Abstract
Background
Cardiopulmonary bypass is widely used in cardiac surgery but often leads to lung ischemia–reperfusion injury, a major cause of morbidity and mortality. Despite advances in critical care, effective prevention remains challenging. Sitaxentan, a selective endothelin receptor antagonist, has shown protective effects in ischemia–reperfusion models, suggesting its potential in mitigating lung ischemia–reperfusion injury. This study investigated the efficacy of sitaxentan in reducing lung ischemia–reperfusion injury during cardiopulmonary bypass.
Methods
Twenty-four female beagles were divided into sham, cardiopulmonary bypass, and endothelin receptor antagonist (sitaxentan-treated) groups. Hemodynamics, arterial blood gas, lung damage scores, wet/dry ratio, and levels of various biomarkers were evaluated.
Results
Lung damage scores in the endothelin receptor antagonist group were lower than those in the cardiopulmonary bypass group but higher than those in the sham group (P < 0.05). The wet/dry ratio was lowest in the sham group and higher in the cardiopulmonary bypass group than that in the endothelin receptor antagonist group (P < 0.05). Caspase-3 and hypoxia inducible factor-1α levels were intermediate in the endothelin receptor antagonist group compared with those in the cardiopulmonary bypass and sham groups (P < 0.05). In contrast, phosphorylated-endothelial nitric oxide synthase, phosphorylated protein kinase B, tumor necrosis factor-α, and interleukin-6 levels were higher in the endothelin receptor antagonist group than in the cardiopulmonary bypass and sham groups (P < 0.05). Malondialdehyde level was higher and superoxide dismutase level was lower in the cardiopulmonary bypass and endothelin receptor antagonist groups than in the sham group (P < 0.05).
Conclusions
Sitaxentan may offer a novel therapeutic approach to attenuate lung ischemia–reperfusion injury in clinical settings by regulating the hypoxia inducible factor-1α/phosphorylated protein kinase B/phosphorylated-endothelial nitric oxide synthase pathway.
Introduction
Cardiopulmonary bypass (CPB), a common technique used in cardiac surgery, can lead to lung ischemia–reperfusion injury (LIRI). LIRI is associated with significant morbidity and mortality after CPB.1–3 Several mechanisms can increase the risk of LIRI with CPB, including suspension of ventilation and mechanical perfusion, and trigger cytokine-mediated systemic inflammatory response.4,5 Pulmonary hypoxia and reoxygenation can activate an inflammatory cascade that damages the pulmonary endothelial and alveolar epithelia barriers, resulting in neutrophil recruitment. 6 Although several studies have proposed various protective measures for CPB-associated LIRI,7–9 there are no definitive therapeutic options. Endothelin receptor antagonists (ERAs) promote nitric oxide (NO)–mediated vasodilation, thereby improving organ perfusion.10–12 An endothelin receptor antagonist (ERA) drug, sitaxentan, is commonly used to treat pulmonary arterial hypertension, 13 and improved organ perfusion was reported in previous studies.14,15 ERAs have been shown to inhibit lung inflammation in animal models 16 and modulate cytokine responses after CPB. 17 We therefore sought to evaluate the potential protective role of sitaxentan against CPB-mediated LIRI.
Materials and methods
Animals
The reporting of this study conforms to the Animals in Research: Reporting In Vivo Experiments (ARRIVE) 2.0 guidelines. 18 All protocols were approved by the Animal Care and Use Committee at the Affiliated Hospital of Southwest Medical University (No. 20231021-009), in accordance with the requirements of the Chinese Animal Care Committee. The animal experiments were conducted in compliance with the “Guide for the Care and Use of Laboratory Animals, 8th Edition.” 19 All animals were housed in a temperature (T)-controlled facility (22°C ± 2°C and 50% ± 10% humidity) with a 12-h light/dark cycle (lights on from 07:00 to 19:00). Beagles were pair-housed in stainless steel kennels (2.5 × 1.5 m) containing raised resting platforms and absorbent bedding, provided with standard laboratory chow and water ad libitum as well as environmental enrichment (toys and social interaction) daily. Sample size was determined via power analysis to avoid the recruitment of excess animals. Suffering was reduced via continuous monitoring and appropriate use of anesthesia. Euthanasia was performed by administering an overdose of pentobarbital sodium (100 mg/kg), and death was confirmed via electrocardiography.
