Abstract
Up until the mid-1900s, tricuspid atresia - a birth defect of the tricuspid valve, was once categorized as a “death sentence.” The challenge of achieving positive health outcomes for affected patients was compounded by a hesitancy to operate on children. The main concern was safely administering anesthesia to young patients who were going through a strenuous operation that was often poorly tolerated. Despite these assumed limitations, Francis Fontan, a pediatric cardiothoracic surgeon at the Hospital of Tondu in Bordeaux, was able to redirect blood flow from the superior and inferior vena cava to the pulmonary arteries in 1971, which elucidated the process of advancing clinical practice in medicine. With the support of mentors and a firm belief in this new technique, Fontan pioneered his eponymous procedure and ultimately paved the way for modern cardiovascular surgical techniques that helped to prolong the life of those with single functioning ventricles. The aim of this study is to examine the genesis and the evolution of the Fontan procedure to elucidate the process of advancing clinical practice in medicine by utilizing personal interviews, Fontan's works, associated primary and secondary sources in the context of 20th century cardiothoracic surgery and innovations.
Keywords
Background
Francis Fontan (1929–2018) was born on July 2, 1929 in the foothills of the Pyrenees-Atlantiques mountains in the province of Bearn, France. His father, Victor Fontan, was a professional cyclist who competed in the Tour de France and was nicknamed the “King of the Mountains.” 1 Sport was therefore very much a part of Francis's childhood and he played rugby in his youth while finding a passion for golf and sailing later in life. However, although Fontan enjoyed sporting activities, he was guided to focus his life on an area that bore a great deal more gravitas.
Fontan's ambition to become a physician began when his older sister showed him a children's book that explained various occupations. It was then that he became enamored with the idea of becoming a surgeon. 2 At a local boarding school in Pau, France, Francis applied great focus to his studies, although these were disrupted as the Second World War enveloped Europe. After completing his school education at the age of 17, he was then accepted by the Faculty of Medicine at the University of Bordeaux where he spent six years studying before passing the licensing exam in 1952, and excelling as one of only 13 of 100 students who were afforded the honor of “internes des hopitaux.” 3
His internship at the University of Bordeaux proved to be pivotal to his career as it was here that he met two of his mentors, cardiac surgeon Georges Dubourg (1903–1984) and cardiologist Pierre Broustet (1903–1974). 2 While Fontan's decision to train under Dubourg was unconventional, since he was not a professor at the university, Fontan considered him to be one of the best surgeons in Bordeaux. However, Fontan acknowledged that it was Pierre Broustet, Head of Cardiology, who was the first to draw his attention to tricuspid atresia.
In 1959, Broustet instructed Fontan and one of his co-interns, Modeste Dallocchio, to conduct an autopsy on a cyanotic adolescent who had died from tricuspid atresia. Following this close examination, Fontan felt motivated to seek ways to prevent death from the condition. After a comprehensive review of earlier work and with enthusiastic support from his mentors, Fontan went on to pioneer his eponymous procedure, ultimately paving the way for today's cardiovascular surgical techniques that help to prolong the lives of those with single functioning ventricles. In this study, the origins and the evolution of the Fontan procedure will be examined, which will not only exemplify the process of advancing clinical practice, but also highlight Fontan's influence on the medical community and the value of mentorship in medicine.
Fontan’s early training years
The autopsy that Fontan performed on the young patient with tricuspid atresia during his training ignited his interest in this area of cardiology, particularly since, at that time, congenital heart defects often represented a death sentence, since there were little to no effective treatments. When his mentor, Dubourg, gave Fontan the task of exploring surgical options for those with tricuspid atresia, he discovered that shunts were used for the majority of patients with restricted pulmonary flow. 4 He found that there were two main surgical procedures – a subclavian-pulmonary artery anastomosis (Blalock-Taussig) and a cavo-pulmonary bypass (Glenn) – with surgeons continually experimenting with techniques to improve the outcomes of both. Inspired by Alfred Blalock (1899–1964) and Helen Taussig (1898–1986), in 1954 Herbert E. Warden (1920–2002) attempted a two-stage bypass in dogs which created a tricuspid stenosis and then an anastomosis between the right auricular appendage and pulmonary artery. 5 In 1955, Elliott S. Hurwitt (1913–1966) attempted an atriopulmonary anastomosis in a child with tricuspid atresia, although the patient did not survive. 6
The first attempt to perform a complete bypass of the right heart was in 1954, by John C. Rose (1924–2013), who anastomosed both the superior and inferior vena cava to the main pulmonary artery via vascular heterographs. 7 However, an operation that involved anastomosis of the inferior vena cava to the pulmonary artery inevitably led to severe venous congestion in the splanchnic system, which was incompatible with survival.
