Abstract
A central challenge in vaccination policy, and public health generally, is the tension that arises between the interests of the community and the interests of individuals. Over the previous two centuries, a range of legislative interventions have been used in efforts to improve vaccination rates, with varying degrees of success in navigating that central challenge. Here, we use a historical and sociolegal approach to characterise and evaluate vaccination laws in Australia from 1853 to the present. In doing so, we provide both a descriptive account of how vaccination laws have operated in Australia and a normative argument for what the role of law should be in relation to vaccination. This study highlights that the role of law in creating accessible, trustworthy, and equitable systems for vaccination is more influential in achieving public health goals than coercive approaches.
Introduction
The question of what role law should play in optimising the benefits of vaccination has been a key public health issue in Australia since the first successful vaccinations against smallpox occurred at Parramatta in 1804. 1 Despite ongoing challenges, vaccination has been a major success for public health globally, preventing more than two million deaths a year and protecting many more millions from illness and disability associated with a wide range of diseases. 2
The strength of vaccination as a public health tool is that immunisation protects both the individual and the community by reducing the spread of disease. 3 This is also its weakness: when individuals do not vaccinate, either because they cannot or choose not to, herd immunity is weakened. The proportion of the population required to achieve herd immunity depends on how infectious a disease is and how effective or long-lasting the vaccine is and varies between about 80% and 95%. 4 Eradication is possible once the requisite threshold is exceeded, although not all infectious diseases conform to this traditional view of herd immunity. There is value in herd immunity to individuals who are unable to be vaccinated and to communities whose health resources are used more efficiently. For these reasons, achieving very high rates of vaccination coverage is widely considered a public good. Meeting this target purely through voluntary uptake has proved challenging, and governments have implemented a variety of strategies – including legal interventions – aimed at increasing vaccination coverage.
Even before the Coronavirus Disease 2019 (COVID-19) pandemic, many countries were tightening their vaccination requirements or seriously considering mandatory approaches, 5 in order to meet the targets established by the WHO’s Global Vaccine Action Plan. 6 Calls for compulsory vaccination have historically been motivated by failure of less-restrictive alternatives to achieve sufficiently high vaccination coverage; outbreaks of vaccine-preventable diseases; and situations where elimination is in sight but frustratingly elusive, as with polio. 7 In seeking to advance collective rights to life and health, however, the use of increasingly intrusive legal interventions potentially infringes on individual rights to autonomy, bodily integrity, private and family life, education, and freedom of thought, conscience, or religion. Determining how to balance these competing rights is at the core of public health law and is the inherent problem of vaccination legislation.
Law is often seen as a tool for achieving public health by overcoming the effects of vaccine hesitancy or refusal through compulsion. Mandatory vaccination legislation is not the only way that law can support this goal, however, and nor is it obviously the best option. Here, we investigate the history of Australian vaccination legislation to identify what role the law plays in influencing vaccination uptake and to consider what role the law should play. Employing a sociolegal approach, we examine vaccination laws in their social and historical contexts by drawing on evidence from law, ethics, history, epidemiology, and the social sciences to undertake the legal analysis in four parts. 8
In Part I, ethical and legal principles are assessed to develop criteria for determining when legislative interventions to increase vaccination coverage may be justifiable. This provides a theoretical framework for evaluating the nature and effects of vaccination laws in subsequent sections.
Then, in Part II, we assess the effects of a wide range of legislative interventions aimed at influencing vaccination coverage within their social contexts. No complete history of Australian vaccination laws previously existed, and so this is achieved by comprehensively mapping the Australian legislative landscape relating to vaccination, from 1853 to the present. Through an iterative qualitative coding process, we identify three clear phases in Australia’s vaccination law history before the emergence of the COVID-19 pandemic: (1) experiments with compulsory vaccination in the self-governing colonies; (2) the introduction of conscientious objection in the newly federated nation and an emphasis on provision of community immunisation rather than compulsion; and (3) a return to legislative interventions aimed at reaching the levels required to achieve herd immunity in the low-disease environment prior to the COVID-19 pandemic. These broad vaccination programmes are evaluated against the legislative intervention criteria from Part I with reference to empirical evidence from historical, epidemiological, and social sciences sources.
Part III examines the current phase of vaccination law and evaluates legislative responses to the issue of vaccination coverage since the emergence of the pandemic. Building on the key themes identified in the historical analysis, we argue for a more nuanced role for law in influencing vaccination uptake, with greater attention to issues of access, equity, and public trust. Furthermore, we highlight the need for rigorous evaluation of legislative interventions to ensure their ongoing justifiability and to support adaptation to changing circumstances.
Part IV explains the dynamics of vaccination legislation over time in terms of continuity and discontinuity in the resolution of the tension between community and individual interests, and the normative and historical analyses provide the foundation for identifying and critically assessing potential law reform targets. The pandemic emerges as a clear break point challenging the law to re-evaluate the balance between community and individual interests. Historical break points mark the development of Australian vaccination legislation, prompting reflection on the role of law in influencing vaccination, as well as the nature and purpose of public health law more broadly.
Public health criteria in vaccination laws
The case of vaccination is exemplary of the interdependence of population and individual in public health: its benefits are unlikely to be fully realised if left entirely to individual discretion. A considerable body of ethical and legal literature has been developed regarding the extent to which it is justifiable for the state to interfere with an individual’s decision to vaccinate or not, and under what circumstances. This section synthesises this literature to identify the prevailing normative arguments and to develop criteria for assessing Australian vaccination laws.
Ethical principles for vaccination policy
The ethical literature relating to vaccination predominantly deals with frameworks for identifying whether particular interventions for encouraging vaccination are ethically justifiable and can be divided into three streams.
The first stream is strongly influenced by Mill’s harm principle and emphasises the degree of restriction on individual liberty as the key consideration. 9 These models are frequently represented as a ladder, with more restrictive legal interventions placed on a higher rung and requiring greater justification. They have been criticised for this linear focus, which encourages the adoption of the least-restrictive alternative by default and discourages consideration of values other than liberty, including public acceptability, distributive justice, and proportionality. 10
A second stream focuses on the obligations of individuals to the common good, arising as a corollary of an individual’s potential role as a vector as well as a victim of disease, as a basis for justifying state interventions, at least until herd immunity is reached. 11 Under the utilitarian perspective, the moral obligation to vaccinate has been founded on the idea that individuals ought collectively to do what brings about the best outcome benefitting the greatest number of people. 12 This stream is especially concerned with the conditions under which coercive interventions may be justified. For example, Savulescu and colleagues have argued that coercion is justified when the threat to public health is grave, mandatory vaccination is expected to result in greater utility than alternatives, and the coercive measures are proportionate. 13 Some commentators also argue conscientious objection to vaccination should not be available, 14 or conscientious objectors should make a proportionate contribution to society in lieu of vaccination, increasing in burden as the population rate of conscientious objection increases. 15
Ethical arguments for mandatory vaccination tend to proceed on the basis that vaccines are safe, effective, and accessible to all. This is not unreasonable as, while safety and efficacy vary depending on the specific vaccine, all vaccines approved for use in Australia have been assessed as within acceptable limits. 16 Existing evidence relating to accessibility, however, suggests that it remains one of the biggest barriers to vaccination coverage. 17 When rights to work, travel, or access benefits are tied to vaccination status, and access is not guaranteed, inequalities are likely to be exacerbated. 18
The focus on if and when mandatory vaccination might be justifiable unduly narrows the debate. Considerations such as safety, access, efficacy, and justice risk being sidelined as assumptions or preconditions, when they are more usefully thought of as goals in their own right, that change over time, that are dependent on context, and that should be advanced by any state intervention aimed at altering individual behaviour.
This limitation is addressed within the third stream, in which ethical frameworks such as those proposed by Verweij and Dawson or Isaacs encourage systemic thinking and permit multiple strategies for protecting public health to be considered, as well as potential interactions between strategies, rather than more narrowly considering whether the conditions for justifying a particular public health intervention have been met. 19 These frameworks identify key considerations for evaluating the appropriateness of vaccination programmes, including potential benefit; safety and efficacy; minimal burdens to participants; just distribution of benefits and burdens; avoiding coercion; providing compensation for harm; and promoting public trust. 20 Significantly, authors in this stream emphasise the need to interpret and balance ethical principles in light of the ‘relevant empirical facts’ relating to the disease, the vaccine, and the specific context for which the vaccine use is proposed. 21
The key considerations that consistently emerge from the ethical literature when evaluating regulatory interventions for improving vaccination coverage are the gravity of the public health risk; the efficacy and safety of both the vaccine and the intervention; the balance of benefits and burdens imposed by the intervention and the distribution of their effects; and the effect of the intervention on public trust.
Principles for vaccination policy from public health law
The ethical principles articulated in these frameworks are prominently echoed in the legal literature. The public health law literature, an area normatively driven by human rights and public health ethics discourses, 22 focuses on how best to assess the human rights burdens arising from interventions that, by their nature, involve restrictions on the freedoms of individuals in the interests of the population. Public health intervention assessment tools integrate similar considerations to those identified within the ethical literature (such as efficacy, ratio of benefits to burdens, and equity), while providing a structured approach to balancing competing rights and interests by introducing a modified least-restrictive alternative test. 23
The self-consciously multidisciplinary approach adopted within the public health law literature results in frameworks for assessing policies that are both theoretically-informed and empirically-dependent. This public health law approach retains the least-restrictive alternative test but subordinates it to efficacy, proportionality, and justice. The resultant frameworks highlight that designing public health interventions that comply with, and advance, human rights obligations has practical as well as ethical import. When public health laws enjoy widespread public trust, they achieve greater compliance and require less enforcement. 24
Evaluating legislative interventions in a way that prioritises public trust promotes transparency, encourages evidence-based decision-making, and allows preventive action to be taken under conditions of uncertainty. To this end, available regulatory frameworks are based either on the precautionary principle, which is predominantly applied in environmental law, or on the risk-based approach, which governs several areas of health law, such as therapeutic products. 25 A detailed examination of regulatory theory is beyond the scope of this analysis. However, these frameworks provide useful insights. The precautionary principle emphasises caution in the absence of extensive scientific knowledge about innovations with a potential for causing harm. 26 Conversely, the pillar of risk-based regulation is risk assessment, which is used to design flexible responses. 27 Here, it is argued that, in the context of vaccination policy, a nuanced approach that integrates elements of these frameworks can better ensure public trust even in the face of scientific uncertainty, as it addresses both safety and adaptability in response to emerging scientific evidence and public concerns and prioritises transparency, responsiveness, and proportionality.
Such an approach also highlights the importance of being clear about what risk is being addressed, as well as what interventions are being compared – including comparison with choosing to do nothing. When looking at regional human rights standards, for example, the positive obligations arising from the right to life under Article 2 of the European Convention of Human Rights (ECHR) support the argument that, from a public health law perspective, human rights obligations should operate to prevent both inaction and lack of preparation, as well as a restraint on government overreach. 28
The vaccination discourse in international human rights law largely reflects the approaches taken within the public health law and ethical literature, and while some of these sources are not binding on Australia, they are nonetheless an important source of guidance in how to balance competing rights. Significantly, the provision of vaccination is described as a core obligation arising from the rights to life and health. 29 State Parties have a responsibility to ensure equitable access, supported by systems that promote procedural fairness and justice, but human rights treaty bodies are less prescriptive about strategies to influence vaccination uptake. Restrictions on the right to bodily integrity, ordinarily protected through requirements for informed consent, 30 may be permissible for the protection of public health or for the protection of the rights and freedoms of others. 31 However, if individuals are injured as a result of mandatory vaccination programmes, Acosta argues that states have an obligation to provide an effective remedy for such injuries, and that remedies need to be suitable, effective, and respectful of due process. 32 Such an obligation aligns with the principle of minimising burdens and finds some support in ECHR case law. 33
The 2021 case of Vavřička and Others v the Czech Republic saw the European Court consider whether a vaccine mandate under Czech law was consistent with the right to bodily integrity within the right to private life under Article 8 of the Convention. 34 This judgement raises two issues relevant to the development of criteria for assessing legislative interventions.