Beagles served as a suitable large-animal model to study LIRI, considering their anatomical and physiological similarities to humans with respect to lung structure and function as well as their manageable size and ease of handling, which facilitate surgical procedures and postoperative monitoring.
Twenty-four female beagles (2–4 years old, 10–15 kg, Beijing Johnsen International Trade Co, Ltd.) without anemia, hypoproteinemia, heart disease, or lung damage were used in this study. The beagles were randomly assigned to groups using a computer-generated randomization sequence. Pathologists performing histological analysis and laboratory technicians conducting biochemical assays were blinded to group allocation. The beagles were randomly divided into the following three groups (n = 8; Figure 1(a)): (a) sham group, which underwent sternotomy followed by 4 h of observation and subsequent chest re-approximation; a sternal retractor was not used during this period; however, continuous ventilation was maintained to ensure normal lung function; (b) CPB group, which underwent 2 h of CPB followed by 2 h of observation; and (c) ERA group, which received 1 h infusion of 0.792 mg/kg sitaxentan (BOC Sciences, USA) prior to 2 h of CPB followed by 2 h of observation.

(a) Flow scheme of the animal experiment; (b–g) physiological parameters measured at five timepoints: T1 (initiation of CPB), T2 (1 h after CPB initiation), T3 (end of CPB), T4 (1 h after CPB), and T5 (2 h after CPB); (b) core temperature; (e) mean arterial pressure (MAP); and (c, d, f, g) blood gas values (pH, PaO2, PaCO2, and Hb). Data are presented as mean ± SD (n = 8 per group). Statistical analyses were performed using two-way repeated measures ANOVA with Tukey post-hoc tests. (a) P < 0.05 versus baseline (T1) and (b) P < 0.05 versus sham group. ANOVA: analysis of variance; CPB: cardiopulmonary bypass; ERA: endothelin receptor antagonist; Hb: hemoglobin; PaO2: partial pressure of oxygen in arterial blood; PaCO2: partial pressure of carbon dioxide in arterial blood.
Drug administration
The selected dose of sitaxentan (0.792 mg/kg) was based on both preclinical evidence and pharmacokinetic considerations in canine models. Previous studies have demonstrated that sitaxentan, a selective ERA, exhibits dose-dependent vasodilatory effects in large animals, with therapeutic efficacy in mitigating ischemia–reperfusion (IR) injury when administered at doses ranging from 0.5 to 1 mg/kg.14,15 In a porcine model of renal IR, a comparable dose (0.7 mg/kg) of sitaxentan improved microcirculatory perfusion without causing significant hypotension, a critical safety consideration during CPB. 15
For canine pharmacokinetics, although direct data on sitaxentan are limited, extrapolations from related ERAs (e.g. macitentan) and small-animal studies indicate that the oral bioavailability of sitaxentan in dogs is approximately 89%, with peak plasma concentrations achieved within 2–3 h of intravenous administration. 13 The elimination half-life in canines is estimated to be 4–6 h, supporting our 1-h pre-CPB infusion protocol to ensure sustained therapeutic levels during the 2-h CPB period and subsequent 2-h observation phase. This dosing strategy aligns with the drug’s pharmacodynamic profile wherein maximal endothelin receptor blockade is maintained for 6–8 h after administration. 13
Surgical procedures
Anesthesia was induced intravenously using ketamine (2 mg/kg), fentanyl (4 μg/kg), and vecuronium (0.2 mg/kg). The animals were orally intubated and ventilated using a mechanical control mode (tidal volume, 10 mL/kg; respiratory rate, 16–20 breaths per minute; GE, DATEX-OMEDA 7100, USA). Propofol (100 μg/kg/min), fentanyl (0.2 μg/kg/min), and vecuronium (0.1 μg/kg/min) were used to maintain the animals. A vertical incision was made along the sternum, and the sternum itself was divided. Systemic CPB flow was established using aortic and venous cannulas in the right atrial appendage, with a priming volume of 0.5 L consisting of 0.2 L Ringer’s solution, 0.1 L colloid, 0.15 L blood from another beagle, and 0.05 L 5% sodium bicarbonate. Heparin (3 mg/kg) was administered, and once