Abstract
Accurate diagnosis is an essential component of healthcare delivery. However, research on the delivery of diagnostic services is lacking in low- and middle-income countries. This article examines the issues related to the provision of medical laboratory services in India, including licencing, geographic distribution, charging practices, quality, personnel requirements, information sharing, and newer technologies that impact the sector. The challenges and gaps in regulatory mechanisms governing these services are discussed, highlighting the need for improvements. Legislation to regulate medical laboratories in many Indian states is either outdated or non-existent, with some states recently updating or enacting their laws. A registry of medical laboratories in the country will assist in assessing and meeting the shortfall. Universal adoption of external and inter-laboratory quality control mechanisms will help in standardization and ensuring quality. Clarity and consensus on who can operate medical laboratories and the responsibility of different cadres of technical staff are required. The provision for making information on registered laboratories publicly available under the Central and some state legislations can be very useful to the users in choosing laboratories. Mandatory reporting to the government by both public- and private-sector laboratories will help in maintaining data on disease burden and in planning health services. However, the lack of data protection laws in the country creates the potential for violation of the users’ privacy when laboratories store data digitally and report it online on government web portals. Caution is warranted in the use of newer technologies until the regulations governing these matters are strengthened. A combination of voluntary and statutory mechanisms such as accreditation and regulation would be useful instruments for ensuring quality in diagnostic services. Research on the effectiveness of existing provisions at the state level would help in understanding their impact and suggest ways of further improvement.
Introduction
‘Test, Test, Test’ was the key message from the World Health Organization in March 2020 on the premise that trying to control the pandemic without accurate and timely testing for COVID-19 infection would be like fighting a fire blindfolded. 1 India increased its testing capacity from 2,000 tests a day in March 2020 2 to almost 2 million tests a day in May 2021. 3 However, as the pandemic progressed, a number of issues emerged, such as the non-availability of test kits, lack of trained staff, frequent changes in testing norms, variable testing costs, lack of transparency, large-scale buying of test kits by the state – without regulatory approval – and fake COVID-19 test reports. 4 Testing in rural areas was particularly inadequate due to a lack of test kits and laboratories. 5 Although these issues came to the fore in the context of the pandemic, many of them are not new to the provision of diagnostic services in India.
A physical examination and symptomatic diagnosis may not always provide precise information to make a prognosis and to guide treatment, 6 thereby making diagnostic investigations crucial. It is believed that 60%–70% of the clinical decisions are based on diagnostic investigations, 7 which can be divided into two categories: laboratory medicine, which deals with the examination of body tissues and body fluids, 8 and imaging technology, which involves the use of techniques such as X-rays, magnetic resonance imaging, and ultrasound to view the interior of the body not visible to the naked eye. 9 The two categories differ in their requirements in terms of personnel, infrastructure, equipment, and the regulations that govern them.
Research on the delivery of diagnostic services is scarce, compared with that on other aspects of healthcare delivery in low and middle-income countries. The article discusses the concerns related to medical laboratories in the context of healthcare delivery in India, including the regulatory environment in which they operate.
The way medical laboratories in India provide services raises many concerns regarding their licencing, geographic distribution, access, charging practices, quality, human resources, information sharing, reporting, and the technologies used. How these concerns affect service delivery and the pertinent regulations – with their strengths and inadequacies – is discussed below.
Licencing and registration
For 7 years in the city of Delhi, a ‘laboratory’ – operated by someone with a graduate degree in the arts – continued to generate reports without testing the samples. 10 A similar ‘laboratory’ in the same city produced 30,000 fake reports within 1 year. 11 Many states in India have had no entry barriers or minimum requirements to set up medical laboratories. Given such a lack of regulation, the industry has flourished, recording rapid growth, 12 along with the spread of several substandard and fake laboratories.
The Clinical Establishments (Registration and Regulation) Act, 2010 – hereafter referred to as the CEA – was enacted by India’s central (federal) government to correct this anomaly. The Act provides for the registration of state-run and private health facilities, including medical laboratories, 13 and prescribes minimum standards for regulating such entities. As can be seen in Table 1, the Act is in force in 12 states and 6 union territories (UTs, which are administrative units typically smaller than a state). 14 However, the implementation of the Act has been sluggish.
Regulation of clinical establishments in states and UTs: March 2023.

CEA: Clinical Establishments (Registration and Regulation) Act.