Having completed his literature review, Fontan published the findings and the results of the autopsy in the
Early contributors set the stage
There were several important innovations that set the stage for Fontan's ground-breaking work. It was believed by many that re-routing venous return to the heart was not possible. Indeed, in the 16th century, the influential English physician William Harvey (1578–1657) declared “those who believe that one ventricle can drive blood full of spirit into the body and lungs, likewise, are heretics. They forget that nature, being divine, never gave a heart to any where there was no need.” 9 Challenges to the centuries old concept of hemodynamics, where the absence of a working right ventricle was incompatible with life, were rebuffed by German physician, Ludwig Lichtheim (1845–1928) 4 who, in 1876, was able to prove that inspiration and insufflation under positive pressure have opposite effects in the pulmonary artery. By the 1900s, over 40 different procedures to bypass the heart had been tested experimentally and some had achieved an element of success, rendering Harvey's firm stance against cardiac bypass obsolete. 4
The American surgeon, Dwight Harken (1910–1993), had been able to successfully remove foreign bodies within the heart of more than 100 soldiers during World War II, thus proving that the heart was not as fragile as once thought, and that it could be surgically manipulated. 10 Subsequently, in 1949, Simon Rodbard (1911- 1975) and David Wagner (1905–1989) were the first to attempt a bypass of the right ventricle in dogs, 11 an operation that took one hour. Although only one animal survived for 12 hours, it negated “the idea that the entire venous return could be propelled through the pulmonary system without a pumping chamber seemed outlandish, ill-advised, unrealistic and perhaps in some people's mind crazy.” 12
In the mid-1900s, there was a generally heeded warning not to operate on children, particularly those with cardiovascular disease, severe cyanosis, and hypoxia. 10 The main concern being safely administering anesthesia to young patients undergoing major operations. Helen Taussig, the influential pediatric cardiologist who co-developed the Blalock-Taussig shunt operation that Fontan studied, believed that a physician should not operate on a child younger than four years with tetralogy of Fallot, or a child younger than eight years with aortic coarctation. 10
A new idea
After completing his training at the University of Bordeaux, Fontan worked with Gerard Brom (1915–2003) at the University of Leiden in the Netherlands where he familiarized himself with the Glenn procedure. In this procedure, an anastomosis is performed between the superior vena cava and the right pulmonary artery. The concept of a novel cavo-pulmonary connection immediately triggered Fontan's imagination: “one day I said to myself that, what we did with the SVC that we drained in the right pulmonary artery, we could maybe do it with the IVC or atrium toward the left pulmonary artery… this would do away with mixing of oxygenated and deoxygenated blood.” 13 He began to consider the possibility that the inferior vena cava could be channeled to the remainder of the pulmonary trunk to perfuse the left lung. This extracorporeal revision was performed on dogs at the Hospital of Tondu in Bordeaux between 1963 to 1964, but with little success, since the dogs only survived for a few hours following the procedure, and tolerated extra corporeal membrane oxygenation (ECMO) very poorly. 13 In retrospect, it is somewhat shocking that Fontan's next step was to attempt the same procedure on a human subject.
The Fontan procedure
In March 1968, Pierre Broustet consulted Fontan regarding a 12-year-old patient with tricuspid atresia. While Fontan found himself at a pivotal crossroads in deciding which surgical intervention to employ, his mentor had no such doubt, saying: “I have confidence in you, you decide.” 13 The available surgical options were a partial cavo-pulmonary, a Taussig-Blalock shunt, or a total cavo-pulmonary bypass. Weighing the options and aware of the significance should his approach be successful, Fontan took a leap of faith for this patient's operation. First, he performed a Glenn procedure by disconnecting the superior vena cava and attaching this to the right pulmonary artery. Fontan then connected the right atrium to the left pulmonary artery, thereby completely bypassing the right ventricle (Figure 1). 14 Although this decision held certain risks, the operation was considered to be Fontan's first success, although post-operative complications, including pleural effusions and anuria, resulting from volume overload and congestion. However, after several dialysis sessions prior to discharge, the patient survived into her thirties. 15 Although this procedure had been a success, it would be several more years, and two more patients, before Fontan published the results of his procedure, forever changing the lives of patients with tricuspid atresia.