First, in determining the proportionality of the Czech vaccination policy, the Court’s reasoning demonstrated significant congruence with principles from public health ethics and law. The following features were identified as relevant: the existence of exemptions for medical and secular objections, as well as a degree of choice regarding the vaccine and timing of vaccination (distributing benefits and burdens equitably); sanctions were found to be at the moderate end of the scale, or for protective rather than punitive purposes (minimising burdens while retaining efficacy); administrative and judicial appeals were available (procedural fairness and transparency); and safety concerns were appropriately addressed through checking for contraindications and a system for monitoring adverse events (minimising burdens). 35
Second, the Court struggled to integrate empirical evidence into this reasoning in a rigorous way, preferring to subsume deference to expertise within the margin of appreciation afforded to states in the area of healthcare policy. 36 Determining whether a less-restrictive, yet effective, alternative existed in the Czech context would have required an assessment of empirical evidence for which the Court is not well-equipped, and so the test of least-restrictive alternative was instead rejected. 37 Furthermore, in considering the suitability of this law for achieving the legitimate aim of protecting public health as part of its proportionality analysis, considerable evidence was presented that vaccination was an effective tool in public health, 38 but there was far less consideration given to whether compulsory vaccination laws were an effective means of protecting health. This highlights the importance of being clear about which intervention is being evaluated: here, the compulsory vaccination law should have been the focus, not the efficacy of vaccination generally.
Although the text and supporting documentation of international human rights instruments appear more supportive of voluntary state vaccination programmes, the law provides little protection from overly-restrictive public health policies because of barriers to engaging with empirical evidence, including limitations on what empirical evidence is brought before the court, compounded by the point-in-time nature of judicial assessments that cannot account for the way risk-benefit ratios may change over time.
Legislative intervention criteria for the historical analysis
The following principles are derived from the preceding ethical and legal analyses and provide criteria for assessing legal interventions to improve vaccination coverage:
Efficacy: does the available evidence suggest that the intervention is likely to be effective in achieving its objective?
Necessity: will it achieve an important objective?
Proportionality: will the benefits of the intervention outweigh the burdens?
Equity: are the benefits and burdens distributed fairly?
Least-restrictive alternative: are burdens minimised while retaining efficacy?
Public acceptability: will the population to whom the intervention will be applied find it broadly acceptable, and are there mechanisms for facilitating public feedback and procedural fairness?
The following section explores the history of vaccination legislation in Australia and applies these intervention criteria with the aim of identifying features of continuity and discontinuity in the legislative responses to the vaccination problem. The search for continuity and discontinuity is a topos in the historiographical literature that is mirrored in legal history scholarship. 39 Drawing on the theory of scientific revolution that explains the history of science through changes of paradigm, 40 this literature advances an evolutionary, albeit nonlinear, vision of history in terms of ‘challenge and response’, 41 in which discontinuous elements – whether internal or external – disrupt the evolutionary direction. 42 This structure of continuity and discontinuity is frequently employed to explain the origin of Western legal tradition and to frame comparisons among European legal systems. 43 Here, we use it to understand the history of vaccination legislation and to provide insights into potential future developments.
Australian vaccination laws
The importance of assessing legislative interventions for public health in context is emphasised in the ethical and legal literature. The Nuffield Council on Bioethics notes that evaluation of a given intervention requires consideration of ‘the interaction of many factors that affect the vaccination uptake rate, including cultural, historical, political and social factors that influence attitudes towards healthcare, as well as educational background and the accessibility of health services’. 44 Without this context-specific evidence, arguments for or against the justifiability of particular interventions will rely on assumptions about the likely effects of these interventions.
To this end, this section takes the legislative intervention criteria derived earlier and applies them to the history of vaccination laws from self-government to the present. First, the legislative landscape was systematically mapped. For each jurisdiction, legislation meeting the definition of Australian vaccination law, here defined as ‘a legislative measure capable of influencing human vaccination uptake in Australian jurisdictions’, was identified, and key provisions were then tabulated by year across each jurisdiction. These jurisdictions include the five Australian states (previously colonies) and two territories, plus the Commonwealth, following Federation. 45
The resultant dataset was then inductively coded, with reference to supporting documentation, such as explanatory memoranda and second reading speeches, where required to determine the objectives of particular laws. The resultant themes aligned with three clear phases plus an emergent fourth, each characterised by a distinctive approach to encouraging vaccination: experiments with compulsory vaccination in the self-governing colonies (1853–1900); the introduction of conscientious objection in the newly federated nation and an emphasis on provision of community immunisation rather than compulsion (1901–1995); and a return to legislative interventions aimed at reaching the levels required to achieve herd immunity prior to the pandemic (1996–2020). The emergent fourth phase, from the pandemic to the present, is examined separately in Part IV.
Next, each period was written into a historical narrative, situating the legislative interventions in their social, political, and cultural context and drawing on primary (e.g. parliamentary papers, contemporary newspaper reports) and secondary (e.g. historical, epidemiological, and social sciences literature) sources. These narratives were then deductively coded using codes derived from the ethical and legal literature and categorised against the legislative intervention criteria. The following three subsections present a summary of the findings of this process, structured to reflect the legislative intervention criteria, while the fourth subsection draws together the key themes to identify targets for law reform.
Self-government, compulsion, and the limits of state intervention (1853–1900)
Faced with outbreaks of smallpox and inadequate voluntary vaccination among the public, some of the newly self-governing Australian colonies’ earliest statutes included compulsory vaccination legislation, requiring parents to get their infants vaccinated or face a fine up to £5 (see Figure 1). 46 Characterised by experiments in compulsion, this period is significant for revealing access, equity, and public trust as the three key preconditions for public compliance with vaccination mandates.

Vaccination laws, 1853–1900.
The main objective of vaccination legislation was to control disease outbreaks through mass vaccination. Despite early suggestions that the colonies were merely copying English legislation in a ‘mechanical and unenlightened’ manner, 47 contemporary accounts make it clear that the colonial legislatures were actually responding to local conditions, events, and people. For example, a smallpox outbreak in Sydney in 1853 impelled Hobart doctor Edward Bedford to appeal to the Lieutenant-Governor of Van Diemen’s Land for a state-sponsored vaccination programme and to suggest that ‘it would be desirable to have an enactment in this Colony’ similar to the English Vaccination Bill under consideration at that time. 48 Bedford had hoped that increasing public awareness of the availability of vaccination would be sufficient to address the very low rates of vaccination and was disappointed when this failed to happen. In his view, compulsory legislation would send a signal that would overcome the ‘carelessness’ of some parents and bring to their attention the value of an overwhelmingly positive intervention. 49
Notably, New South Wales (NSW) never passed compulsory vaccination laws, despite experiencing the greatest incidence of smallpox in the colonies.
50
Vaccination rates in NSW, as elsewhere, were governed by cycles of panic and apathy, with high rates when smallpox was present in the colony and negligible uptake when it was absent.
51
When asked to investigate whether compulsion would overcome this pattern, Registrar-General Christopher Rolleston expressed his belief in the value of vaccination but also noted its limitations in the local context:
. . . the advantages of compulsory Vaccination Laws in old and thickly populated countries are indisputable, [yet] it does not follow that they would operate with like success in a widely scattered population like ours, and I confess that I see so many difficulties in the way of applying the system to the peculiar features of this country, that I cannot venture to recommend its adoption as a general measure, under present circumstances.
52
He noted that the implementation of the Victorian legislation had been very successful in urban areas, but difficulties had arisen in more sparsely populated areas, and that these difficulties would be exacerbated in NSW, where the necessary resources and infrastructure were not available. 53 Rolleston’s arguments, founded on practical consideration of local conditions, resulted in NSW being one of the few Western governments not to at least attempt to mandate vaccination.
The Australian colonies, except for NSW, thus introduced compulsory vaccination laws with the objective of achieving sustained vaccination uptake, irrespective of the presence of smallpox.
Efficacy and necessity
The effectiveness of colonial efforts in encouraging vaccination varied significantly. Although Victoria was able to maintain consistently high vaccination rates through effective administration of the Act, rates in the other colonies continued to be subject to cycles of panic and apathy. 54 Part of the reason for the discrepancy between colonies is the effect of the gold rush in the 1850s on public health, which caused the Victorian government to establish Central and Local Boards of Health and to enact public health legislation well in advance of the other colonies. 55 Victoria’s success was largely a consequence of its investment in effective bureaucratic structures for identifying and prosecuting cases of non-compliance and the consistency with which the Victorian Acts were enforced.
By contrast, the other colonies enforced compulsory provisions only sporadically, usually during outbreaks or smallpox scares, and no mechanism was instituted for enforcement beyond periods of emergency. 56 This inconsistency contributed to resistance during periods when smallpox was absent; unlike in Victoria, vaccination was never normalised as a routine operation, and when there was no immediate threat, it seemed rational not to risk potential harm from the vaccine.
Furthermore, most of the colonies had difficulty maintaining supplies of vaccine lymph. Cumpston drew attention to the particularly poor rates of vaccination in Western Australia (WA), Queensland, and NSW, observing that ‘the gravity of the position rests upon the fact that these are the three States which are most often attacked from without by small-pox’.
57
This was not always the result of a lack of political will: in WA, for example, the minister responded to recommendations from the Central Board of Health in 1874 for improving vaccination rates by noting that:
These provisions seem unobjectionable, nay absolutely necessary, to insure the completeness of such a measure as general vaccination, subject, however, to this remark: The Colony is sparsely peopled, and on long lines of road on vast areas of country. Doctors are very rare. Will not mere distance be almost an insuperable obstacle? . . . How can it be enforced?
58
While there was broad agreement as to the necessity of measures to ensure high vaccination coverage, the efficacy of compulsion depended upon the ability to facilitate access (i.e. supplies of lymph and doctors to perform the operation), the development of effective bureaucratic structures for administration, and consistency of enforcement.
Proportionality, equity, and least-restrictive alternative
The effects of evolving legislative efforts to increase vaccination rates were experienced unevenly, as the vaccination debate inevitably intersected with aspects of class, gender, and race identity. Working class families were associated with a reluctance to vaccinate, frequently attributed by the medical profession to ignorance, best remedied through education and compulsion. The penalty for non-compliance was a fine, and as Victorian parliamentarian James Hume Cook noted:
These penalties were a tremendous infliction upon the poor of the colony. Those who could afford to pay continued to pay, but those who could not afford to pay were imprisoned under the 329th section of the Act.
59
More specifically, it was working-class women who were blamed for poor vaccination rates, and the legitimacy of their concerns was consistently undermined. As Francis Campbell, the Superintendent of Vaccinations in NSW, complained: ‘The legitimate cause is ignorance – primordially the ignorance of mothers’. 60
Anti-vaccinationists commonly alluded to their rights as English subjects, using the language of citizenship to bolster claims to conscience and autonomy. 61 This contrasts with the way vaccination was experienced by those framed as non-citizens, most notably Aboriginal and Chinese communities. While there is some evidence of efforts to offer vaccination to Aboriginal people, particularly during outbreaks, it appears to have occurred infrequently. 62 Systematic efforts to reach Aboriginal populations were not made by governments, and no legislation addressed the issue of vaccination of Aboriginal and Torres Strait Islander peoples specifically, in contrast to the United States’ approach to Native American peoples. 63 This was partly because Aboriginal communities were not viewed as a significant source of infection and exacerbated by contemporary views of Aboriginal people as an inferior and ‘dying race’. 64
Chinese immigrants, by contrast, were the subject of a number of targeted legislative provisions, including restrictions on immigration, compulsory vaccination, and severe quarantine requirements upon arrival. 65 Chinese people were depicted throughout this period as carriers of disease, as unsanitary, and as being unable to integrate into colonial society. 66 Significantly, vaccination policies for Chinese immigrants were tightened at the same time as opposition to compulsory vaccination strengthened among the European population. That is, as public debates emphasised the importance of the rights of individuals to make decisions about their own bodies, this very right was denied to the Chinese population, framing them as non-citizens and outside society.
The effects of early compulsory vaccination laws were highly inequitable, which worked to undermine the efficacy of the laws as an intervention to improve vaccination coverage. As public opposition to compulsion swelled towards the fin de siècle, it was increasingly clear that the benefits of these laws were not outweighing the burdens, and that alternative approaches were necessary.