the activated coagulation time reached 480 s, the aorta was cross-clamped. During CPB, cardioplegia was administered to arrest the hearts. The type of myocardial protection used was cold blood cardioplegia with a volume of 15 mL/kg. CPB was performed using a CPB machine (Stockert, Stork-2, Germany) for 2 h (32°C–34°C, 70–100 mL/kg/min) with the ventilation stopped. Mean arterial pressures (MAPs) were maintained at 50–70 mmHg; hemoglobin (Hb) levels were also maintained at >70 g/L throughout the experiment. Therapeutic interventions were carried out if necessary. At the end of the CPB, the aortic clamp was opened, and reperfusion was initiated. The beagles were weaned from CPB as the flow of the CPB was consecutively reduced. They were then given 3 mg/kg of protamine to reverse the effects of heparin and observed for another 2 h. Core T, MAP, and arterial blood samples were collected at the following five defined time points (T1–5): initiation of CPB, 1 h after initiation, end of CPB, 1 h after CPB, and 2 h after CPB (Figure 1(a)). Finally, the lungs were harvested and perfused with 100 mL sterile saline via the pulmonary artery. Arterial blood samples (0.5 mL each) were collected from the left femoral artery at each time point (T1–5). The right lungs were used to calculate the wet/dry (W/D) ratio due to their anatomical accessibility and ease of handling during the surgical procedure, whereas a portion of the left lungs was excised for identifying pathological changes. The remaining portion was stored at −80°C for further enzyme–linked immunosorbent assay (ELISA) and western blot analysis. All procedures were conducted in strict adherence to aseptic technical guidelines, and proper sterilization of equipment and surgical instruments was ensured.
Measurement of blood gas values
Arterial blood samples were collected from the left femoral artery using a polyethylene catheter. Blood gas parameters, including pH, partial pressure of oxygen in arterial blood (PaO2), partial pressure of carbon dioxide in arterial blood (PaCO2) and Hb, were measured immediately using the alpha-stat mode on a blood gas analyzer (Abbott, i-stat300G, USA).
W/D ratio
The amount of interstitial fluid in the lung was estimated by calculating the W/D ratio specifically as follows. The wet weight of the right lungs was measured; then, the lungs were dried in an oven at 60°C for 24 h to obtain the dry weight.
Pathological scoring
The lungs were fixed with 4% paraformaldehyde for 24 h and then embedded in paraffin wax. Sections were cut and stained with hematoxylin and eosin (H&E). A blinded pathologist evaluated the specimens and assessed the following parameters histologically: intra-alveolar and interstitial edema, hyaline membrane formation, inflammation (classified as histiocytic, lymphocytic, neutrophilic, or mixed), fibrosis, atelectasis, intra-alveolar hemorrhage, and overall classification. All the indicators were graded on a scale of 1 to 4 as follows: 1 for absent, 2 for mild, 3 for moderate or scattered, and 4 for severe or frequent.
ELISA
The concentrations of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), malondialdehyde (MDA) and superoxide dismutase (SOD) were measured using ELISA kits (R&D Systems, Minneapolis, Minnesota, USA), and experiments were conducted following the manufacturer’s instructions. For ELISA measurements of TNF-α, IL-6, MDA, and SOD, intra-assay variability was assessed by running six replicate samples (from a pooled canine lung tissue homogenate) within each assay plate. The coefficient of variation (CV) for each analyte was as follows: TNF-α, 4.2%; IL-6, 3.8%; MDA, 5.1%; and SOD, 4.7%. All CV values were <6%, indicating acceptable intra-assay precision. Blank wells (without tissue homogenate or standards) were included to account for nonspecific binding, with absorbance values consistently <0.1 optical density, ensuring minimal background interference. A reference canine lung tissue lysate (supplied by R&D Systems, Minneapolis, Minnesota, USA) with known analyte concentrations was run in triplicate per plate, yielding measurements within 10% of the expected values, validating assay accuracy.