Many states have neither adopted the CEA nor updated their own state legislation. Legislation in Delhi and Maharashtra does not include laboratories within its ambit. 15 A similar situation prevailed in Gujarat, Kerala, and Punjab until recently. 16 In Andhra Pradesh, Karnataka, Madhya Pradesh, Manipur, Meghalaya, Nagaland, Odisha, and West Bengal, the relevant regulations are applicable only to private laboratories: state-run laboratories are exempt. 17
Collection centres
Several laboratories rely on collection centres that operate on the franchise model to achieve high business volumes and economies of scale. 18 For instance, Metropolis Healthcare and SRL Diagnostics (two among the top five diagnostic companies in revenue terms), respectively, plan to add 1,800 and 1,500 collection centres through franchisees to their existing 3,006 and 2,400 collection centres in the country. 19 However, collection centres are regulated differently across states. For instance, Chhattisgarh and West Bengal require the centres to be registered independently of the parent laboratory, whereas Goa does not. Similarly, under the CEA, collection centres are part of the parent laboratory, which is held responsible for their compliance with the minimum standards. 20
Online health service aggregators and collection of samples from home
Online health service aggregators 21 act like franchisees without physical collection centres. 22 However, they have no legal standing and public-interest litigation was filed in the Delhi High Court in August 2020 to ban them. 23 The Ministry of Health and Family Welfare (MoHFW), Government of India (GoI), directed states in January 2021 to take action against online aggregators that offer laboratory facilities. 24 Nevertheless, they continue to function, and concerns have been raised about the quality of their services. 25 Collection of samples from home is not explicitly mentioned in either the central or any state legislation except in the West Bengal Rules that provide for it, in case the person is ‘unable or unwilling to attend the clinical establishment’. 26
Distribution of and access to medical laboratories
A comprehensive database of medical laboratories in India is not available: different sources provide different numbers and distribution across the country and are incomplete (Table 2). Although the number of medical laboratories in India is estimated at 100,000, 27 the largest available database, the National Health Resource Repository (NHRR) 28 features 38,738 entries, nearly one-third of the estimated number. 29 The National Register of Clinical Establishments maintained under the CEA does not specifically mention how many of the registered 34,337 allopathic clinical establishments are medical laboratories across the states and UTs in which the Act is in force. 30 A third source is the National Accreditation Board for Testing and Calibration Laboratories (NABL), which puts the number at 2,374 nationally. 31 A more recent database of laboratories that report COVID-19 test results to the Indian Council of Medical Research (ICMR) lists 3,388 laboratories. 32
Distribution of medical laboratories in India, by state.
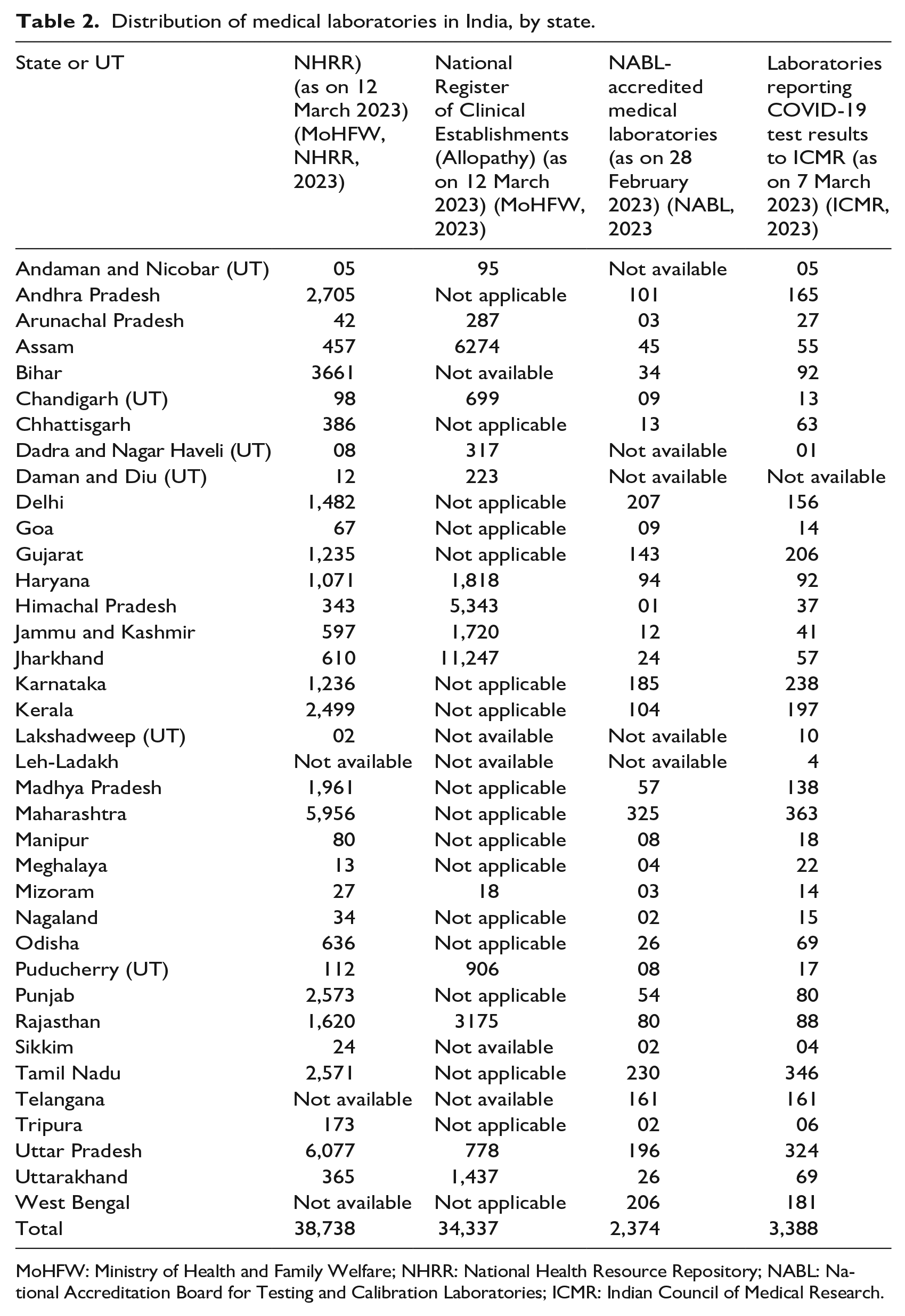
MoHFW: Ministry of Health and Family Welfare; NHRR: National Health Resource Repository; NABL: National Accreditation Board for Testing and Calibration Laboratories; ICMR: Indian Council of Medical Research.
