Abstract
The complementary advantage of carbon nanotubes (C) and graphene oxide sheets (G) was used to form composites with Fluoroelastomer (FKM) via melt solvent/solid mixing techniques and their properties were studied. The hybrid compositions demonstrated better physico-mechanical properties than the single-filled, at lower concentration of the G-loading. At high content of G-sheets, formations of agglomerates were observed, resulting in inferior properties. The hybrid composition FCG (1:1) recorded the highest tensile strength (∼47% > FKM), slightly higher than FCGa (0.1:0.3) by ∼1.9%. FCGa in turn, recorded the fastest cure properties (lowest T90 and highest CRI), higher cure rheo-mechanical strength indicators (ML, MH and ΔM), high thermal stability (84.3% > FKM based on weight residue (%)). FCGa also recorded the lowest energy dissipation (tanδ), and highest dielectric constant (ɛʹ), which were about 57.7% > FKM. It was observed that enhancement in properties of FCG was mainly due to high content of physical networks (—C—C— or —G—G— and —C—G—) while those in FCGa were mainly as results of chemical links (C—FKM—
Keywords
Introductions
Elastomers are known for their excellent flexibility, elongation, tensile strength, chemical resistance, water resistance and other interesting properties. These properties make elastomers useful in many sectors including: agriculture, military, transportation, oil and gas and medical field.1–4 Example of these elastomers include: natural rubber (NR),5–8 ethylene propylene-diene monomer (EPDM),9,10 fluorinated rubber (FKM)11,12 and acrylonitrile butadiene rubber (NBR)13,14 etc. For some time now, elastomer blends have been greatly investigated by taking advantage of the complementary effect of the two or more matrix systems. The blend system has been reported to be better than individual matrices.15,16 Examples of these blends are: the dual matrix system: NR-styrene-butadiene rubber (SBR) 17 and SBR-polybutadiene rubber (BR)15,16 etc., the ternary matrix systems; NR-SBR-BR 18 and NR-BR-SBR 19 etc.
The unfilled state of elastomeric matrix (single or blend), is not desired for practical applications, unless reinforced with specific fillers for an intended application.20,21 The common fillers hugely investigated include: carbon black (CB), silica (SiO2), nanoclays (NC), nickel zinc ferrite, Fe3O4 and alumina nanoparticles etc.,22–30 These fillers are used to enhance strength, wear, thermal stability and other interesting properties etc.20,21,31 One major disadvantage is that high concentrations (≥20 phr) of these traditional fillers are required in order to achieve high reinforcing effect.20,21,31 Drawbacks for the high filler concentration include: difficulty in mixing, elevation of energy for mixing and subsequently an increase in production cost.20,21,31 Moreover, at these high concentrations, fillers have shown evidence of poor dispersions with high tendency of forming agglomerates within the matrix. This results in poor physico-mechanical properties.5–8 Functionalization of these reinforcing fillers, with different kinds of compatibilizers and coupling agents such as: silane and maleic-anhydride etc.,20,21,31 has been proposed to overcome these limitations.
The discovery of nanotubes (NT) and their counterpart graphenes/derivative graphene sheets (graphene oxide (G), reduced graphene oxide (rGO), graphene nano platelets (GNP) etc.) have brought a huge revolution in rubber science and nano-technology. Unlike the traditional fillers (CB, SiO2, Alumina and NC etc.), with NTs and graphenes, only a small volume fraction (0.1∼5phr) is required to improve dispersions and increase the physico-mechanical properties. This has been linked to their unique properties like: high aspect ratio, electrical conductivity, and thermal conductivity etc.1–4 Several elastomeric-graphenes composites 2 and elastomer-NT composites 1 have already been reported for their versatile applications in sensors, dielectric, electrical, and thermal resistance.32–38 For instance, smaller amount of GNP (2wt%) was incorporated into thermoplastic natural rubber (TPNR) containing polyaniline by melt compounding. This caused an increase in the electrical conductivity, thermal and mechanical property of TPNR, reported by Zailan et al. 39 Also, Flaifel et al. 40 recently developed a bio-nanocomposites by incorporating GNP and polyaniline into a polylactide/liquid NR matrix, achieving improved mechanical, electrical, thermal and radiation shielding capabilities.
Nowadays, a hybrid of G, rG or NT fillers and sometimes with CB is under serious investigation, aimed at taking advantage of the complementary effect of these fillers. Typically, in the area of strain sensor applications, rubber containing CB-G,3,41,42 CB-NT,43,44 G-NT45,46 have recorded a sensitivity or gage factor (GF) as high as ∼104 ∼ 105 at 0∼44% elongation, even with small concentrations (0.1∼10phr), as reported by Mensah and Song et al..3,4 This performance exceeded that of elastomers filled with single fillers which recorded as low as of 0.06⁓5
In this present work, the microstructure of FKM rubber (known for its resistance to harsh chemical and thermal environment), was controlled with the NT, G and hybrid NT-G using a combine method of solvent and open milling techniques for the first time. The compounds were then cured using peroxide and coagent Triallyl Isocyanurate (TAIC) curing system at 160°C with a hot-press machine. A peroxide-cured system was used in this work because it is widely known for its relatively high bonding energy, thermal stability and higher resistance to chemicals like alkaline and amine etc.48,49 The physico-mechanical properties of the cured composites, namely rheometric cure, thermodynamic parameters of mixing, state of dispersions/morphological properties, tensile and dielectric properties etc., were all investigated and clearly presented.
Experimental
Materials and chemicals
Samples of C, G and C-G filled in FKM matrix.