Western blot analysis
Equal amounts of protein from the tissue extracts were separated via 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes through wet electrophoretic transfer. The membranes were washed twice with Tris-buffered saline with Tween 20 for 10 min each and incubated overnight at 4°C with the following primary antibodies: anti-caspase-3: rabbit polyclonal (Catalog number: ab4051; RRID: AB_304697; Clone: EPR17687; Lot: GR3350449-1; Abcam, Cambridge, UK; 1:200); anti–hypoxia inducible factor-1α (HIF-1α): rabbit monoclonal (Catalog number: ab228649, RRID: AB_2893141; Clone: EPR16897; Lot: GR3351486-2; Abcam; 1:1000); anti–phosphorylated protein kinase B (p-Akt) (Ser473): rabbit polyclonal (Catalog number: ab8805, RRID: AB_306736; Clone: N/A; Lot: GR3361221-7; Abcam; 1:500); and anti–phosphorylated-endothelial nitric oxide synthase (p-eNOS) (Ser1177): rabbit monoclonal (Catalog number: ab215717, RRID: AB_2893154; Clone: EPR25134; Lot: GR3365218-3; Abcam; 1:1000). Following washing, membranes were incubated for 1 h at RT with horseradish peroxidase-conjugated goat anti-rabbit secondary antibody (Catalog number: ab97051, RRID: AB_10679352; Clone: Polyclonal; Lot: GR3367122-4; Abcam; 1:5000). After the final washes, protein bands were visualized using an enhanced chemiluminescence kit (Clarity™ Western ECL Substrate, Bio-Rad, USA), and protein quantification was performed using an image analysis software (ImageJ, National Institutes of Health, Bethesda, MD, USA). All Western blot experiments included three biological replicates (tissue from three independent beagles per group) and two technical replicates per sample.
Statistical analyses
Data were analyzed using IBM Statistical Package for Social Sciences (version 20.0) software. Data were presented as mean or percentage values. Physiological data were assessed for time effect and treatment using two-way repeated measures analysis of variance and Tukey post-hoc tests. Lung injury score, W/D ratio, ELISA results, and western blot analysis findings were compared using a two-tailed t-test. A significance level of P < 0.05 was considered statistically significant.
Results
Basic characteristics
There were no differences in the baseline values of T, MAP, pH, PaCO2, PaO2, and Hb among the groups. In the CPB and ERA groups, T, MAP, and PaO2 were lower at T2 than at T1 (P < 0.05); moreover, these values were lower in the CPB and ERA groups than in the sham group at T2 (P < 0.05). The Hb levels in the CPB and ERA groups were lower at T2–5 than at T1 (P < 0.05); moreover, the Hb levels at T2–5 were lower in the CPB and ERA groups than in the sham group (P < 0.05) (Figures 1(b) to (g)).
Histological analysis and lung W/D ratio
Lung damage scores were assessed using H&E staining. Figures 2(a) to (c) show H&E staining at 400× magnification, whereas Figures 2(d) to (f) show H&E staining at 100× magnification. The ERA group exhibited significantly lower lung damage scores than the CPB group but higher scores than the sham group (P < 0.05) (Figure 2(g)). Additionally, the ERA group had a significantly lower W/D ratio than the CPB group; the lowest W/D ratio was observed in the sham group (P < 0.05) (Figure 2(h)).

Microscope images of the lung, lung damage scores, and wet/dry ratio. (a–f) Representative H&E-stained lung sections (400× and 100× magnification) from Sham, CPB, and ERA groups. (g) Lung damage scores evaluated based on intra-alveolar edema, inflammation, and hemorrhage (scored 1–4). (h) Lung wet/dry (W/D) ratio. Data are presented as mean ± SD (n = 8 per group). Statistical analysis was performed using unpaired two-tailed t-tests. (a) P < 0.05 versus sham group. (b) P < 0.05 versus CPB group. CPB: cardiopulmonary bypass; ERA: endothelin receptor antagonist; H&E: hematoxylin and eosin.