Public acceptability
The acceptability of colonial compulsory vaccination laws can be gauged by the evolution of the legislation. Throughout the second half of the nineteenth century, colonial governments amended their respective vaccination legislation. Early amendments comprised administrative adjustments, such as defining proof of vaccination 67 or providing for the appointment of district vaccination officers, 68 in response to difficulties encountered in achieving the goals of the legislation and reflecting increasing administrative maturity. Some amendments reflected the growing body of vaccination case law developing in Victoria, which cumulatively described the incremental working-out of administrative and logistical issues in a novel public health intervention. 69 The brevity of the earlier statutes gave way to increasing detail and complexity, peaking just prior to the end of the century, by which stage it was relatively standard to offer a choice of glycerinated calf lymph (a comparatively hygienic source of vaccine material), to include detailed exemptions for medical conditions or insusceptibility, and to make provision for data collection as a basis for prosecutions for non-compliance and for informing government decision-making. 70
The depth of detail in these amendments hints at the rise of more organised opposition to compulsory vaccination during this period. Even in Victoria, cracks were beginning to appear as anti-vaccinationist sentiment gained momentum and vaccination rates dropped. By the end of the nineteenth century, it was evident that compulsory vaccination legislation on its own was insufficient to meet the vaccination goals of the colonial governments, and that compulsion was increasingly politically counterproductive. One by one, amendments were passed introducing conscientious objection clauses – first in Tasmania in 1898, followed by Queensland in 1900, South Australia in 1901, WA in 1911, and finally Victoria in 1919 – rendering vaccination legally (and not just practically) optional in all Australian jurisdictions. 71
Immunisation in a new nation (1901–1995)
Informed by the experiences of the previous century, Australian legislators increased their focus on encouraging public trust in vaccination, by improving access, efficacy, and safety. The twentieth century saw a proliferation of vaccines against a wide range of diseases, including tetanus, diphtheria, pertussis, polio, measles, rubella, and mumps, among others. Despite – or perhaps because of – the increased availability of preventive measures against many of the most prevalent and life-threatening diseases, there were no legislative efforts overtly compelling vaccination during this period. Rather, government efforts during this period focused more on developing infrastructure for the provision and regulation of vaccines (see Figure 2).

Vaccination laws, 1901–1995.
This was achieved through legislating for the establishment of the Commonwealth Serum Laboratories (CSLs), for the local development and production of vaccines; the establishment of the National Health and Medical Research Council (NHMRC, formerly Federal Health Council), providing strategic direction; and the introduction and refinement of national bodies, processes, and standards for safety and efficacy through the Therapeutic Substances Act 1953 (Cth) and, later, the Therapeutic Goods Act 1989 (Cth). 72 Towards the end of the twentieth century, these strategies were supplemented by the implementation of the National Immunisation Strategy and by data collection facilitated by state and territory legislation requiring documentation of immunisation status for enrolment in primary schools and childcare centres, as well as jurisdictional vaccination registers in Queensland and the Northern Territory. 73
The key objectives through this period appear to be ensuring access to safe and effective vaccines, thereby indirectly encouraging vaccination. Towards the end of the century, there was increased attention given to the need for reliable data on vaccination coverage, both to support these objectives administratively and to assess the efficacy of this less-intrusive approach.
Efficacy and necessity
Regulation of vaccines to ensure safety and efficacy was clearly necessary for sustaining public trust, as the 1928 Bundaberg tragedy in Queensland demonstrated. Eighteen children vaccinated from a contaminated batch as part of a diphtheria immunisation campaign fell ill, and 12 died. 74 Diphtheria immunisation was effectively halted for the next 2 years, and it took years to rebuild public confidence. Importantly, it resulted in significant tightening of protocols for ‘testing, dispensing, labelling, packaging, and issuing products’ and contributed to the development of an improved vaccine and use of single-dose vials. 75
The Second World War provided the conditions for some restoration of public trust in vaccination. Mass vaccination of Australian troops against tetanus was outstandingly successful, and effective influenza vaccines were being produced in Australia for Australian and British troops within a year of being developed in the United States. 76 These experiences demonstrated efficacy and safety on a broad scale and cemented CSL’s place in vaccine development and production.
It also became increasingly evident that access was a continuing issue in vaccination uptake. Vaccine schedules and pricing varied across the country, and this fragmentation contributed to inefficiencies, disparities, and gaps in coverage that allowed repeated outbreaks of diseases such as pertussis and measles. 77 In 1989–1990, surveys indicated that only 53% of children were adequately immunised. 78 This led to the introduction of the National Immunisation Strategy in 1993. The Strategy established a common schedule and consistent vaccine pricing across all jurisdictions, identified distinct responsibilities for the different levels of government through the National Childhood Immunisation Agreement, improved recording and reporting of coverage, and enhanced coordination between public and private care providers. 79
The focus for achieving target vaccination rates centred on ensuring sufficient access through community and mass vaccination campaigns. Community vaccination programmes focused on periods of increased contact with health practitioners, such as early childhood and pregnancy, or delivery through schools. 80 The facilitative approach taken during this period was effective in earning public trust and achieved good voluntary rates of vaccination, albeit insufficient coverage to reach herd immunity.
Proportionality, equity, and least-restrictive alternative
Throughout this period, vaccination was encouraged but entirely voluntary, imposing minimal burdens. While this facilitative approach was effective in getting vaccination to a point where it was widely trusted and reasonably normalised, it was ineffective in achieving coverage sufficient to prevent outbreaks of vaccine-preventable diseases. Notably, the National Immunisation Strategy explicitly ruled out compulsory vaccination, endorsed a role for conscientious objection, and characterised the role of schools as a mechanism for identifying and protecting unimmunised children in the event of an outbreak, rather than as a site for policing immunisation. 81 This meant that the burden was then shifted to those who were vulnerable but could not be vaccinated, such as immunocompromised individuals or infants too young to be vaccinated, to exclude themselves from social activities during outbreaks.
Public acceptability
To some extent, legislative interventions to encourage vaccination were not necessary during this period: the public were generally disposed towards vaccination and optimistic about its potential to curb the ravages of common diseases, especially those affecting children. 82 The risks of contracting endemic diseases such as poliomyelitis were so present and immediate that people placed substantial faith in the promise of vaccines, even when there were good reasons to have doubts about its safety and efficacy. For example, in 1955, the demand for a polio vaccine was so great that even a disaster of the scale of the US Cutter vaccine – in which live virus present in a batch of vaccine resulted in 204 cases of poliomyelitis – had little impact on public appetite for polio vaccination. 83
The twentieth century was a period of exceptional progress in terms of both the proliferation of available vaccines and the consequent reduction in mortality and morbidity from vaccine-preventable diseases. Through the establishment and support of the CSL for development and production of vaccines, the improvement of delivery of vaccinations through community and school-based programmes, and expansion of regulatory mechanisms relating to vaccines and other therapeutic substances, Australian governments worked to improve access, efficacy, and safety in relation to vaccines. 84 These efforts, combined with the threat posed by these preventable diseases, were sufficient to achieve considerable vaccine coverage for much of this century.
However, by the mid-1990s, concern was growing about suboptimal vaccination coverage and the re-emergence of diseases such as measles, pertussis, and rubella. 85 Having laid strong foundations for public trust by developing a regulatory environment that facilitated good access to safe and effective vaccines and a system of data collection that indicated the need for more to be done to improve vaccination coverage, the mid-1990s marked a pivotal transition in vaccination laws from a focus on provision, efficacy, and safety to more concerted efforts to increase uptake.
Achieving herd immunity in a low-disease environment (1996–2020)
The period from 1996 to the outbreak of the coronavirus pandemic witnessed a surge in vaccination legislation, creating a system of increasing integration and sophistication (see Figure 3). This period was characterised by the development of systems for the collection, management, and analysis of data as a basis for increasingly interventionist approaches to promoting vaccination uptake.
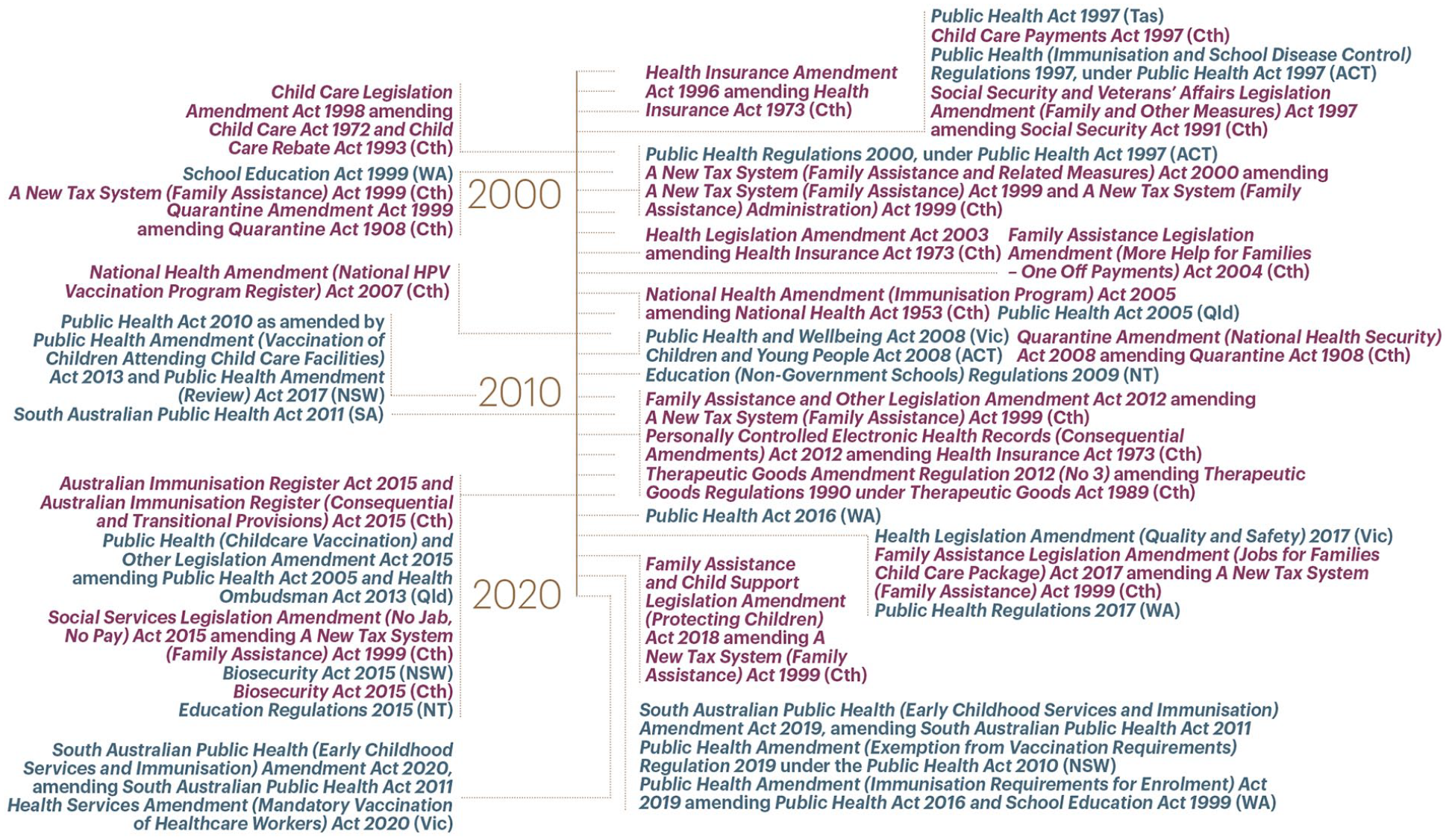
Vaccination laws, 1996–2020.
Central to this programme was the establishment of the Australian Childhood Immunisation Register (ACIR), the world’s first comprehensive and purpose-built national childhood immunisation register, which replaced the incomplete and unwieldy system of regional registers and periodic national household surveys. 86 Then, in 1997, the Immunise Australia Program was launched, with a Seven-Point Plan to improve Australian childhood vaccination coverage, comprising:
Initiatives for parents, including a maternity immunisation allowance (MIA) and childcare rebate for fully immunised children; 87
An expanded role for general practitioners, supported by incentives for each immunisation encounter and practice-based coverage outcomes;
Monitoring and evaluation of targets using ACIR;
Immunisation days;
Measles eradication through a school-based Measles, Mumps and Rubella (MMR) programme;
Education and research, including the establishment of the National Centre for Immunisation Research and Surveillance (NCIRS); and
Immunisation requirements for school entry. 88
This approach was consolidated in the mid-2000s with the expansion of the ACIR to capture vaccinations received overseas, 89 removal of means-testing from the MIA, 90 and amendment of the National Health Act 1953 to bring vaccine funding decisions within the established and trusted framework used for medications. 91
Subsequently, however, legislative interventions took a markedly more restrictive turn: several states introduced ‘no jab, no play’ policies, preventing children from attending childcare unless fully vaccinated, in a catch-up programme, or medically exempt; and the Commonwealth introduced a national ‘no jab, no pay’ policy in 2016, further tightening vaccination requirements for family assistance payments by removing the conscientious objection exemption. 92 This was complemented by the extension of the ACIR, first up to 20 years of age, and then to include all immunisations across the lifespan, in the renamed Australian Immunisation Register (AIR). 93
The dramatic shift from a focus on provision, efficacy, and safety to more concerted efforts to increase uptake was motivated by the failure of the voluntary system to prevent outbreaks of vaccine-preventable diseases. As the Federal Minister for Health and Family Services, Dr Michael Wooldridge, explained in relation to ACIR, this shift aimed to:
address the continuing epidemics of vaccine-preventable diseases which indicate that current immunisation rates are not sufficient to prevent the transmission of diseases such as measles, whooping cough and rubella.
