Abstract
Judson Chesterman (1903–1987) was a surgeon working in Sheffield, United Kingdom in the mid-20th century. Born in Bath, Somerset, he attended Bristol Medical School before completing junior doctor positions around England. He developed his skills in thoracic surgery during a Fellowship with Evarts Graham (1883–1957) at Barnes Hospital, St Louis, Missouri and by the mid-1950s was also performing a large number of closed cardiac procedures. In 1955, he performed the first mitral valve replacement in the world, using a prosthesis of his own design, but the patient only survived for around 18 hours. Recognising the limitations of off-pump surgery, he visited the University of Minneapolis before building his own bypass machine and used it in two patients, the first in February 1957, one of the earliest outside the United States of America to do so. In retirement he established an osteoarchaeology laboratory and made additional contributions to that field.
Introduction
During the 19th century, developments such as general anaesthesia, antisepsis and asepsis revolutionized the feasibility and scope of safe surgical treatment. By the turn of the 20th century, procedures were essentially being undertaken inside every body compartment except the pericardium. Despite the success of some early cardiac procedures such as Ludwig Rehn's (1849–1930) repair of a cardiac stab wound, operating on the heart was generally unacceptable. 1 Theodor Billroth (1829–1894) is said to have made remarks along the lines that any surgeon attempting cardiac surgery would lose the respect of his colleagues. In fact, this is probably misattributed but the sentiment seems a reasonable reflection of the prevailing attitude at the time. 2 Yet by the end of the 20th century, cardiac surgery was routine and indeed into the 21st continues to offer definitive treatment for a broad range of cardiovascular diseases, which together remain the world's commonest cause of death. The decades of the 1950s and 1960s probably saw the most sustained period of rapid development in cardiac surgery. At the start of the 1950s, cardiac surgery was rarely performed and when it was, only closed procedures without the aid of cardiopulmonary bypass (CPB, using a ‘heart-lung’ machine) were contemplated. 3 In contrast, by the end of the 1960s, a wide range of operations were taking place all over the world, including coronary artery bypass grafting, valve surgery and cardiac transplantation, all possible only because of the availability of successful CPB. Much has been written about pioneering figures in the field during those times, particularly when high profile, such as Christiaan Barnard (1922–2001), Russell Brock (1903–1980) and Michael DeBakey (1908–2008).3–5 But as well as the more well-known individuals, there are those less acknowledged, often found working in smaller provincial institutions. Recognising the contribution of these individuals is not just about giving them due credit, but moreover leads to greater understanding of how cardiac surgery as an entity evolved, illustrating how small steps, often initially unsuccessful, were made by many practitioners before wide and successful major changes could be implemented. This article surveys the life and work of one such individual, Judson Chesterman (1903–1987), with a focus on his contributions to cardiac surgery.
Early life and influences
Judson Tyndale Chesterman (Figure 1) was born on 16th February 1903 at Woodland Place, Bathwick Hill in Bath, Somerset, England. 6 His father, William Thomas Chesterman (1856–1923), was a prominent local solicitor. Educated at Monkton Combe School along with his seven elder brothers, Chesterman's schooldays traversed the World War I, during which he volunteered as a Medical Auxiliary at the Bath War Hospital, and the injuries he witnessed left him ‘fascinated and appalled’. 7 Another key influence was his next eldest brother, Clement Clapton Chesterman (1894–1983), who had attended Bristol Medical School ahead of Judson and later became a prominent physician and medical missionary, specialising in tropical diseases and was later knighted for his achievements. 8

Judson Tyndale Chesterman (1903–1987). Image courtesy of the Chesterman family.
