Abstract
Objective
To evaluate an immunochromatographic (IC) strip assay for Hb Bart's as a routine screening test for α-thalassaemia in area with a high prevalence of thalassaemia and haemoglobinopathies.
Methods
A total of 300 adult screen positive blood specimens were collected at an ongoing thalassaemia screening programme in northeast Thailand. Routine screening was done using red blood cell indices, osmotic fragility, and dichlorophenolindophenol tests. The IC strip assay for haemoglobin Bart's was performed on all samples. The result was evaluated against thalassaemia genotypes determined using standard haemoglobin and DNA analyses.
Results
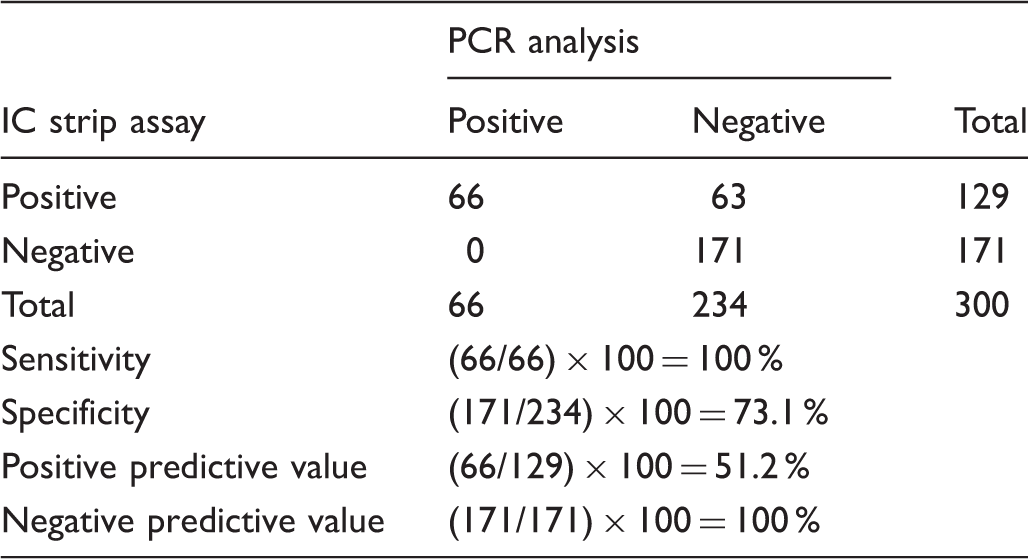
Of 300 subjects investigated, Hb and DNA analyses identified 32 with normal genotype. The remaining subjects carried thalassaemia with as many as 16 different genotypes. Hb Bart's was detected in all cases, with several α0-thalassaemia (SEA type) related disorders. Of cases with α+-thalassaemia, 86.1% showed a positive result; 100 out of 103 Hb E carriers, all homozygous Hb E and β-thalassaemia trait were negative. Nine out of 17 cases with β-thalassaemia/Hb E disease, and one case of double heterozygote for Hb Q-Thailand and Hb E returned positive results. The overall sensitivity and specificity of the IC strip assay for detecting α0-thalassaemia were 100% and 73.1%, respectively.
Conclusion
The results showed a high sensitivity for screening for α0-thalassaemia using IC strip assay for Hb Bart's. This simple method, used in combination with conventional screening protocols, should lead to a significant reduction in the number of referral cases for DNA analysis. Cost effectiveness in each population should be taken into consideration.
Introduction
The gene frequency of α-thalassaemia, including both α0-thalassaemia and α+-thalassaemia, in Thailand is 20--30%. The most severe form is α0-thalassaemia which, in its homozygous state, can lead to the fatal condition haemoglobin (Hb) Bart's hydrops fetalis.1,2 Interaction of α0-thalassaemia and α+-thalassaemia leads to the Hb H disease, and interaction of Hb H disease with Hb E, the most common Hb variant among Southeast Asian populations, leads to the complex syndromes AEBart's, EFBart's, and EEBart's disease, with thalassaemia intermedia phenotypes. In addition, co-inheritance of α-thalassaemia with β-thalassaemia can ameliorate the clinical severity of β-thalassaemia disease.3–6 Identification of α-thalassaemia is essential for providing appropriate genetic thalassaemia counselling to patients.