C: carbon nanotubes, G: graphene oxide, FKM: floroelastomer.
Sample preparations
The preparation of the FKM, FKM-C, FKM-G and FKM-C-G compounds was done by combined method of solution mixing and an open two-roll milling techniques. The FKM was cut into pieces and dissolved in acetone under 50°C temperatures for ∼12 h. The respective amounts of G and C as shown in Table 1, were individually dispersed homogeneously in Dimethylformamide (DMF) using ultrasonication for ∼2.5 h, in order to disentangle the sheets and tubes from each other. The solutions of G-DMF and C-DMF were each mixed with FKM-acetone mixture separately. The mixtures were then stirred vigorously on a magnetic stirrer at ⁓60°C for ∼12 h until homogenous mixtures were obtained. To avoid phase separation between the matrix and the fillers, de-ionized water was added to the homogeneous mixture while still stirring. The resulting uncured composites (FKM-C, FKM-G and FKM-C-G) were oven dried at ⁓80°C for about ⁓6 h in order to eliminate entrapped liquid in the mixtures. The same process was used for the compounds with hybrid fillers (C-G), except that the dissolved FKM solution was mixed with C and G in the same container, as depicted in Figure 1 Later, the curing ingredients (Triallyl Isocyanurate (TAIC) and peroxide) were gradually and gently added one after the other and milled to obtain solid masses by using a two-roll mill (Farrel 8422, USA). The homogeneously milled samples were sheeted out and allowed to cool overnight. Image of the solution mixing steps for the composites formulation (a) C dispersed solution (b) G dispersed in DMF solution (c) KFM dissolved in Acetone (d) FKM-C-G solvent before coagulation, and (e) FKM-C-G after coagulation.
The different formulations were cured in a square metallic mold (15 cm × 15 cm with ∼0.1 cm thickness) in-between Mylar film, with the help of a hot press machine (Caver WMV50H, USA), at the optimum cure conditions (A pressure of ∼11 MPa and a temperature of 160°C). The samples were allowed to cool overnight, and were later cut into standard shapes and dimensions for analysis and characterizations.
Characterization
Rheometric cure property
The cure properties of the FKM samples were measured at 160°C by a cure rheometer (ODR 2000, Alpha Technologies, USA). The cure properties which include: onset of curing time (ts2), optimum curing time (t90), cure rate index (CRI) = 100/(t90-ts2)), maximum (MH) and minimum torque (ML), and change in torque (ΔM = MH-ML) were all collected from the respective rheo-curves and analyzed.
Scanning electron microscopy (SEM)
The morphologies and state of dispersions of carbon nanotubes (C), graphene oxide (G)-sheets or C-G blend in FKM were studied by using SEM technique. The composites were initially cryogenically fractured and then sputter-coated with platinum metal. This was to increase the electrical conductivity at the observing surfaces of fractured samples. Field emission SEM/energy dispersive x-ray spectroscopy (EDS) (JEOL, JSM 599, Japan) was used to examine the surface morphologies. The test was completed within ∼40 min.
Transmission electron microscopy (TEM)
Ultrathin specimen (thinner than 100 nm) of cured FKM samples for the TEM observation were cryogenically cut with a diamond knife using an ultrathin microtome (UCT, Leica Ultracut, EMFC7) and collected on 200-mesh copper grids. A TEM was used to observe state of dispersions of carbon nanotubes (C), graphene oxide (G)-sheets or C-G in FKM rubber (TEM, JEOL, JEM2100).
X-ray diffractometry (XRD)
A wide angle X-ray diffraction measurement (PANalytical X-PERT Powder diffractometer) was carried out to characterize the state of dispersions of carbon nanotubes (C), graphene oxide (G)-sheets or C-G in FKM with Cu-Kα radiation (40 kV, 100 mA, λ = 0.154 nm). X-ray diffraction patterns were obtained at room temperature with a continuous scan type.
Crosslinking density by equilibrium swelling test
The cured FKM samples were equilibrated in methyl-ethyl ketone (MEK) (molar volume of 72.11 mL/mol) for ⁓72 h at room temperature. The degree of swelling (Qr) was calculated using the fractional change in the amount of MEK (Ws-Wi) absorbed and the dried weight (Wdr) of samples by using equation (1);
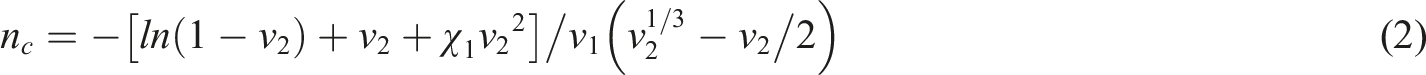

Gibbs free energy
In order to understand the mixing efficiency of the fillers and FKM matrix, a thermodynamic study was conducted. The change in Gibbs free energy (ΔG) was calculated by Flory-Huggins equation (4)
54
using the equilibrium swelling information.
Tensile test
The tensile properties of FKM and its composites with carbon nanotubes (C), graphene oxide (G)-sheets, and C-G were measured at room temperature by using universal tensile testing machine (Lloyd Instrument, UK), obtained from IPNL, Department of Polymer-Nano-science and Engineering, Jeonbuk National University (South Korea), with dumb-bell shaped specimen (ASTM D412). The cross-head speed was 500 mm/min. The elongation at break Ebr (%) and tensile strength TS (MPa) were deduced from the stress-strain curves. At least four samples were tested for each composition and averaged.