It is estimated that 65%–75% of laboratory services are located in urban areas, 33 although around 65% of the country’s population resides in rural areas. 34 According to the NHRR data, nearly 80% (33,829 out of 42,105) of the enumerated laboratories are in urban areas with a few being in rural areas. 35 Such disproportionate distribution limits access and leads to a significant proportion of illnesses remaining undetected and untreated. 36
The large number of medical laboratories in urban areas leads to their excessive use. The ‘preventive or wellness packages’ offered as routine health check-ups comprise nearly 90 tests, 37 many of which are not recommended by any guidelines and have little impact on morbidity and mortality. Besides the unnecessary financial burdens on customers and the waste of resources, such overuse may lead to over-diagnosis and over-treatment. For instance, there is an increased diagnosis of small thyroid nodules, which are otherwise harmless, but once detected are treated for papillary carcinoma. 38 Another reason for overuse is greater reliance on laboratory results by clinicians to reassure and protect themselves from legal liability. 39
Quality and accuracy of results
Lack of standardization, with different laboratories adopting different standards, and supplier-induced demand fuelling the industry, 40 raises concerns regarding the reliability of test results. Unqualified professionals, proxy signatories, and use of substandard equipment, consumables, and reagents are some other reasons for the questionable quality of laboratory services. 41 Such shortcomings can have serious consequences, such as false positives (e.g., misdiagnosis of breast cancer or a life-threatening condition when none exists) and false negatives (e.g., missed diagnosis of leukaemia). 42 A couple in Bangalore would have put the life of their newborn at risk had they not taken a repeat test for thalassemia. 43
The data on quality and accreditation of medical laboratories in India are inadequate. 44 Accreditation, which involves periodic third-party audits, is a voluntary mechanism to attain pre-defined standards that ensure the reliability of results. 45 Participating in external quality assurance schemes (EQAS) 46 , a measure of quality control, is mandatory for accreditation by NABL. 47 However, very few laboratories in India are thus accredited. 48 During the COVID-19 pandemic, inter-laboratory quality control mechanism was used by the ICMR to ensure the reliability of COVID-19 tests. 49
The CEA makes it mandatory to adopt minimum standards for the calibration of equipment and reagents and standard operating procedures. However, quality control mechanisms such as EQAS and inter-laboratory comparison are listed as desirable rather than essential. 50 Some states such as Tamil Nadu 51 and West Bengal prescribe more stringent quality standards than the central Act, whereas others such as Delhi, Gujarat, Madhya Pradesh, Maharashtra, Meghalaya, Nagaland, Odisha, and Punjab are less strict.
In a national-level survey of 29,000 people, only 20% fully trusted the laboratories they used, and 32% reported receiving one or more incorrect laboratory reports. 52 The health minister of Uttar Pradesh acknowledged that the state was unable to ensure quality in over 90% of the laboratory services that were in the private sector. 53
Human resources
Numerous cases have been reported from across the country in which unauthorized personnel generate reports, sometimes without performing the tests – a practice known as ‘sink tests’. 54 The issue of who can operate medical laboratories is a grey area in several states. Pathologists 55 and laboratory technicians 56 have been embroiled in a legal battle claiming their authority to operate medical laboratories. 57 A 2010 Gujarat High Court ruling, upheld by a 2017 Supreme Court ruling, barred technicians from operating laboratories independently and mandated them to work under the supervision of pathologists. 58 However, many laboratories that operate only with technicians circumvent this, by ‘purchasing’ signatures from qualified professionals for INR 15,000–20,000 a month. The signature of a pathologist employed with the government of Gujarat was used by nine private laboratories. 59
In states such as Tamil Nadu, West Bengal, and those where the CEA is in force, requirements for personnel along with their minimum qualification are prescribed. However, the regulations in Andhra Pradesh, Madhya Pradesh, Meghalaya, Nagaland, and Odisha are ambiguous and inadequate in this regard. The Directorate of Medical Education and Research in Maharashtra cited a shortage of qualified pathologists as the reason to permit technicians to operate laboratories by themselves, particularly in rural areas. 60 Taking cognisance of the short supply of pathologists, 61 the CEA, in contrast to the Supreme Court ruling, permitted those with an MSc or PhD in pathology or a related field to sign the test reports with a mandatory disclaimer ‘reports are for the use of medical practitioners and are not medical diagnoses as such’. 62 However, a pathologist filed a petition in the Delhi High Court seeking abrogation of these rules. 63
Cost and charging practices
The cost of diagnostic investigations comprises a significant proportion of the total healthcare cost, a reason why people may forego necessary investigations. 64 In the National Sample Survey 75th Round (2017/18), the average out-of-pocket expenditure on diagnostics is reported to be 12.6% of the total healthcare cost for outpatient care. 65 In cases of hospitalization, the cost ranges from 9.5% in the private sector to 17.7% in the public sector. 66 There is little or no cost control on diagnostics in India. Although the CEA prescribes the fixing of rates, none of the states or UTs where the Act is in force has done so. In states such as Andhra Pradesh and Madhya Pradesh, charges must be disclosed to the registering authority and cannot be revised more than once a year. In Meghalaya, the charges need to conform to the norms prescribed by the state government. In addition to the provisions of the CEA, the legislation in some states such as Karnataka, Kerala, and West Bengal mandate that the rates be publicly displayed.
