Abstract
Objective
To estimate the distribution of the FMR1 alleles and the prevalence of the premutaion (PM) and full mutation (FM) of the FMR1 gene in Korean women of reproductive age.
Methods
Using polymerase chain reaction and Southern blot, 5829 women of reproductive age were screened (low-risk group n = 5470 and high-risk group n = 359) and 11 prenatal diagnoses were completed between September 2003 and December 2011.
Results
Of the 5829 women screened, normal FMR1 alleles (11,607) had a bimodal distribution with most alleles having 29 (37.87%) and 30 (31.87%) CGG repeats. Of the 5470 women in the low-risk group, 7 PM were identified, giving a PM carrier frequency of 1:781; none of the women had Fragile X syndrome. We also identified 38 intermediate alleles, with a reported incidence of 1:143. Of the 11 prenatal diagnoses, five were normal, five had a premutation, and one had a full mutation allele.
Conclusions
The carrier frequency is 1/781 (0.13%) in Korean women of reproductive age. This is lower than among Caucasians, but relatively higher than in other Asian populations. Although there may be a founder effect, these results might be valuable in understanding Fragile X syndrome in Koreans and Asians as a whole.
INTRODUCTION
Fragile X syndrome (FXS) is the most common cause of inherited intellectual and developmental disability after Down's syndrome, affecting approximately 1:6000 to 1:4000 men. 1,2 Because it is an X-linked dominant disorder, the prevalence of FXS in female population is approximately half the prevalence in males. Its clinical features present as mild to severe mental retardation, with large ears, an elongated face, a prominent jaw, macroorchidism and autistic behaviour in affected males. Due to X inactivation, however, affected women usually show a milder phenotype. 3
FXS is caused by an amplification of a CGG repeat in the 5′-untranslated region of the FMR1 gene, located at Xq27.3. 4,5 Fragile X alleles are classified according to guidelines from the American College of Medical Genetics. Normal alleles have 6–44 CGG repeats, which encompasses most people in the general population; they are also very stable. Alleles of 45–54 CGG repeats have the possibility of instability, and are defined as intermediate or grey zone. Premutation (PM) alleles have 55–200 repeats, and full mutation (FM) alleles have more than 200 CGG repeats. 6, 7 PM alleles, are unstable and at risk of expanding into FM alleles when transmitted from mother to offspring. The risk of expansion for an FM allele depends on the number of CGG repeats in a PM allele. Alleles with more frequent sequences are at higher risk for the expansion than less frequent ones. 8–10 In addition, there are risks associated with transforming into a PM carrier such as fragile X-associated tremor/ataxia syndrome (FXTAS) and fragile X-associated primary ovarian insufficiency (FXPOI). 11,12 Around 40% of male PM carriers are at risk of developing FXTAS by the age of 50. 13 Female PM carriers also have a 20% risk of developing FXPOI. 12 In a case of FM, the expansion of the CGG repeat is accompanied by abnormal hypermethylation of a neighbouring CpG island at the 5′ of the FMR1 gene, which represses transcription. This results in lack of FMR1 protein, which subsequently accounts for the abnormal phenotype. 14
Several studies have estimated prevalence of the PM and FM of the FMR1 gene in various populations and ethnicities. The reported PM carrier frequencies are between 1:113 and 1:157 in Israeli women of child-bearing age; 1:259–1:397 in French-Canadian women; 1:151–1:382 in the USA and 1:246 in Finland. 15–22 Recently, some studies have screened newborns for FXS mutations in the general population. The prevalence of the FM was 1:5161–1:7321 in US males, 0:6895 in US females, 1:6209 in Canadian males, and 0:12032 in Canadian females, whereas the frequency was 1:2633 in newborn Spanish males. 18,23–25
Although several population-based screening programmes have established the prevalence of PM and FM alleles in women of reproductive age, newborns, and fetuses in western countries, few studies have reported the prevalence of PM and FM alleles for Asians. In Korea, there have been no reports for population-based studies on FXS; reports have been based on studies in selected groups only, such as patients with mental retardation or autism. 26,27 We aimed to ascertain the distribution of the FMR1 alleles and the frequency of PM and FM alleles in Korean women of reproductive age.
