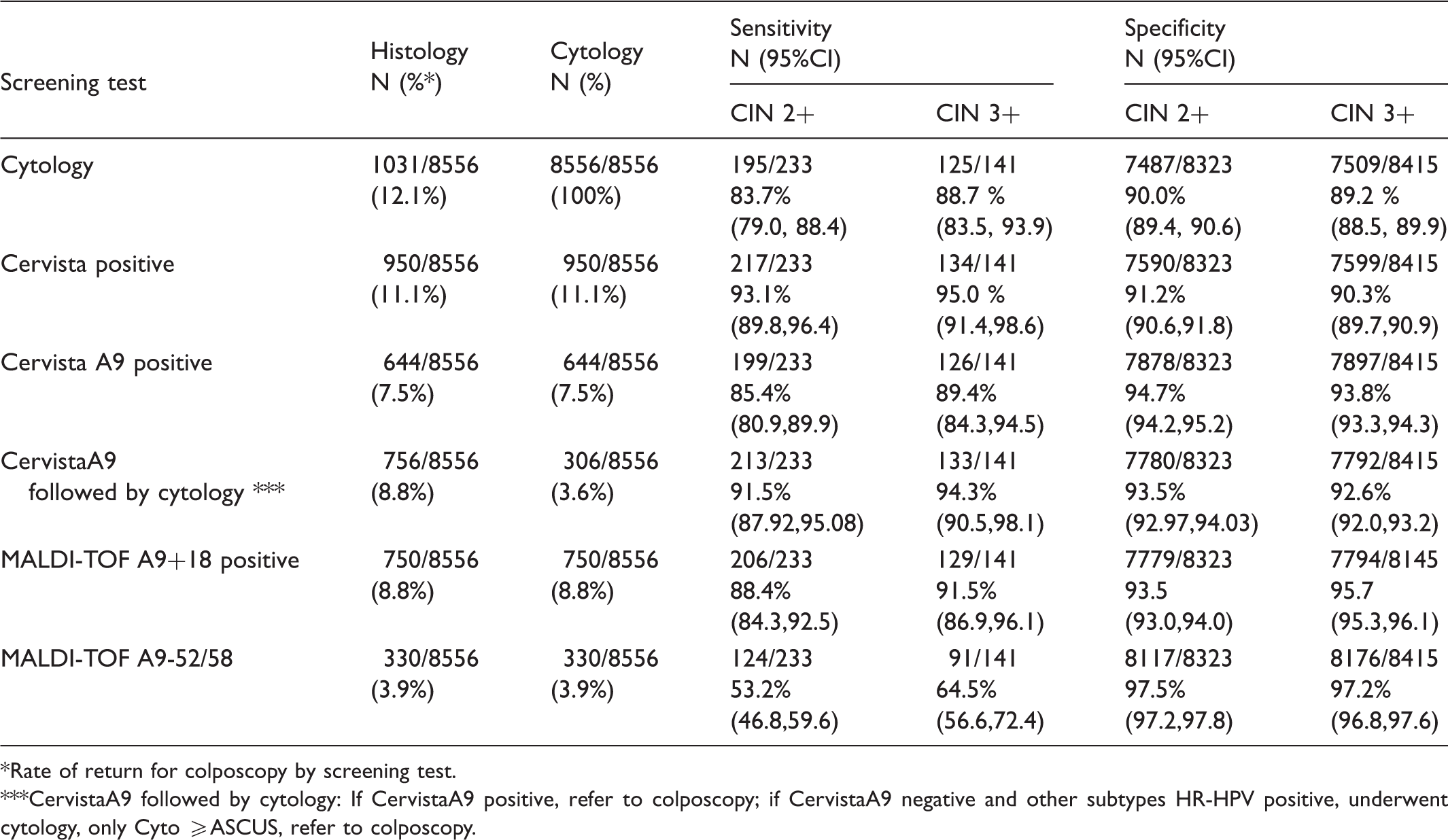
Rate of return for colposcopy by screening test.
CervistaA9 followed by cytology: If CervistaA9 positive, refer to colposcopy; if CervistaA9 negative and other subtypes HR-HPV positive, underwent cytology, only Cyto ≥ASCUS, refer to colposcopy.
According to the above results, the following algorithm is proposed: If the woman tests positive for the Cervista group A9 in her primary screen she is directly called back for colposcopy. If the woman tests negative for A9 but positive in groups A5/A6 and/or A7 she is triaged by cytology and then referred for colposcopy if the cytology ≥ASCUS. This algorithm produces a sensitivity for the diagnosis of CIN2 + of 91.5%, CIN3 + of 91.5%, and a specificity of 93.5%, 92.6%. The percentage of women referred to colposcopy would decrease from 11.1% to 8.8%, the percentage requiring cytology would decrease from 11.1% to 3.6%, and the sensitivity of detecting CIN 2+ (91.5%), CIN3+ (94.3%) would be similar to the full Cervista assay for CIN 2+ (93.1%), CIN3+ (95.0%).