However, the diagnostic industry has been pushing back the government’s attempt to regulate charges and to bring in greater transparency. The industry contends that some laboratories offer cheaper services by compromising on quality standards. That argument, however, does not explain the wide variation in costs of the same investigations by different laboratories. For instance, an MRI scan can cost from INR 1,000 to INR 6,000, 67 and a thyroid profile test can cost from INR 100 to INR 3,100. 68 Such wide disparity makes it difficult to ascertain whether the differences in cost are on account of quality or a lack of price control. Taking cognizance of the inflated charges, the West Bengal Clinical Establishment Regulatory Commission ordered two private hospitals to refund the excess amount to the patients, including the inflated charges for liver function tests. 69 Although the cost of Real-time reverse transcriptase-polymerase chain resction (RT-PCR) and rapid antigen tests was regulated during the COVID-19 pandemic in several states under the Epidemics Diseases Act 1897, 70 under routine circumstances the costs remain unregulated.
What adds to the cost of diagnostics is the practice of a ‘cut’, or commission, offered by laboratories to referring clinicians, which can account for nearly 40%–50% of the cost. 71 This is despite the Indian Medical Council (Professional Conduct, Etiquette, and Ethics) Regulations, 2002, prohibiting such practices by doctors. 72 In the Local Circles survey mentioned above, the majority of the 29,000 people suspected a corrupt nexus between laboratories and doctors and 59% believed that their doctors had ordered more tests than were needed. 73
The legislation in some states such as Meghalaya and West Bengal provides for ‘poor and indigent people’ to have access to free investigations. The central and state governments have been taking measures to provide free diagnostic services. 74 However, no definitive data are available on the utilization of the central government’s Free Essential Diagnostics Service Initiative or state-specific initiatives and their impact on out-of-pocket expenditure. 75
Information provision and reporting
Medical laboratories are liable to provide information to users and the state to maintain transparency and accountability. In states or UTs where the CEA is in force, laboratories need to display their charges publicly, and hospitals need to provide information about investigations in the discharge summary. In Chhattisgarh and Kerala, inspection reports of the laboratories need to be placed in the public domain. In West Bengal, information regarding available services, personnel, mechanism to avail concession, and the charter of the patient’s rights needs to be displayed. A discharge summary including laboratory investigations needs to be provided even if the patient leaves against medical advice. For the benefit of users, the MoHFW is required to publish a list of registered clinical establishments including medical laboratories for those states and UTs where the CEA is in force. Among other states, Tamil Nadu and Kerala have similar provisions.
The increasing use of digital reports is convenient and quick, but there are concerns about the safety of data. In a breach of privacy, data regarding laboratory investigations of millions of people were freely available in the public domain. 76 Similar breaches have occurred repeatedly. 77 Medical data storage does not enjoy adequate legal protection in India. 78
All public and private laboratories registered under the CEA must provide monthly data on all tests performed, sorted by specialty, and those related to the 22 listed communicable diseases, to the District Registering Authority. 79 Similar provisions are prescribed in states such as Andhra Pradesh, Chhattisgarh, Goa, Karnataka, Kerala, Tamil Nadu, and West Bengal. Further, a failure to report notifiable diseases is a criminal offence under Section 188 of Indian Penal Code (45 of 1860). 80 During the COVID-19 pandemic, GoI invoked the Epidemic Diseases Act of 1897 to make the reporting of all positive COVID-19 cases mandatory. 81 At the same time, concerns have been raised that such notifications can have adverse consequences for patients suffering from diseases that attract a social stigma. 82 Under the Integrated Diseases Surveillance Programme (IDSP), public-sector laboratories and private-sector laboratories that are part of the IDSP reporting network provide weekly information on more than 20 epidemic-prone diseases. 83
Newer technologies
Simpler tests and a more diverse range of tests are available with the advent of new technologies, all of which contribute to wider and more frequent testing. Demand for at-home diagnostics has increased manyfold since the COVID-19 pandemic, including those for monitoring infections, cholesterol, blood pressure, oxygen, and heart rate, among others. 84 However, neither the accuracy nor the reliability of these test kits is necessarily assured. 85
The situation in India is more alarming because of the country’s weak regulations governing diagnostic kits and devices. Medical devices were minimally regulated as drugs under the Drugs and Cosmetics Act 1940, until the Medical Devices Rules 2017 and 2020 were notified to bring all medical devices under regulation by October 2023. 86 Hence, several diagnostic kits and devices entered the Indian market without regulation. 87
Another aspect of concern is the increasing use of artificial intelligence (AI) in medical diagnosis and how it may impact the structure and functioning of the sector by virtue of its level of accuracy, efficiency, and ability to analyse large data sets, among other factors. The security of patient data is a legitimate concern in the absence of appropriate regulatory provisions. 88 AI-enabled self-testing kits are vulnerable to cybersecurity breaches. For instance, the communication chain between an automated insulin pump and the attached blood glucose metre can be hacked, disrupting insulin delivery to the body and potentially leading to hypoglycaemia or hyperglycaemia. 89
In addition, the use of self-test kits, although convenient, may reduce reporting to the state (as witnessed with home-testing of COVID-19), 90 thereby impacting data-based planning and management.