In training
Judson Chesterman entered Bristol University Medical School in 1920 but he did not complete his undergraduate medical studies for seven years, two longer than the standard five-year course. The delay was due to illnesses, including tuberculosis, and he spent some time in a Swiss sanatorium. 7 This perhaps seeded his lifelong enthusiasm for mountaineering, regularly going back to climb the Alps, reportedly including the Matterhorn with fellow Bristol-educated surgeon Trevor Hellier Berrill (1905–1982). 9 Clearly a gifted undergraduate, he won multiple prizes, including the Faculty Gold Medal upon graduation, and his illnesses did not prevent him being awarded university blues in swimming and water polo. 10
After qualifying, Chesterman started house jobs at Bristol Royal Infirmary, including working for Professor Arthur Rendle Short (1880–1953). 11 Short had a particular interest in the treatment of shock, and Chesterman recalled performing experiments on different intravenous fluids. 11
Subsequently, he rotated through various Senior House Officer appointments. For a time he worked in London, including at St Paul's Hospital and Great Ormond Street Hospital, but later reported that although the teaching was ‘excellent’ he found ‘even with the English FRCS discussion with the chiefs was frowned upon … I said adieu to the Gods in London without regret’. 7 During this time he completed both the Fellowship of the Royal College of Surgeons of England and Membership of the Royal College of Physicians of London. 10
After a spell as Resident Surgical Officer at Sheffield Royal Hospital, he became Surgical Registrar there in January 1935, staying until August 1936. 10 Although new to Sheffield himself, it was a city with which the Chesterman family had a strong connection. Judson's great-grandfather, James Chesterman (1804–1876) had moved there in 1829 to develop a tool manufacturing business, having invented the flexible steel tape measure. Judson's father had, in fact, been born on Rockingham Street just round the corner from the Sheffield Royal Hospital. 6
Flourishing general and thoracic surgeon
Chesterman developed a particular interest in thoracic surgery and in 1936 secured a Medical Research Council travelling fellowship to the United States to focus on this. On arriving in New York on 4 September 1936 on the RMS Berengaria, he visited a number of local medical establishments including Mount Sinai and the New York Medical Center. 12 In Philadelphia, he worked for a short period at the Jefferson Hospital and also under the supervision of the ‘Father of Endoscopy’ Chevalier Jackson (1865–1958) at the Temple Hospital. 13 After brief visits to Baltimore, Washington and Cincinnati, he arrived at Barnes Hospital, Washington University, St Louis, Missouri, where he would spend five months as a fellow to the influential surgeon Evarts Graham. Graham had the distinction of performing the first pneumonectomy for lung cancer in 1933, 14 and had also performed trailblazing animal work proving the feasibility of cardiac surgery. 15 Other visiting fellows had recently included Russell (later Lord) Brock. Graham had a great influence on Chesterman, who later admitted to keeping a signed photograph of him on his desk ‘lest I am tempted to take “short cuts” in work’. 7 They maintained regular written correspondence, throughout the war, and up to Graham's death, and Graham stayed with Chesterman when he visited the United Kingdom in 1947, 1954 and 1956. 16 From his experience in the United States, he felt three features contributed to the success of hospital medicine, which he resolved to emulate in his own practice: first, the emphasis on understanding disease in the context of the underlying physiology; second, enthusiasm for new ideas; and third, ready access to high-quality research facilities. 7
In 1938, Chesterman secured a Consultant post as Surgeon to the Municipal Service, a regional body providing medical care to those unable to gain admission to the more prestigious voluntary hospitals. He was appointed to the City General in Sheffield, an acute referral hospital founded in 1912 to complement the Fir Vale Infirmary, a former workhouse located on the same site. In 1967, the two institutions merged to form the Northern General Hospital.
There was not enough work though to justify specialising in thoracic surgery full-time, and he remained a general surgeon in the broadest sense, treating abdominal cases, orthopaedics and even respiratory medicine. He typically had around 100 inpatients under his name at any one time, rising as high as 500 during World War II. Chesterman developed a particular interest in acute intestinal obstruction leading to the publication of a monograph that remained an authoritative textbook for some time. 17 He showed his pioneering spirit, for example performing the first total gastrectomy for haematemesis, which was reported in the British Medical Journal in 1954. 18
As a guide to his caseload at this time, handwritten slips containing his cases from various years remain in existence. The summaries for the years 1955 to 1958 are shown in Table 1.
Breakdown of Chesterman's patient numbers and deaths at the City General Hospital, Sheffield, 1955–1958.
