Abstract
Objective
To evaluate clinicopathological differences between screen-detected (SD) and interval (IC) breast cancers diagnosed in women enrolled in an organized breast screening programme in 2000--2007.
Setting
Breast Cancer Screening Programme of the north region of Portugal.
Methods
Using data from the screening programme and from the population-based North Region Cancer Registry, SD and IC were identified. Information on screening history, age, date of diagnosis, tumour size, histological type and grade, lymph node status, tumour stage, biomarkers, and treatment was obtained from the cancer registry and from clinical and pathological reports. Association between mode of detection and these clinicopathological characteristics was estimated by unconditional logistic regression.
Results
A total of 442 SD and 112 IC were identified in women aged 50--69. Compared with SD, IC were diagnosed in younger women (60.0 ± 5.8 years and 58.4 ± 6.0 years, respectively), were larger (tumour size >20 mm: 60.2% versus 25.1%), lobular (6.3% versus 16.1%), with a higher differentiation grade (grade 3: 17.7% versus 38.9%), had more lymph node metastases, more advanced stage, and oestrogen receptor (ER) negative (12.9% versus 29.0%) and progesterone negative, and HER2 positive. After multivariable analysis, compared with SD, IC were more likely to be larger than 20 mm, lobular, of grade 3 and negative for ER.
Conclusion
Our results are consistent with other studies. IC’s have a more aggressive biology than SDs. Our findings did not show any unexpected pattern requiring changes to our screening procedures, but continuous identification and characterization of IC is advisable.
Introduction
Good practices in organized breast cancer screening include the identification and characterization of interval breast cancers (IC), those cancers that occur after a negative mammogram and before the scheduled date for the next examination. 1 In comparison with the evaluation of other routine performance indicators of screening, the identification of IC is a more complex task, relying on the availability of a population-based cancer registry with high coverage, operating continuously over time.2–4 In the absence of a cancer registry, it is also possible to consult hospital data (including that from pathology units) in order to identify and characterize all breast cancers in the screened population.2,5,6
IC are related both to the sensitivity of the screening programme and the length of interval between screening rounds, and are expected events in any screening programme.1,2,7,8 In general, compared with screen-detected cancers (SD), IC consist of larger tumours, with lymph node invasion, more advanced stage, and higher grade9,10, but not all studies are consistent and some have shown different patterns.5,11,12 Although there may be some controversy on the interpretation of the findings, IC are an heterogeneous group of tumours, the clinical and pathological features of which should be characterized in order to assess their relative contribution to the efficacy of breast cancer screening.13–15
The organized population-based breast cancer screening programme, conducted by the north branch of the Portuguese Cancer League (Liga Portuguesa Contra o Cancro – LPCC), was initiated in the Northern Region of Portugal in 1999. Preliminary results from the programme have been published, and considered satisfactory. 16
The aim of this study was to evaluate the clinicopathological differences between SD and IC diagnosed in the northern region of Portugal, in women enrolled in the screening programme between 2000 and 2007, within the context of evaluation of the screening programme.
Methods
By the end of 2009 the screening programme had expanded to 43 municipalities, covering 17% of the 454666 eligible women resident in the Northern Region of Portugal. 17 Women aged between 45 and 69 were offered biennial screening with two-view mammography, with double independent reading of the examinations by trained radiologists and, in case of discrepancy, a final reading by a third independent and experienced radiologist.
SD was defined as a breast cancer diagnosed after a positive screening result. IC was defined as a breast cancer diagnosed in a woman who had a screening test, with or without further assessment, which was negative either before the next screening invitation or within a time period equal to a screen interval for women who have reached the upper age limit. 1 IC diagnosis occurred as a result of opportunistic screening examinations, or in the course of a medical examination sought by women due to symptoms (eg. a lump, nipple discharge, pain).
To identify SD cases in women with a positive mammogram, we conducted a search for diagnosed breast cancers from hospital clinical files, pathology reports, and the North Region Cancer Registry (Registo Oncológico Regional do Norte – RORENO), a population-based registry with high completeness. 18 Information on identified SD cases was added to the Breast Cancer Screening Programme database. To be included in this study, screening must have occurred in the period 2000–2007.