Discussion and conclusions
Medical laboratory services in India have essentially grown in an unregulated environment. With no mandatory registration and minimum standards, they have been operating as any other commercial enterprise with negligible quality control. Consequently, news reports about fake laboratories and inaccurate test results continue to appear in the media. It is only in the past few years that regulations pertaining to medical laboratories have come into existence, though they are still non-existent or outdated in some states. Whereas legislation in states such as Chhattisgarh, Karnataka, Kerala, Tamil Nadu, and West Bengal is more comprehensive, Delhi and Maharashtra have made no progress in this regard, and legislation in other states such as Madhya Pradesh, Meghalaya, Nagaland, and Odisha is deficient in content. Coverage of state-run laboratories, laboratories within hospitals, collection centres, and home collection services varies across state regulations. Online health service aggregators continue to provide laboratory services illegally despite court orders and GoI directives to discontinue such operations. The users, oblivious to such breach, are attracted by the convenience and discounts that such operators offer.
Data on the availability of medical laboratories in the country indicate inequitable services across regions, although their exact number and distribution remain unknown. A registry of medical laboratories in the country giving information on their number and geographical spread will assist in assessing their adequacy and in planning to meet the shortfall.
Due to the lack of a quality control mechanism, there is no assurance that the results of a sample tested in two laboratories would be the same. Universal adoption of external and inter-laboratory quality control mechanisms that are mandatory for accreditation by NABL, but not a statutory requirement, in all states will help in standardization and ensuring quality, make the reports generated by different laboratories more credible, and reduce the burden of repeat tests on users in cases of referrals or a second opinion.
Ambiguity about who can operate medical laboratories and the responsibility of different cadres of technical staff remains unresolved. The insistence on pathologists operating medical laboratories restricts access in under-served areas. The graded level of human resources prescribed in the CEA (Amendment) Rules, 2020, depending on the scope of services offered by a laboratory is practical and realistic.
There are few studies on the costing of diagnostic services in India. However, media reports, including those during the COVID-19 period, are indicative of the inexplicably high costs. 91 The Economic Survey 2017–18 reveals disparity in the average cost of diagnostic tests and the wide range of costs across various cities in the country. Unethical practices and resistance by the industry to submit to price control contribute to the high costs of diagnostics. It is necessary to maintain transparency in the costs charged to gain the users’ trust and confidence. The action taken by the West Bengal Clinical Establishment Regulatory Commission on overcharging by hospitals is noteworthy and needs to continue and be consistent.
The provision for making information on registered laboratories publicly available under the CEA 2010, the Kerala Clinical Establishments (Registration and Regulation) Act 2018, and the Tamil Nadu Clinical Establishments (Regulation) Rules 2018 can be very useful to the users in choosing laboratories.
Reporting to the government by both public- and private-sector laboratories will help in maintaining data on disease burden and in planning health services. However, given that regulations across states differ in their reporting requirements, it is not comprehensive. The increasing emergence of new diseases makes data sharing with the authorities imperative in the detection of outbreaks and taking necessary control measures in a timely fashion. It is recommended that reporting under IDSP, which is currently optional for private laboratories, should be made mandatory.
While harnessing technological advancements is to be welcomed, caution is warranted in the use of newer technologies such as self-testing tools, AI-assisted testing, point-of-care testing, and digitization of data until the regulations governing these matters are strengthened. The Medical Devices Rules 2017 and 2020 do not regulate all diagnostic kits and devices currently. The lack of data protection laws in the country also creates the potential for the violation of users’ privacy when laboratories digitally store data and report it online on government web portals.
The Indian diagnostic market is expected to witness a compound annual growth rate of nearly 12% and offer substantial financial returns to investors in the next 10 years and is attracting both established and new investors. 92 Considering the industry’s keen interest in expanding markets, the need for effective regulation of medical laboratories to protect the users’ interests and promote rational medicine cannot be overemphasized. A combination of voluntary and statutory mechanisms such as accreditation and regulations would be useful instruments for ensuring quality in diagnostic services. A greater push towards updating outdated legislation and their implementation in letter and spirit is warranted. Research on their effectiveness at the state level would help in understanding the impact and suggest ways of further improvement.
Footnotes
Acknowledgements
The authors would like to thank the following colleagues at Health Systems Transformation Platform, Mr. Rajeev Sadanandan, Dr. Kumaravel Ilangovan, and Dr. Pratheeba John for their valuable comments on the earlier drafts of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
1.
2.
3.
4.
M. Krishnan, ‘COVID: How Is India Tackling a Surge in Fake Test Reports?’, DW, 17 September 2021, available at https://p.dw.com/p/40SR7; S. Sharma, ‘Why Were Coronavirus Testing Kit Approvals Hidden From Public View?’, Scroll.In, 7 April 2020, available at ![]()
5.
6.
F. A. Delwiche, ‘Mapping the Literature of Clinical Laboratory Science’, Journal of the Medical Library Association 91(3) (2003), p. 303–310; M. L. Wilson, K. A. Fleming, M. A. Kuti, L. M. Looi, N. Lago, and K. Ru, ‘Access to Pathology and Laboratory Medicine Services: A Crucial Gap’, The Lancet 391(10133) (2018), pp. 1927–1938.