In Thailand and other Southeast Asian countries, thalassaemia prevention and control programmes target three severe diseases: homozygous α0-thalassaemia, β-thalassaemia major, and β-thalassaemia/Hb E disease. Such programmes aim to offer carrier screening to all pregnant women and their husbands, in order to identify carrier couples and inform them of their risks and options. A screening protocol based on electronic blood cell counting, osmotic fragility (OF), and dichlorophenolindophenol (DCIP) tests has been developed and implemented. Individuals with low MCV (< 80 fl) and MCH (< 27 pg), and a positive OF or DCIP test are initially considered screen positive, and usually have further investigation by Hb analysis using HPLC or electrophoresis (to identify β-thalassaemia and Hb E) and DNA analysis (to identify α0-thalassaemia). This screening protocol is simple, cheap, and straightforward, and has high sensitivity (ie. usually more than 95 %), although specificity is rarely over 80 %.7–9 The high false positive rate is due to other non-targeted abnormalities (eg. α+-thalassaemia or other hypochromic microcytic anaemia, especially iron deficiency anaemia, which is common in the regions 10 ), and leads to unnecessary operating costs for further Hb and DNA analyses. 11
A sandwich-type immunochromatographic (IC) strip assay, based on monoclonal anti-Hb Bart's antibody has been developed for rapid screening of α0-thalassaemia. This IC strip assay can detect the small amounts of Hb Bart's present in red blood cells of α-thalassaemia carriers. 12 We here evaluate whether this relatively new assay is practical and helpful in improving routine screening of the target population in northeast Thailand, where there is a high prevalence of thalassaemia and haemoglobinopathies.
Methods
Ethical approval of the study protocol was obtained from the Institutional Review Board of Khon Kaen University, Khon Kaen, Thailand (HE562059).
Subjects, haematological and DNA analyses
We determined that, with an α-thalassaemia gene frequency of approximately 20% in the Thai population, a sample size of 245 would be appropriate in this study. A total of 300 adult EDTA-anticoagulated blood specimens were obtained from our thalassaemia service unit at Khon Kaen University in northeast Thailand. All samples were selected from couples who had positive initial screening results using automated blood cell counting, OF test, and DCIP test as previously described7,8, and who were at risk of having severe thalassaemia diseases in the fetuses. Hb analysis is performed at our laboarory using an automated capillary electrophoresis system (Capillarys 2 Flex Piecing, Sebia, France). β-thalassaemia heterozygosity is diagnosed in individuals with Hb A2A; Hb A2 ≥ 4.0%.13,14 Polymerase chain reaction (PCR) and related methods are routinely utilized to identify β-thalassaemia mutations and six α-thalassaemia alleles common in Thailand (ie. α0-thalassaemia SEA & THAI deletions, α+-thalassaemia 3.7 & 4.2 kb deletions, Hb Constant Spring and Hb Paksé15–17). Thalassaemia genotypes were defined.
Identification of Hb Bart's by immunochromatographic strip assay
We used the i + LAB αTHAL IC Strip test kit, applying the IC strip, reagents, and a protocol provided by the manufacturer (i + MED Laboratories Co., Ltd., Bangkok, Thailand). In brief, 200 µl of whole blood was mixed with lysis solution containing 1% Triton X-100 to make red blood cell lysis. The IC strip was then immersed vertically (with the arrow pointing down) in the haemolysate for 2--5 minutes. The strip was removed from the blood samples and washed with washing buffer, provided with the kit, until the background was clear. The result was read by naked eyes. A positive result appears as two pink bands at the control and test zones; a negative sample has only one band at the control zone (see Figure 1).