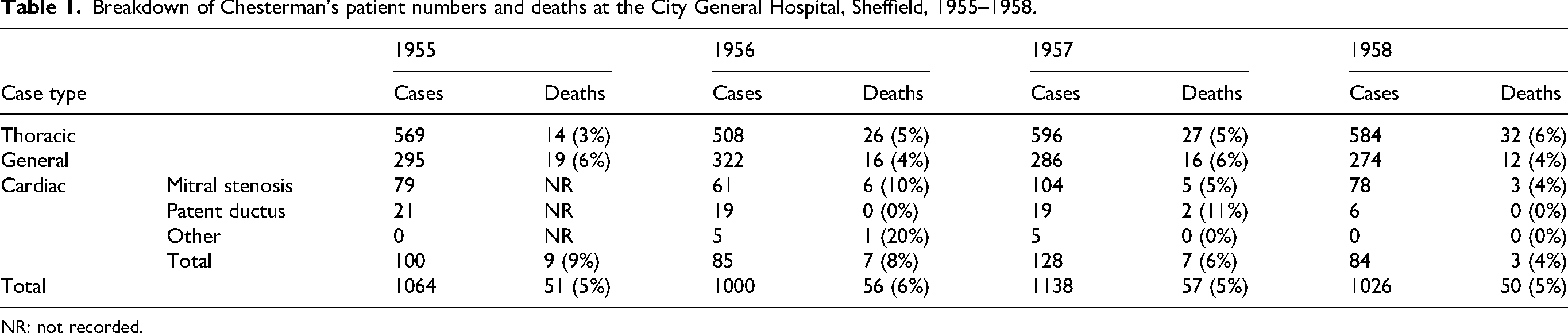
NR: not recorded.
These indicate a brisk turnover of patients with a mix predominantly of thoracic and general patients. Of note, however, is that he was by now performing a significant number of cardiac procedures.
Chesterman as cardiac surgeon
After the first mitral valvotomy had been performed in 1923 by Elliott Cutler (1888–1947) at the Peter Bent Brigham Hospital, Boston, USA, there was a flurry of these ‘closed’ cardiac procedures, in which the heart is not fully opened but instead a small incision is made to allow passage of a finger or an instrument. Though there were notable early successes, enthusiasm quickly waned, and it was not really until the late 1940s that systematic renewed attempts were made. The renaissance of interest in cardiac surgery in the United Kingdom was led by Russell Brock and Thomas Holmes Sellors (1902–1987), both working in London and Chesterman himself was regularly undertaking mitral valvotomy by the early 1950s. 19
It is also clear Chesterman was trying to advance his own practice in cardiac surgery during this period. His busy working life gave him little time for research, but he applied, unsuccessfully, for Professorial positions not only in Sheffield but also in Cape Town. 10 He was clearly keeping up with the literature, as evidenced by his ongoing correspondence now held in U.S. university archives with Evarts Graham and Thomas Burford (1907–1977) of Barnes Hospital.20,21 He also visited important European units to observe state-of-the-art procedures. This included to Brock at Guy's Hospital London and it is recorded that he attended at least one meeting of the famous Peacock Club on 27th November 1950. 22 He also travelled to observe Clarence Crafoord (1899–1984), an early adopter of CPB, and his team at the Karolinska Institute in Stockholm, Sweden.
First intracardiac total valve replacement in man
Chesterman sought to develop his own innovations in cardiac surgery. He worked on new surgical instruments, for example publishing his design for a new valvulotome in the Lancet in July 1955, 23 and performed some of the earliest tricuspid valvotomies for rheumatic stenosis reported in the literature, including the first known combined mitral and tricuspid valvotomy procedure. 24
At Georgetown Hospital, Washington D.C., Charles Hufnagel (1916–1989) and colleagues had successfully implanted prosthetic valves to treat patients with aortic regurgitation, reporting these in print by 1954. 25 The Hufnagel valve, made from polymethyl methacrylate (also known as Perspex), included a ball within a housing that was arranged to allow anterograde but not retrograde blood flow. Hufnagel placed his valves in the descending aorta. This avoided the need to open the heart itself to implant the prosthesis and made for, in relative terms, a more straightforward procedure that could be conducted with hypothermia and speed.
Regurgitation of the mitral valve, however, could not be dealt with using an extracardiac prosthetic valve. Few details are known, but Chesterman presumably knew about Hufnagel's work as he was in regular contact with other members of the international surgical community. In addition, quite remarkably his colleague at the Sheffield Royal Infirmary, Alan Wordsworth Fawcett (1896–1969), was carrying out Hufnagel valve procedures by 1955, just one year after the technique appeared in the literature. 26 What is clear is that some time before July 1955 Chesterman began working on a mitral valve prosthesis, settling on a poppet design (Figure 2).

Chesterman's sketch of his mitral valve prosthesis design (left) and high-resolution image generated by 3D scanning of the original prosthesis (right).