Differential scanning calorimetry (DSC)
A DSC analysis was conducted on the FKM and it’s composite with carbon nanotubes (C), graphene oxide (G)-sheets and C-G fillers. The test was conducted using a Universal V4.7 A, TA Instruments, Q20 V24.9 Build 121 with different temperature rates. All samples were cooled to −80°C and reheated up to 80°C at 10°C/min. Some of the most important properties deduced from the DSC analysis include: melting temperature (Tm), glass transition temperature (Tg) and crystallization temperature (Tc). These data enabled further understanding of the filler dispersions and interactions with the matrix material.
Thermal degradation and kinetics study
Thermal gravimetric analysis (TGA-DSC) (NETZSCH, STA409PC, Germany) was used to determine the thermal stability of FKM and its composites with carbon nanotubes (C) and graphene oxide (G). The tests were conducted under N2(g) atmosphere in ambient temperature of 25°C to 1000°C at a heating rate of 5°C/min. The thermal stability behavior of FKM and its composites with C and G-sheets were studied based on the temperature at 10% weight (T10) or onset of degradation temperature, optimum degradation temperature at 90% weight loss (T90) loss, maximum degradation temperature (Tmax) from the derivative thermograph (DTG), and the weight residue (%) from the TGA curves.
Dielectric spectroscopy
The dielectric constant (ɛʹ) of FKM and its composites with carbon nanotubes (C), graphene oxide sheets (G), was investigated by an LCR meter (VHR-200, USA). The conditions used include; frequency of 1∼106 Hz, at 1 Hz with steps and an optimum voltage of 0.5 V.
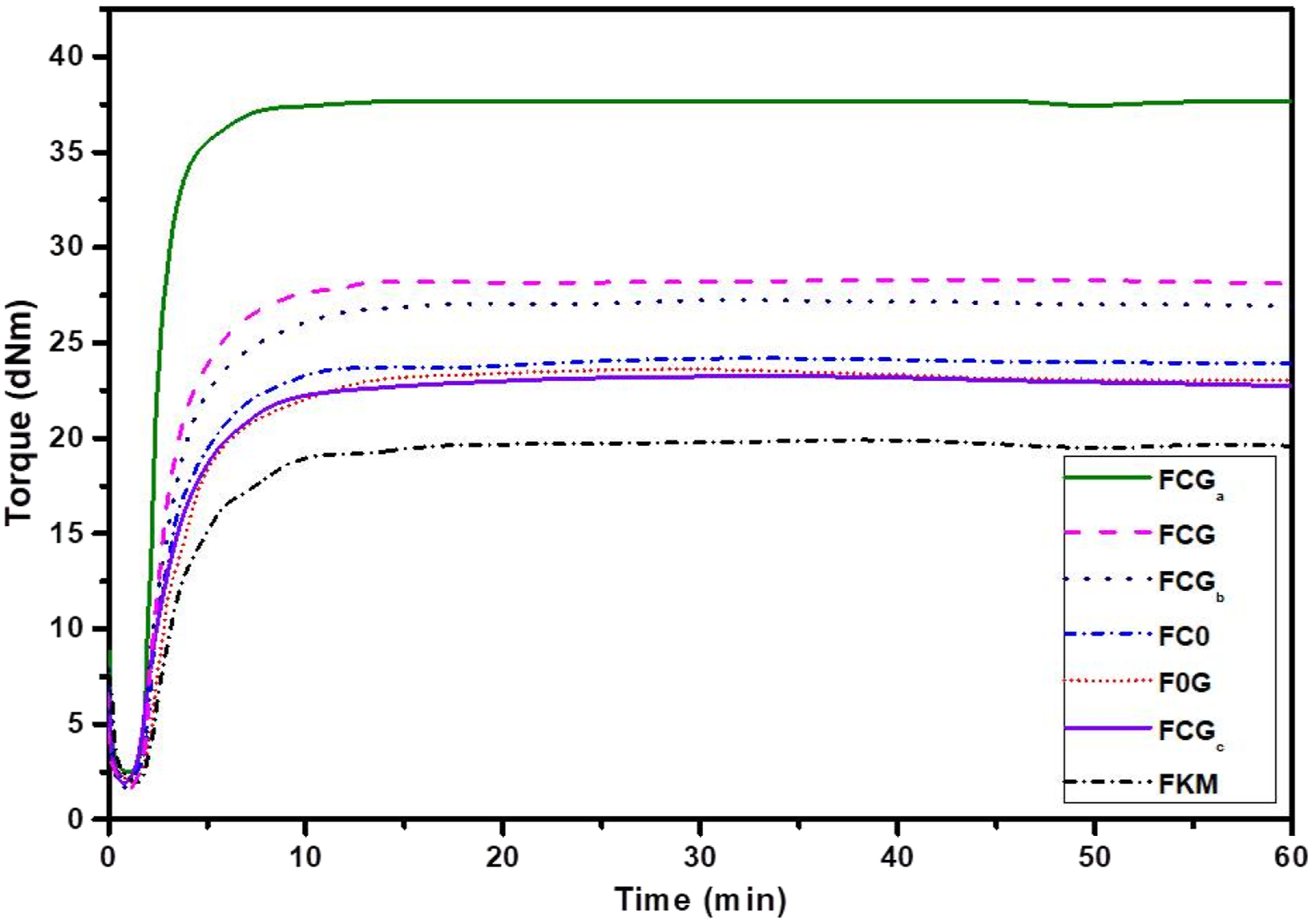
Both sides of the samples were coated with silver grease electrode to improve the surface resistance to the same degree for specimen to avoid differences in thickness. The average thickness of the samples used was ∼1 mm. The study was done at room temperature and the prepared sample is as shown in Figure 2. The samples were connected to the applied field through the copper electrode (tape) as shown. The real part of complex dielectric constant ( Sample prepared for dielectric constant testing. The rheo-curves of the various vulcanizates.