Representative IC strip assay for detection of Hb Bart's. P and N are positive and negative controls, respectively. 1 & 2 are normal and Hb E heterozygote subjects with negative results. 3 & 4 are positive subjects with α0-thalassaemia trait and double heterozygote for α0-thalassaemia and Hb E, respectively.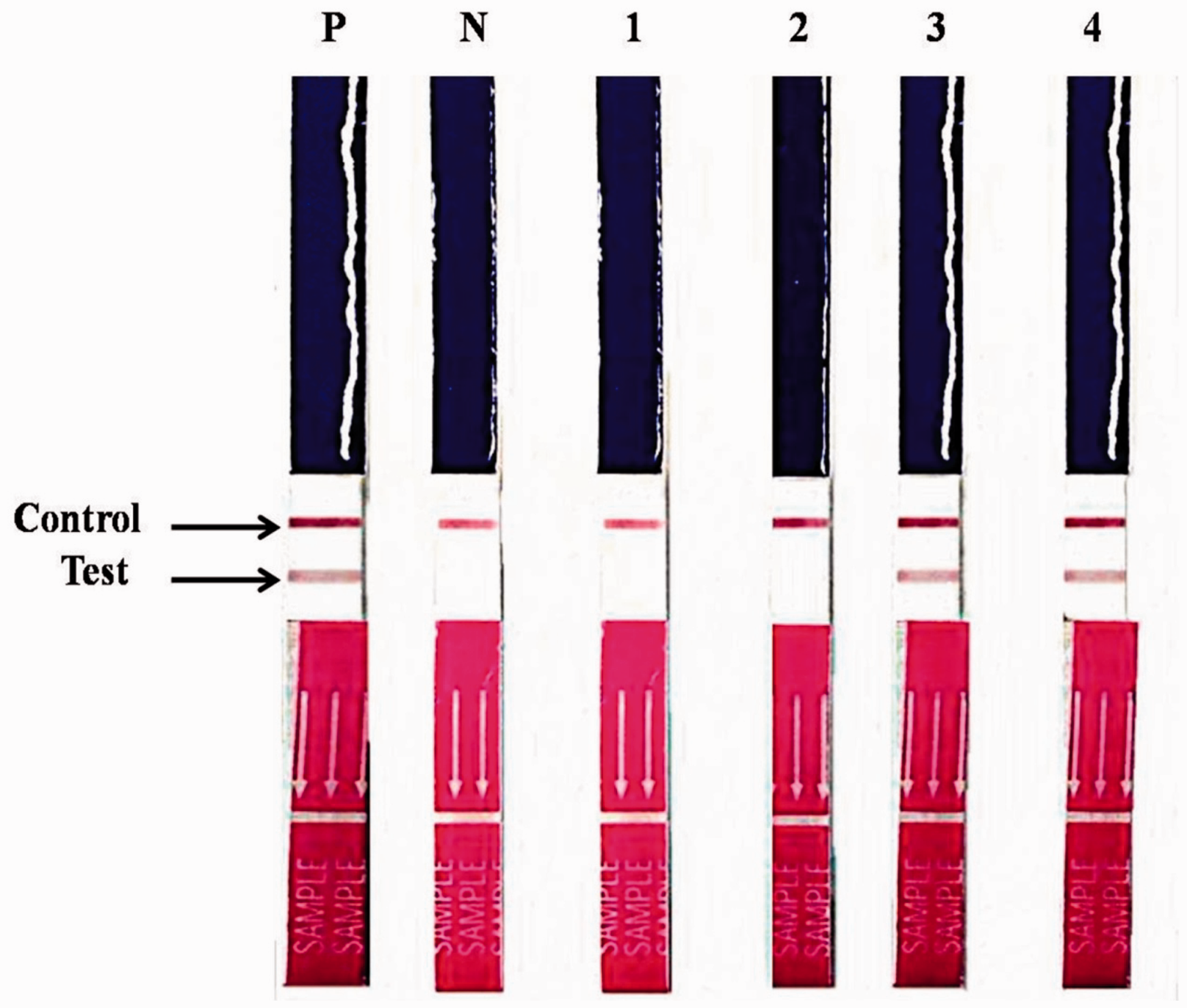
Results
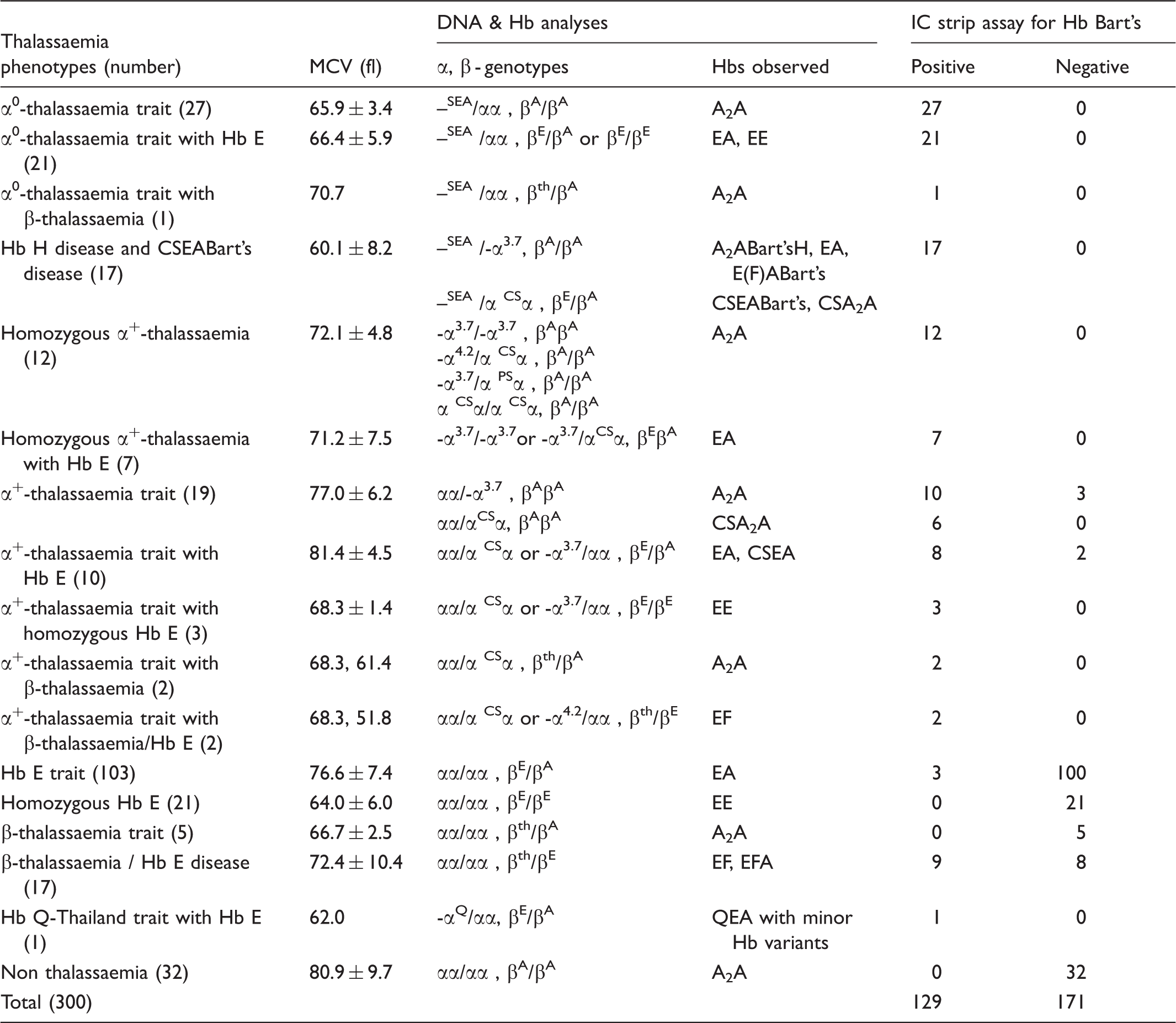
Thalassaemia phenotypes and genotypes of the whole subjects with positive and negative IC strip assay for Hb Bart's. MCV values are presented as mean ± S.D. or as raw data where appropriate.