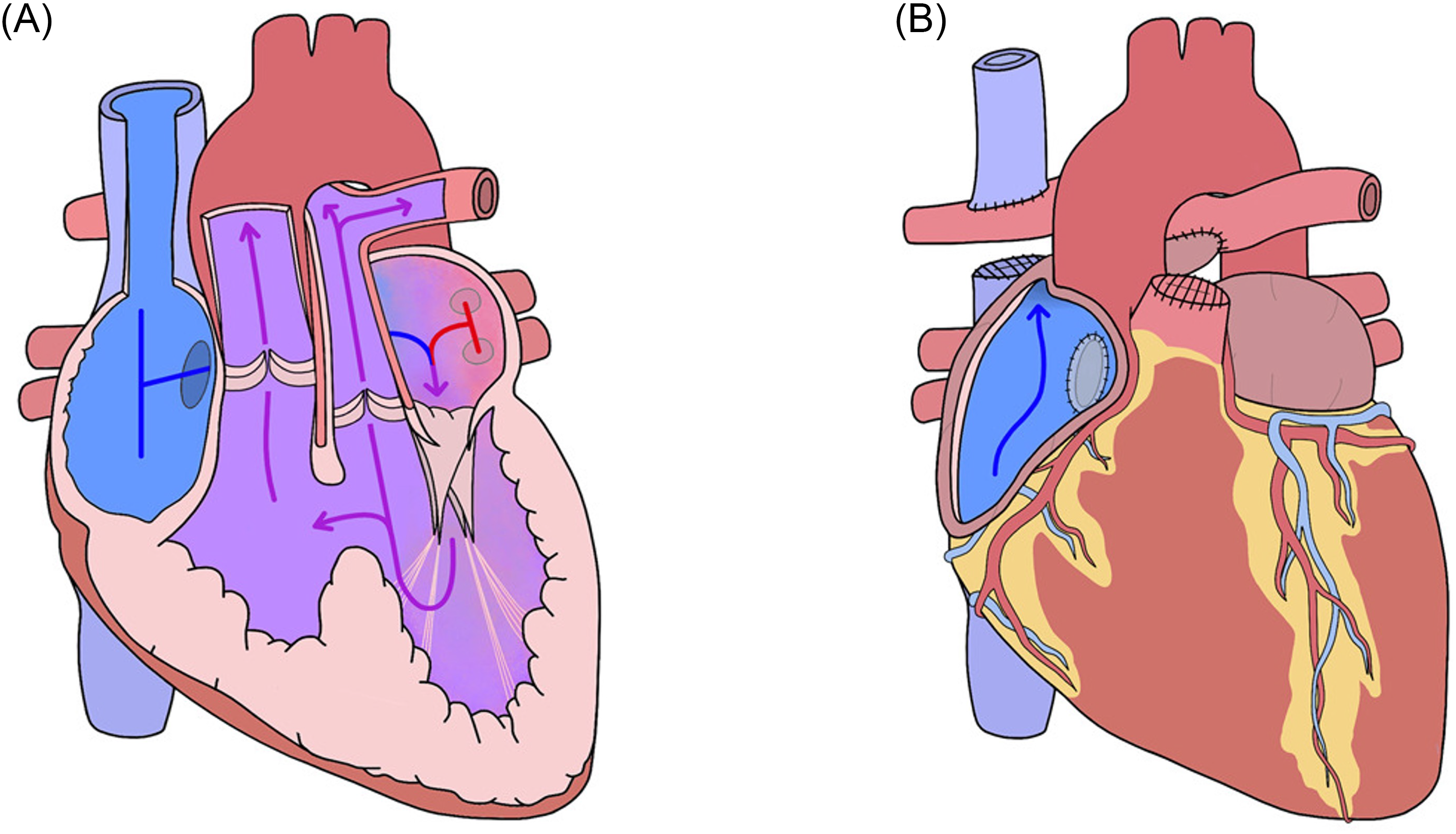
Depiction of the original Fontan procedure performed on a heart with tricuspid atresia. 14 (Creative Commons CC By license).