Information on IC was gathered by merging the RORENO and Breast Cancer Screening Programme databases.The woman’s name, date of birth, and national health service number were used to link records. Inconsistencies and mismatching were checked manually. IC were those breast cancers registered in RORENO which matched with negative screening results in the previous two year period. This ascertainment was limited to women participating in the screening programme from 2000–2007. Because of the IC definition, breast cancers gathered might have been diagnosed in the period 2000–2009. As RORENO completeness is limited to invasive cancers, analysis was restricted to invasive SD and IC. Ductal carcinomas in situ were not considered. Following European Guidelines, 1 only women aged 50–69 were considered in the analysis.
The data collected was used to build a specific database for this study, including only cases of SD and IC. Information extracted from the screening programme database included variables such as date and round of screening, outcome of screening, woman's age at last screening examination, and residence. Information was also extracted from RORENO. To obtain additional data not routinely collected at the cancer registry, hospitals were contacted and clinical files and pathology reports were accessed. Overall, the following variables were collected: date of diagnosis, tumour size, histological type (using the International Classification of Diseases for Oncology-3rd edition; 19 8500, 8521 coded as ductal; 8520, 8522, 8524 coded as lobular; 8211, 8480, 8510, 8530, 8540 coded as other), histological grade (according to Nottingham Grading System 20 ), lymph node status, tumour stage (TNM classification - AJCC 21 ), and first treatment (mastectomy, breast conserving surgery, chemotherapy). IC and SD tumour size were classified in two categories (≤20 mm or >20 mm; the cut-off between T1 and T2, T3, T4 21 ). Information on biomarkers such as oestrogen (ER) and progesterone (PR) receptor status, and detection of overexpression and/or amplification of the human epidermal growth factor receptor 2 (HER2) were registered according to the pathology reports.