94
Similarly, the Seven-Point Plan was explicitly linked to the objective of achieving higher than 90% coverage ‘to stop epidemics’ 95 , and the ‘no jab, no pay’ policy ostensibly ‘aimed at boosting childhood immunisation rates’ to ‘reinforce the importance of vaccination in protecting public health’. 96 Despite these aims, this period demonstrates that when equity and access are not assured, coercive interventions are less effective and may act to erode public trust.
Efficacy and necessity
Good data are crucial to health policy, and ACIR rapidly demonstrated its efficacy. It served to counteract the decentralised nature of vaccination provision by linking vaccination data, including conscientious objections (until 2016) and medical contraindications, sent by immunisation providers to data uploaded nightly from the Medicare database (from 2003), enabling nearly complete records to be maintained. 97 Furthermore, initiatives in 2003–2004 led to significant improvements in the recording of Indigenous status (from 42% in 2002 to 95% in 2005), consequently increasing confidence in estimates derived from ACIR data and providing a better basis for programme and policy planning. 98 The detailed and comprehensive nature of this dataset also supports the delivery of vaccination programmes either nationally or targeting identified regions or populations, as well as facilitating recall and reminder letters to individuals and enabling opportunistic vaccination.
The system of incentives under the Seven-Point Plan led to improvements in coverage, when combined with complementary initiatives. The effect of these comprehensive interventions was to dramatically increase vaccination coverage, rising to over 90%. 99 In particular, the MIA, worth over $200 per child, was considered ‘substantial enough to provide motivation both to complete immunisation and for parents to prompt their provider to notify any outstanding reports to the ACIR before the child reaches 24 months of age’, thereby contributing to increases in vaccination coverage and the completeness of the ACIR. 100
After initial dramatic improvements in childhood vaccination coverage between 1997 and the early 2000s, coverage was very high by international standards but continued to fall short of the national target of 95%. 101 The more restrictive penalties-based approach of the ‘no jab, no pay’ programme removed conscientious objection exemptions and ostensibly aimed to further increase coverage. Subsequent evaluations found its implementation was associated with increased coverage, with greater effects in lower socioeconomic areas and among Indigenous children than among non-Indigenous children. 102 Indeed, the figures for the annual period ending March 2021 show Australian children entering their first year of school had, as a cohort, achieved herd immunity for diseases with vaccines listed on the National Immunisation Program Schedule (NIPS). 103
These figures appear to provide compelling evidence that this suite of measures was effective. However, an analysis by Attwell and colleagues found ‘vaccine coverage continued on its positive trajectory without any conclusive evidence of the impact of mandatory policies’ and instead emphasised ‘the contribution of pre-existing and parallel nonmandatory interventions employed by Australian governments to the country’s enhanced coverage’. 104 A more recent study by Beard and colleagues used individual-level data to further interrogate the impact of the ‘no jab, no pay’ policy and found that the increased stringency of this approach was associated with modest increases in vaccination uptake, likely attributable to the additional eligibility assessments more than the removal of non-medical exemptions. 105 These findings point to the value of epidemiological assessments of legal interventions to avoid overly-simplistic correlative interpretations of trends.
Furthermore, there remained concerns about timeliness given that target coverage was not achieved until school age. This is particularly evident when regional variation is examined: for example, only 87.09% of 2-year-old Aboriginal and Torres Strait Islander children are fully vaccinated in WA, and just 90.78% of all 2-year-olds in Tasmania are fully vaccinated. Furthermore, there are wealthier regions with even lower rates that are identifiable when AIR data are analysed at more granular levels. 106
Thus, although this system has achieved the highest vaccination coverage in Australian history, much of its success is likely due to pre-existing interventions, and coverage remains uneven, with stubborn pockets remaining in predominantly higher socioeconomic areas where herd immunity has not been achieved. 107
Proportionality, equity, and least-restrictive alternative
The complexity of the multifaceted system developed during this period makes it challenging to assess. Nevertheless, it is clear that some aspects of this more integrated and sophisticated data-driven system serve to minimise burdens. For example, rigorous assessments of efficacy and safety are built in at multiple stages: technical advice from the Australian Technical Advisory Group on Immunisation informs the Pharmaceutical Benefits Advisory Committee’s decision whether a vaccine will be listed on the National Immunisation Program (NIP) or Pharmaceutical Benefits Scheme, and ongoing monitoring for safety is undertaken by NCIRS and Therapeutic Goods Administration (TGA). 108 Furthermore, this funding model is predictable and transparent, decision-making is based on considerations of equity of access, and high-quality clinical and public health evidence is built into the process. 109
Other aspects, however, demonstrate highly inequitable outcomes. Soon after general practitioner incentive payments for childhood and human papillomavirus (HPV) immunisations were discontinued, the MIA was split into two payments (for a fully immunised child at 18–24 months and then at 4–5 years) in 2009 and then replaced in 2011 with the linking of childhood immunisation status to the existing Family Tax Benefit Part A supplement.
110
This change marked a significant turning point: from incentives to penalties, and from a policy that included all families to one that affected under three-quarters of families, with significant implications for the distribution of benefits and burdens, as noted by Ward and colleagues:
For many of these families, there will now be more dollars at stake for ensuring that their children are fully immunised. For the minority of higher income families who are not eligible for this and/or the childcare-related benefits, there will no longer be any financial incentive for immunisation. Other families will be eligible for some or all of these incentives in part but, due to means-testing of these payments, the dollar value may not be as great as that previously provided by the MIA.
111
That is, while these policy changes were likely to continue to encourage vaccination uptake, the effect would be less equitable and less likely to influence hesitant parents in higher-income areas.
Significantly, the human rights assessment for the ‘no jab, no pay’ legislation did not consider issues of equity. It found the Bill to be compatible with human rights because although rights to freedom of thought, conscience, and religion and to social security were potentially limited, these limitations were asserted to be necessary and proportionate to advance the legitimate aim of protecting the right to health. 112 At the time, experts worried that financial penalties of this kind were unlikely to have the desired effect, given (a) they targeted vaccine refusers rather than the numerically more important vaccine-hesitant parents, and (b) there was a lack of evidence supporting their introduction, compared to interventions facilitating access. 113 Commentators also noted that this policy was forecast to result in savings of more than $500 million, of which only $26 million was earmarked for immunisation programmes. 114 This is particularly notable when contrasted with the cost of vaccination incentives under previous policies and perhaps indicates that economic considerations were more influential in this instance than considerations of equity or efficacy.
The effect of this policy was disproportionately experienced according to socioeconomic status. 115 This is ethically problematic: it disproportionately infringes the autonomy of less well-off families; children of vaccine refusers miss out on both the vaccination and financial support from the government; and it perpetuates disadvantage, and distrust of the government and health system, while distracting from established barriers to vaccination, including problems relating to access. 116 Furthermore, this policy failed to target the major group contributing to vaccine-refusal rates, was unlikely to have an effect on many vaccine objectors, and removed the incentive to engage in discussions with health practitioners while increasing pressure on practitioners to provide unjustified medical exemptions. 117
In addition to the troubling consequences of the ‘no jab, no pay’ legislation, the inability of existing systems to encourage adult vaccination was identified as a key failing. 118 Of particular concern are international travellers, migrants, and refugees who have not received recommended vaccines and may introduce disease on entry to the country; adolescents and adults who fail to receive NIPS vaccinations, especially in high-risk categories; and people recommended to receive occupational vaccines, such as healthcare workers, laboratory personnel, and people in childcare and aged care. 119 The extension of the ACIR to record vaccinations administered across the lifespan was an important initial step, but the removal of incentives for health practitioners and the lack of additional strategies for encouraging adult vaccination means this remains a live issue. For all these reasons, the penalties-based system is not the most-effective/least-restrictive alternative and is further undermined by producing highly inequitable outcomes.
Public acceptability
This period is noteworthy for its very high vaccination rates and the increasing complexity of the regulatory frameworks supporting this achievement. In many respects, Australia has been a world-leader in the encouragement of vaccination compliance, and the efficacy and acceptability of the system appear to be borne out in the statistics captured by the AIR.
Changes implemented during this period contributed to, and combined with, bipartisan support for vaccination, strong public confidence in vaccination, and generally pro-vaccination media coverage to form a relatively robust position for vaccination in Australia. 120 This became evident after Wakefield and colleagues published a case series in the Lancet suggesting a causal association between MMR vaccination and autism. 121 Although the study was subsequently fully retracted by the journal for research integrity concerns, MMR vaccination rates in the UK dropped significantly in the wake of publicity around Wakefield’s work. 122 In Australia, however, MMR coverage remained unaffected, which commentators have attributed to the systems and culture that had developed around vaccination in Australia just prior to this controversy, supported by good public communication and engagement. 123
However, while it is evident that the comprehensive national system that has developed over the last few decades for the provision and monitoring of vaccination in Australia has contributed to a situation where routine childhood vaccination is normalised and broadly accepted by the public, it is not universally accepted. There is, for example, evidence that individuals who might previously have sought exemption on the basis of conscientious objection sometimes sought medical exemptions from sympathetic medical practitioners to avoid vaccination but remain eligible for the associated benefits. 124
In the last few years, mandates by employers have been introduced for vaccination against influenza within high-risk workplaces, such as healthcare, aged care, and childcare. In a number of instances, employees who have refused vaccination, either for medical or personal reasons, have had their employment terminated and subsequently applied to the Fair Work Commission for an unfair dismissal remedy, although none has yet been successful. 125 Together, these cases indicate a willingness of tribunals to find mandatory vaccination lawful and reasonable in the context of operations involving potentially vulnerable individuals from a relatively superficial analysis that does not involve much in the way of empirical evidence or considerations beyond the medical perspective. As Lake DP concluded in Barber v Goodstart, ‘the Employer made a logical and legal analysis of the risks and hazards in the workplace, developed a response and implemented a policy to target that risk’. 126 More recently, however, a Queensland case found COVID vaccination mandates for police officers were unlawful because it was ‘more likely than not that the [Police] Commissioner did not consider the human rights ramifications’ of the mandate. 127 This judgement does not find that the mandates were inconsistent with the Human Rights Act 2019 (Qld), but rather that incorrect procedure was followed in failing to consider the human rights implications, suggesting that this case is unlikely to indicate that courts may find future mandates unreasonably restrictive.
The experiences of the minority seeking to avoid vaccination highlight the failure, at various points in the legislative lifecycle, of legal scrutiny to effectively engage with the principles of necessity, proportionality, and justice, as a consequence of the challenges involved in integrating empirical evidence into legal reasoning and the resultant deference to medical orthodoxy. As a result, the law provides little protection against the overly-restrictive vaccination policy.
Legislative interventions in context: key themes
The history of vaccination legislation in Australia reveals a number of factors important for improving vaccination coverage: ensuring equitable access and minimising barriers to access; enhancing public confidence through trustworthy regulatory systems that ensure safety and efficacy of vaccine technologies; and if the preceding conditions are in place, interventions to incentivise or compel vaccination may be effective, but care must be taken to ensure the effects of such interventions are experienced equitably across the population.
This analysis highlights that the existing legal mechanisms, either in the form of human rights compatibility statements or legal challenges to specific mandates, are not well-suited to applying these criteria in a meaningful way. This is at least partly because of a siloing of law and epidemiology, such that epidemiological evidence is not brought before the court by the parties, or the litigation is not framed in terms that require the court to consider epidemiological evidence. Even when it is, the problem remains as to how to integrate empirical evidence into legal analysis, as well as to account for changing risk-benefit ratios as evidence for the efficacy and impact of a particular intervention emerges over time. This gap could usefully be filled by legal epidemiology, in which epidemiological methods are used to empirically assess the effect of legal interventions on public health. 128 Bringing together legal and public health scholars, this approach bridges the gap between disciplines in a way that would be more useful to courts trying to evaluate the impacts of specific laws on human rights. 129 Requiring interventions that restrict rights to undergo rigorous post-implementation evaluation of this kind may help mitigate the problems identified in this section, by normalising empirical assessment of the effect of laws and increasing the accessibility of such evidence. Moreover, when applied to law-making, this approach makes it possible to shape legislative interventions designed to enable an open exchange between scientific, ethical, and social components that, by responding adaptively to changes in both the scientific and the ethical or social paradigm, enjoy greater legitimacy.