Results and discussions
Vulcanization behavior
The rheometric cure curves for samples cured at 160°C are shown in Figure 2 while the cure properties; viscosity index (ML) and maximum torque or crosslinking density index (MH), strength index (ΔM), optimum and minimum curing time (t90 and ts2) and the cure rate index (CRI) are compared in Figure 5(a)–(d) respectively. Generally, both the onset of curing time (ts2) and optimum curing time (T90) of the FKM dropped when the carbon-based fillers were incorporated. However, when the content of the graphene oxide (G) was increased, the curing parameters (ts2 and T90) were seen increasing significantly. At 0phr of fillers, FKM recorded T90 of about 8.5min, but when the level of G was increased to 2phr with 0.1phr of carbon nanotubes (C), the T90 shot up to ∼8.7min for composition (FCGc). It was also observed that G-based compounds showed slightly higher (ts2 and T90) than the C based systems.
The high thermal conductivity behavior of nanotubes than the wrinkled graphene oxide sheets could be a reason for such observation.55,56 At lower concentration level of the hybrid (∼0.1phr), the carbon-base fillers acted as cure accelerators and promoted faster curing. 2 However, at higher carbon base fillers loading, especially at high G level (0.1 to 2phr), the hybrid carbon base fillers acted as curing scavengers or cure retarders, 2 and thus delayed crosslinking reactions.
This effect could be due to the formation of agglomerated phases which inhibits crosslinking reactions. Several mechanisms have been proposed to explain delays in curing of rubbers, some include: poor dispersions, adsorption of cure accelerators by fillers, and aspect ratio effect etc.
.2
For further understanding of this phenomenon, Scheme 1 is proposed. Clearly, unfolded/unbroken carbon base fillers filler like G-sheets can bond well to different sections of rubber chains for high crosslinking to proceed, in the presence of curatives compared to the folded, corrugated and broken fillers as depicted in Scheme 1U. Illustration of how agglomerated fillers (C, G or C-G) leads to delayed crosslinking reactions in rubber matrix; (U) folded/defective and unfolded G-sheets attached to a rubber chain (V) agglomerated G-sheets in a matrix (W) homogenously dispersed G-sheets in a matrix (X) well aligned G-sheets for high conductivity (Y) Fillers blocking active/reactive sites on a chain/molecule and (Z) Crosslinking additives entrapped or adhered in the core of fillers.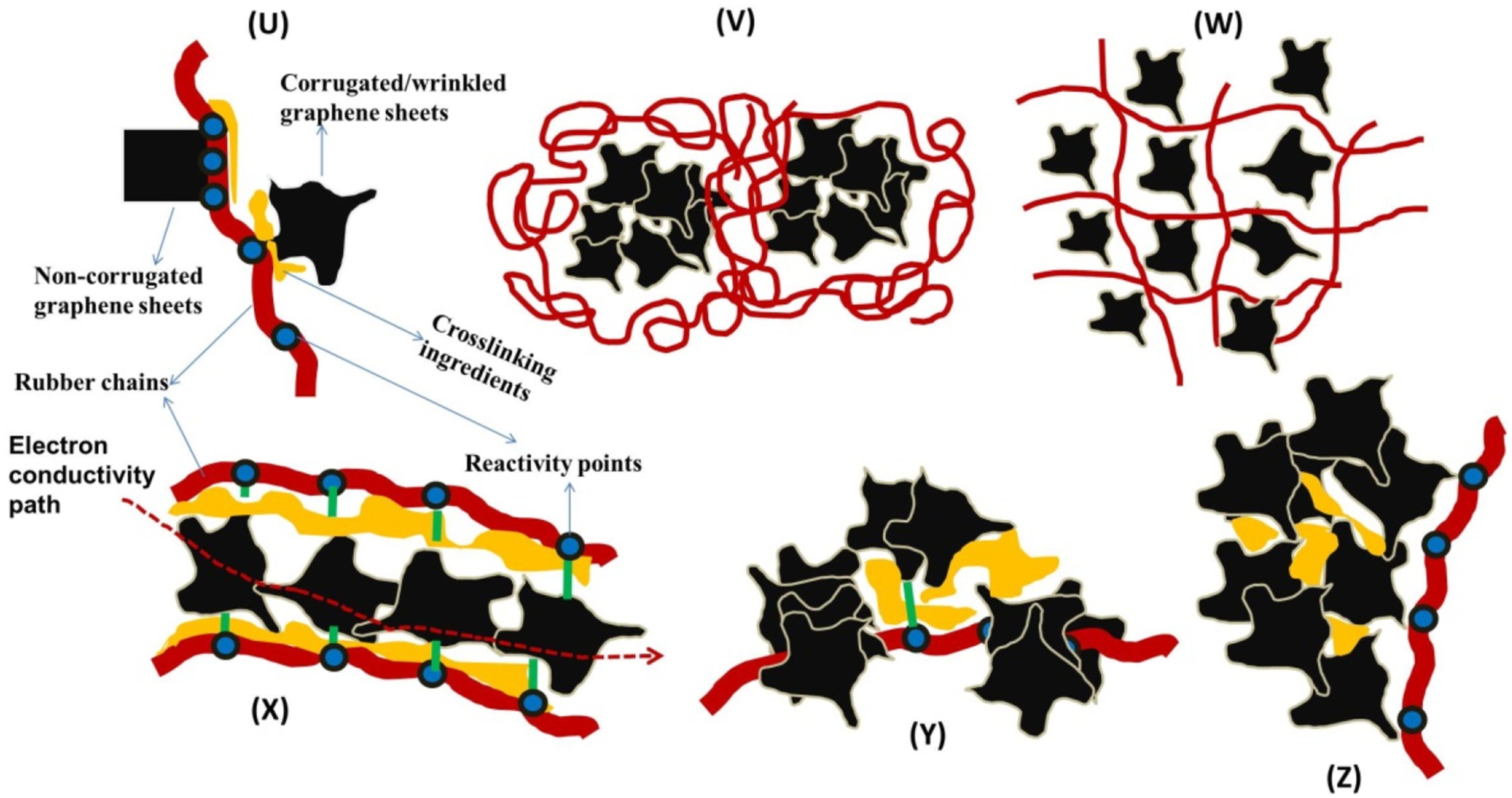
When agglomerates (Scheme 1V) are formed other than well dispersed fillers (Scheme 1W), it will be difficult for aggregated fillers to transport heat, to speed up melting/dissolving of the curatives within the bulk matrix to initiate chemical crosslinking (see Scheme 1X, for enhanced heat conduction for well dispersed fillers). Again, with curatives around, agglomerate could block the active or reactive sites for crosslinking reaction by physical adherence to the sites on the chains (Scheme 1Y). Further, it is also possible for agglomerates to entrap curatives within their core (Scheme 1Z), thereby slowing down or leading to poor overall crosslinking reactions.