The two types of breast cancer (IC versus SD) were compared for each of these variables. To analyse the contribution of the different parameters to the risk of IC versus SD, an unconditional logistic regression was performed to calculate crude odds ratios (ORs) with corresponding 95% confidence intervals (CI-95%). T-test and χ 2 test were also used to compare differences between IC and SD, depending on the type of variables considered. After multivariable analysis (with all variables), the final model included only parameters significant to the risk of IC versus SD. HER2 status and triple negative assessment variables were not used in the multivariable analysis, due to the small number of cases. A P < 0.05 was considered to be statistically significant.
Results
In total, 554 invasive breast cancers were diagnosed in the study population, 442 (79.8%) classified as SD, and 112 (20.2%) as IC.
Odds ratio of interval breast cancers relative to screen detected cancers according to clinicopathological characteristics.
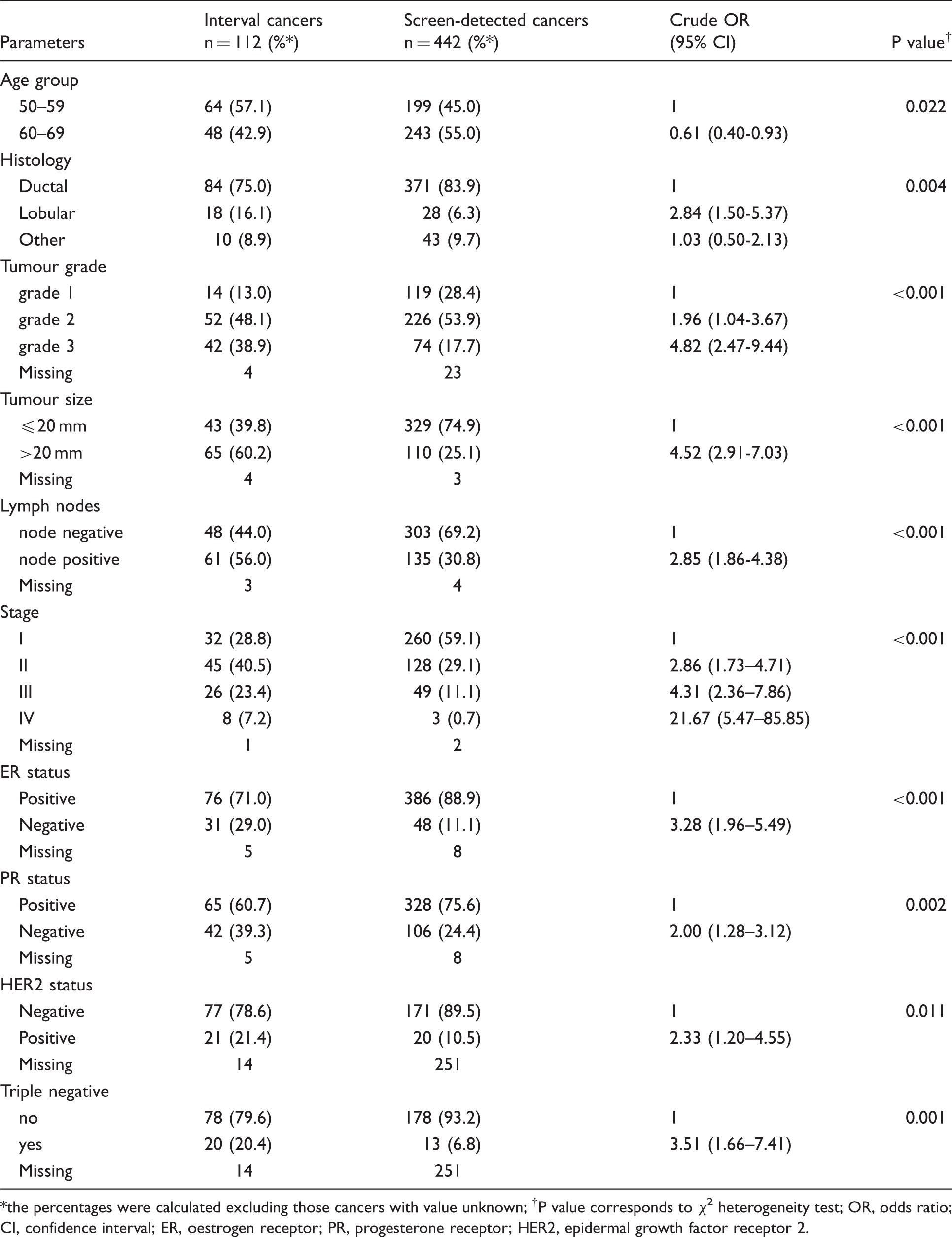
the percentages were calculated excluding those cancers with value unknown; †P value corresponds to χ 2 heterogeneity test; OR, odds ratio; CI, confidence interval; ER, oestrogen receptor; PR, progesterone receptor; HER2, epidermal growth factor receptor 2.
The predominant histological type in both IC and SD cancers was ductal invasive, but the relative proportions of ductal invasive and lobular were significantly different; lobular type was more likely to occur among IC (OR = 2.84, table 1). In this series of cases there were four inflammatory cancers, three in the IC group and one in the SD. In 27 cases, histological grade was not classified (table 1), either because the tumour had micro-invasion or because grade was not reported by the pathologist. In the remaining cancers, grade 3 was reported for 17.7% of the SD and for 38.9% of IC.
Women with IC presented more frequently than women with SD at diagnosis with lymph node invasion, and also with stage IV presentation (7.2% versus 0.7%). Evaluation of ER and PR status showed that SD more often had positive receptors than IC.
HER2 expression was evaluated in only 52.2% (n = 289) of the studied cancers; the proportion of HER2 positivity was higher in IC than in SD (21.4% and 10.5%, respectively) and the difference was significant. The phenotype triple negative for oestrogen, progesterone, and HER2 expression was more frequent in IC.
Odds ratio of developing an interval cancer relative to screen detected cancer. Final model, after multivariable analysis.