However it was conceived, he produced a design that included a waisted annulus in which the poppet sat, moving up and down with the direction of blood to allow forward flow but restrict backward flow. Two button-like structures, also of Perspex, were sewn onto the outside of the heart around the valve ring to hold it in place. It remains unclear if any preclinical testing was performed, though reportedly Chesterman did have access to the laboratory facilities at Lodge Moor Hospital, Sheffield, which had been established by Sir Edward Mellanby (1884–1955). 27
Early prototypes were made from copper, some of which are held in the Baxter Edwards valve museum in California, USA, having been donated by the Sheffield pathology technician Clifford Lambourne (1930–2017), but these were never implanted.
28
Lambourne produced the final version from Perspex (Figure 2). He recounted to the hospital newspaper many years later I had to turn the Perspex valve – which measures about an inch in diameter – on a huge metal-turning lathe … Turning left a roughish surface and it had to be smooth. I tried various sandpapers but the valve had to be smoother and I realised it was going to take a lot of work … At that time, 1955, the cinema was still showing some very good films … and my wife and I were keen cinema-goers. We sat in the cinema, watching films, me with a piece of silk cloth in my hands, polishing the pieces. I put a fine abrasive on the cloth and it took about three feature length films to get the valve smooth enough.
28
What is apparent is that the valve was implanted on 27th July 1955 in a 34-year-old male. This was of course before Chesterman had access to CPB, so the operation would have been performed very quickly and with severely limited access and visualisation. The only account in the literature comes from a letter written to the Annals of Thoracic Surgery in 1991 by Mrs Anita Norman, theatre sister at the Northern General Hospital,
29
published after Chesterman's death. Norman's account includes the following: The patient was cooled with ice-packs and the use of a wind tunnel. The inferior vena cava and the superior vena cava were clamped to give venous inflow occlusion. The valve was totally excised and the manufactured valve was inserted through the atrium and pushed into the mitral valve orifice. The valve was secured with several sutures and two Perspex buttons on the outside of the heart. The operation was performed on a beating heart, from which air was removed with a syringe and needle.
In his memoir, Chesterman does not mention the valve replacement at all, save for a brief apparent reference when he stated ‘Closed methods for some forms of valve disease were inadequate in spite of ingenious methods such as … insertion of prosthetic mitral valve (TJ. 34 22755)’, the numbers presumably referring to the 34-year-old and the operation of the 22 July 1955, and TJ being the initials of the patient. In 1985 Chesterman wrote ‘My patient T … J … age 34 had a mitral valve replacement 22/7/55 … He read the evening paper and was comfortable at night when I last saw him. He was found dead by the night at about 3.30 am – autopsy valve had dislocated (sic)’. 30
Though only successful in the very short-term, this is now believed to be the first intracardiac total valve replacement in the world.29,31,32 The first ‘successful’ mitral valve replacement has generally been credited to Nina Braunwald (1928–1992) in 1959, with the patient surviving around four months. 33 The ‘ball and cage’ Starr-Edwards valve heralded the era of routine intracardiac valve replacement. 34
The mitral valve prosthesis implanted by Chesterman was removed at post-mortem and preserved in the hospital pathology department. As far as is known, Chesterman did not perform another mitral valve replacement although there is evidence that he and Lambourne worked on further designs. Intriguingly, this included some featuring Perspex spheres bringing to mind the successful Starr-Edwards prosthesis though further details are not known. 28
Early adoption of cardiopulmonary bypass
To retell the story of the development of CPB is beyond the scope of this paper and excellent reviews already exist. 35 In summary, a great deal of effort was put into developing safe and effective CPB in the 1950s to enable open heart surgery and therefore make feasible the more complex cardiac procedures such intracardiac valve replacement or repair, and in due course coronary artery bypass grafting and cardiac transplantation. CPB machines fundamentally consist of a pump and an oxygenator. Though other approaches such as adult donor cross-circulation or use of animal lungs as oxygenators were also tried,36,37 wholly mechanical solutions were felt to be most promising in the longer-term and largely, but not exclusively, the key developments were occurring in the United States. In essence, two distinct approaches were emerging for mechanical oxygenation. 35 On the one hand was the technically complex and very expensive screen oxygenator type of system (often known as the ‘Mayo-Gibbon’ machine) initially developed by John Gibbon (1903–1973) of Philadelphia with support from International Business Machines (IBM) and subsequent refinement by John Kirklin (1917–2004) at the Mayo Clinic. 38 On the other hand, Walton Lillehei (1918–1999) and his fellow Richard DeWall (1926–2016) had taken a simpler approach of bubbling oxygen through mechanically circulated blood, which was then passed through a debubbling helix of tubing (often known as the ‘bubble oxygenator’). 39
Chesterman became determined to obtain a form of CPB for his own practice. He wrote In January 1956 after much argument the Regional Health Board gave me £300 to go to the States for 6 weeks to study open heart surgery and a promise of £250 for equipment on return! With such inadequate financial backing it was no good going to the Mayo Clinic or the Jefferson Hospital Philadelphia where the minimum apparatus ran to $60,000 so I went to Minneapolis where both hypothermia and pump oxygenation were in use, the latter being of a semi-disposable and low cost invented by DeWall. I was given every opportunity to watch, to assist in the dog lab and see the human patients.