When compared, the composition FCGa(Figure 5(a)) exhibited the fastest crosslinking reactions, with corresponding high CRI followed by FCG, as shown in Figure 5(b). The effect of faster crosslinking reactions may be due to effective carbon base fillers dispersions within the matrix. In Figure 5(c), it can be seen that incorporation of the G-sheets into the FKM matrix, generally reduced the ML but increasing the G-sheet does not hugely change this trend, unlike C (FC0) which was seen increasing ML significantly for the pure FKM as compared to F0G (Figure 5(c)). With regards to the crosslinking index (ΔM), addition of the mono or hybrid C-G of the carbon base fillers, especially for FCGa (0.1phr NT-0.3phr G) generally increased the ΔM = MH-ML to a critical point (∼37 dNm) where further increment in the G-sheets to 2phr (sample FCGc) dropped drastically to ∼22 dNm, as seen in Figure 3(c). This trend was similar to the mechanical strength index, MH as shown in Figure 3(d). The improved rheo-strength properties (ML, MH, and ΔM) for the hybrid filler systems may promote high mechanical properties. The curing properties of FKM filled carbon base fillers; (a) initial torque (ts2) & optimum curing time (T90) (b) minimum torque (ML) & change in torque (ΔM) (c) and (d) maximum torque (MH).
Thermodynamic of mixing
The thermodynamic parameters; Gibbs free energy ( Thermodynamic parameters; (a) Gibbs free energy, 
Clearly, the phase stability of the gum (FKM) was more feasible or negative, however when the fillers (carbon nanotubes (C), graphene oxide (G)-sheets and C-G) were incorporated, the composition attained less negative
State of dispersions and surface morphology
(SEM &TEM analysis)
The Figure 7(a)-(c) is the representative SEM images of samples FKM, F0G and FC0 respectively. In Figure 7(a), as seen, the cryo-fractured surface of FKM is smooth while the wrinkled graphene oxide (G)-sheets coated with rubber molecules are seen on the surface of F0G indicated by arrows in Figure 7(b). Also, the tube-like structures seen at the interface of FKM in Figure 7(c) is a clear presence of the carbon nanotubes(C). Earlier, Nah et al.57,58 reported that compressed rubber-C composite forced the tubes to slip and migrate to the cryo-interface. After a day, these nanotubes were completely reverted backwards deeply into the matrix. When the nanotubes were later functionalized, and subjected to the same test, some of the nanotubes did not migrate back into the matrix while others delayed in reverting back into the matrix, even after 24 h. This was a clear indication of strong matrix-filler bonding.57,58 A similar observations was made for graphene sheets in NBR matrix by Mensah et al.,
51
with SEM. In this case, even though no compressive stress was imposed, however the sheets and nanotubes at the cryogenic fractured interface did not revert back into the bulk rubber matrix, instead they remained at the interface throughout the SEM observations. Similarly, the current observation could also confirm stronger FKM-C or FKM-G interactions, likely to result in improved mechanical properties, as suggested by Nah and Mensah group.51,57,58 When the hybrid C-G carbon based fillers were incorporated into the FKM matrix, dispersion of G-sheets and C were seen at the cryo-interface (higher magnification in Figure 7(d) and lower magnification in Figure 7(e)). And this was associated with a strain induced rougher interface, probably due to tight structures. This effect could promote resistance to chain mobility, for higher physico-mechanical properties. State of dispersions and surface morphology of composites by SEM (a) FKM (b) F0G (c) FC0 (d) FCGa at higher magnification (e) FCGa at lower magnification (f) FCGc at lower magnification and by TEM; (g) FCG (h) FCGa and (i) FCGc.