In 1970, Fontan was presented with another patient, a cyanotic woman with tricuspid atresia and discordant ventriculoarterial connections who had miraculously reached adulthood without a cardiac procedure. A similar version of the first procedure was performed with an aortic homograft sewn into the right atrium and pulmonary trunk. 3 Unlike the first patient, the woman suffered only minor post-operative complications and was able to go on to marry and give birth. This success heralded a brighter future for those born with tricuspid atresia.
Based upon data and hypotheses that had been two centuries in the making, the Fontan procedure represented both monumental progress in the world of cardiac surgery and a beacon of hope for those with single functioning ventricles. When asked during an interview to reflect on his decision to perform a high-risk new procedure, and whether he had believed it would work, Fontan replied, “Oh yes! Otherwise, I wouldn’t have done it. The first time I did it, it was in 1968. I had previously done experimental research on dogs, here in Bordeaux, at the Tondu hospital. The dogs survived only a short time, several hours. They tolerated ECMO very poorly. And then we did not have a transfusion reserve.”
13
In 1971, Francis Fontan and Eugene Baudet published the findings of three cases with two successes in the medical journal
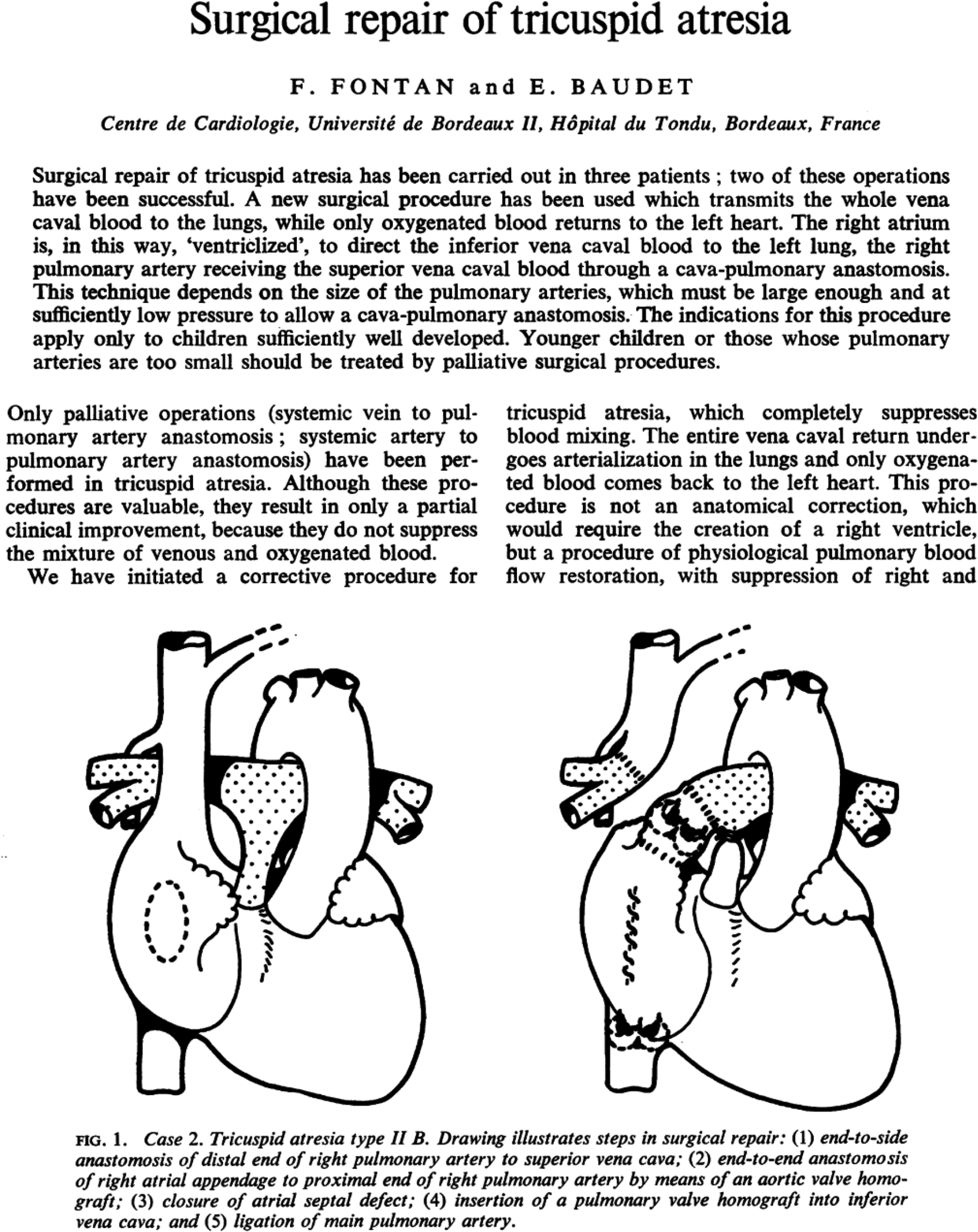
The first three Fontan procedures published in Thorax in 1971. 15
Worldwide response
By the 1950s, advances in the realm of cardiac surgery had created an openness to innovation within the medical community. Although several techniques were explored and introduced to the world – including the Robert Gross (1905–1988) atrial well, Clarence Walton Lillehei's (1918–1999) cross circulation technique and Richard DeWall's (1926–2016) bubble diffusion system – Fontan's novel approach remained a source of amazement.5,16 Former colleague, friend, and surgeon, Marko Turina, as described the impact of Fontan's procedure as substantial, “previously, very little could have been done for those children, they were sentenced to a miserable short life as a cyanotic baby with heart failure.” 17 Fontan's own recollection of the world's perception was that of astonishment and disbelief. He realized that his work had truly made an impact as he recalled, “I knew it while reading the book written by cardiac surgeon Christiaan Barnard (1922–2001) at Groote Schuur Hospital in Cape Town, South Africa who performed the first successful heart transplant in the world. He wrote that one day, his first assistant remarked to him: “Boss, we have been roasted by a Frenchman!” 13 Once the news had reached overseas, Stanley Kirkpatrick described the new procedure as “one of the major medical advances we have in our surgical armamentarium to correct congenital heart problems. In the past, we were only able to palliate with shunts and banding. Once the shunts and bands didn’t work anymore, the patients died. That was all we had.” 18 Dennis Pupello (1939–2018), Director of the Cardiac Surgical Unit at Tampa General Hospital, reflected on the innovation, saying, “for years, experts have been trying to come up with cures for kids with these particular problems… it was a matter of applying old principles to new techniques.” 19