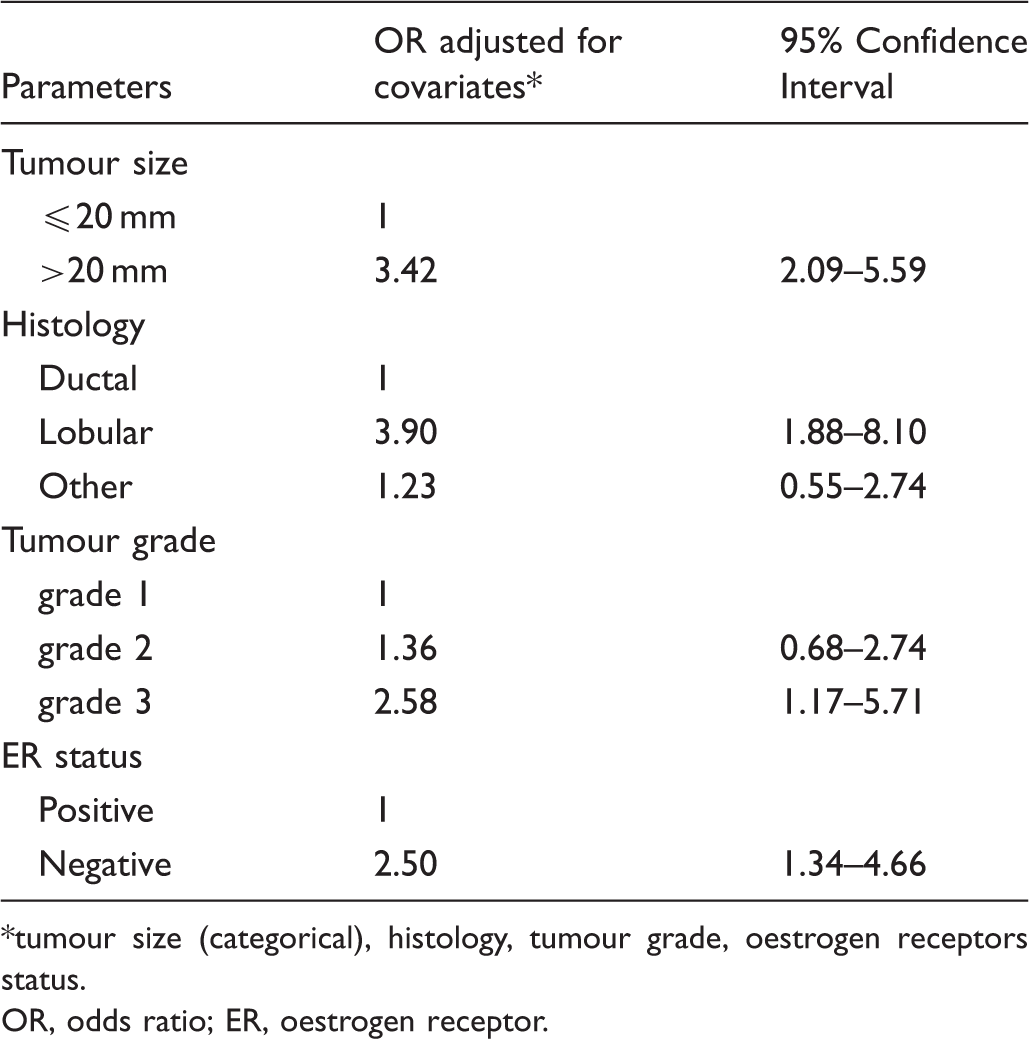
*tumour size (categorical), histology, tumour grade, oestrogen receptors status.
OR, odds ratio; ER, oestrogen receptor.
First treatment modality was significantly different between the two groups. Only 1.1% of SD cases had upfront chemotherapy compared with 13.8% of IC cases (p < 0.001). For those 525 women whose first treatment was surgery, breast conserving surgery was more commonly used in SD (261 in 431 cases, 60.6%) than IC (36 in 94 surgeries, 38.3%).
Discussion
This is the first report on the clinicopathological characteristics of IC versus SD within this screening programme.