Intestinally, when the content of G-sheets was increased to 2phr, a much larger agglomerates were seen sitting at the various interfaces of the cryo-fractured surfaces. To further understand the state of dispersions of the individual reinforcing particles or the hybrid effect, TEM technique was adopted to study the compounds. Figure 7(g)–(i) are TEM images for representative samples FCG, FCGa and FCGb respectively. As seen, it is difficult to use the TEM technique to observe the G-sheet probably due to the similar contrast between the matrix and the transparent G-sheets, however the presence of the nanotubes is clearly shown in Figure 7(f)–(h), as indicated by arrows. Another trend which can be seen is that, increment in the content of the G-sheets led to agglomeration formations as encircled with red colour. This agglomerate was very low in the case of Figure 7(f) and very higher for composition with higher amount of G-sheets. An agglomerated interface has been reported to yield poor physico-mechanical properties, by acting as defective sites in the matrix of the composites.1,2,59
WAXD analysis
Figure 6 presents the WAXD graphs, for the state of dispersions of the carbon-based fillers dispersed within the FKM matrix, the FKM matrix demonstrated relatively sharper peak intensity around Bragg’s diffraction angle (2θ) of 16.7° ( The carbon nanotubes (C) and graphene oxide (G)-sheets dispersions within FKM matrix characterized by WAXD.
The sharp peak that appeared around 26.5° (
Crosslinking density via equilibrium swelling
Crosslinking density and Young’s modulus properties of FKM and its composites.

To further understand the nature of the crosslinks (filler-filler or filler-matrix), the total network density (NT = E/3RT) based on the Young’s modulus,
Now, when
The dual filler system of C-G improved the
Glass transition temperature
The Figure 9(a) compares the glass transition temperatures (Tg) curves recorded in the DSC analyses for representative samples while the Figure 9(b) are the Tg values collected from Figure 9(a). Generally, incorporation of the fillers into the FKM matrix increased the Tg of FKM, FC0 recorded very low Tg value as seen in Figure 9(b). Glass transition temperature (Tg) of the gum (FKM) and representative composites containing carbon nanotubes (C), graphene oxide (G) and C-G, conducted by DSC analysis.
From Figure 9(b), the order of increment in the Tg can be written as; FCGa> FCG > F0G > FCGc > FKM > FC0. The synergy effect of the C and G-sheets improved the Tg of FKM more than the single-filler systems, by restricting the mobility of chains, reducing flexibility and increasing stiffness of the FKM. For instance, the FCGa increased the Tg of FKM above 16.5%. The Tg slightly dropped on increasing the G-sheets to 2phr (FCGc), which was suspected to be the results of increased number of filler-filler networks rather than stronger interactions (filler-matrix). Higher Tg do not only suggest higher mixing efficiency of fillers within the rubber matrices but also predicts higher strength of the compositions, hence the composites, typically FCGa is expected to show higher physico-mechanical properties than the pure matrix. With reference to the gum, the current composites, especially those with hybrid fillers have recorded higher Tg values than those reported for G and reduced graphene oxide (rG) in EPDM, NBR and EPDM-NBR blends reported earlier by Mensah et al. 69 The current results for the composites are also better than those reported for NR which recorded a rise of Tg by 5.8%. 