METHODS
Subjects and procedure
This study was approved by the Institutional Review Board at Cheil General Hospital (#CGH-IRB-2011–45). From September 2003 through December 2011, a total of 5829 women, either at preconception stage or already pregnant, were screened for FXS at the Department of Medical Genetics, Cheil General Hospital & Women's Healthcare Center. They applied for testing on their own initiative, or on the advice of their physician. Each woman received a leaflet including information about FXS and was asked to complete a questionnaire assessing any history of FXS (n = 2, 0.65%), mental retardation (n = 138, 38.44%), developmental problems (n = 112, 31.20%), autism (n = 96, 26.74%), or premature ovarian failure (n = 10, 2.92%) in the extended family. The 359 (6.16%) women with a family history were separated from the remaining 5470 (93.84%) low risk women for subsequent analysis. A total of 5311 (91.11%) women were tested during pregnancy, 5014 (91.66%) in the low risk group and 297 (82.73%) in the high-risk group. The mean gestational age of women tested in pregnancy was eight weeks six days and nine weeks three days in the low-risk and high-risk groups, respectively.
All samples were assayed by a polymerase chain reaction (PCR) to determine the CGG repeat length. If the PCR results were uninformative (homozygous allele or amplification failed), Southern blot was performed to identify the presence of PM and FM alleles. All women diagnosed as PM or FM carriers were referred for genetic counselling and offered prenatal diagnosis during current or subsequent pregnancies using amniocentesis.
CGG repeat analysis
Using the GC-Rich PCR System (Roche Diagnostics, Indianapolis, USA) PCR was performed following the procedures of O'Connell et al. 28 To measure size of each PCR product, an ABI 3130 XL Genetic Analyzer (Applied Biosystems, Foster City, CA, USA) was used. Analysis was carried out with GeneMapper software (version 4.0; Applied Biosystems) according to the manufacturer's instructions.
Southern blot analysis
Southern blots were analysed using a biotin labelled PCR probe (StB12.3: forward 5′-CGC CAA GAG GGC TTC AGG TCT CCT-3′, reverse 5′-GAG ACT GTT AAG ATA AAC GCG GG-3′) according to Gold et al. 29 DNA (7–10 μg) was digested with EcoRІ and EagІ (New England Biolabs, Ipswich, MA, USA). Hybridization and detection were performed using the AP Chemiluminescent Blotting Kit (KPL, Inc., Gaitherburg, MD, USA) according to the manufacturer's instructions. The percentages of normal FMR1 alleles on the active X chromosomes and normal FMR1 alleles on the inactive X chromosomes were ascertained by densitometry using a G:box Ichemi XL (SynGene, Inc. Cambridge, UK). The FMR1 activation ratios were calculated on the basis of the following formula: absorbed density of normal active X/combined densities of normal active X and normal inactive X, as described previously. 30
CGG/AGG interspersion analysis
We performed the CGG RP PCR using an Amplidex™FMR1 PCR kit (Asuragen, Inc., Austin, TX, USA) to determine the number and location of the AGG interruptions in the FMR1gene of PM mothers. The preparation and analysis of CGG RP PCR was performed according to the kit instructions.
Statistical analysis
Differences in carrier frequency between various ethnic groups were compared by using Fisher's exact test. For all statistical analyses, P value <0.05 was considered statistically significant. Statistical analysis was performed with the Statistical Package for Social Sciences version 12.0 (SPSS Inc., Chicago, IL, USA).