7
Thus, Chesterman was awarded funds to travel once again to America. The modest grant of £250, equivalent to around £8000 (c.$10,000) in 2024, while not a small sum of money, did not represent a large budget to set up a state-of-the-art open-heart surgery centre.
He arrived in Boston by air, travelling on Pan American's ‘Clipper de Soto’ on 10th January 1956 then went on to Minneapolis. His notes record the cases he observed, including on the 31 January 1956 an aortic valvotomy performed by Walton Lillehei using the DeWall oxygenator, which he sketched (Figure 3).
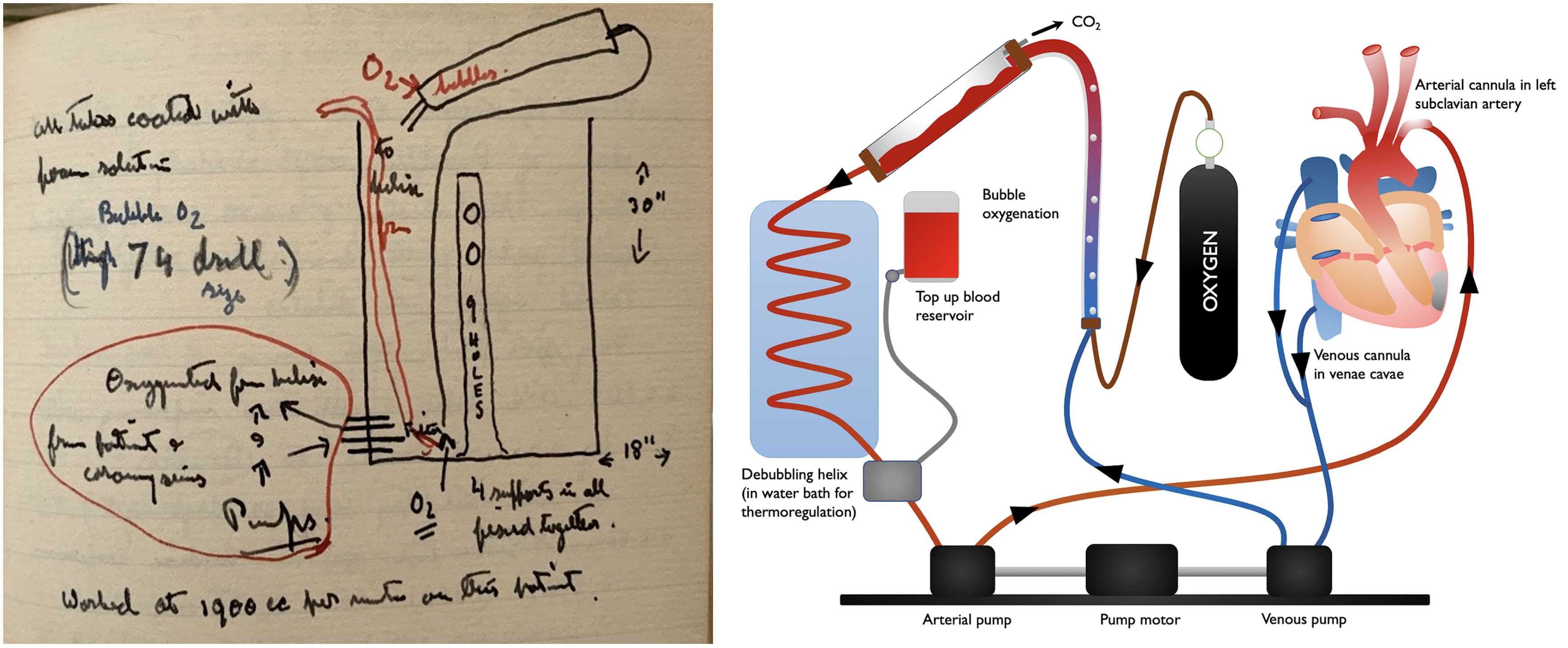
Chesterman's notebook sketch of the DeWall oxygenator (left), drawn after seeing Lillehei and Varco using it first-hand (from the Chesterman family), and a diagrammatic representation of its design (right).