Western blot analysis
The levels of caspase-3 and HIF-1α in the ERA group were lower than those in the CPB group (P < 0.05) but higher than those in the sham group (P < 0.05). Additionally, the levels of p-eNOS and p-Akt in the ERA group were higher than those in both CPB and sham groups (P < 0.05) (Figure 3).

Expression analysis of caspase-3, HIF-1α, p-Akt, and p-eNOS in Different Groups. (a) Western blot bands for caspase-3, HIF-1α, p-Akt, and p-eNOS; (b) the bar graph presents the quantified data from the Western blot bands. Data are presented as mean ± SD (n = 8 per group). Statistical analysis was performed using unpaired two-tailed t-tests. (a) P < 0.05 versus sham group. (b) P < 0.05 versus CPB group. HIF-1α: hypoxia inducible factor-1α; p-Akt: phosphorylated protein kinase B; p-eNOS: phosphorylated-endothelial nitric oxide synthase.
Expression levels of TNF-α, IL-6, MDA, and SOD
The expression levels of TNF-α and IL-6 in the ERA group were lower than those in the CPB group but higher than those in the sham group (P < 0.05). The MDA levels in the CPB and ERA groups were higher than those in the sham group (P < 0.05), whereas the SOD levels in the CPB and ERA groups were lower than those in the sham group (P < 0.05) (Figure 4).

Expression levels of TNF-α, IL-6, MDA, and SOD in the lung tissue of beagles. The TNF-α (tumor necrosis factor-α), IL-6 (interleukin-6), MDA (malondialdehyde), and SOD (superoxide dismutase) levels were measured to assess the inflammatory and oxidative stress responses. Data are presented as mean ± SD (n = 8 per group). Statistical analysis was performed using unpaired two-tailed t-tests. (a) P < 0.05 versus sham group. (b) P < 0.05 versus CPB group.
Discussion
Hypoperfusion is a significant contributing factor to complications related to CPB. Studies have shown that decreased bronchial arterial blood flow during CPB can lead to extensive LIRI.20,21 Our previous studies have indicated that improving renal microcirculatory perfusion during CPB can help prevent acute kidney injury. 14 In this study, we aimed to investigate whether sitaxentan, a selective ERA drug, can enhance lung perfusion and play a role in protecting against CPB-associated LIRI.
We used a classical animal CPB model and managed the flow rate to ensure adequate organ perfusion. However, despite maintaining the MAP at 50–70 mmHg, LIRI occurred. Histological analysis revealed higher lung injury scores in the CPB group than in the sham group. Although after sitaxentan infusion, there was noticeable reduction in lung injury, there was no significant difference in the MAP of the ERA and CPB groups. The occurrence of LIRI was unlikely to be associated with Hb levels because the Hb levels of all the beagles in each of the three groups were above the safe threshold. 22 As per a previous study, sitaxentan infusion may benefit beagles in terms of LIRI protection due to its vasoconstrictor-blocking effect and improved organ perfusion. 10 This mechanism was previously identified in our study which demonstrated that improved renal perfusion aids renal protection. 14
The lung W/D ratio was significantly increased in the CPB group compared with that in the sham group. However, due to the presence of multiple potential causes, it was challenging to determine the primary cause of pulmonary edema based on the available data. Nonetheless, pretreatment with sitaxentan resulted in a relatively lower lung W/D ratio, indicating a potential protective effect against CPB-associated LIRI.