RESULTS
FMR1 allele frequencies in females screened between 2003 and 2011
Data are expressed as n (%)
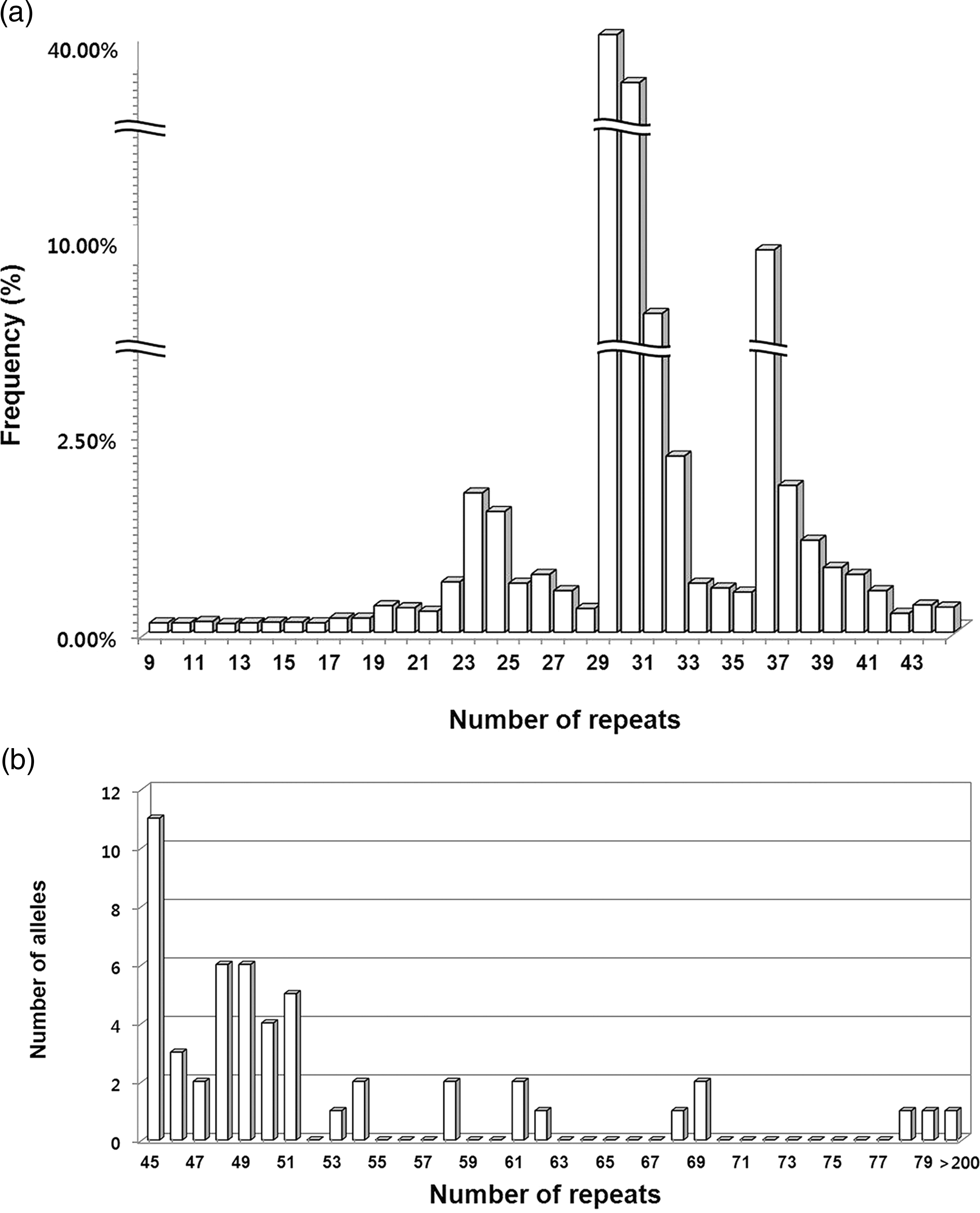
(a) Distribution of CGG repeats in 11,607 normal FMR1 alleles from 5829 Korean women of reproductive age; x-axis, number of CGG repeats; y-axis, percent of total. (b) Distribution of CGG repeats in 51 intermediate, premutation and full mutation FMR1 alleles from 5829 Korean women of reproductive age; x-axis, number of CGG repeats; y-axis, number of alleles
List of FMR1 repeat transmissions in prenatal diagnosis of FXS
NF, normal female; NM, normal male; PF, premutation female; PM, premutation male; FF, full mutation female
The high-risk group is shown in bold
*Size mosaic full mutation female (122, 170, >200 repeats)
DISCUSSION
We screened 5829 antenatal and preconceptional Korean women for FXS. Within this population, the distribution of normal FMR1 alleles (11,607) was bimodal with a major peak at 29 repeats and a minor peak at 30 repeats. To estimate the prevalence of the PM and FM of the FMR1 gene in the low-risk group, the high-risk group were separated from the total screened women. Thus, 7 PM were identified from the 5470 women in the low risk group, giving a PM carrier frequency of 1:781; none of the women had FXS. We also identified 38 intermediate alleles, with a reported incidence of 1:143. Among high-risk women, one in 90 was a carrier; among low risk women 1 in 781 was a carrier. As expected, the carrier frequency was significantly higher in the high-risk group than the low-risk group (P = 0.003). In addition, we analysed the difference of testing during pregnancy versus preconception in women with/without a family history (data not shown here). In comparing family history with pregnancy status, there were no significant differences for the intermediate/PM allele frequencies.
