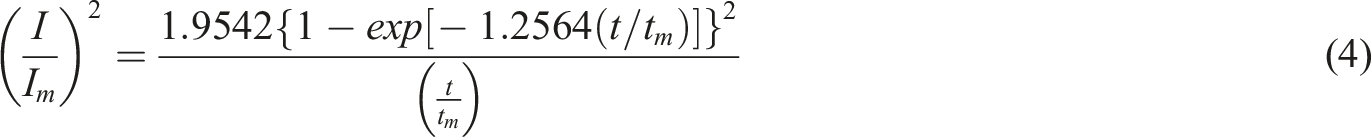Abstract
The photoelectrochemical route has emerged as an attractive alternative to produce H2O2 in a sustainable way since O2, H2O, solar energy and electricity (which can be obtained from a renewable energy source) are used as inputs. Currently, this route is in an early stage of research and several challenges must be overcome, mainly associated with the materials used as photoelectrodes in the process. In this study, polypyrrole coatings doped with the anionic porphyrin meso-tetra-(4-sulfonatophenyl)-porphyrin (TPPS) were electrochemically synthesized and their nucleation and growth mechanisms were studied. Then, the photoelectrochemical performance of the synthetized materials towards the oxygen reduction reaction (ORR) was examined by chronoamperometry tests and by measuring the H2O2 produced. It was found that in an initial stage of the electrosynthesis the coatings present an instantaneous nucleation with three-dimensional growth. Besides, it was observed that the coatings perform as electrocatalysts for the ORR and that their performance depends on their thickness. Interestingly, it was shown that the insertion of cobalt into the photoelectrodes increases the photocurrent by 100% and the total current by 74%, but this increase is not reflected in a larger production of H2O2 because the oxygen reduction does not take place via a two-electron pathway when cobalt is present. These results contribute to understanding the role of polypyrrole-based coatings in the photoelectrochemical process and also provide evidence on their stability.
Introduction
The electrocatalytic oxygen reduction is attracting attention in the field of energy transition into a sustainable and fossil-free economy. This reaction has been investigated for the development of devices such as hydrogen-based fuel cells, metal-air batteries, and recently for the design of hydrogen peroxide production cells.1–4
Hydrogen peroxide is considered as a green and environmentally friendly oxidant and is also a possible energy carrier due to its energy density (3.0 MJ/L in a 60 % w/w H2O2 solution), which is similar to the energy density of H2 compressed at 35 MPa (2.8 MJ/L).5,6 Currently, H2O2 is used for bleaching, disinfection, wastewater treatment and synthesis processes, 7 but its production is carried out by the anthraquinone route, which requires a large amount of energy, the use of expensive palladium catalysts and the implementation of several separation and purification steps.8,9 Furthermore, global H2O2 demand is expected to be 5.7 millions of tons by 2028, representing an annual growth of 4.6 %.10,11 These aspects are the motivation to investigate alternative and environmentally friendly technologies to synthesize H2O2.
A promising substitute to the anthraquinone route is the electrocatalytic process, which has the potential to be environmentally sustainable if an alternative energy source is used. In particular, the photoelectrochemical system (where at least one of the electrodes is available to absorb solar radiation) has gained attention in recent years for the production of hydrogen peroxide. Here, the desired reactions are the oxygen reduction by a two-electron path on the cathode equation (1) and the water oxidation by a four-electron path on the anode equation (2).