To further investigate the role of sitaxentan in vascular alterations in the lungs, the level of hypoxia was considered as a factor associated with perfusion. HIF-1α, a reliable indicator of tissue hypoxia, 23 was used to evaluate the hypoxia state of the lungs. HIF-1α expression was significantly increased in the CPB group, indicating that the lung was experiencing hypoxia even under normal pressure. Hypoxia was associated with increased permeability of the lung capillary and alveolar walls, possibly due to altered cytoskeletal morphology of endothelin receptors in the endothelial cells. This increased permeability is believed to have led to the formation of small intercellular pores and tissue edema. 19 Consequently, the beagles were more susceptible to pulmonary edema, as deduced from the lung W/D ratio after CPB. Sitaxentan has demonstrated favorable interspecies pharmacokinetic consistency: canine studies show 89% oral bioavailability and 10-h elimination half-life, closely mirroring the trend in human profiles (100% bioavailability, 7–10 h half-life). This translational reliability suggests that our observed protective effects at 0.792 mg/kg (achieving plasma concentrations of 15–20 µg/mL) would correspond to clinically effective human dosing (100 mg/day). 24 Importantly, sitaxentan’s selective endothelin type-A receptor blockade preserves endothelin type-B receptor activity, reducing the risk of fluid retention observed with the use of nonselective ERAs, making it preferable for pulmonary edema prevention.
In this study, we focused on the role of NO in the imbalance between vasoconstrictor factors (endothelin types 1, 2, and 3) and vasodilator factors (NO) in pulmonary microperfusion. 25 We assessed the levels of p-Akt and p-eNOS to identify alterations in pulmonary vascular function. p-Akt is widely associated with cell metabolism and apoptosis inhibition, whereas p-eNOS promotes NO production by activating the eNOS enzyme. After CPB, we observed significant decreases in the expressions of p-Akt and p-eNOS. However, p-eNOS expression increased after sitaxentan administration. Previous studies have shown that eNOS expression decreased after IR; 26 however, this was not observed in those who had not undergone CPB. It has been demonstrated that NO is produced by p-eNOS in the endothelial cells; p-eNOS can be activated by the HIF-1α/p-Akt pathway. 27 This activation is known to increase the cyclic guanosine monophosphate concentration, resulting in vasodilation through the activation of soluble guanylate cyclase, leading to improved organ microcirculation and oxygenation.28–30 Our proposed mechanism of sitaxentan’s protective effect is illustrated in Figure 5. Therefore, sitaxentan may upregulate the expression of p-eNOS via the HIF-1α/p-Akt pathway, potentially improving pulmonary microperfusion, reducing inflammation and apoptosis, and ultimately mitigating LIRI.
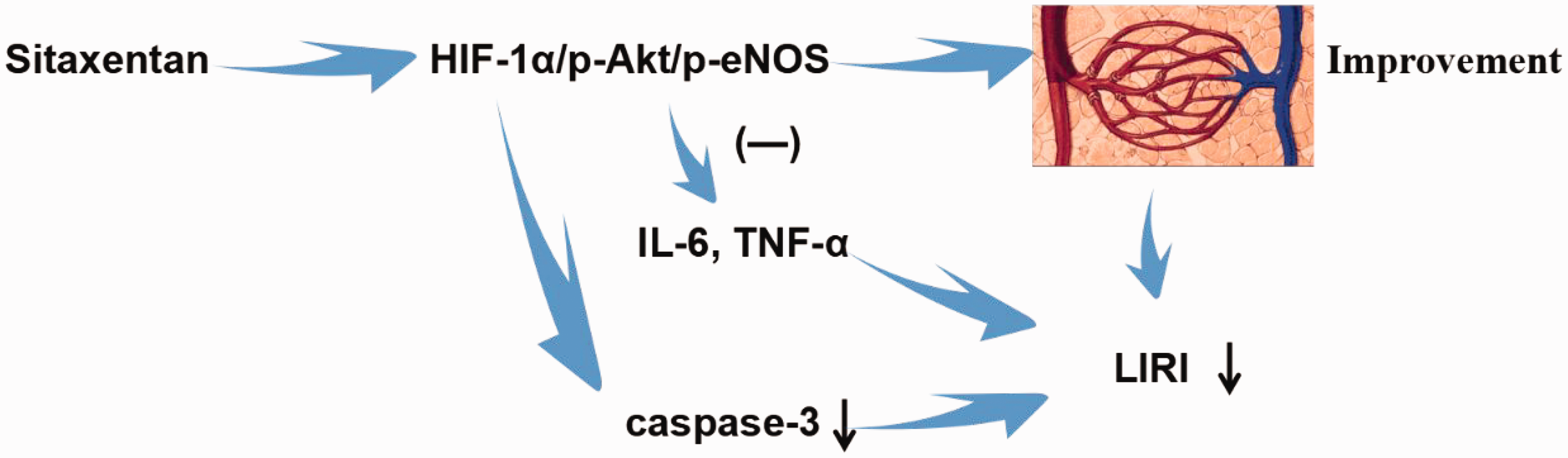
The graphical abstract in this figure illustrates the proposed mechanisms of the protective effect of sitaxentan on cardiopulmonary bypass-associated lung ischemia–reperfusion injury (LIRI). The figure shows how sitaxentan may reduce LIRI by regulating the HIF-1α/p-Akt/p-eNOS pathway, thereby improving pulmonary microcirculation and reducing inflammation and apoptosis. HIF-1α: hypoxia inducible factor-1α; p-Akt: phosphorylated protein kinase B; p-eNOS: phosphorylated-endothelial nitric oxide synthase.