The response to the pandemic
While the effects of the coronavirus pandemic continue to unfold, it is difficult to analyse the period from 2020 using the aforementioned methods. Nevertheless, the recurrent themes of access, trust, and equity are readily apparent in the Australian response to COVID-19 as vaccines became available. While a well-established regulatory system was already in place, the pandemic combined high urgency with significant uncertainty and evolving information to present a situation that strained the existing regulatory system, requiring both empirical data and considered communication strategies to secure public trust. 130
With the development of effective vaccines came a focus within the global public health agenda on mass vaccination. Within this context, Australia’s focus on vaccine sovereignty led to significant procurement issues. 131 The government opted to initiate the mass immunisation campaign by rolling out the AstraZeneca vaccine over other available vaccines because it could be produced in Australia and so neglected to order adequate supplies of the Pfizer vaccine. 132 As evidence of adverse reactions to the AstraZeneca vaccine emerged, confusing communications from health authorities and the unavailability of alternative vaccines hindered the rollout. Consistent with earlier experiences, these events highlighted that when safety, access, and equity could not be assured, public trust plummeted, and voluntary vaccination failed to reach the necessary targets.
Although no general mandate for COVID-19 vaccination was introduced at the federal level, all states and territories adopted some mandatory vaccination requirements for specific targeted groups and for limited periods. 133 As Wilson and Rudge have described, Tasmania, Queensland, and South Australia introduced obligations for health care and aged care workers, while NSW and Victoria introduced vaccine mandates combined with a vaccine passport system. 134 These mandates were implemented in the context of a progressive reopening linking the relaxation of restrictions to the achievements of specific targets. These interventions were introduced in an environment of rapidly evolving knowledge about the disease, its effects, public attitudes, and the efficacy and long-term effects of vaccines and other interventions. These uncertainties presented significant challenges for maintaining public trust in vaccination, as well as for meeting goals for access and equity.
Problems with access and equity particularly affected those in rural and remote areas. However, the proactive response to the pandemic led by Aboriginal Community Controlled Health Organisations (ACCHOs) was highly successful in protecting remote Aboriginal communities from outbreaks and preventing infections and deaths. 135 ACCHOs were also able to influence the success of the vaccination rollout to some extent, and as Soares and colleagues observed, ‘In the face of unilateral, centralised, and poorly coordinated measures adopted by governments in Australia, ACCHOs were capable of promoting self-determination through community-based interventions’. 136 The success of partnership models that facilitated local Indigenous leadership highlighted the importance of flexible and dynamic approaches in achieving high engagement and increasing community trust. 137 This is particularly significant in light of the devastating and ongoing legacy of colonisation which has, as Matthews and Menzel argue, created a context in which implementation of mandatory vaccination policies would result in structural injustice and exacerbate existing mistrust and avoidance of health services. 138 They advocate for future programmes founded on cultural safety, humility, and responsiveness, with an emphasis on self-determination, as was demonstrated to be successful during the pandemic. The need for such a trust-based approach is most acute in communities already structurally disadvantaged, but the underlying principle is more widely generalisable.
Regulation is an important mechanism by which the trustworthiness of vaccines may be created and maintained. Although historically the Australian health regulatory system enjoys high levels of public trust, the pandemic revealed some weaknesses. An example of this is the way the pandemic revealed shortcomings in the ethical review and monitoring of clinical trials. This role is fulfilled by Human Research Ethics Committees (HRECs).
When reviewing applications, HRECs tend to be very risk-averse, which can lead to situations where vulnerable groups, such as pregnant women or Aboriginal and Torres Strait Islander peoples, end up excluded from some types of research because of the perceived difficulties of undertaking research with those groups or of obtaining ethical approval to undertake research involving those groups. 139 Regardless of intention, this can have far-reaching consequences. For example, early clinical trials for COVID-19 vaccines did not include pregnant or lactating women, and so initially the WHO cautioned pregnant women against receiving the vaccine unless at high risk of exposure. 140 Women aged 30–39 years were identified as the most COVID-19 vaccine-hesitant group in Australia. 141 Although guidelines for inclusion of pregnant women in vaccine development were recently released, 142 individual HRECs are not always up to date with these developments and are poorly placed to push back against applications for international multisite clinical trials.
The implementation and monitoring of COVID-19 vaccination outside clinical trials was largely a continuation of existing systems. While COVID-19 vaccines are not currently part of the National Immunisation Program Schedule, and thus not linked to family assistance payments, the need for accurate data relating to COVID-19 vaccinations led to amendments to the Australian Immunisation Register Act 2015 (Cth), introducing mandatory reporting of all COVID-19, influenza, and NIP vaccinations. 143 These improvements to data collection have been welcomed and also criticised for not going far enough. 144 In particular, the AIR lacks the ability to monitor at-risk groups so that targeted educational or access interventions can be implemented, undermining efforts to improve equity systemically.
The pandemic also highlighted the troublesome absence of an equitable system to redress harm following vaccination and revived the debate on vaccine injury compensation schemes. Although Australians suffering from vaccine injuries can access medical care subsidised by Medicare and disability supports through the National Disability Insurance Scheme, significant gaps remain, and a serious adverse event may result in ongoing financial, physical, and psychological burdens for the affected family. Furthermore, these schemes do not address suffering caused by a vaccine-induced death. Compensation for harm may also be sought through an action in tort law, although this does not provide an equitable solution given the costs involved in pursuing such an action, as well as the uncertainty in outcome and payout, particularly where there is no obvious negligent party. 145 This issue is particularly acute, given that contracts with suppliers of COVID-19 vaccines included indemnity against legal claims arising from vaccine injury. 146
No-fault vaccine injury compensation schemes offer a solution to this problem. 147 The human rights case law and ethical and social sciences literature all strongly suggest that in the current Australian context, a general system of compensation for vaccine injury is warranted, and commentators have consistently called for its introduction. 148 In spite of this consensus, no such scheme was introduced until the pandemic. Partially the result of a political compromise after the rejection of the anti-mandate legislation at the senate, 149 the COVID-19 Vaccine Claims Scheme came into being under exceptional circumstances to redress the harm caused by the newly developed vaccines with no clear aspiration for continuing beyond the pandemic. 150
The scheme provides compensation to individuals who suffered harm as a result of receipt or administration of COVID-19 vaccine on or after 22 February 2021 until September 2024. 151 The limited scope and the temporary nature of the scheme undermine its efficacy, considering that the materialisation and final diagnosis of harms from vaccination may take time. 152 Furthermore, the scheme has significant exclusions, including psychological or psychiatric harm and secondary injuries. The narrow range of the COVID-19 compensation scheme, coupled with the absence of any equitable measure to redress harms resulting from the other vaccinations listed in the NIP, leaves a key equity issue unresolved. Once the regulation for the COVID-19 compensation scheme currently in force comes to an end, the pandemic will represent a major missed opportunity to enhance the equity of Australia’s vaccination programme.
Despite an unpromising beginning, Australia eventually achieved a comparatively high COVID-19 vaccination rate, which Gillespie and colleagues attribute to the ‘agile stability’ of the existing health structures. 153 Significantly, these structures were most successful when their agile stability promoted public trust through improving access, equity, and safety in various forms.
Continuity, discontinuity, and the way forward
The history of vaccination laws in Australia reveals patterns that are useful both in understanding the interaction of law and society and for informing future policy. The aforementioned historical analysis identified several significant periods of continuity, disrupted at clearly identifiable junctures. These disruptions were essentially episodes of challenge and response: external factors, usually either epidemiological or sociological in nature, prompting a re-evaluation of the balance between community and individual interests and a consequent shift in the character of vaccination policy. In each phase, the balance between public health and liberty, and between community and individual interests, is renegotiated, and a new equilibrium is found, with each new phase exhibiting increasing complexity in the legislative responses to the problem of vaccination.
The first phase, from the beginning of self-government to the start of the twentieth century, is the simplest. The compulsory vaccination laws introduced during this period were a response to the threat posed by epidemic disease and the perception that the populace lacked the knowledge or motivation to vaccinate voluntarily. These laws contrast starkly with the liberal ideology of the period and are illustrative of the pragmatism tempering the political influence of colonial liberalism. While this approach made sense in times of outbreak, it was difficult to justify – or even to administer – the subordination of individual choice to public health in the longer term. It was the inability of the system to deal with rising anti-vaccination sentiment that led to the first break point and saw the introduction of conscientious objection clauses across the continent.
Building on the lessons of the first phase, the second phase focused on the development of bureaucratic structures that supported a system that encouraged voluntary vaccination. These structures addressed key barriers to vaccination by making vaccines safer and more effective, by improving access, and by developing reliable data-collection systems to support efficient administration. The success of these and other public health measures led to the second break point, in which the lack of epidemic disease created a sense of complacency among the public, undermining efforts to reach herd immunity and allowing the re-emergence of previously rare diseases. This situation caused a re-evaluation of the facilitative approach and the adoption of progressively intrusive laws in the third phase: first incentives, then substantial penalties, to increase vaccination coverage. These laws were criticised for their inequitable effects and uncertain efficacy, 154 and had this phase continued longer, it is possible that diminishing public support would have created its own break point.
Instead, the pandemic emerged as the next major challenge. It diverted attention from the childhood schedule and saw a flurry of coercive measures implemented and justified within the context of a public health emergency. It is too early to determine how these experiences will shape the next phase. The immediate concern was to save as many lives as possible and avoid the collapse of the healthcare system under the pressure of the unprecedented spread of the infection, leading to decisions that deprioritised individual choice and undermined public trust. However, the temporary COVID-19 vaccine compensation scheme represents a feature of discontinuity aimed at addressing public concerns and also represents an opportunity to address broader concerns, by widening its scope to include other vaccine injuries. So far, this opportunity has been missed.
Beyond compensation schemes, the historical analysis reveals several opportunities for improving the way vaccination is regulated in Australia and to optimise its uptake before moving to mandates. Here, we identify opportunities for reform that advance the aims of improving access, enhancing trustworthiness, and achieving equity in vaccination.
Regarding access, the establishment of essential infrastructure, including the AIR, TGA, and NCIRS, for assessing and monitoring vaccines in Australia means that, theoretically, Australians generally have excellent access to effective and safe vaccines. Access in practice is somewhat more complicated. Research by Beard and colleagues found that unregistered objections to vaccination accounted for only a third of children with no record of vaccination, and that higher rates of incomplete vaccination among children in the bottom 10% of socioeconomic status postcodes indicated that delayed vaccination was ‘caused by problems related to disadvantage, logistic difficulties, access to health services, and missed opportunities in primary, secondary and tertiary health care’. 155 There is therefore considerable potential for appropriately resourced health services and vaccination programmes to facilitate access and prompt uptake within this group.
To be effective, these strategies require reliable and complete data, and this has led to calls for changes to the AIR. 156 In February 2021, the Australian Immunisation Register Act 2015 (Cth) was amended to make it mandatory for vaccination providers to report to the AIR all NIP, influenza, and COVID-19 vaccinations given, replacing the previous voluntary system. 157 Providers found in breach are to be provided education and support in the first instance, with civil penalties available for continued non-compliance. 158 This approach is difficult to enforce given the lack of processes to identify providers that, albeit meeting the reporting obligations, record vaccinations inconsistently. 159 Based on previous experiences, a system of practice-based or provider-based incentives for comprehensive recording of vaccinations would likely be more effective.
A current limitation of the AIR is the inability to identify individuals who possess medical risk factors qualifying them for targeted vaccine programmes. While amending the AIR to include medical risk factor data fields has been suggested as a potential solution, ensuring these are completed and updated routinely would require substantial investment in provider education and is poorly suited to penalty-based compliance approaches given difficulties in identifying non-compliance. 160 Integration of the AIR with practice management software, such that alerts can be automated for at-risk patients but additional personal data are not accessible to others outside the practice, offers one alternative. Linkage with other national datasets, such as Medicare Benefits Schedule and Pharmaceutical Benefits Scheme or hospitalisation datasets, is another. Research into the effectiveness of different approaches, supported by appropriate stakeholder consultation, is needed to identify the best method in this context.
Although vaccination in Australia is technically – and emphatically described by its ministers as – voluntary, 161 there is no doubt that the penalties for failing to comply with the NIPS have the practical effect of making vaccination mandatory, at least for that proportion of the population who cannot afford to lose the associated benefits. 162 The key features of the current childhood vaccination system are the removal of the option of conscientious objection and the use of penalties rather than incentives. If penalties are to be retained, consideration should be given to the reintroduction of conscientious objection exemptions so that children of vaccine refusers are not excluded from government support and access to early childhood learning opportunities. Such an approach should be supported by a vaccine counselling scheme, which would also reduce pressure on doctors to provide inappropriate medical exemptions and avoid polarising hesitant parents into vaccine refusal. Matters of access would be critical to such a scheme: it would be important, for example, that vaccine counselling incurs no cost, that regional areas are adequately serviced, and that waiting times are reasonable.