12
Since the oxygen reduction reaction can also occur by a four-electron path equation (3), great effort is required to fabricate suitable materials as cathodes to drive the desired reaction. Among the materials currently being investigated are intrinsically conducting polymers (ICPs). These are molecular materials that combine characteristics of metals and plastics, which exhibit electronic conductivity (according its doping level) and ionic conductivity (because of the presence of compensating ions). 13
Winther-Jensen B. et al., reported for the first time that poly [3,4-ethylenedioxythiophene] (PEDOT) catalyzes the oxygen reduction reaction. 5 After that, it has been demonstrated that PEDOT doped with poly (sodium 4-styrenesulfonate) (PSS) allows obtaining H2O2 as a primary product during the oxygen reduction reaction independently of the synthesis route of the polymer. 14 In addition, P. D. Nayak et al. found that the variation of the dopant and the presence of a conductivity enhancer during the synthesis of PEDOT influence on the performance of the polymer for the electrocatalytic H2O2 production. They showed that higher doping levels lead to higher reduction currents and larger H2O2 production. 15
Other ICPs that have been investigated as cathodes for hydrogen peroxide generation are: poly (3-hexylthophene) (P3HT), which although promotes the reaction of interest has the limitation of suffering photo-oxidative degradation. 16 Polyindole (PIN), which has been studied as an electrocatalyst but its properties as a photocathode have not been explored. 17 Polyaniline (PANI), that has shown a good performance for the reaction, especially when accompanied by nickel and subjected to a calcination treatment. 8 And Polypyrrole (PPy), which exhibits electrocatalytic properties for the H2O2 production and also prevents the further reduction of H2O2 to H2O. 18 Among PANI and PPy, it has been reported that although both polymers can act as electrocatalysts for the oxygen reduction, PPy allows obtaining larger currents. 18
In this work, we are particularly interested in PPy due to its favorable characteristics, such as good electrical conductivity, high visible light absorption, environmental stability and good redox properties.19,20 Furthermore, in a previous report, Cortés et al., showed that PPy has good stability as photocathode for the oxygen reduction reaction, 21 but aspects such as the effect of material thickness were not addressed nor was the reaction product analyzed. In this way, here we delve into these aspects and also study the mechanism of nucleation and growth of the polymer on a conductive transparent glass substrate, fluorine-doped tin oxide (FTO). The latter because the understanding of the electro-polymerization process is useful for the use of these polymers in thin film applications, where homogeneous coatings, adequate thickness control and electronic properties in accordance with the application are desired.
Materials and methods
Materials
FTO substrates (25 mm × 12.5 mm, Ossila, TEC 8) were cleaned at 55°C in a 10 % w/w NaOH (Carlo Erba, ≥97 %) solution. After that, FTO substrates were sonicated twice in ultrapure water for 15 min each. Pyrrole (Sigma Aldrich, 98 %) was distilled under a nitrogen atmosphere and stored in dark conditions. (CH3COO)2Co.4H2O (Sigma Aldrich, ≥98 %), H2SO4 (Sigma Aldrich, 98 %), H2O2 (Milipore, 30 %), TiO2 powder (Sigma Aldrich, ≥99 %) and meso-tetra-(4-sulfonatophenyl)-porphyrin, also known as TPPS (Sigma Aldrich, ≥95 %) were used without further purification.
Electro-polymerization of pyrrole to obtain TPPS-doped PPy
Electro-polymerizations were carried out using a three-electrode cell. For this, a platinum foil was used as counter electrode and an Ag/AgCl (3 M NaCl) electrode (MF-2052, BASi) as reference electrode (RE). The precursor solution was composed of 0.025 M pyrrole, 0.1 mM TPPS and 0.5 mM H2SO4 in ultrapure water (this solution was deoxygenated by bubbling with nitrogen for 10 min before each synthesis). Electrochemical polymerizations were performed on clean FTO substrates using potentiostatic signals (0.65, 0.70, 0.75 and 0.80 V vs Ag/AgCl). The thickness of the polymeric coatings was modified by the supplied electric charge. For all the electrochemical experiments was used an Autolab potentiostat (PGSTAT302 N).
Characterizations
The polymeric coatings were examined by scanning electron microscopy (SEM) using a TESCAN LYRA3 equipment. Photoelectrochemical characterizations were performed in a 0.1 M Na2SO4 solution using a solar simulator (ABET technologies 10500, equipped with an A.M. 1.5 filter and calibrated with a light intensity of 100 mW/cm2). Electrochemical impedance spectroscopy (EIS) data were obtained at −0.4 V versus Ag/AgCl using a frequency range of 0.1 Hz-10 kHz and an AC potential of 0.01 V. UV-Vis measurements were carried out with an Analytik Jena SPECORD 50 PLUS equipment. Raman measurements were carried out with a HORIBA Scientific XploRA equipment using a 532 nm laser. IR spectra were performed in a IRTracer-100 FTIR Shimadzu spectrophotometer. X-Ray photoelectron spectroscopy (XPS) measurements were carried out in a Specs equipment with a PHOIBOS 150 1D-DLD analyzer. A monochromatic Al-Kα source (1486.7 eV, 13 kV) was used with a pass energy of 85 eV for general spectra and 20 eV for high-resolution spectra. Binding energies were calibrated using the C 1s peak at 284.6 eV as a reference. The quantification of the hydrogen peroxide was carried out using a reported colorimetric method. 22 For this purpose, a titanium sulfate reagent was prepared by digesting 1 g of TiO2 powder with concentrated sulfuric acid for 16 h on a sand bath at a temperature of 150°C, then the obtained solution was made up to 500 mL using deionized water and filtered before use. In each H2O2 quantification, 10 mL of the titanium sulfate reagent were combined with 30 mL of the solution resulting from the photoelectrochemical process. The absorbance of each mixture was measured at a wavelength of 432 nm and compared with the absorbances of a calibration curve previously prepared with a standarized H2O2 solution.