Discussion
Cervical cancer precursors can be effectively detected and cancer prevented through screening with an HPV test and cytological examination of the cervix. 17 The false negative rate of an HPV test for high-grade lesions will be less than 5%, with a negative predictive value over 99%. 18 However 80-90% of these HPV infections are transient and disappear without inducing cervical cancer precursors, and therefore require no treatment. 3 As a result, using an HPV test for primary screening creates a large number of HPV-positives and a huge burden for the medical system to evaluate those women who, ultimately, need no intervention. Management of the high-risk HPV infected woman has become a major issue of common concern. Many studies now show that HPV genotyping can serve as an indicator of risk, and thereby be used for the triage of patients who test high-risk HPV positive. With some geographic variation in genotype distribution, the geographically important types can guide the triage for specific regions of the world.19–25
Our data from SHENCCAST II show the prevalence of the HR-HPV genotypes in the general population of Shenzhen, China and its surrounding communities as HPV 52(21.3%) followed by HPV 16(13.4%), HPV 58(12.1%), HPV33 (7.1%), HPV39 (6.8%), HPV51 (6.7%), HPV45 (4.9%), HPV66 (4.8%), HPV31 (4.6%), and HPV18 (4.1%). 26 Furthermore, in our Chinese population, types 16, 58, 18, 31, 33, and 52 had the highest risk of predicting CIN 2+ and CIN3+, and the sensitivity decreased to 53.2% (46.8,59.6) for a diagnosis CIN 2+ and 64.5% (56.6,72.4) CIN3+ by using the genotyping assay MALDI-TOF for the A9 types minus 52/58 compared with Cervista A9 positive, 85.4% (80.9,89.9) for diagnosis CIN 2+ and 89.4% (84.3,94.5) CIN3+. Applying MALDI-TOF A9 + 18 as a triage had less impact on increasing the sensitivity and specificity for both CIN 2+ and CIN 3+ lesions our population compared with Cervista A9, confirming that for Chinese women HPV 18 is less important, and HPV52/58 is relatively more important.
These data are similar to the study reported by Zhao et al, where 1,064 eligible women participating in routine cervical cancer screening in three hospitals in Beijing, China, were enrolled in a multi-centre clinical trial. 27 The sensitivity of Cervista HPV HR for CIN 2+ in this study population was 98.48%; the clinical sensitivity and specificity of just the group A9 were 87.97% and 78.3% respectively. The odds ratio of a positive result with group A5/A6, A7 and A9 was 1.611(0.976, 2.660), 1.819 (1.107,2.988), and 26.390 (15.300,45.520) respectively. Their data provide further evidence that the Cervista group A9 plus cytology triage for the non-A9 HPV positives is a reasonable screening protocol. The Cervista group A9 can be used for the triage of a high-risk positive group directly to colposcopy after primary screening with Cervista HPV.
Identifying the infections most likely to be associated with high-grade disease or cancer can help to reduce the high-risk population and make a rational triage of clinically HPV infected women. Hopefully, new screening and triage algorithms can reduce the overall economic burden to a healthcare system, by reducing the number of women requiring referral for colposcopy. Our analysis shows that by combining positive Cervista A9 primary screening and referral, with negative A9, positive A5/A6 and/or A7 cytology triage, some key programmatic advantages are realized. These include: 1) maintaining the desired high sensitivity and specificity, 2) reducing the number of women referred for colposcopy, and 3) reducing the number requiring cytology. This algorithm seems particularly suited for a Chinese population. This will clearly have an impact on the healthcare system, by reducing the workload, and hopefully these advantages will also be realized in the form of decreased costs for the patients. The strength of our study is that it is from a widely published and well-validated database, with minimal verification bias.
A proper cost analysis of this proposed algorithm is planned for the future.
Footnotes
Conflict of interest
None of the funding organizations made decisions concerning the data analysis, the content of this manuscript, or the decision to publish this report.
Support
This investigator initiated study was funded by Shenzhen Technical Innovation Committer under the Foundation Medicine Research Projects: A Study on Function and Functional Adjustments of the Carcinogens related to Canceration from HPV Infection to Cervical Cancer, Preventive Oncology International Inc. (Cleveland Heights, Ohio), Hologic Inc. (Bedford, Mass.) and the Shenzhen Female Doctors Assoc. (Shenzhen, P.R. China)