7.
U. P. Rohr, C. Binder, T. Dieterle, F. Giusti, C. G. M. Messina, E. Toerien, H. Moch, and H. H. Schäfer, ‘The Value of In Vitro Diagnostic Testing in Medical Practice: A Status Report’, PLoS ONE 11(3) (2016), e0149856.
8.
Allied Health Section, ‘Ministry of Health and Family Welfare, “Model Curriculum Handbook, Medical Laboratory Science”’, Government of India (2015–2016), p. 162, available at https://main.mohfw.gov.in/sites/default/files/Model%20Curriculum%20Handbook-Medical%20lab%20sciences.pdf; M. L. Bayot, G. D. Brannan, P. Naidoo, Clinical Laboratory, StatPearls [Internet]. Treasure Island (FL), (StatPearls Publishing LLC, 2022), available at ![]()
9.
10.
12.
K. Kahol, ‘India’s Diagnostics Industry: Opportunity or Challenge?’, CNBCTV18.Com, 17 June 2021, available at https://www.cnbctv18.com/healthcare/indias-diagnostics-industry-opportunity-or-challenge-9694131.htm; NATHEALTH, ‘An Assessment of India’s Laboratory Diagnostic Industry’, 2022, available at ![]()
13.
The CEA, 2010, section 2, c (ii) defines laboratories as places connected ‘with the diagnosis or treatment of diseases where pathological, bacteriological, genetic, radiological, chemical, biological investigations or other diagnostic or investigative services with the aid of laboratory or other medical equipment, are usually carried on’. Medical laboratories run by India’s armed forces are exempt.
14.
15.
Delhi Nursing Homes Registration Act, 1953; Bombay Act No. XV of 1949 (Maharashtra Nursing Homes Registration Act) as amended by Mah. 24 of 2012, s. 2, Sch, entry no. 36, with effect from 1 May 1960.
16.
Gujarat Clinical Establishments (Registration and Regulation) Act, 2021, replaced the Gujarat Nursing Homes Registration Act, 1949; newly enacted Kerala Clinical Establishments (Registration and Regulation) Act, 2018, and Punjab Clinical Establishments (Registration and Regulation) Act, 2020.
17.
Andhra Pradesh Allopathic Private Medical Care Establishments (Registration and Regulation) Rules, 2007; Karnataka Private Medical Establishments (Amendment) Rules, 2018; Madhya Pradesh Upcharyagriha Tatha Rujopchar Sambandhi Sthapanaye (Registrikaran Tatha Anugyapan) Rules, 1997; Manipur Nursing Home and Clinics Registration Act 1992; Meghalaya Nursing Home (Licensing and Registration) Rules, 2010; Nagaland Health Care Establishments Rules, 2002; Odisha Clinical Establishments (Control and Regulation) Rules, 2018; West Bengal Clinical Establishment (Registration, Regulation and Transparency), Rules, 2017.
18.
19.
V. Pilla, ‘Diagnostic Chain Metropolis Says Ramping Up Labs, Collection Centres’, The Economic Times, 25 November 2021, available at https://economictimes.indiatimes.com/industry/healthcare/biotech/healthcare/diagnostic-chain-metropolis-says-ramping-up-labs-collection-centres/articleshow/87912061.cms?from=mdr; PTI, ‘SRL Diagnostics Allocates Rs 300-500 cr to Expand Lab, Collection Centres Network’, BusinessToday.In, 5 September 2021, available at ![]()
20.
Directorate General of Health Services (DGHS), Minutes of 11th Meeting of the National Council for Clinical Establishments, 2018.
21.
Technology platforms that facilitate collection of samples from users and transport them to the collaborating laboratories.
22.
23.
24.
R. Bhushan, ‘Department of Health & Family Welfare Order No. D.O. No.-C.18018/12/2020-MS dated 19 January 2021’, MoHFW, GoI (2021).
25.
26.
West Bengal Clinical Establishment (Registration, Regulation and Transparency), Rules, 2017.
28.
A joint project of the Central Bureau of Health Intelligence, DGHS, MoHFW, and the National Remote Sensing Centre, Indian Space Research Organisation, initiated in 2017.
30.
31.
NABL, ‘Directory of Accredited Medical Laboratories’, NABL 600 (2023).
32.
33.
R. RamPrakash and J. Arun, ‘Access to Diagnostic Services Through Health Insurance Schemes: Lessons from a Southern Indian State for Post-COVID Era’, Journal of Health Management 24(3) (2022), pp. 347–355; ReportLinker, ‘India Diagnostic Labs Market, By Provider Type, By Test Type, By Sector, By End User, By Region, Competition Forecast & Opportunities, 2027’, 2022, available at https://www.reportlinker.com/p06128276/India-Diagnostic-Labs-Market-By-Provider-Type-By-Test-Type-By-Sector-By-End-User-By-Region-Competition-Forecast-Opportunities-FY.html?utm_source=GNW; A. Misra, ‘Diagnostic Industry in India’, Digital First Magazine, 25 October 2021, available at ![]()
35.
36.
D. Barik and A. Thorat, ‘Issues of Unequal Access to Public Health in India’, Frontiers in Public Health, 3 (2015), p. 245; V. Yellapa, N. Devadasan, A. Krumeich, N. Pant Pai, C. Vadnais, M. Pai, and N. Engel, ‘How Patients Navigate the Diagnostic Ecosystem in a Fragmented Health System: A Qualitative Study From India’, Global Health Action 10(1), (2017), p. 1350452.