Results and discussion
Initially, electrochemical polymerizations at different potentiostatic signals were performed until an electric charge of 25 mC/cm2 was supplied (typical current-time transients obtained during the process are shown in Figure S1). It was observed that all the transients presented a maximum current, which has been attributed to the expansion and collapse of polymeric nuclei, and also with the occurrence of a diffusion-controlled synthesis.23–25
It’s accepted that nucleation and growth mechanism of ICPs can be analyzed by theoretical models derived by Harrison and Thirsk. In these models, both instantaneous and progressive nucleation, as well as 2D and 3D growth, are considered and expressed in terms of dimensionless values using the following equations equation (4)–(7),23,26,27 where I and t indicate the current and time, respectively, while Im and tm refer to the coordinates at the top of the current.
Growth in three dimensions (3D) and instantaneous nucleation:
Growth in three dimensions (3D) and progressive nucleation:
Growth in two dimensions (2D) and instantaneous nucleation:
Growth in two dimensions (2D) and progressive nucleation:
Comparations between theoretical models and experimental transients of PPy electrosynthesis are shown in Figure 1. Here, it was found that the nucleation and growth mechanism of TPPS-doped PPy (PPy-TPPS) is similar under all applied potentials. Furthermore, it was observed that the experimental data fit better to the instantaneous nucleation model with three-dimensional growth, which was supported by calculating the mean absolute percentage error (MAPE) associated with each comparison (Table 1). Herein, MAPE values were obtained considering the range 0 < t/tm < 2.5, and the smallest magnitudes corresponded in all cases to the fit with the afore mentioned model. The instantaneous nucleation is characterized by having a constant number of nuclei during polymerization, this means that all nuclei form at the same time. In this way, an advance in the polymerization process implies the growth of the initially formed nuclei without the formation of new nuclei. Instantaneous nucleation leads to obtaining nuclei with large diameters and rough surface morphologies. The 3D growth implies that the expansion of nuclei is practically the same in the parallel and perpendicular directions to the electrode, leading to the formation of an homogenous film.27,28 Plots with no dimensions showing experimental transients and theoretical curves of instantaneous and progressive nucleation during electropolymerization of PPy-TPPS with (a) growth in three dimensions and (b) growth in two dimensions. Mean absolute percentage error values calculated by fitting the experimental data of PPy-TPPS electropolymerization on FTO to the theoretical nucleation and growth models.

It is worth mentioning that notable deviations of the experimental transients with respect to the theoretical ones could be seen after a t/tm > 1.5, that is, when the polymer has already completely covered the surface of the electrode and the polymerization leads to the thickening of the coating. These deviations can be attributed to the fact that electrochemical polymerization involves the formation of oligomers, and therefore Faraday’s law (used in theoretical models) does not exactly predict the process.24,29
The morphology of the PPy-TPPS coatings obtained with different supplied electric charges was examined by SEM micrographs (Figure 2), for this, 0.7 V versus Ag/AgCl was selected as the synthesis potential. It was observed that bare FTO presented grains of different sizes and shapes, which were covered with the polymer as the polymerization took place. When an electric charge of 3 mC/cm2 was supplied (Figure 2(b) and (g)), small nuclei distributed homogeneously are appreciated. At this early stage of the synthesis there is no evidence of overlapping of the nuclei. With a charge of 5 mC/cm2 the polypyrrole nuclei are larger and it can be seen that the nuclei begin to get closer to each other (Figure 2(c) and (h)), but in general the process leads to an ordered coating on the surface of the electrode. Now, with an electrical charge of 10 mC/cm2, some polypyrrole microstructures (microwires) appeared on the surface (Fig. d and l), which presented a disordered arrangement and their location was not distributed uniformly. These microstructures were more evident when an electric charge of 30 mC/cm2 was supplied (Figure 2(e) and (j)). In this case, the polypyrrole nanowires tended to form networks that largely covered the original morphology of the bare substrate. At this point, it is worth mentioning that the formation of polymer microstructures at a certain stage of the polymerization may be related to the observed deviation between the experimental transients and the theoretical models after a t/tm of 1.5 (equivalent to 9.3 mC/cm2 for 0.7 V as synthesis potential). SEM micrographs of bare FTO (a and f) and PPy-TPPS on FTO synthesized at 0.7 V versus Ag/AgCl with (b and g) 3 mC/cm2, (c and h) 5 mC/cm2, (d and i) 10 mC/cm2 and (e and j) 30 mC/cm.2.