70 Earlier, atomistic molecular dynamics simulation was used to predict the Tg of polysisoprene rubber reinforced with graphene sheets and a rise of 4.3% in Tg was observed. This value was closed to the experimental findings 71 but the value was lower as compare to the value obtained in this current composites, especially for FCGa sample.
Tensile strength
The tensile properties of FKM containing graphene oxide (G), carbon nanotubes (C) and C-G hybrid were also studied, in order to evaluate the hybrid effect of the fillers compared with single fillers in relation to the neat FKM. Figure 10(a) shows stress–strain curves of the compounds. As seen, the addition of G, C or C-G into the FKM matrix mostly enhanced the tensile properties compared to the pure FKM matrix. Interestingly, the hybrid filler systems showed improved TS as compared to single filler systems. The order of decreasing TS is written as: FCG > FCGa> FCGb > FC0 > FCGc > FKM > FC0, with compostion FCG recording the highest. FCG recorded TS of over 52, 39, 59,3.7, 3.7, and 55% higher than FKM, FC0, F0G, FCGa, FCGb and FCGc respectively. However, a higher content of G-sheets was observed to reduce the TS, causing a dramatic decrease in the elongation at break, as seen in Figure 10(b). This was observe to be due to the stiffening effect (see Figure 10(c)) based on the Young’s modulus Tensile properties of FKM and its composites with C, G and C-G; (a) the stress-strain curves (b) Young’s modulus at lower strains (c) elongation at break (%) and reinforcement index (RI = M200/M100).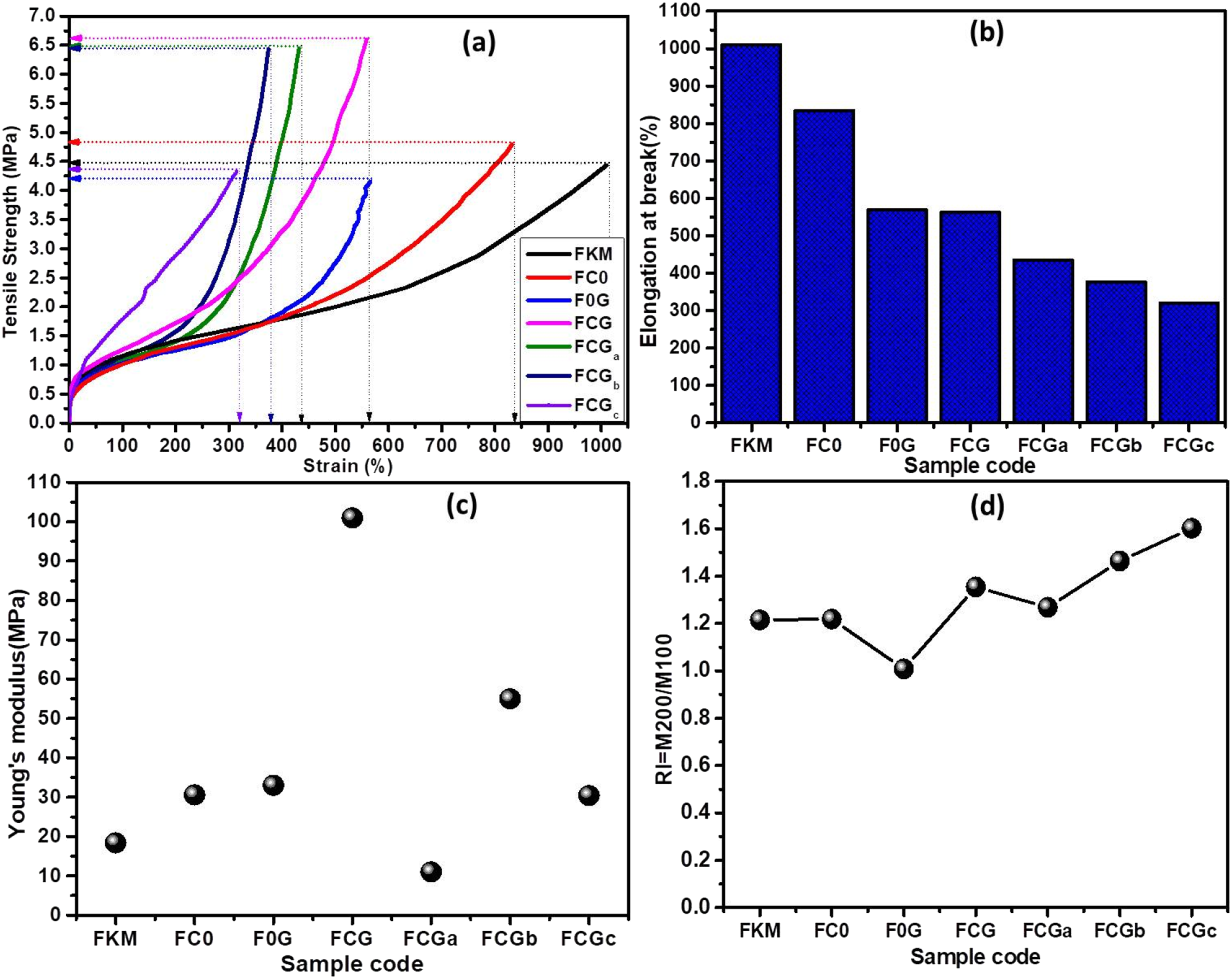
Wet and dry mixing techniques was used by Chen et al. 74 to prepare a composites of NR-G-sheets and NR-C. It was reported that the wet mixing techniqe yielded better physico-mechanical properties than the dry mixng method in most of the properties tested. Even so, the current sample, FCG showed relatively higher modulus at 300% and an improved elongation at break (%) than both technques reported by Chen et all. 74 This confirms that the hybrid effect of the C-G-sheets was superior compared to the individual nanoparticles in rubber matrices for applications where high strength will be needed.
Thermal stability study
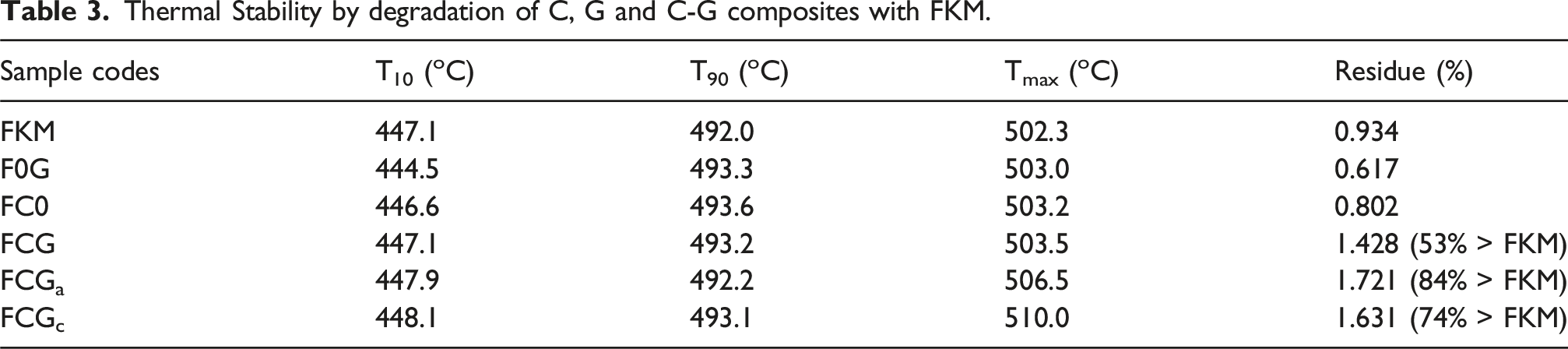
The thermal degradation behavior of FKM at different concentration of graphene oxide (G) carbon nanotubes (C) and C-G loading for representative samples are compared in Figure 11. The temperature corresponding to the 10 % weight loss was taken as the initial degradation temperature (Tonset), and the temperature corresponding to 90% weight loss was (T90%) while derivative thermograph was used as the maximum degradation temperature (Tmax). TGA/DTG results for representative samples of C, G, C-G in FKM in air N2(g) medium; (a) neat FKM, (b) FC0, (c) F0G, and (d) FCG.