Sensitivity and specificity of the IC strip assay for Hb Bart's in detection of α0-thalassaemia, using PCR analysis for α0-thalassaemia as a gold standard.
Discussion
Detection of α-thalassaemia, especially α0-thalassaemia, is crucial for genetic counseling of couples at risk, because couples who both carry α0-thalassaemia have a 25% risk of producing offspring with Hb Bart's hydrops fetalis, an emerging health care problem in many populations. 18 α0-thalassaemia is characterized generally by a deletion of two α-globin genes in cis on chromosome 16 and a mark reduction in α-globin chain production. Accurate diagnosis requires DNA analysis of the deletion by PCR, which cannot be performed in rural areas. The DNA deletion results in an excess of γ- and β-globin chains, which can polymerize to form γ4 tetramer (Hb Bart's) and β4 tetramer (Hb H). Therefore, α0-thalassaemia carrier status may be detected by an Hb H inclusion test. 1 This test, used in combination with a conventional screening protocol, could be considered as a secondary screening test for α0-thalassaemia. While the test is useful, it is relatively labour intensive, and lacks sensitivity for those with double heterozygosity for α0-thalassaemia with Hb E and β-thalassaemia, because the β4 tetramer could be minimal.19,20 Methods for detecting the small amounts of Hb Bart's using anti-Hb Bart's monoclonal antibody have been developed for use in screening of α-thalassaemia.12,21 Although these methods are easy to perform, their use in population screening is not straightforward in Southeast Asian countries, where thalassaemia and haemoglobinopathies are exceptionally common and heterogeneous. We therefore evaluated this in the real setting of our ongoing thalassaemia screening programme.
As shown in Tables 1 and 2, results on 300 subjects indicated that the IC strip assay for Hb Bart's could provide excellent sensitivity (100%) in α0-thalassaemia screening. All cases with α0-thalassaemia heterozygosity, Hb H disease (α0-thalassaemia/α+-thalassaemia) and EABart's disease (α0-thalassaemia/α+-thalassaemia with Hb E) could be detected. However, the specificity of the test was found to be 73.1%. In fact this is not unexpected as a number of false positives from other genotypes were noted: ie. 12 of 12 (100%) homozygous α+-thalassaemia, 16 of 19 (84.2%) heterozygous α+-thalassaemia, 8 of 10 (80.0%) double heterozygotes for α+-thalassaemia / Hb E, 3 of 3 (100%) α+-thalassaemia with homozygous Hb E, 9 of 17 (52.9%) β-thalassaemia/Hb E, and 1 of 1 (100%) double heterozygote for Hb Q-Thailand and Hb E. The results probably indicate that red blood cells in some of these α-thalassaemia syndromes contain small amounts of Hb Bart's (γ4), which could be detected by the IC strip assay. In contrast, the positive results in some cases of β-thalassaemia/Hb E disease might alternatively indicate the cross reactivity of the test with Hb F (α2γ2), which is variably presented in most cases. Identification of Hb Bart's (γ4 tetramers) in the absence of elevated Hb F (α2γ2) probably indicates a response to the α-globin chain depletion due to α-thalassaemia alleles, as well as a high detection sensitivity of the IC strip assay. This is also the case for the AEBart's disease (or Hb H disease with Hb E trait), in which only marginally elevated Hb F is noted. 22 A positive result associated with a case of Hb Q-Thailand / Hb E syndrome could be explained as Hb Q-Thailand is one of the most common Hb variants in Thai population, and is caused by a mutation (α74Asp-His) on an α1-globin gene on a chromosome with α+-thalassaemia (4.2 kb deletion). This leads to a production of small amount of Hb Bart's which could be detected by the IC strip assay, but not by the automated Hb analyzer. 23