The optic characteristics of PPy-TPPS were analyzed using UV-Vis spectroscopy (Figure 3). As the electric charge of synthesis was raised, there was an increase in absorption in practically the entire range of wavelengths that can be explained by an increase in the mass of the synthetized coating on the substrate. In addition, from UV-Vis spectra is possible to corroborate that TPPS was entrapped in the polymer matrix since the Soret band (430 nm) and Q bands (524, 561, 598, 660 nm) of TPPS are recognized.21,30,31 Furthermore, it can be noted that from a very early stage of polymerization (3 mC/cm2) the polypyrrole was doped with TPPS, which is deduced by the presence of the Soret band in the spectrum. Regarding the Q bands, these were observed more easily as the amount of electric charge supplied was larger. As a complement, the absorption spectrum of TPPS porphyrin in aqueous solution is presented in Figure S2. Although all porphyrin signals appear in PPy-TPPS, the signals have a red-shift that is attributed to the interaction between the polymer and the porphyrin.
21
UV-Vis spectra of PPy-TPPS synthetized on FTO substrates under control of the supplied electric charge.
The photoelectrochemical response of the coatings was evaluated by chronoamperometry measurements under a constant applied potential (−0.4 V vs Ag/AgCl). To this end, PPy-TPPS photoelectrodes were synthetized with different electric charges to analyze the effect of the polymer thickness on its performance (Figure 4). The characterizations were carried out using chopped light with lighting and darkness periods of 5 min each. In order to verify the activity of the coatings towards the oxygen reduction reaction, the solutions were bubbled with N2 or with air for 15 min before each experiment. As results, all the photoelectrodes presented negative (cathodic) currents when the solutions were previously bubbled with air, however, the magnitude of the current was very small when the characterization was carried out using a nitrogen atmosphere (maintained over the electrolyte during the measurement). Therefore, the observed cathodic currents can be assigned to the oxygen reduction reaction on the photoelectrodes. On the other hand, it was found that the amount of the coating influences the magnitude of the photocurrent, thus, photocurrents of about 0.006, 0.011, 0.015, 0.018 and 0.017 mA/cm2 were obtained for the coatings synthesized with 10, 20, 40, 80 and 160 mC/cm2, respectively. In this sense, the results showed that for thin coatings, an increase in the thickness of the coating leads to obtaining a larger photocurrent, however, this effect is lost in thick coatings. Thicker films are known to absorb more light and this would be expected to lead to larger photocurrents, but the oxygen reduction reaction is also known to occur at the interface where the semiconductor/oxygen/water meet (the photoelectrode surface). In thicker coatings, the distance the charge carriers must travel to reach the surface and the substrate is greater, which can increase the probability of recombination.
32
Chronoamperometry characterizations of PPy-TPPS under intermittent illumination. 0.1 M Na2SO4 as electrolyte and −0.4 V versus Ag/AgCl as applied potential. Measurement in a deoxygenated solution is marked by the notation “N2 atmosphere”.