At screening examination, women with IC were younger than women with SD, as verified in other studies.14,22–25 This has also been associated with higher prevalence of mammographic density in younger cases.10,23,26
Size of the breast cancer has been interpreted as reflecting the chronological age of the tumour.14,27 In this study, IC were significantly larger than SD, in agreement with the findings from other studies.10,12,28–30
Lobular histological type was more frequent among IC than SD breast cancers. This has also been observed in some studies,5,14,22,31 but others have found no difference in the histology type between IC and SD.10,11,28 For the purposes of our analysis, “lobular” carcinoma and “mixed lobular and ductal” carcinoma were considered the same entity, as their characteristics and outcomes have been considered similar. 32 The growth patterns detected in lobular tumours make these more difficult to detect with mammography. 14
Lymph node involvement in breast cancer has been considered to reflect both tumour chronology and biology. 27 In our study, after adjustment for the other variables, lymph node metastases were not significantly more frequent in IC than in SD. The small size of our studied population may explain this finding, but this lack of association has also been observed in other studies, after adjustment for size of the tumour.7,14 This excess of lymph node metastases among IC depended not only on the chronological age and biology of the tumour, but also on other unexplained variables.22,27
Grade is an important indicator of biology and prognosis, considered by some as time independent,13,33,34 but others have shown a phenotypic drift in cancer grade as tumour progresses.35,36 With mammographic screening, a high proportion of tumours is stage T1N0M0 and grade is considered of particular utility in the evaluation of small tumours. 33 In our dataset the distribution of high grade tumours was more frequent in IC than SD, and this association was independent of the size of the tumour. This result is consistent with previous studies reporting a more aggressive behaviour of IC.10,11,14,22,24,30,31
This pattern of a more aggressive behaviour associated with IC was also expressed in the biomarkers results. The proportion of cases that were negative for RE or triple negative for RE, RP, and HER2, was higher in IC compared with SD.7,24,37
In the final model (after multivariable analysis), tumours larger than 20 mm, histological type lobular, high grade histology and RE negative were the independent predictors of the presence of interval cancers.
Primary treatment for women with IC was more likely to be extensive surgery, or systemic therapy, compared with SD cases. This is also in accordance with other published results, and reflects the more advanced stage at diagnosis for IC cases. 30
Radiological review of last screening/assessment imaging and diagnostic mammography of the IC was not performed; this may be considered a limitation. However, although some authors have used a radiological category to distinguish between true interval and missed interval breast cancers,28,38–41 others have not considered this assessment essential, and some have even thought it to be misleading.11 Mammographic density has been considered a risk factor for IC,23,26 but in this study the retrospective analysis of the mammographic pattern was not performed.
The sample size of our study may have hampered the detection of some associations of smaller magnitude. As mentioned in other studies, the existence of unregistered opportunistic screening can introduce some misclassification on the detection mode (SD versus IC).29,42,43 In the northern region of Portugal, opportunistic screening coexists with the organized screening programme, but it is difficult to quantify with precision. 44
We believe that limitations mentioned above do not seriously affect the internal validity of our study. Furthermore, the methodology used in data collection and management support the validity of the associations identified. Data were retrieved from the population-based breast cancer screening organized programme, ascertainment of IC cases was made through a population-based cancer registry (RORENO) to ensure complete capture of breast cancer cases in the population served by the screening programme, additional data not routinely collected at the cancer registry was actively sought in hospital clinical files and pathology reports, and this procedure was identical for SD and IC.
Variable by variable, these results are consistent with other studies conducted in different settings and with different screening methodologies. IC constitute a subset of breast cancers with a more aggressive biology than SD. They occur in any screening programme, but our findings did not show any unexpected pattern leading us to changes in the screening procedures. Continuous ongoing identification and characterization of IC, as we have carried out in this study, is advisable.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
Ana Aguiar is the Head of the Breast Cancer Screening Programme in the North Region of Portugal and Vítor Veloso is the President of the Portuguese League Against Cancer, North Branch. The remaining authors have no conflicts of interest to disclose.
Acknowledgements
The authors thank Manuel António Silva and Joana Bastos from the Centre Region Cancer Registry, Alice Coelho from São Sebastião Hospital, Isabel Carvalho from S. João Hospital and pathologists Vicente Gonçalves, Eduardo Ferreira and Fernando Pardal for their valuable contribution to data collection.