37.
Dr. Dangs Lab, ‘Towards Better Health -Test Packages’, available at https://www.drdangslab.com/Packages.aspx; Dr. Lal PathLabs, ‘Test List’, 2018, available at ![]()
38.
S. Bhagwat and S. A. Pai, ‘Medical Ethics in Laboratory Medicine: A Review, With an Oath for Pathologists’, Indian Journal of Medical Ethics 5(1), (2020), pp. 39–44; B. Honnekeri, A. Vyas, D. Lokhandwala, A. Vaishnav, A. Vaishnav, M. Singhal, P. Barwad, G. K. Panicker, and Y. Lokhandwala, ‘Routine Health Check-Ups: A Boon or a Burden?’, National Medical Journal of India 29(1), (2016), pp. 18–21.
39.
S. Dudeja and N. Dhirar, ‘Defensive Medicine: Sword of Damocles’, The National Medical Journal of India 31(6), (2018), pp. 364–365.
40.
41.
42.
S. S. Dutta, ‘How Diagnostic Labs Are Failing Patients in India in Terms of Poor Standards’, The New Indian Express, 2 March 2020, available at https://www.newindianexpress.com/thesundaystandard/2020/mar/01/how-diagnostic-labs-are-failing-patients-in-india-in-terms-of-poor-standards-2110382.html; P. Raju, ‘Illegal Path Labs: India’s Open Secret’, Express Healthcare, 30 July 2019, available at https://www.expresshealthcare.in/lab-diagnostics/illegal-path-labs-indias-open-secret/413141/; A. S. Rozario, ‘Pathless Pathology: Why Delhi’s “Unethical” Labs Love Lawlessness’, The Quint, 2 October 2019, available at ![]()
43.
44.
Ministry of Finance, Economic Survey 2017-18 (GoI, 2018), II, p. 177.
45.
The NABL, an autonomous body under the Quality Council of India, offers a voluntary accreditation system for laboratories in accordance with the International Organization for Standardization, ISO 15189:2012: Medical laboratories: Requirements for quality and competence.
46.
EQAS involves evaluation of laboratories by an outside agency based on their analytical performance of tests on samples supplied by the external agency.
47.
S. Yerram, D. V. Sripad, and V.S. Prabodh, ‘External Quality Assurance Scheme (EQAS): Criteria for Evaluating Performance of a Laboratory’, IOSR Journal of Biotechnology and Biochemistry 4 (4) (2018), pp. 16–20; NABL, ‘NABL 112: Specific Criteria for Accreditation of Medical Laboratories’, Issue No. 4, Issue date: 11-Feb-2019 Amendment No. 01, Amendment date: 26-Apr-2019.
48.
NABL, ‘Directory of Accredited Medical Laboratories’, NABL 600 (2023).
49.
H. Kaur, L. Mukhopadhyay, N. Gupta, N. Aggarwal, L. Sangal, V. Potdar, F. Y. Inbanathan, J. Narayan, S. Gupta, S. Rana, N. Vijay, H. Singh, J. Kaur, V. Kumar, N. Kaundal, P. Abraham, and V. Ravi, ‘External Quality Assessment of COVID-19 Real Time Reverse Transcription PCR Laboratories in India’, PLoS ONE 17(2) (2022), p. e0263736.
50.
Clinical Establishments (Central Government) Amendment Rules, 2018.
51.
Tamil Nadu Clinical Establishments (Regulation) Rules, 2018.
52.
53.
Dutta, ‘How Diagnostic Labs Are Failing Patients in India’.
54.
55.
Doctor of Medicine or Diplomate of the National Board in Pathology or related field or MBBS with PhD in pathology or a related field.
56.
Diploma or bachelor’s in science with specialization in medical laboratory technology or a postgraduate in related fields.
57.
S. J. Mukhopadhyay and K. M. Thaker, Association of Pathologist of Bhavnagar & 17 - petitioner(s) vs Secretary Health Department and eight other respondent(s), High Court of Gujarat judgement dated September 17, 2010 (Application No. 7999 of 1998, 2010).
58.
59.
60.
61.
Dutta, ‘How Diagnostic Labs Are Failing Patients in India’.
62.
Clinical Establishments (Central Government) Amendment Rules, 2020.
63.
PharmaBiz Bureau, ‘HC Seeks Responses of Govt & NMC on Repeal of CEA Rules, Allowing Non-Pathologists to Certify Medical Reports’, PharmaBiz.Com, 22 December 2020, available at http://www.pharmabiz.com/NewsDetails.aspx?aid=134289&sid=1
64.
Yellapa et al., ‘How Patients Navigate the Diagnostic Ecosystem’.
65.
National Statistics Office, Ministry of Statistics and Programme Implementation, Key Indicators of Social Consumption in India: Health, NSS 75th Round (New Delhi, GoI, November 2019). p. 23 (Statement 3.22).
66.
Op. cit., p. A-21 (Table A19). The total cost of care in private sector (INR 31,845) is nearly seven times that in the public sector (INR 4,452) and the cost of diagnostics (INR 3,038) is nearly four times that in the public sector (INR 791).
67.