Inflammatory responses and apoptosis were significantly reduced after sitaxentan administration, as evidenced by the levels of inflammatory cytokines (TNF-α and IL-6, Figure 4) and apoptosis factor (caspase-3) expression (Figure 3). Caspase-3 is a key enzyme and initiator of cell apoptosis, playing a crucial role in the caspase cascade reaction. Increased activity of caspase-3 is shown to promote apoptosis. 31 The reasons for the decrease in lung inflammation may vary. Although we cannot discount the possibility that improved whole body perfusion with sitaxentan contributed to its anti-inflammatory effects, sitaxentan may also have a protective effect on LIRI via the downregulation of inflammation and cell apoptosis. First, this may be associated with the downregulation of HIF-1α. HIF-1α is expressed in hypoxic cells 32 and plays a critical role in TLR4-mediated production of inflammatory cytokines (TNF-a and IL-6), apoptosis (as examined by caspase-3 expression), and hypoxia-induced disease and inflammation pathogenesis.33,34 Furthermore, the generation of NOS inhibitors can be induced after pulmonary IR; 35 NO also inhibits the interaction between the endothelium and inflammatory cells by preventing the accumulation of neutrophils and leukocyte adhesion.36,37
Compared with the sham group, the CPB group showed an increase in the MDA level and a decrease in the SOD level. This finding suggests that CPB can induce oxidative stress response. As per research, MDA, a marker of oxidative stress, is a lipid peroxide product released through oxidation from prostanoid metabolism and polyunsaturated lipids. 38 SOD is an antioxidant enzyme that is widely present and is observed to convert superoxide anion free radicals into hydrogen peroxide. 39 The observed increase in oxidative stress response may be attributed to IR injury associated with CPB. 40
The effect of NO on LIRI is controversial. Some studies have shown that NO does not decrease pulmonary edema during lung reperfusion and does not provide any benefit in terms of IR injury incidence. 41 However, Ardehali et al. 42 demonstrated the benefit of postoperative NO inhalation in a subgroup of patients who developed IR injury. In terms of basic research, NO has been used as an additional substance for lung preservation in several studies and has been found to effectively reduce reperfusion injury in various animal models. 43 It is noteworthy that clinical conditions in patients are complex and variable, whereas animal research is conducted under controlled and simplified conditions. Therefore, more robust data are needed in the future to determine the protective effect of NO on LIRI and to support clinical medical care. We believe that even small adjustments in research methodologies may lead to significant changes in patient outcomes.