On 21st February he also recorded performing his first operation using the oxygenator, supervised by DeWall himself, in the dog laboratory. He was subsequently able to purchase a Sigmamotor pump, the hospital records noting a price of £243, 40 and it must have been with some excitement that he returned with this to Sheffield on 25th February 1956.
Once home, he compiled notes on various British suppliers for the rest of the components needed to assemble the machine. He recalled The £250 offered by the Board paid for the sigma motor (out there), the rest was “found”, glass was given by the manufacturers … and I added about £300. After 6 months working on the machine during which time we found and corrected a 1/200 inch fault in the alignment of the motors we were ready to go.
7
A set of official photographs taken by the medical illustration department exist within Chesterman's personal papers, showing an elegant machine of the DeWall design (Figure 4).

Chesterman's pump oxygenator installed in the operating theatre at the City General (left) and the atrial myxoma it was used to remove (right).
The machine was first used on 26th February 1957. The patient was a 42-year-old umbrella maker who had been referred by her general practitioner (GP) with a progressive history of shortness of breath and a murmur consistent with mitral stenosis. In view of her significant symptoms, Chesterman recommended mitral valvotomy. This would not need the bypass machine as it was a closed procedure and she was taken to theatre on 15th January 1957. At operation the mitral valve was in fact found to be normal, but a large tumour in the left atrium was discovered, the ‘size of [a] golf ball’. Chesterman noted in the medical records that ‘it was considered too dangerous to attempt removal … hence closure with nothing done – for resection under pump oxygenator’. She remained in hospital awaiting a second procedure before returning to theatre on 26th February 1957. Though atrial myxoma excision had been attempted by others without CPB, only with CPB had successful cases been reported, though the global experience of managing this rare condition remained very scarce in 1957. 41 Cannulae were placed in the left subclavian artery, superior vena cava and inferior vena cava and connected to the machine. The notes record that bypass was started at 12.48 pm at 2.2 L/min. He opened the atria and removed the tumour successfully. Chesterman must have worked quickly as the patient came off bypass by 1.14 p.m. Shortly afterwards, there was a period of cardiac arrest but this responded to cardiac massage and noradrenaline. There had been significant blood loss of around 30 oz (850 mL) during bypass set up, 63 oz (1790 mL) from the coronary sinus (which drains blood from the heart muscle), 26 oz (739 mL) of other losses plus a total of around 100 swabs. In all, the patient received around 19 pints (10.8 litres) of transfused blood. At 3.45 p.m. she was recorded as being sat up with a good blood pressure of 120/90 mmHg however all must not have been well as Chesterman wrote ‘has definite tracheal tug, query cerebral lesion’ and then the entry at 6.45 p.m. simply stated ‘collapsed and died’. 42
A post-mortem was conducted and the cause of death was felt to be acute on chronic cerebral hypoxia, to which the large blood loss and period of cardiac arrest may have contributed. Chesterman was obviously disappointed and wrote back to the GP that We reopened her with an artificial heart lung machine attached. The tumour was removed and she left the table for recovery ward with a better pulse and blood pressure than she had when she came, but unfortunately she died some four hours later … I very much regret this as I thought we had probably got away with successful resection and she would have been perfectly well afterwards. I feel that she had not more than a few months to live if the condition had been left.
42
The spectre of post-operative mortality was ever present in this era of cardiac surgery. Lillehei noted that of the first 18 reported cases of CPB in the literature, 17 were unsuccessful. 35 Even in the most experienced units, mortality after open-heart surgery was running at around 25% into the 1960s. 43 Today, in-hospital mortality from open heart surgery is around 3%, 44 this progress being owed to the patients who underwent these early procedures as well as the practitioners devising and performing them.