The dependence of the amount of polymer deposited on the electric charge of synthesis was observed at the macroscopic level as a result of the light absorption properties of the material (Figure 5(a)). The thickness of the coatings was estimated using SEM microscopy, although it should be noted that due to the granular morphology of the FTO and the formation of polymer microstructures, the thickness is not completely uniform (Figure 5(b)-(f)). Thus, it was found that electric charges of 10, 20, 40, 80 and 160 mC/cm2 lead to approximate thicknesses of 0.06, 0.09, 0.16, 0.24 and 0.38 μm, respectively. These values can be compared with the theoretical thicknesses (calculated assuming flat substrates)
33
: 0.02 μm for 10 mC/cm2, 0.04 μm for 20 mC/cm2, 0.08 μm for 40 mC/cm2, 0.16 μm for 80 mC/cm2 and 0.33 μm for 160 mC/cm2. In general, it is observed that the measured thicknesses are greater than the theoretical ones, which can be attributed to the fact that the theoretical calculation assumes that the dopants are small (such as ClO4-), which leads to more compact coatings. In addition, it should be considered that only local thickness values are obtained by SEM microscopy. Photograph and micrographs of the photoelectrodes. (a) Photograph of the coatings synthesized with different electric charges in mC/cm2. SEM images of cross-sections of coatings synthesized with (b) 10 mC/cm2, (c) 20 mC/cm2, (d) 40 mC/cm2, (e) 80 mC/cm2, (f) 160 mC/cm.2.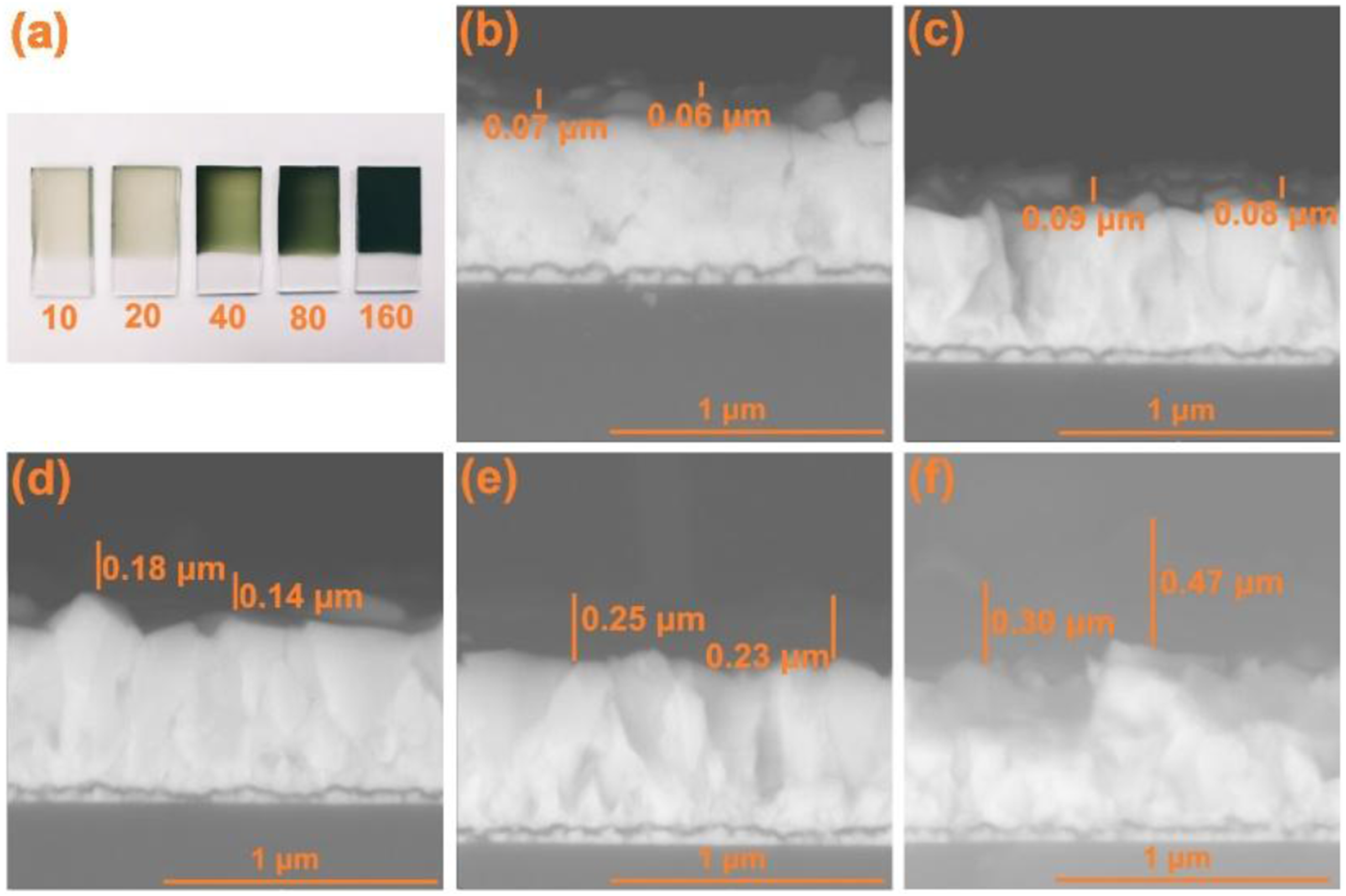
EIS measurements were performed in order to analyze the charge-transfer mechanism at the electrodes. In general, two apparent semicircles were observed in the impedance spectra (Figure 6(a)). The high-frequency semicircle is attributed to the charge transport within the photoelectrode, while the low frequency semicircle is related to reaction kinetics on the photoelectrode surface. It is worth mentioning that for the coatings synthesized at 80 mC/cm2 and 160 mC/cm2 the semicircle corresponding to high frequencies was large enough to overlap with the second semicircle. The experimental data were fitted to an equivalent circuit that has been reported to simulate electrochemical reactions at different electrodes (Figure 6(b)).34–36 In this circuit, Rs is the ohmic resistance of the electrolyte, Rc is the charge transport resistance in the coating, Cc is the coating capacitance, Rct is the charge transfer resistance at the electrode-electrolyte and the Cdl is the double-layer capacitance. The fitted parameters with their corresponding errors are listed in Table 2. (a) Nyquist plots obtained at −0.4 V versus Ag/AgCl under dark conditions. (b) Graphical illustration of PPy photocathodes with the equivalent circuit used for EIS analysis. Results of the fit of the experimental data of PPy photocathodes to the equivalent circuit model shown in Figure 6(a).