Whilst the world applauded Fontan for his innovation, he did not bask in the glory because, as soon as the original cases were published in 1971, Fontan realized that there were limitations to the procedure that contributed negatively to the outcomes. Although the Fontan procedure represented a significant advance, it was not perfect, and Fontan and Baudet both recognized that certain conditions were associated with better outcomes. These included early tracheal extubation, post operative hemodynamic stability without requiring large volumes of fluid, low pulmonary airway pressures, and normal left ventricular and mitral valve function.15,20 As Fontan later reminisced, “we tried to improve [the procedure], creating a connection between the right auricle and small VD and closing the IVC. These are the patients who had the better result, probably because there was still a small dynamic ejection by the small residual RV.” 20 As more Fontan procedures were performed, numerous modifications were applied that contributed to the contemporary use of the procedure.
Procedure modifications
In 1973 in Buenos Aires, Argentina, Guillermo Kreutzer suggested that the inferior vena caval valves and Glenn shunts were not essential parts of the procedure and believed that they had resulted in pulmonary hypertension in two of his patients. 21 Instead of the homograft that Fontan originally used, Kreutzer successfully utilized an end-to-end anastomosis between the atrial appendage and pulmonary annulus using the patient's own pulmonary artery. This was a major contribution to the procedure since post-operative pulmonary hypertension had been a concern cited by Fontan and numerous others.
By 1977, Fontan and Choussat et al. had published a list of the “Ten Commandments,” which described the ideal candidate for the Fontan procedure, thereby establishing guidelines for the use of this imperfect albeit invaluable procedure. 22 They defined that the best outcomes were achieved in patients who were > 4 years old, had left-ventricular ejection fraction > 0.60, mean pulmonary artery pressure < 15 mm Hg and the absence of pulmonary artery distortion. 22
In 1978, Frederick O. Bowman (1928) introduced a technique, which connected the right atrium to the right ventricle with a valved conduit. 23 Building upon Bowman's refinement of the Fontan procedure, Viking O. Bjork (1918–2009) devised an operation in 1979 that used a pericardial patch in lieu of a conduit. 24 Since younger patients could outgrow implants and conduits, the use of a pericardial patch was an important modification for pediatric patients.