The uptake of invasive prenatal diagnosis was 100% in women found to be pregnant carriers in our screening. In mother-offspring transmission, the segregation ratio of the expanded allele was 0.54, which means that female carriers have a 50% risk of transmitting the abnormal allele to the offspring in each pregnancy. There have been suggestions that the risk for increase in the size of the expansion of maternal premutate alleles depends on the number of CGG repeats and the presence of AGG triplets embedded in the CGG repeat segment. 31 Nolin et al. 32,33 reported on intermediate and small PM alleles with 45–69 repeats, the number of AGG interruptions and the length of uninterrupted 3′CGG repeats associated with the instability of maternal alleles and the risk for expansion of CGG repeats on transmission to offspring. Even though the sample size was insufficient, our results tend to support these studies. This would be important and appropriate for women with intermediate and small PM alleles to estimate the repeat stability.
Prevalence of the premutation in different populations
Current studies not only address carrier frequency in the general population of women, but also in newborns. 15–25 Hill et al. 37 showed that studies of preconception and prenatal screening for FXS are supported by health professionals and families of patients with FXS. In Korea, few population-based studies of carrier frequency have been carried out; most studies have been on selected groups. Song et al. 38 reported that the mean CGG repeat (26.9 repeats) number of FMR1 in 1000 Korean women. We present a distribution of FMR1 alleles and the frequency of PM carriers in Korean women. Our study may have overestimated carrier frequency. Because women had to pay for the test themselves, our study may have been biased toward a higher socioeconomic group. In addition, subjects who self-referred or were referred by a doctor may not represent the whole population, which can bias the results towards higher frequencies.
A PM can expand to a FM when passed to offspring through female carriers. Therefore, carrier screening in women of reproductive age is required for primary care. Carrier screening for FXS occurs predominantly during pregnancy, and this screening is supported. 11,15,16,21,36,39 Carrier screening during pregnancy has been offered as part of standard prenatal care. If the pregnancy is affected, specific treatment strategies can improve some of the physical and behavioural symptoms, even though FXS is not curable. Health professionals, however, suggest that preconception is the most appropriate time to offer population-based screening for FXS. 37,40
Because of the noticeable benefits, preconception screening has been offered in primary care, and an expanded range of reproductive options are available to those who receive a positive carrier result. Preconception screening allows more time for deliberation and is less stressful than screening during pregnancy. Therefore, it is crucial that future studies offer screening before conception, without a selection bias, and that they address the prevalence of FXS and carriers in the general population.
CONCLUSION
Although we noted several limitations to our study, to our knowledge, this is the first report of an FXS cohort study in Korea. Our results estimate that the PM carrier frequency is 1/781 (0.13%) in Korean women of reproductive age, which is lower than that among Caucasians but relatively higher than in other Asian populations. Although there may be a founder effect, these results might be valuable in understanding FXS in Korea, and in Asian as a whole.