A preferable option would be to revert to an incentives-based programme. By providing a MIA without means-testing, the previous system resulted in more equitable effects across the socioeconomic spectrum. Furthermore, removing the option to conscientiously object is arguably more ethically acceptable in relation to incentive schemes than penalties, as it would not have the effect of infringing rights to social security. 163 Although the decision to forgo an incentive payment may be taken more lightly by wealthier families, it is unlikely to be less effective than the current system, which leaves wealthy parents unaffected. Incentives for vaccination providers and medical practices were also shown to be an effective mechanism for improving vaccination, as it encourages health practitioners to engage with eligible patients and promoted good record-keeping and offers an important complementary intervention.
For adults, it is likely that mandates by employers, similar to those already in place for some caring-related jobs, will become more common. The case law to date suggests that such mandates are reasonably likely to be found to be ‘lawful and reasonable directions’, in circumstances where efficacy, necessity, and proportionality can be established, such as within higher-risk industries, including childcare, aged care, and health care. 164 However, from a public health law perspective, employers should be encouraged to consider issues of equity and the potential effects on public trust arising from overly stringent mandates. In situations where reassignment of duties is not feasible, the use of personal protective equipment may, in some circumstances, offer a less restrictive but equally effective alternative. Critically, if conscientious objection exemptions are not available, employees refusing vaccination will need to be afforded procedural fairness prior to termination of employment.
Reintroduction of conscientious objection also requires the ability to exclude unvaccinated individuals from childcares, schools, and workplaces during outbreaks, with low thresholds for initiating. This is already the case for unvaccinated school children and adults with medical exemptions, although whether access to leave entitlements is reasonable may be fact-dependent, including how regularly outbreaks occur and the impact on the particular workplace.
These measures, if appropriately supported by interventions to increase access, improve public trust and implement evidence-based measures to engage with vaccine-hesitant individuals, are likely to be both more equitable and as effective as the current system. While these recommendations are specific to the Australian context, analogous reforms can readily be identified in other countries by using access, trust, and equity as guiding principles.
Any amendments should be subject to rigorous evaluation to monitor ongoing efficacy and to identify any emergent effects that may have implications for assessment against the legislative intervention criteria. Assessments against these criteria depend upon the available evidence and highlight the role of legal epidemiology in policy-making. This is crucial to ensure that public health laws continue to be evidence-based and justified in the specific political and cultural context in question. Considering the legal scrutiny of public health policies, there is scope for improvements in the way empirical evidence is integrated into judicial reasoning, to better protect and promote human rights at the individual and population levels.
Conclusion
We have taken a historical and sociolegal approach to the problem of vaccination, with the aim of bringing together theoretical and empirical perspectives to identify the appropriate role for law in influencing vaccination uptake. In doing so, we provide both a descriptive account of how vaccination laws have operated in Australia and a normative argument for what the role of law should be in relation to vaccination.
Drawing on ethical and legal principles, a set of criteria for analysing the operation of vaccination laws was developed. These criteria formed the basis of a content analysis of Australian vaccination laws in their historical context. The survey of legislative interventions revealed three clear phases, and an emergent fourth, in the Australian approach to encouraging vaccination uptake over two centuries: from experiments in compulsion, to a focus on provision and voluntary vaccination, to increasingly restrictive interventions to achieve herd immunity, to the current phase, defined at its outset by the disruptive force of the pandemic. From these different approaches, key themes consistently emerged, demonstrating critical roles for law in improving access, enhancing trustworthiness, and ensuring equity of burdens and benefits from vaccination. The analysis also provided evidence of the lack of protection provided by legal scrutiny of legislative interventions against a poorly justified public health policy, resulting both from problems assessing empirical data and from an inability to assess changing risk calculi over time.
When assessed against the legislative intervention criteria, coercive measures that directly target individual behaviour are difficult to justify, given the challenges they raise for equity and justice. Rather, a comprehensive systems-wide approach, aiming to both elicit and deserve public trust, emerges as most congruent with the goals of public health. Regulatory measures to ensure the efficacy and safety of vaccines registered in Australia are foundational to this system. Before any further legislative interventions may be justifiable, it is critical that issues of access, public trust, and equitable outcomes are thoroughly addressed.
By reading the development of vaccination legislation through the lenses of continuity and discontinuity in legal history, we identify the tension between community and individual interests as the red thread of the development of vaccination legislation. It illustrates that considerations of justice have been counterbalancing the public health utility over time, according to a pattern of challenge and response in vaccination law-making, with the pandemic marking the emergence of a new phase with the potential to move in a markedly new direction.
On these bases, a number of potential targets for law reform were proposed, aimed at improving access, trustworthiness, and equity in vaccination. Although each can be justified against the legislative intervention criteria with reference to anticipated outcomes, this analysis provided strong support for a role for legal epidemiology in providing continual evaluation of the efficacy and effects of legislative interventions. Evaluations based on data collected, analysed, and interpreted using research methods and theoretical frameworks from both epidemiological and legal traditions would facilitate effective use by policymakers and courts, enhancing legal protections and promoting public health laws that are evidence-based and justified within the relevant political and cultural context.
It is tempting, when armed with a legislative hammer, to view the problem of vaccination coverage as a public health nail. There are, however, other options in the toolbox which, depending on the context, may be better suited to the task. The role of law in vaccination policy is to create conditions that are conducive to widespread uptake of the vaccine, and only when these are in place might more restrictive interventions be useful in sending a normative signal without risking exacerbating inequalities. In this way, this study of Australian vaccination laws provides a case study for public health law more broadly, indicating that rather than a tool for coercion, public health law is more productively conceived of as a tool for creating conditions conducive to public health.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
1.
M. Bennett, War Against Smallpox: Edward Jenner and the Global Spread of Vaccination (Cambridge: Cambridge University Press, 2020), pp. 339–342.
2.
M. Chan, Ten Years in Public Health: 2007-2017 (Geneva: World Health Organization Report, 2017), p. 82.
3.
Note that vaccination is the process of receiving a vaccine, whereas immunisation involves both receiving the vaccine and developing immunity as a result.
4.
See, D.M. Morens, G.K. Folkers, and A.S. Fauci, ‘The Concept of Classical Herd Immunity May Not Apply to COVID-19’, Journal of Infectious Diseases 226 (2022), p. 195.
5.
N.E. MacDonald, S. Harmon, E. Dube, A. Steenbeek, N. Crowcroft, D.J. Opel, D. Faour, J. Leask, and R. Butler, ‘Mandatory Infant & Childhood Immunization: Rationales, Issues and Knowledge Gaps’, Vaccine 36 (2018), p. 5812.
6.
World Health Organization, Global Vaccine Action Plan: 2011-2020 (Geneva: World Health Organization, 2013).
7.
MacDonald et al., ‘Childhood Immunization’, p. 5812.
8.
See, D.N. Schiff, ‘Socio-Legal Theory: Social Structure and Law’, Modern Law Review 39 (1976), p. 287.
9.
See, e.g., Nuffield Council on Bioethics (‘NCB’), Public Health: Ethical Issues (NCB, 2007), pp. 41–42; O.M. Bradfield and A. Giubilini, ‘Spoonful of Honey or a Gallon of Vinegar? A Conditional COVID-19 Vaccination Policy for Front-Line Healthcare Workers’, Journal of Medical Ethics 47 (2021), p. 467.
10.
A.J. Dawson, ‘Snakes and Ladders: State Interventions and The Place of Liberty in Public Health Policy’, Journal of Medical Ethics 42 (2016), pp. 512–513.
11.
See, e.g., A. Dawson, ‘Herd Protection as a Public Good: Vaccination and Our Obligations to Others’ in A. Dawson and M.F. Verweij, eds., Ethics, Prevention and Public Health (Oxford: Oxford University Press, 2007), pp. 177–178; A. Giubilini, T. Douglas and J. Savulescu, ‘The Moral Obligation to Be Vaccinated: Utilitarianism, Contractualism, and Collective Easy Rescue’, Medicine, Health Care and Philosophy 21 (2018), p. 559.
12.
See M. Otsuka, ‘The Paradox of Group Beneficence’, Philosophy & Public Affairs 20 (1991), p. 132; D. Parfit, Reasons and Persons (Oxford: Oxford University Press, 1984).
13.
J. Savulescu, A. Giubilini, and M. Danchin, ‘Global Ethical Considerations Regarding Mandatory Vaccination in Children’, Journal of Paediatrics 231 (2021), p. 10; J. Savulescu, ‘Good Reasons to Vaccinate: Mandatory or Payment for Risk?’ Journal of Medical Ethics 47 (2021), p. 78.
14.
R. Pierik, ‘Mandatory Vaccination: An Unqualified Defence’, Journal of Applied Philosophy 35 (2018), pp. 395–396.
15.
S. Clarke, A. Giubilini, and M.J. Walker, ‘Conscientious Objection to Vaccination’, Bioethics 31 (2017), p. 161.
16.
The Therapeutic Goods Administration assesses safety and efficacy of vaccines before adding them to the Australian Register of Therapeutic Goods, and even in the case of provisional approvals, preliminary clinical data demonstrating the likelihood of the vaccine providing a major therapeutic advance and significant improvement in efficacy or safety over existing alternatives must be provided. See Therapeutic Goods Regulations 1990 r 10L(1).
17.
K. Ward, M.Y.K Chow, C King, and J. Leask, ‘Strategies to Improve Vaccination Uptake in Australia, a Systematic Review of Types and Effectiveness’, Australian and New Zealand Journal of Public Health 36 (2012), p. 369; F.H. Beard, B.P. Hull, J. Leask, A. Dey, and P.B. McIntyre, ‘Trends and Patterns in Vaccination Objection, Australia, 2002-2013’, Medical Journal of Australia 204 (2016), p. 275.e1.
18.
See, e.g., S. Burns, R. Bhoyroo, J.E. Leavy, L. Portsmouth, L. Millar, J. Jancey, J. Hendriks, H. Saltis, J. Tohotoa, and C. Pollard, ‘The Impact of the No Jab No Play and No Jab No Pay Legislation in Australia: A Scoping Review’, International Journal of Environmental Research and Public Health 20 (2023), p. 6219.
19.
See, e.g., M. Verweij and A. Dawson, ‘Ethical Principles for Collective Immunisation Programmes’, Vaccine 22 (2004), p. 3123; D. Isaacs, ‘An Ethical Framework for Public Health Immunisation Programs’, NSW Public Health Bulletin 23 (2012), p. 112.
20.
Verweij and Dawson, op. cit.; Isaacs, op. cit.
21.
Verweij and Dawson, ‘Ethical Principles for Collective Immunisation Programmes’, p. 3123.
22.
See, e.g., L.O. Gostin, Public Health Law: Power, Duty, Restraint, 2nd ed. (Berkeley, CA: University of California Press, 2008), pp. 43–70.
23.
Gostin, Public Health Law, pp. 43–70; L.O. Gostin and J.M. Mann, ‘Towards the Development of a Human Rights Impact Assessment for the Formulation and Evaluation of Public Health Policies’, Health and Human Rights 1 (1994), p. 58.
24.
See, Gostin and Mann, ‘Human Rights Impact Assessment’, p. 77.
25.
R. Baldwin, M. Cave, and M. Lodge, eds., Understanding Regulation: Theory, Strategy, and Practice, 2nd ed. (Oxford: Oxford University Press, 2012).
26.
N. de Sadeleer, ‘The Precautionary Principle’, in N. de Sadeleer, ed., Environmental Principles: From Political Slogans to Legal Rules, 2nd ed. (Oxford: Oxford University Press, 2020); J. Zander, The Application of the Precautionary Principle in Practice: Comparative Dimensions (Cambridge: Cambridge University Press, 2010); A. Janssen and N. Rosenstock, ‘Handling Uncertain Risks: An Inconsistent Application of Standards? The Precautionary Principle in Court Revisited’, European Journal of Risk Regulation 7 (2016), p. 144; M. Rogers, ‘Risk Management and The Record of The Precautionary Principle in EU Case Law’, Journal of Risk Research 14 (2011), p. 467; M. Martuzzi, ‘The Precautionary Principle: In Action for Public Health’, Occupational and Environmental Medicine 64 (2007), pp. 569–570.
27.