The relatively high proportions of false positives caused by other thalassaemias, especially α+-thalassaemia, indicate that using the IC strip assay for Hb Bart's directly as a primary screening test could be problematic. In general, the frequency of α+-thalassaemia is much higher than that of α0-thalassaemia; overall frequencies of 0.107 for α+-thalassaemia and 0.029 for α0-thalassaemia have been reported in the northeast Thai population. 24 A high screening false positive rate therefore would be expected. Another method, based on the detection of embryonic ζ-globin chain, is an effective alternative means for α0-thalassaemia screening. 25 The method has high sensitivity and specificity for screening of α0-thalassaemia with SEA deletion in a Chinese population, with lower false positive rates from α+-thalassaemia.26,27 Unfortunately, this seems to be inappropriate for use in Southeast Asian populations (including the Thai population), where α0-thalassaemia could result from both SEA deletion (ζ-globin gene intact) and THAI deletion (ζ-globin gene deleted).28,29
It is noteworthy that applying a conventional screening protocol using MCV (cut-off 80 fl)8,30 to the data in Table 1 would also produce a sensitivity of 100 % for α0-thalassaemia screening, as all cases with α0-thalassaemia had an MCV less than 80 fl. However, the specificity would be much lower (ie. 29.2%, data not shown). In addition, MCV could detect cases with other thalassaemia targets, including β-thalassaemia carrier, homozygous Hb E, and some cases of Hb E trait. 8 Nonetheless, although this IC strip assay could not replace PCR for identification of α0-thalassaemia, it could be applied as a preliminary screening test, prior to molecular testing.
Cost effectiveness of the IC strip assay preliminary screening test must be considered before implementation in any population. In this study for example, additional screening with the IC strip assay on 300 initially screen positive subjects would be expected to lead to an exclusion of 171 cases for PCR analysis. In our setting, the IC strip test costs approximately USD 5 per sample, whereas a PCR test for α0-thalassaemia is approximately USD 10 per test. Without the IC strip test, the cost of PCR analysis would be 300 × 10 = USD 3,000. In comparison, using the IC strip test on all 300 subjects would cost USD 1,500. Excluding the 171 cases with negative IC strip test results, PCR analysis of the remaining 129 cases with positive IC strip test results would cost another USD 129 × 10 = 1,290. The overall cost of this protocol, with IC strip assay, would be USD 1,500 + 1,290 = USD 2,790, slightly lower than that of the protocol without the IC strip test.
Limitations and implications
The first limitation of the study is that we do not know with certainty the total population from which the 300 sample subset is derived. All these 300 subjects were selectively recruited from couples at risk of having severe thalassaemia diseases in the fetuses, who had had positive initial thalassaemia screening using CBC, OF, and DCIP tests. If we consider that this initial screening protocol would usually have approximately 50% positive results in a Thai population, the estimated total population could be extrapolated to approximately 600, but the exact total number of samples could not be obtained. Secondly, an accurate IC strip test result is obtained on fresh rather than aged blood specimens, and the test is designed to detect Hb Bart's at concentrations >5 µg/ml.
The cost effectiveness of the protocol with the IC strip test may not be very attractive, but because of its simplicity and the lack of a requirement for sophisticated equipment, the test could be utilized in remote areas where molecular testing is not available. The proposed screening protocol should therefore significantly reduce the number of referral cases for molecular testing and the PCR workload from such remote areas. It is conceivable that this protocol would be more useful and cost effective when applied to populations with a lower prevalence of α+-thalassaemia.
We therefore recommend, as shown in Figure 2, the use of the IC strip assay with an existing OF/DCIP or MCV/DCIP protocol7,8 for thalassaemia screening in the regions. This assay should be used only in a group of subjects with positive initial screening using an OF/DCIP or MCV/DCIP protocol. Hb analysis would be needed for all subjects, but those with a negative IC strip assay result can be excluded for α0-thalassaemia, and no further PCR analysis is necessary.