Also in 1979, the Norwood procedure introduced a surgical correction for children born with hypoplastic left heart syndromes with the goal of using the right ventricle as the primary pumping chamber. 25 Most notably today, the Fontan procedure is used after the Norwood procedure to treat hypoplastic left heart syndromes.
In 1988, Marc de Leval (1941) contributed the lateral tunnel technique which consisted of three parts: “(1) end-to-side anastomosis of the superior vena cava to the undivided right pulmonary artery; (2) construction of a composite intraatrial tunnel with the use of the posterior wall of the right atrium; and (3) use of a prosthetic patch to channel the inferior vena cava to the enlarged orifice of the transected superior vena cava that is anastomosed to the main pulmonary artery.” 26 By integrating these techniques in a single operation, de Leval was able to demonstrate that redirecting all venous blood to the left side resulted in better outcomes and reduced the risk of arrythmias and atrial thrombosis. 26 His work ultimately made the procedure simpler and more reproducible.
In 1989, Nancy Bridges (1954) introduced fenestrated atrial baffles (by surgically created tunnels in vessels or heart) which reduced the incidence of postoperative pleural effusions, reduced mortality, and shortened hospitalization time. 27
In 1990, Carlo Marcelletti (1944–2009) further improved the procedure by utilizing a prosthetic valveless conduit between the inferior vena cava and pulmonary artery in elderly patients. 28 This avoided aortic cross-clamping and reduced suture lines, resulting in fewer arrythmias.
De Leval's lateral tunnel technique, Marcelletti's extracardiac conduits, and fenestrated atrial baffles are the modifications to the Fontan procedure that are currently used.
Late complications
Although the Fontan procedure was initially successful in extending the lifespan of patients with one ventricle, it was nevertheless associated with significant morbidity as the patients aged.16,29–32 Jack Rychik (1959) observed that “[survival] appears to come at a cost, with progressive decline and circulatory failure in a growing number of subjects as they enter their second and third decades of life.” 29 In longitudinal studies, patients having undergone the Fontan procedure were found to have elevated central venous pressure, suboptimal cardiac output, decreased exercise capacity, atrial arrhythmia, and thromboembolism.16,29,32 After having undergone the Fontan procedure, children were found to have significantly higher factor VIII levels which could contribute to the risk of thrombosis. 33
A large case series established silent pulmonary thromboembolism as the cause of death in 8% of survivors.29,30,34 Several case reports also described the development of hepatic fibrosis, elevated liver enzymes, and cirrhosis. 35 Cross sectional studies of long term survivors of Fontan type operations found that at least 40% of patients who were categorized as functioning well in fact had elevated liver enzymes, high levels of coagulation factors, and low serum albumin. 16 Protein loss enteropathy was also cited as a significant mortality risk.29,30,36 In light of these complications, management strategies were developed to improve outcomes. Low dose aspirin was recommended to decrease the risk of thromboembolism, and some patients required long term anticoagulation medication. 37 Patients with protein losing enteropathy were found to respond to low dose oral budesonide. 36 Most importantly, the long-term monitoring of cardiac and liver function was found to be essential to identify post-Fontan complications.
Since Fontan's report was published in 1971, controversies surrounding survival outcomes have arisen, with several studies that assessed the results of the Fontan operation finding decreased 15-year survival.9,38 In 1999, Thomas Yeh Jr. (1961) examined survival in 435 patients with cavopulmonary shunts performed with or without the Fontan procedure, and found the 20-year survival rate to be 55–65% in both groups. 39 A 2006 study compared 177 patients who underwent a bidirectional Glenn procedure with 149 patients who underwent a Fontan procedure. Complications were found to be markedly lower with a bidirectional Glenn procedure (7%) compared to a Fontan (49%). 40 The authors concluded that the bidirectional Glenn procedure may be better suited for single functional ventricle patients. With contemporary improvements, however, one report describes the 20-year survival rate in 305 patients to be 84%. 41
Discussion
Ultimately, it was the assignment to perform an autopsy on a young patient who had died of tricuspid atresia that was the catalyst for Fontan's relentless pursuit to find a solution to a supposedly unsolvable problem. During his training, Fontan realized that tricuspid atresia represented an almost universal fatal diagnosis, offering a grim prognosis even with treatment. Fontan was then encouraged by his mentors to study previous work on this subject, using this information to guide his own experimentation.