Where does Chesterman's use of CPB fit in on the global stage? Clearly, use in North America was reasonably well established in the key units by 1957. Outside America, attempts at mechanical CPB were more infrequent, and often like Chesterman's were quickly abandoned after early poor results. Considering the chronology of use in Europe, for example, is made challenging by whether to count acutely unsuccessful cases or where CPB was used to support closed rather than open cardiac procedures. An apparent chronology of notable European instances of CPB use up to February 1957 is shown in Table 2. It should be noted, however, that just as Chesterman's cases were not published, it is plausible that use in other units may similarly be undiscoverable using this method. Nevertheless, it appears that Chesterman's may have been only the second unit in Europe to use the De Wall-Lillehei method of pump oxygenation and potentially only the third or fourth in Europe to perform open-heart surgery using CPB. Chesterman's first patient was essentially concurrent with that of fellow Yorkshire surgeon, Geoffrey Wooler (1911–2010) of Leeds, who successfully carried out a mitral valve repair using a Melrose-type machine in February 1957, though the exact date is not apparent. 45
Significant uses of cardiopulmonary bypass in Europe before February 1957.

Chesterman used the pump oxygenator once more, for a patient with a pre-operative diagnosis of a ventricular septal defect, but on opening the heart was in fact found to be a complex mix of congenital and rheumatic heart disease. 7 Chesterman did place the patient on bypass but on finding the true diagnosis did not attempt correction. The pump was stopped and the patient woken up, so in that sense the machine performed well, but the patient died during a subsequent procedure for cardiac correction at the Middlesex Hospital in London. 7 Chesterman's pump oxygenator was not used again and he wrote on reflection: ‘a younger man should be brought in to tackle the problem of open-heart surgery and … more accurate diagnostic ability was equally necessary on the medical side’. 7 The former was dealt with by attracting the London trainees Desmond Taylor (1925–2014) and Geoffrey Smith (1937–2016) as new Consultants in 1960 and 1969 respectively.50,51 The next phase of open-heart surgery in Sheffield came with the purchase of two Guy's-Ross bypass machines in 1961. 52 The problem of inaccurate pre-operative diagnosis was seemingly pervasive during this period. For example, in the USA Gibbon's first CPB patient died on the table because the pre-operative diagnosis of atrial septal defect was incorrect. 53 In time, cardiac catheterisation and imaging improved cardiac diagnosis and made for fewer surprises when embarking on open heart surgery. For Chesterman, more cardiological support came in the form of David Verel (1919–2009) and later James Fleming (1931–1992).54,55 The unit flourished, culminating in the establishment of a successful heart and lung transplant programme by Smith in 1989.
Chesterman retired in 1969 but kept busy until his death in 1987. Building on a lifelong passion and no doubt benefiting from his huge surgical experience, he was appointed Lecturer to the University of Sheffield Archaeology Department, establishing the now internationally renowned osteology laboratory there and contributing to field-leading research in the area of palaeopathology. He contributed to important projects, including analysing bone specimens from Neolithic settlements excavated on the Orkney Isles, working with Colin Renfrew who was a Lecturer at the University of Sheffield in the 1960s and later was appointed Disney Professor of Archaeology at the University of Cambridge. 56
Chesterman's legacy
Judson Chesterman's career spanned the period that saw cardiac surgery transform from being perceived as impossible to near-routine. Innovations in medical engineering, such as CPB and valve prostheses, played a large part in ensuring the rapid expansion and success of heart surgery. Chesterman's contributions added to this tapestry of achievement. Like others, early disappointments dissuaded him from further pursuing novel approaches, but subsequent developments showed that his proposed solutions were valid and, with time, became mainstream and successful.
Chesterman's CPB machine was for many years on display in the Hunterian Museum at the Royal College of Surgeons of England. Locally, he was honoured in 1997 when the Northern General Hospital's new cardiothoracic wing was named in his memory, the family connection to the steel industry happily also fulfilling the hospital's tradition of naming buildings after prominent figures in that field.
Footnotes
Acknowledgments
The author wishes to thank Dr Jake Alderson, Dr Mike Collins, Mr Steven Hunter, Rosemary Stow (neé Chesterman), the late Professor Kim Suvarna and the staff of the Sheffield City Archives for help in obtaining primary sources.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