R. Baldwin, M. Cave, and M. Lodge, ‘Risk-Based Regulation’, in R. Baldwin, M. Cave, and M. Lodge, eds., Understanding Regulation: Theory, Strategy, and Practice, 2nd ed. (Oxford: Oxford University Press, 2012), pp. 281–295; A. Burgess, A. Alemanno, and J. Zinn, eds., Routledge Handbook of Risk Studies (New York: Routledge; Taylor & Francis Group, 2016); National Research Council, Science and Decisions: Advancing Risk Assessment (Washington, DC: The National Academies Press, 2009); OECD, OECD Regulatory Policy Outlook 2021 (Paris: OECD, 2021).
28.
Convention for the Protection of Human Rights and Fundamental Freedoms (European Convention on Human Rights, as amended) (ECHR) art 2. See also, e.g., R. Frowde, E.S. Dove, and G.T. Laurie, ‘Fail to Prepare and You Prepare to Fail: The Human Rights Consequences of the UK Government’s Inaction During the COVID-19 Pandemic’, Asian Bioethics Review 12 (2020), p. 1.
29.
See, e.g., International Covenant on Economic, Social and Cultural Rights, opened for signature 16 December 1966, 993 UNTS 3 (entered into force 3 January 1976), Art 12(2)(c); UN Committee on Economic, Social and Cultural Rights, General Comment No 14 UN Doc E/C.12/2000/4 (11 August 2000) [16]; UN Committee on the Rights of the Child, General Comment No 15 UN Doc CRC/C/GC/a5 (17 April 2013) [41].
30.
See, e.g., Universal Declaration on Bioethics and Human Rights (UNESCO, 19 October 2005), Art 6, (‘UDBHR’); Council of Europe, Convention on Human Rights and Biomedicine (CETS 164, 4 April 1997), Art 5.
31.
See, e.g., UDBHR, Art 27.
32.
J.I. Acosta, ‘Vaccines, Informed Consent, Effective Remedy and Integral Reparation: An International Human Rights Perspective’, Vniversitas 131 (2015), p. 19.
33.
Baytüre and Others v. Turkey (dec.) (ECtHR, Second Section, no. 3270/09, 12 March 2013); Vavřička and Others v. the Czech Republic (ECtHR, Grand Chamber, no 47621/13 and five others, 8 April 2021) [302] (‘Vavřička’).
34.
Vavřička.
35.
Vavřička, [290]–[309].
36.
R. McWhirter and M. Clark, ‘Expertise, Public Health and the European Convention on Human Rights: Vavřička v Czech Republic’, Modern Law Review 86 (2023), pp. 1037–1038.
37.
Vavřička, Dissenting Opinion of Judge Wojtyczek [14].
38.
Vavřička [147]–[151].
39.
J.M. Broekman, ‘Changes of Paradigm in the Law’, in A. Peczenik, L. Lindahl, and G.C. van Roermund, eds., Theory of Legal Science: Proceedings of the Conference on Legal Theory and Philosophy of Science Lund, Sweden, 11–14 December 1983, Synthese Library 176, (Dordrecht: Springer, 1984), pp. 133–144.
40.
T.S. Kuhn, The Structure of Scientific Revolutions, 2nd ed. (Chicago, IL: University of Chicago Press, 1970), vol. I.
41.
A. J. Toynbee, A Study of History (Oxford: Oxford University Press, 1962), vol. I, pp. 271–339.
42.
A. Gerschenkron, ‘On the Concept of Continuity in History’, Proceedings of the Am. Philosophical Society 106 (1962), p. 195.
43.
See H.J. Berman, Law and Revolution (Harvard University Press, 1983); G. Santucci, Diritto romano e diritti europei: continuità e discontinuità nelle figure giuridiche (New York: Il Mulino, 2018).
44.
NCB, Public Health, p. 58.
45.
Australia’s federal structure entails different but overlapping legislative powers at state and national levels, which can lead to divergent approaches, causing challenges to effective management of public health. See, e.g., B. Bennett, I. Freckelton, and G. Wolf, COVID-19, Law & Regulation: Rights, Freedoms, and Obligations in a Pandemic (Oxford: Oxford University Press, 2023), p. 553.
46.
An Act for the Prevention of Small-Pox by Rendering Vaccination Compulsory in Certain Cases 1853 (VDL/Tas); Vaccination Act 1853 (SA); An Act to Make Compulsory the Practice of Vaccination 1854 (Vic); An Ordinance to Make Compulsory the Practice of Vaccination 1860 (WA); Vaccination Act 1867 (Qld).
47.
J.H.L. Cumpston, ‘The Development of Public Health in Australia’, Medical Journal of Australia 1 (1928), p. 333.
48.
49.
Op. cit.
50.
R. McWhirter, Lymph or Liberty: Responses to Smallpox Vaccination in the Eastern Australian Colonies (PhD Thesis, University of Tasmania, Australia, 2008), p. 28.
51.
J.Y. Rutter, ‘Vaccine Institution’, NSWV&P, LC 1 (1853), pp. 411–412.
52.
C. Rolleston, ‘Vaccination. Report of the Registrar-General’, NSWV&P, LA, 2 (1856–1857), p. 694.
53.
Op. cit.
54.
McWhirter, Lymph or Liberty, pp. 60–79; J.H.L. Cumpston, The History of Smallpox in Australia, 1788-1908 (Melbourne, VIC, Australia: Government Printer, 1914) (‘Smallpox in Australia’), pp. 130–146.
55.
R. Hicks, Rum, Regulation and Riches (Deakin West, ACT, Australia: Australian Hospital Association, 1981) p. 10.
56.
McWhirter, Lymph or Liberty, pp. 80–85.
57.
Cumpston, History of Smallpox, p. 133.
58.
Quoted in op. cit., p. 143.
59.
‘Non-Compulsory Vaccination Bill’, Victorian Parliamentary Debates: Legislative Assembly, 78 (1895–1896), p. 2482.
60.
F. Campbell, ‘Vaccination’ NSWLCJ (No 67, 1867–1868 Part 1) p. 833. Emphasis in original.
61.
McWhirter, Lymph or Liberty, pp. 312–314.
62.
J. Campbell, Invisible Invaders: Smallpox and Other Diseases in Aboriginal Australia, 1780-1880 (Melbourne, VIC, Australia: Melbourne University Publishing, 2002), pp. 129–162.
63.
S. Archer, S., ‘Vaccination, Dispossession, and the Indigenous Interior’, Bulletin of the History of Medicine 97(2) (2023), pp. 255–293.
64.
Op. cit., p. xii.
65.
See, e.g., Chinese Immigration Act 1887 (Tas); ‘Influx of Chinese Restriction Bill’, NSWPD, LA, 1881, Vol. 1, p. 266; ‘Small Pox’, VPD, LA, 1884, Vol 46, pp. 735–736.
66.
K. Cronin, Colonial Casualties: Chinese in early Victoria (Melbourne, VIC, Australia: Melbourne University Publishing, 1982), pp. 69–70.
67.
Vaccination Act 1854 (SA).
68.
Vaccination Act 1881 (Tas); Vaccination Act 1871 (Qld).
69.
See, e.g., O’Loughlin v. McCay [1898] 23 VLR 650; Hibberd v. Kelleher (1901) 27 VLR 474; Thompson v. King (1905) VLR 375; Thornton v. Kelly (1910) VLR 156; O’Malley v. Russell (1908) VLR 545; Akeroyd v. Phillips (1890) 29 VLR 662.
70.
Vaccination Act 1898 (Tas); Health Act 1890 (Vic); Vaccination Act 1892 (SA).
71.
Vaccination Act 1898 (Tas); Health Act 1900 (Qld); Compulsory Vaccination Abolition Act 1901 (SA); Health Act 1911 (WA); Health Act 1919 (Vic).
72.
Quarantine Act 1908 (Cth) s 87; Commonwealth Serum Laboratories Act 1961 (Cth); National Health and Medical Research Council Act 1992 (Cth).
73.
Including the Northern Territory Immunisation Register in 1991 and the Vaccination Information and Vaccination Administration System in Queensland in 1993.
74.
C.H. Kellaway, Report of the Royal Commission of Inquiry into Fatalities at Bundaberg (1926-28/IV/1085-1211), available at https://parlinfo.aph.gov.au/parlInfo/download/publications/tabledpapers/HPP052016005415/upload_pdf/HPP052016005415_1926-27-28_243.pdf;![]() .
.
75.
A.H. Brogan, Committed to Saving Lives: A History of the Commonwealth Serum Laboratories (Melbourne, VIC, Australia: Hyland House, 1990), p. 26; H.F. Gidding, M.A. Burgess, and A.E. Kempe, ‘A Short History of Vaccination in Australia’, Medical Journal of Australia 74 (2001), p. 37.
76.
Gidding et al., ‘Short History’, p. 37.
77.
T.A. Ruff, K. Taylor, and T. Nolan, ‘Australia’s Contribution to Global Immunisation’ Australian and New Zealand Journal of Public Health 36 (2012), p. 566.
78.
Australian Bureau of Statistics, ‘1989–90 National Health Survey, Children’s Immunisation, Australia’, Cat. No. 4379.0, 1992.
79.
National Health and Medical Research Council, ‘National Immunisation Strategy’, Canberra, April 1993, available at https://webarchive.nla.gov.au/awa/20170819044412/
![]() .
.
80.
Gidding et al., ‘Short History’, p. 38; K. Ward, H. Quinn, R. Menzies, and P. McIntyre, ‘A History of Adolescent School-Based Vaccination in Australia’, Communicable Diseases Intelligence 37 (2013), p. E168.
81.
National Health and Medical Research Council, ‘National Immunisation Strategy’, pp. 6, 9.
82.
Brogan, Committed to Saving Lives, p. 118.
83.
Op. cit.
84.
Op. cit., p. 26; Gidding et al., ‘Short History’, pp. 37–38.
85.
Explanatory Memorandum, Health Insurance Amendment Bill 1996 (Cth).
86.
Piloted under the Health Insurance Commission Regulations and later established by amending the Health Insurance Act 1973. B.P. Hull, S.L. Deeks, and P.B. McIntyre, ‘The Australian Childhood Immunisation Register – A model for Universal Immunisation Registers?’ Vaccine 27 (2009), p. 5054.
87.
Child Care Payments Act 1997 (Cth); Social Security and Veterans’ Affairs Legislation Amendment (Family and Other Measures) Act 1997 (Cth) amending the Social Security Act 1991 (Cth).
88.
K. Ward, B.P. Hull, and J. Leask, ‘Financial Incentives for Childhood Immunisation – A Unique but Changing Australian Initiative’, Medical Journal of Australia 198 (2013), 590; Ruff et al., ‘Australia’s Contribution’, p. 566.
89.
Health Legislation Amendment Act 2003 (Cth) amending Health Insurance Act 1973 (Cth).
90.
Family Assistance Legislation Amendment (More Help for Families - One Off Payments) Act 2004 (Cth).
91.
National Health Amendment (Immunisation Program) Act 2005 amending National Health Act 1953 (Cth).
92.
Social Services Legislation Amendment (No Jab, No Pay) Act 2015 (Cth) amending A New Tax System (Family Assistance) Act 1999 (Cth) s 19(3).
93.
Australian Immunisation Register Act 2015 (Cth); Australian Immunisation Register (Consequential and Transitional Provisions) Act 2015 (Cth).
94.
Explanatory Memorandum, Health Insurance Amendment Bill 1996 (Cth).
95.
Commonwealth, ‘Parliamentary Debates’, House of Representatives, 25 February 1997, p. 1178.
96.
Commonwealth, ‘Parliamentary Debates’, House of Representatives, 16 September 2015, p. 10329.
97.
Hull et al., ‘The Australian Childhood Immunisation Register’, p. 5055; C. Rank and R.I. Menzies, ‘How Reliable Are Australian Childhood Immunisation Register Coverage Estimates for Indigenous Children? An Assessment of Data Quality and Coverage’, Communicable Diseases Intelligence 31 (2007), p. 283.
98.
Rank and Menzies, ‘ACIR Coverage Estimates’, pp. 283–286.
99.
B. Hull, A. Dey, D. Mahajan, R. Menzies, and P.B. McIntyre, ‘Immunisation Coverage Annual Report, 2009’, Communicable Diseases Intelligence 35 (2011), p. 135.
100.
Op. cit., p. 133.
101.
B.P. Hull, F.H. Beard, A.J. Hendry, A. Dey, and K. Macartney, ‘“No Jab, No Pay”: Catch-Up Vaccination Activity During Its First Two Years’, Medical Journal of Australia 213 (2020), p. 364.
102.
Op.cit.; A. Li and M. Toll, ‘Removing Conscientious Objection: The Impact of ‘No Jab No Pay’ and ‘No Jab No Play’ Vaccine Policies in Australia’, Preventive Medicine 145 (2021), p. 106406.