The early stages of Fontan's career coincided with several advances in cardiothoracic surgery, thus promoting a culture of innovation and hope. In 1953, John Gibbon (1903–1973) had been the first surgeon to successfully utilize cardiopulmonary bypass in a patient with an atrial septal defect, thus popularizing the use of extracorporeal circulation. 42 In 1961, the technique of priming the heart-lung machine with 5% dextrose allowed for a safer and more practical alternative in open heart surgery. 43
Although Fontan's first attempts at surgical treatment in dogs had been unsuccessful, the support and trust of his mentor encouraged him to continue. Most importantly, Fontan's ability to recognize the deficiencies in his approach and to find solutions to the problems that arose as the result of his work enabled him to improve the outcome for subsequent patients. The overall impact of Fontan's work is immeasurable, since his procedure paved the way for further procedures in the field of pediatric cardiovascular surgery, most notably the Norwood procedure, which would have been impossible without Fontan's contribution. 44
Contributions to modern medicine
As a man of many talents, Francis Fontan's accomplishments extend beyond his eponymous procedure. For example, in 1965 he established the first homograft bank in France, he was the first surgeon in France to perform the Senning procedure (baffle prepared from the patients’ own tissue) and the fourth to perform a heart transplant. 1 Described as a wine aficionado, a golf lover and a beloved mentor, his contributions to the world of cardiothoracic surgery have proved to be integral to the modern techniques that are used today. A former Fontan mentee, Thomas Spray, said “since the initial description of the Fontan cavo-pulmonary connection in 1971, countless patients have benefited from his surgical innovation and dedicated exploration of the boundaries for success in the procedure.” 45
Mentorship was integral to Fontan's journey, enabling him to navigate skepticism and long-standing ideologies within the surgical community. In describing his own experience in the discovery of his eponymous procedure, Fontan remarked, “you may have experienced being misunderstood because your ideas were too advanced for the time, because they conflicted with well-established convictions, or because they were not expressed convincingly.” 2 Fontan believed his role as a mentor and teacher to be his most important and proudest accomplishments, and, therefore his legacy is not limited to pediatric cardiothoracic surgery. When asked about the happiest or proudest moments of his career, he replied “professionally: my students, my successors. I shaped a good 10.” 13 Close friend and mentee, Robert Anderson, described Fontan's impact, saying “as one of my most important mentors… it provided me with immense support.” 44 Fontan's personal conviction to pursue the unknown inspired many, especially his mentees.
In an address to the American Association for Thoracic Surgery in 1990, Fontan explained that in order to continue advances in medicine, any innovator “must permanently improve his personal knowledge through his own ideas while keeping an open mind to others.” He believed that “the holder of knowledge has a difficult task and great responsibilities. Of course, he must be willing to transfer his knowledge, not to keep it for himself.” 2 This ethos motivated him to help establish the European Association of Cardiothoracic Surgery in 1986, one of the largest cardiothoracic associations in Europe (Figure 3), which uses English as its main language to standardize and maximize the dissemination of knowledge.

Dr. Fontan one of the founding fathers of the EACTS in 1982 with his friends, Gerard Brom on his right and John Kirklin on his left. 13
Conclusion
In March 1968, Francis Fontan boldly attempted a novel surgical technique to improve the life of a patient with tricuspid atresia. At that time, he faced a clinical impasse since no other satisfactory treatment options were available. While medical progress often takes a steady and incremental path toward improved clinical care, the process is sometimes interrupted by a revolutionary concept that changes medical management forever. The introduction of antibiotics, the discovery of anesthesia, the concept of the artificial kidney and renal transplantation are good examples of this phenomenon.
Although Fontan's concept and his original operation may have undergone modification and improvements, he introduced a new approach to a previously untreatable condition and one which could be applied in a myriad of clinical scenarios. The career of Francis Fontan exemplifies the fortitude and determination necessary to solve a seemingly unsolvable clinical problem and thus advance the practice of medicine (Figure 4). 46

Portrait of Dr. Francis Fontan (1929–2018). Courtesy of Dr. Marko Turina. 46
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Massachusetts Chan Medical School-Intramural funding.