One distinct degradation peak pattern was observed upon addition of C and G (Figure 11(b)–(c)); the Tonset showed scattered results but T90% and Tmax shifted to higher temperature compared to the pure FKM. It was observed that the low Tonset peak for the FKM containing single fillers recovered when the hybrid fillers (C-G) were added, giving one distinct peak ∼437°C. Tmax shifted slightly to higher temperature at 503°C for F0G compare to the neat FKM (502.3°C). The value was slightly lower than that of FC0 (503.2°C) and FCG (503.5°C) respectively. Thermal degradation temperature has been greatly characterized by the maximum weight loss rate in TGA.75–77
Thermal Stability by degradation of C, G and C-G composites with FKM.
Dielectric constant and loss factor
The effects of frequency and filler type on the dielectric constant (ɛʹ) of FKM have been compared in Figure 12(a) while the loss factor is shown in Figure 12(b) respectively. Generally, when the fillers were incorporated into polar FKM, the value of ɛʹ for FKM was seen improving significantly. Clearly, the hybrid systems containing smaller amount of graphene oxide (G)-sheets showed higher ɛʹ than the single filler systems and the compounds with high concentration (2 phr) of G-sheets (FCGc). The order of increment in ɛʹ, at lower frequency (<600 Hz) is: FCGa > FCG > F0G > FC0 > FCGc > FKM. Factors like; surface effect (high electrode polarization), ionic nature and the radical nature of the ingredients in the matrix are known to increase the value of ɛʹ.79–81 Presently, it appears improved dispersions of the carbon nanotubes (C)-G sheets in FKM matrix contributed to the improved value of ɛʹ. In particular, the graphene oxide used (G-sheets), although an insulator but contained high concentration of polar moieties such as Dielectric properties of FKM, and its composites with C, G-sheets and C-G in FKM(a) dielectric constant and (b) loss modulus (tanδ).
Another noticeable trend seen is that, the frequency, value of ɛʹ declined for all the samples at higher frequency. It was suggested that low frequency area where ɛʹ is higher occurs to allow enough time for permanent and induced dipoles to align themselves according to the applied field (associated with increased in polarization). 86 Also, at the higher frequency region where lower ɛʹ is observed, is due to dielectric relaxation effect caused by improper dispersion of the inclusions.6,47,86 In contrast, Dang et al., 87 earlier proposed that the low ɛʹ is due to a competition of higher electrical field and with elevated frequency. This phenomenon was reported in poly (vinyl alcohol) (PVA)-graphite and PVA-G composites 86 and poly (vinylidene fluoride)-graphite composites. 88 The loss factor (tan δ) in Figure 12(b) is a reversed trend of ɛʹ, which is seen increasing as frequency increases. Thus, increasing the frequency, led to a reduction of energy dissipation in FKM as a result of the presence of the nanoparticles. This is a common observation in dielectric-to-be materials observed for butyl rubber-C, 84 NBR-G, NBR-1phr reduced graphene oxide (rG) and NBR-1phr GAuNP.47,59
Conclusion
The microstructure of fluorinated elastomer (FKM) was modified using carbon nanotubes (C), graphene oxide sheets (G) and their hybrids (C-G), by using melt solvent/solid mixing techniques. The various physico-mechanical properties: cure properties, thermodynamic mixing effects, state of dispersion of fillers and morphologies by scanning electron microscopy (SEM), transmission electron microscopy (TEM), wide angle X-ray diffraction, tensile properties, thermal and dielectric constant etc., were carefully investigated. It was observed that, the influence of the hybrid effect (C-G) on the properties of FKM, especially at lower concentrations, was very pronounced as compared to the single filler (C and G) systems. The hybrid compositions demonstrated better physico-mechanical properties than the single-filled composites. Noticeably, increasing the content of G-sheets in the C-G blends, led to formation of agglomerates which resulted in poor properties for the highly filled compound, FCGb (0.1:2) observed by the help of SEM and TEM techniques. The hybrid composition FCG (1:1) recorded the highest tensile strength which was ∼47% higher than that of FKM and ∼1.9% higher than FCGa (0.1:0.3). On the other hand, FCGa recorded the fastest cure properties (lowest T90 and highest CRI), higher mechanical rheo-strength indicators (ML, MH and ΔM), high thermal stability (84.3% > FKM) measured by residue (%), lowest energy dissipation (tanδ), and highest dielectric constant (ɛʹ). The improvement in ɛʹ was about 57.7% higher than that of FKM. Based on the crosslinking densities, it was observed that properties improvement in FCG was mainly due to high content of physical networks (—C—C— or —G—G— and —C—G—) while those in FCGa was purely as results of chemical links (C—FKM—
Footnotes
Acknowledgment
We acknowledge Intelligent Polymer Nano Lab (IPNL), Polymer Nano-technology Department, Jeonbuk National University, South Korea for granting us the chance to use their facility for this work. The University of Ghana – Carnegie Next Generation of African Academics (UG-Carnegie NGAA) Project and the Office of Research Innovation and Development (ORID) at the University of Ghana toward the completion of the manuscript.
Consent to participate
Corresponding author has consent from all co-authors.
Author contributions
• Prof. Bismark Mensah (A), Mr Newman-Adjiri, Martey (B), Boniface Yeboah Antw(C). Dr Emmanuel Essien (D). • A: Project administration, Resources, Methodology, Investigation, Formal analysis, Data Curation, Supervision, Conceptualization and Writing – original draft and review/editing. • B: Methodology, Investigation, Data analysis and Writing– review/editing. • C: Writing– review/editing, validation. • D: Resources, Support, Writing – review/editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