103.
Australian Government, ‘Historical Coverage Data Tables for All Children’, 2024, available at https://www.health.gov.au/topics/immunisation/immunisation-data/childhood-immunisation-coverage/historical-coverage-data-tables-for-all-children; Australian Government, ‘Historical Coverage Data Tables for Aboriginal and Torres Strait Islander Children’, available at ![]() .
.
104.
K. Attwell, R. Seth, F. Beard, A. Hendry, and D. Lawrence, ‘Financial Interventions to Increase Vaccine Coverage’, Pediatrics 146 (2020), p. e20200724.
105.
F. Beard, A.J. Hendrya, H.F. Giddinga, A. Deya, K. Macartney, J. Leaska, and P. McIntyre, ‘Impact of Australia’s No Jab, No Pay Policy on Vaccination Uptake – A Before-After Study in Two National Birth Cohorts’, The Lancet Regional Health – Western Pacific 54 (2025), p. 101259.
106.
107.
Op. cit.; Li and Toll, ‘Removing Conscientious Objection’.
108.
T.M. Nolan, ‘The Australian Model of Immunization Advice and Vaccine Funding’, Vaccine 285 (2010), p. A76.
109.
Op. cit., pp. A76–A83.
110.
Ward et al., ‘Financial Incentives’, p. 591.
111.
Op. cit., pp. 591–592.
112.
Explanatory Memorandum, Social Services Legislation Amendment (No Jab, No Pay) Bill 2015 (Cth).
113.
F.H. Beard, J. Leask, and P.B. McIntyre, ‘No Jab, No Pay and Vaccine Refusal in Australia: The Jury is Out’, Medical Journal of Australia 206 (2017), p. 381.
114.
115.
National Centre for Immunisation Research and Surveillance, ‘Immunisation Coverage Data’; Li and Toll, ‘Removing Conscientious Objection’.
116.
Y.T. Yang and D.M. Studdert, ‘Linking Immunization Status and Eligibility for Welfare and Benefits Payments: The Australian “No Jab, No Pay” Legislation’, Journal of the American Medical Association 317 (2017), p. 803.
117.
J. Leask and M. Danchin, ‘Imposing Penalties for Vaccine Rejection Requires Strong Scrutiny’, Journal of Paediatrics and Child Health 53 (2017), pp. 440–441.
118.
R.I. Menzies, J. Leask, J. Royle, and C.R. MacIntyre, ‘Vaccine Myopia: Adult Vaccination Also Needs Attention’, Medical Journal of Australia 206 (2017), p. 238.
119.
A.W. Taylor-Robinson, ‘Letter: Vaccine Myopia: Adult Vaccination Also Needs Attention’, Medical Journal of Australia 207 (2017), p. 406; R.I. Menzies, J. Royle and C.R. McIntyre, ‘Reply: Vaccine Myopia: Adult Vaccination Also Needs Attention’, Medical Journal of Australia 207 (2017), p. 407.
120.
D.T. Smith, K. Attwell, and U. Evers, ‘Majority Acceptance of Vaccination and Mandates Across the Political Spectrum in Australia’, Politics 40 (2020), p. 203.
121.
A.J. Wakefield, S.H. Murch, A. Anthony, J. Linnell, D.M. Casson, M. Malik, M. Berelowitz, A.P. Dhillon, M.A. Thomson, P. Harvey, A. Valentine, S.E. Davies, and J.A. Walker-Smith, ‘Ileal-Lymphoid-Nodular Hyperplasia, Non-Specific Colitis, and Pervasive Developmental Disorder in Children’, The Lancet 351 (1998), p. 637.
122.
J. Leask, R. Booy, and P.B. McIntyre, ‘MMR, Wakefield and The Lancet: What Can We Learn?’ Medical Journal of Australia 193 (2010), p. 5.
123.
Op. cit., p. 6.
124.
Piesse v. Medical Board of Australia [2017] VCAT 1615.
125.
Arnold v. Goodstart Early Learning [2020] FWC 6083; Glover v. Ozcare [2021] FWC 231; Barber v. Goodstart Early Learning [2021] FWC 2156.
126.
Barber, [435].
127.
Johnston v. Carroll (Commissioner of the Queensland Police Service) [2024] QSC 2 at [127]. See also C. Rudge, ‘Challenging Pandemic Law: From Vaccine Mandates to Judicial Review of Vaccine Approvals’, Journal of Law and Medicine 31 (2024), p. 225.
128.
S. Burris, M. Ashe, D. Levin, M. Penn, and M. Larkin, ‘A Transdisciplinary Approach to Public Health Law: The Emerging Practice of Legal Epidemiology’, Annual Review of Public Health 37 (2015), p. 135.
129.
W.E. Parmet, ‘Population-Based Legal Analysis: Bridging the Interdisciplinary Chasm through Public Health in Law’, Journal of Legal Education 66 (2016), p. 100.
130.
J.A. Gillespie, J. Buchanan, C.H. Schneider, and F. Paolucci, ‘Covid 19 Vaccines and the Australian Health Care State’, Health Policy and Technology 11 (2022), p. 100607.
131.
Op. cit.
132.
Op. cit.
133.
See K. Wilson and C. Rudge, ‘COVID-19 Vaccine Mandates: A Coercive but Justified Public Health Necessity’, UNSW Law Journal 46 (2023), pp. 399–401.
134.
Op. cit.
135.
S. Eades, F. Eades, D. McCaullay, L. Nelson, P. Phelan, and F. Stanleyf, ‘Australia’s First Nations’ Response to the COVID-19 Pandemic’ The Lancet 396 (2020), pp. 237–238.
136.
G.H. Soares, J. Hedges, B. Poirier, S. Sethi, and L. Jamieson, ‘Deadly Places: The Role of Geography in Aboriginal and Torres Strait Islander COVID-19 Vaccination’, Australian and New Zealand Journal of Public Health 48 (2024), p. 100130.
137.
T. Riley, J. Guthrie, L. Whop, N. Walsh, A. Jackson, and T. Weeramanthri, ‘A Successful Partnership Model for COVID-19 Vaccination in Aboriginal Communities in New South Wales’, Australian and New Zealand Journal of Public Health 46 (2022), p. 562.
138.
R. Matthews and K. Menzel, ‘Vaccine Mandates and Cultural Safety’, Journal of Bioethical Inquiry 20 (2023), p. 719.
139.
R. McWhirter, D. Nicol, and J. Savulescu, ‘Genomics in Research and Health Care with Aboriginal and Torres Strait Islander Peoples’, Monash Bioethics Review 33 (2015), p. 203.
140.
WHO, ‘Pfizer BioNTech COVID-19 Vaccine: What You Need to Know’, 20 April 2021, available at https://www.who.int/news-room/feature-stories/detail/who-can-take-the-pfizer-biontech-covid-19–vaccine. See also RANZCOG, ‘COVID-19 Vaccination in Pregnant and Breastfeeding Women’, available at ![]() .
.
141.
142.
C.B. Krubine, R.R. Faden, R.A. Karron, M.O. Little, A.D. Lyerly, J.S. Abramson, R.H. Beigi, A.R. Cravioto, A.P. Durbin, B.G. Gellin, S.B. Gupta, D.C. Kaslow, S. Kochhar, F. Luna, C. Saenz, J.S. Sheffield, and P.O. Tindana; PREVENT Working Group, ‘Pregnant Women & Vaccines Against Emerging Epidemic Threats: Ethics Guidance for Preparedness, Research, and Response’, Vaccine 39 (2021), p. 85.
143.
Explanatory Memorandum, Australian Immunisation Register Amendment (Reporting) Bill 2021 (Cth).
144.
J. Tuckerman, C.C. Blyth, F.H. Beard, and M.H. Danchin, ‘COVID-19 and Changes in the National Immunisation Program: A Unique Opportunity to Optimise the Australian Immunisation Register (AIR)’, Medical Journal of Australia 214 (2021), p. 247.
145.
B. Madden, ‘Vaccine Injury Compensation’, Australian Health Law Bulletin 14 (2006), p. 41; C. Looker and H. Kelly, ‘No-Fault Compensation Following Adverse Events Attributed to Vaccination: A Review of International Programmes’, Bulletin of the World Health Organization 89 (2011), p. 371.
146.
S. Halabi, A. Heinrich, and S.B. Omer, ‘No-Fault Compensation for Vaccine Injury – The Other Side of Equitable Access to Covid-19 Vaccines’, New England Journal of Medicine 383 (2020), p. e125.
147.
See, K. Attwell, S. Drislane, and J. Leask, ‘Mandatory Vaccination and No Fault Vaccine Injury Compensation Schemes: An Identification of Country-Level Policies’, Vaccine 37 (2019), p. 2844. See also, D. Fairgrieve, J.S. Borghetti, S. Dahan, R. Goldberg, S. Halabi, S. Holm, G. Howells, C. Kirchhelle, A. Pillay, E. Rajneri, M. Rizzi, M. Sintes, S. Vanderslott, and N. Witzleb, ‘Comparing No-Fault Compensation Systems for Vaccine Injury’, Tulane Journal of International and Comparative Law 31 (2023), p. 77; K. Watts and T. Popa, ‘Injecting Fairness into COVID-19 Vaccine Injury Compensation: No-Fault Solutions’, Journal of European Tort Law 12 (2021), p. 1.
148.
See, e.g., N. Wood, K. Macartney, J. Leask, and P. McIntyre, ‘Australia Needs a Vaccine Injury Compensation Scheme’, Australian Journal of General Practice 49 (2020), p. 49; D. Isaacs, ‘Should Australia Introduce a Vaccine Injury Compensation Scheme?’ Journal of Paediatrics and Child Health 40 (2004), p. 247; Royal Australian College of General Practitioners, No-Fault Compensation Scheme for Immunisations. Position statement, August 2021, available at ![]() .
.
149.
Wilson and Rudge, ‘Vaccine Mandates’.
150.
Covid-19 Vaccine Claims Scheme Policy 2021 (Policy) Under the Financial Framework (Supplementary Powers) Regulations 1997 (Cth) [Version 1.3, 31 May 2022].
151.
The closing date, initially set in April 2024, was extended to September 2024, <COVID-19 Vaccine Claims Scheme | Australian Government Department of Health and Aged Care>. See also B. Madden and T. Cockburn, ‘The COVID-19 Vaccine Claims Scheme: An Overview of the Policy Document’, Precedent 168 (2022), p. 22.
152.
The Sydney Morning Herald reported that ‘[s]ince the scheme began in December 2021, it has paid 126 claims a total of $7.2 million, a fraction of the $76.9 million set aside for possible payments in Services Australia’s portfolio budget statement’. M. Ward, ‘Thousands Left Waiting for Compensation After Claims of COVID-19 Vaccine Injury’ The Sydney Morning Herald, 16 April 2023, available at ![]() .
.
153.
Gillespie et al., ‘Covid 19 Vaccines’, pp. 4–6.
154.
Yang and Studdert, ‘Linking Immunization Status and Eligibility for Welfare and Benefits Payments’, p. 803; J. Leask and M. Danchin, ‘Imposing Penalties for Vaccine Rejection Requires Strong Scrutiny’, Journal of Paediatrics and Child Health 53 (2017), pp. 440–441; Menzies et al., ‘Vaccine Myopia’, p. 238.
155.
Beard et al., ‘Trends and Patterns’, p. 275.e3.
156.
Tuckerman et al., ‘COVID-19’.
157.
Australian Immunisation Register Amendment (Reporting) Act 2021 (Cth).
158.
Australian Immunisation Register Amendment (Reporting) Act 2021 (Cth), art. 11 inserting art. 28A in the Australian Immunisation Register Act 2015 (Cth).
159.
Op. cit.
160.
Tuckerman et al., ‘COVID-19’, p. 248.
161.
Commonwealth, ‘Parliamentary Debates’, House of Representatives, 3 December 2020, p. 10426.
162.
The strongly coercive effects of linking vaccination status to ability to work, for example, was recognised by Chief Justice Beech-Jones as having ‘an encouraging effect or even a coercive effect so far as vaccination is concerned’ without going so far as to authorise vaccination without consent: Kassam v Hazzard; Henry v Hazzard NSWSC 1320, at [59].
163.
International Covenant on Economic, Social and Cultural Rights, opened for signature 16 December 1966, 993 UNTS 3 (entered into force 3 January 1976), Art 9.
164.
See, e.g., Arnold v Goodstart Early Learning [2020] FWC 6083; Glover v Ozcare [2021] FWC 231; Barber v Goodstart Early Learning [2021] FWC 2156.