The study has certain limitations. First, the small sample size and the exclusive use of healthy animals limit the generalizability of the findings. A small sample size is a common limitation in large-animal studies owing to cost and logistical constraints. However, the use of healthy animals allows for a controlled experimental environment, which is crucial for isolating the effects of the intervention. Second, the ventilation strategy during CPB introduced a limitation. The sham group was ventilated throughout, whereas the experimental lungs were deflated during the 2-h CPB. Although this deflation better mimics most CPB operative conditions, it may have detrimental effects on lung function and edema formation. An additional sham group with continuous ventilation during CPB could provide further insights into the effects of cannulation and extracorporeal circulation on lung function. Additionally, the study did not measure the levels of sitaxentan in the circulation and lungs, which could have provided more direct evidence of its protective effect against lung injury. The absence of pharmacokinetic data limits the interpretation of drug exposure and its relation to efficacy. Future studies should include measurements of plasma and tissue drug concentrations. The data suggested that sitaxentan may have a protective effect; however, further assessments are needed to confirm this. In addition, this study did not measure vascular alterations in the lungs related to sitaxentan, although the HIF-1α expression level may indicate the hypoxic state of the lung. Furthermore, the reason for the decrease in lung inflammation is not fully understood. Improved whole-body perfusion with endothelin receptor antagonism (sitaxentan) may have contributed to decreased inflammatory findings in the lungs; however, the potential role of HIF-1α regulation in reducing inflammatory responses cannot be dismissed.
Future research should explore the effects of ERAs in a broader range of animal models and clinical settings; the use of specific pathway inhibitors (e.g. HIF-1α or Akt inhibitors) would help establish causality for the proposed mechanism and enable impact assessment of different ventilation strategies during CPB to address these limitations and enhance the generalizability of the results. Our findings support the potential clinical translation of sitaxentan as a prophylactic agent for high-risk cardiac surgery patients, particularly those with pre-existing pulmonary hypertension, chronic obstructive pulmonary disease, or right ventricular dysfunction who have an elevated risk of acute respiratory distress syndrome after CPB. The drug’s rapid onset (peak plasma concentration within 1–3 h) and sustained activity align with preoperative administration timelines, potentially reducing the LIRI incidence when administered 24 h preoperatively. This approach mirrors successful prophylactic strategies for contrast-induced nephropathy where targeted receptor antagonism precedes insult exposure. Additionally, future studies should include the use of ex-vivo lung perfusion to assess post-transplant LIRI or primary graft dysfunction. This approach would provide valuable insights into the long-term effects of ERAs on lung function after transplantation, which is crucial for clinical applications. Additionally, future studies should investigate the mechanisms underlying the protective effects of ERAs, potentially leading to the development of novel therapeutic strategies.
It is important to note that sitaxentan was previously withdrawn from the market due to cases of severe hepatotoxicity. Although our study demonstrates its potential efficacy in mitigating LIRI, clinical translation would require careful consideration of the risk-benefit ratio and strategies to mitigate liver injury, such as rigorous monitoring of liver function and dose optimization. Other ERAs have been studied in the context of lung injury. For example, bosentan, a dual ERA, has shown protective effects in pulmonary hypertension and inflammation models. Macitentan, another dual ERA, offers a better safety profile and longer duration of action. Future studies could compare the efficacy of selective versus nonselective ERAs in LIRI to determine the optimal therapeutic approach.
Conclusion
Our study demonstrated that the use of CPB may increase the risk of LIRI. We found that sitaxentan infusion effectively reduced inflammation, apoptosis, and LIRI in beagles by regulating the HIF-1α/p-Akt/p-eNOS pathway.
Footnotes
Acknowledgments
The author would like to acknowledge that no individuals or organizations require specific mention for their contributions to this work.
Author contributions
Zhengjia Wang: Conceptualization, methodology, investigation, formal analysis, and writing–original draft.
Renshu Zhan: Conceptualization, methodology, investigation, data curation, writing–original draft.
Liqun Mo: Investigation, resources, and project administration.
Yingying Zhang: Validation, formal analysis, and data visualization.
Yirong Chen: Investigation and resources.
Li Liu: Supervision and writing–review & editing.
Yiping Bai: Conceptualization, supervision, funding acquisition, writing–review & editing, and project administration.
Data availability statement
The data supporting this study’s findings are available from the corresponding author (Yiping Bai) upon reasonable request.
Declaration of conflicting interests
All authors declare no competing financial interests or personal relationships that could influence the work reported herein. No author has received support from commercial entities related to the study, holds relevant patents, or has stock interests in associated companies.
Disclosure
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Funding
This work was supported by the Sichuan Science and Technology Program (2024NSFSC0613) and the Luzhou Science and Technology Program (2023JYJ043).
Statement
The authors confirm that patient consent was not required for this study because this was an animal study.