Proposed strategy using IC strip assay for Hb Bart's as a secondary screening with the existing OF/DCIP or MCV/DCIP screening protocol.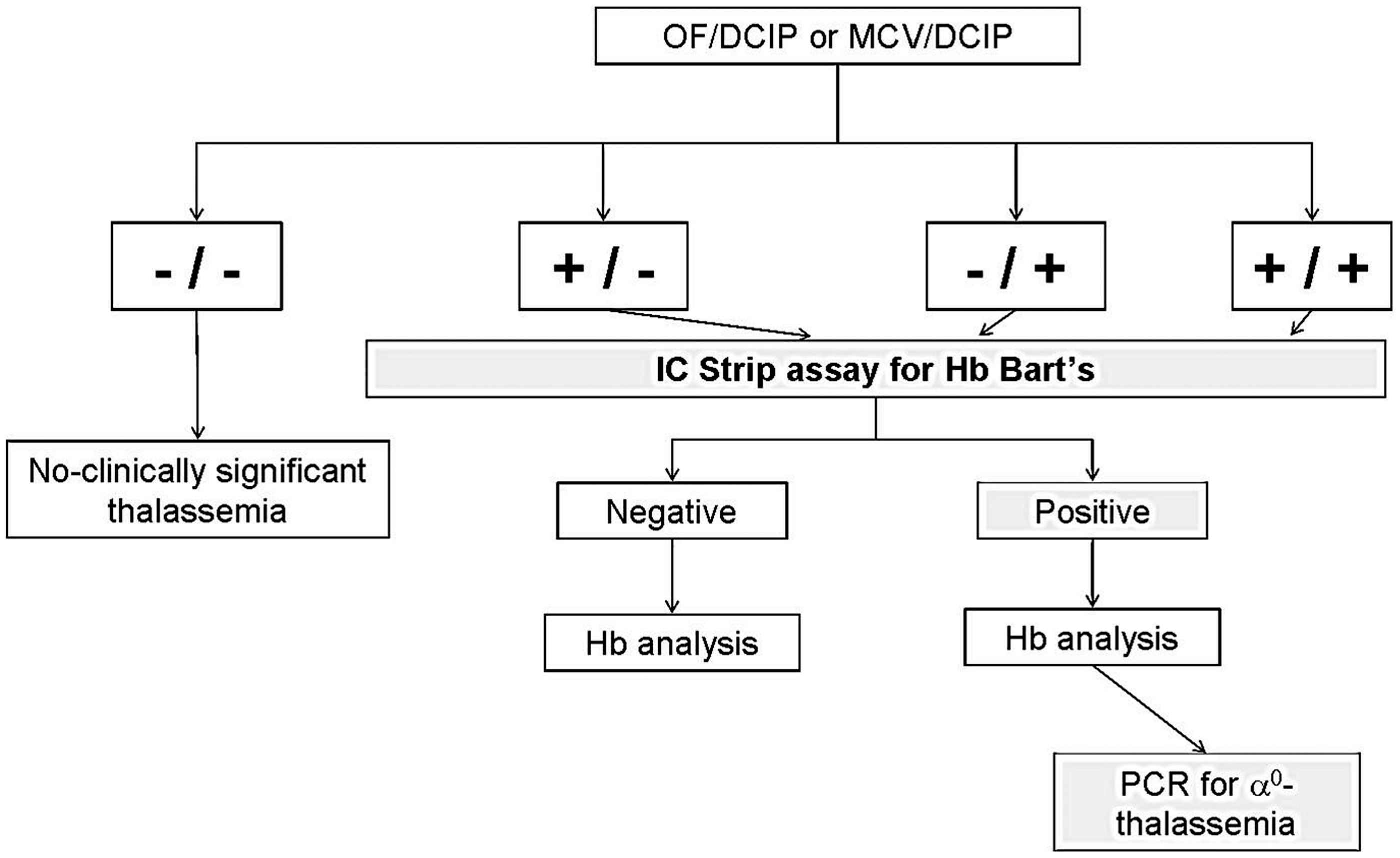
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
This work was supported by a grant from the National Research University (NRU) programme of Khon Kaen University and the Office of the Higher Education Commission, Ministry of Education, Thailand. We thank Dr. Watchara Kasinrerk of Chiangmai University and Dr. Suthat Fucharoen of Mahidol University and the i + MED Laboratories Co. Ltd., Bangkok, Thailand, for providing the IC strip assay kit used in this study.