S. Dey, ‘Lack of Regulation Haunts India’s Test Labs’, Business Standard, 20 April 2014, available at https://www.business-standard.com/article/companies/lack-of-regulation-haunts-india-s-test-labs-114041400536_1.html; L. Yadav, ‘Diagnostic Chains, Pathologists Hail Govt Move to Regulate Diagnostic Sector’, Pharmabiz.Com, 22 May 2017, available at http://www.pharmabiz.com/NewsDetails.aspx?aid=102081&sid=1
68.
Ministry of Finance, Economic Survey 2017-18 (GoI, 2018), II, p. 176.
69.
70.
T. Barnagarwala, ‘Maharashtra: Govt Caps Rates for HRCT Test’, The Indian Express, 25 September 2020, available at https://indianexpress.com/article/india/maharashtra-govt-caps-rates-for-hrct-test-6609772/; Times Now Digital, ‘COVID-19: How Much Does RT-PCR Test Cost? Find State-Wise Price Here’, Times Now Digital, 2 December 2020, available at ![]()
71.
A. Baheti, ‘Cut Practice in Radiology and Medicine’, Cafe Roentgen, 8 October 2021, available at https://caferoentgen.com/2021/10/08/cut-practice-in-radiology-and-medicine/ (accessed 29 August 2022); S. Nagral and S. Nundy, ‘We Need to End “Cut” Practice in Indian Healthcare’, The BMJ Opinion, 7 July 2017, available at ![]() (accessed 29 August 2022).
(accessed 29 August 2022).
72.
A. Jain, S. Nundy, and K. Abbasi, ‘Corruption: Medicine’s Dirty Open Secret’, BMJ 348 (2014), p. g4184; Department-Related Parliamentary Standing Committee on Health and Family Welfare, ‘73rd Report on IMC (Amendment) Bill, 2013’, Parliament of India, Rajya Sabha, 20 November 2013.
73.
74.
National Health Mission, National Health Mission: Free Diagnostics Service Initiative (New Delhi, MoHFW, GoI, 2015), pp. 1–4.
75.
RamPrakash et al., ‘Lessons From a Southern Indian State’, pp. 347–355.
76.
77.
P. Dixit, ‘A Lab Accidentally Released the Medical Reports of 43,000 People, Including HIV Patients’, BuzzFeed News, 2 December 2016, available at https://www.buzzfeednews.com/article/pranavdixit/the-medical-reports-of-43000-people-including-hiv-patients-w; O. C. Kurien, ‘Data, Privacy, Pandemic: India Just Had the Biggest Medical Records Breach Ever’, Observer Research Foundation, 12 January 2021, available at ![]()
78.
A. Abbas and J. Simte, ‘India: Health Data Under the Data Protection Bill, 2021 and Recommendations of the Joint Parliamentary Committee on Data Protection’, Mondaq, 14 January 2022, available at https://www.mondaq.com/india/privacy-protection/1150676/health-data-under-the-data-protection-bill-2021-and-recommendations-of-the-joint-parliamentary-committee-on-data-protection#; V. Lalwani, ‘Explainer: Does India Need Digital Health Records and Can the Draft Policy Protect Sensitive Data?’, Scroll.In, 6 September 2020, available at ![]()
79.
Clinical Establishments (Central Government) Amendment Rules, 2018. C. Information to be collected Monthly from Diagnostic Medical Laboratory under Clinical Establishment Act, 2018.
80.
81.
82.
83.
Central Surveillance Unit, IDSP, ToT Training Manuals for State/District Surveillance Officers, State RRTs & Nodal Officers from Medical Colleges (New Delhi, MoHFW, GoI, 2020), pp. 78–82.
84.
85.
M. L. Millenson, J. L. Baldwin, L. Zipperer, and H. Singh, ‘Beyond Dr. Google: The Evidence on Consumer-Facing Digital Tools for Diagnosis’, Diagnosis 5(3) (2018), pp. 95–105; E. J. Tidy, B. Shine, J. Oke, and G. Hayward, ‘Home Self-Testing Kits: Helpful or Harmful?’, British Journal of General Practice 68(673) (2018), pp. 360–361.
86.
Department of Pharmaceuticals, Approach Paper on draft National Medical Devices Policy 2022 for Consultation (New Delhi, Ministry of Chemicals and Fertilisers, GoI, March 10, 2022), pp. 9–10.
87.
88.
Y. Kumar, A. Koul, R. Singla, and M. F. Ijaz, ‘Artificial Intelligence in Disease Diagnosis: A Systematic Literature Review, Synthesizing Framework and Future Research Agenda’, Journal of Ambient Intelligence and Humanized Computing 14 (2023), pp. 8459–8486; Meticulous Research, ‘Artificial Intelligence in Medical Diagnostics Market’, Globe Newswire, 7 July 2022, available at ![]()
90.
91.
92.
R. V. Vora, ‘Diagnostics, a Test Indian Corporates Take Hoping for Positive Results’, The Hindu Business Line, 21 August 2022, available at https://www.thehindubusinessline.com/specials/pulse/diagnostics-a-test-indian-corporates-take-hoping-for-positive-results/article65726528.ece; P. Vardhan and T. Mukherjee, ‘India’s $9 Billion Diagnostics Industry: A Midsummer Private Equity’s Dream’, ETHealthWorld, 15 October 2022, available at ![]()