It was found that Rc increased as the coating thickness increased, but a strong change in magnitude was evident with the thickness of 80 mC/cm2. This can be related to a partial overoxidation of polypyrrole during the synthesis, due to this process breaks the electronic conjugation of the polymer and decreases the conduction of charge carriers. 37 It has been reported that overoxidation of polypyrrole can occur during its electrochemical synthesis. 38 Furthermore, it is known that this phenomenon can occur under anodic potentials as low as 0.58 V versus SCE and that the longer the time under these potentials, the greater the overoxidation. 39
Concerning Rct, as the electric charge of synthesis increased the value of Rct decreased, except for the highest electric charge (160 mC/cm2). This behavior can be related to the increase in surface area, as a result of the growth of the polymeric microstructures (Figure S3), which favors the oxygen reduction reaction at the electrode-electrolyte interface. However, in the case of the coating synthesized at 160 mC/cm2, the overoxidation of the polymer may have been high enough to affect the electrical conductivity of the material at its surface. With regard to Cdl, it is expected that higher electric charges lead to larger surface areas (due to the formation and growth of microstructures) and consequently be reflected in higher double-layer capacitances. However, our results show that the Cdl magnitude is lower for 80 mC/cm2 and 160 mC/cm2, which can be explained by the loss of conductivity of the polymer due to overoxidation. In view of these results, it can be suggested that among the electric synthesis charges evaluated, the most convenient is that of 40 mC/cm2.
Considering that metallized porphyrins are attractive for various electrocatalytic applications, among these, hydrogen evolution reaction (HER),
40
carbon dioxide reduction,
41
oxygen evolution reaction (OER)
42
and recently for oxygen reduction reaction (ORR),
43
the coatings of PPy-TPPS were subjected to a metallization step with cobalt ions (PPy-CoTPPS) and tested by chronoamperometry under illumination to perform the ORR. For metallization, the PPy-TPPS coatings were immersed for 10 min in a 0.1 M cobalt acetate solution heated to 90°C, following a previously reported work.
44
The resulting electrodes were characterized by UV-Vis (Figure S4), FT-IR (Figure S5) and XPS (Figure 7). XPS characterization of PPy-TPPS and PPy-CoTPPS. (a) PPy-TPPS survey. (b) High resolution of N 1s in PPy-TPPS. (c) High resolution of Co 2p in PPy-TPPS. (d) PPy-CoTPPS survey. (e) High resolution of N 1s in PPy-CoTPPS. (f) High resolution of Co 2p in PPy-CoTPPS.
In the case of UV-Vis, after the metallization step the Q bands of the porphyrin showed a slight overlap, suggesting a partial complexation of the total TPPS molecules trapped in the polymer, given that the TPPS porphyrin complexed with cobalt only presents two absorption bands.21,45 With regard to FT-IR, in the metal-free coating two absorption bands appear in the wavenumbers of 1035 and 963 cm-1, but these bands are not observed after the metallization process, but a new signal is evident in 957 cm-1. For other porphyrins, this metal-dependent band was previously identified as an in-plane porphyrin deformation mode.46,47 Concerning the XPS characterization, the survey of PPy-TPPS (Figure 7(a)) showed the presence of C 1s, N 1s, O 1s and S 2p, while the PPy-CoTPPS survey (Figure 7(d)) showed the presence of cobalt (Co 2p) in addition to all the elements of PPy-TPPS. The high-resolution N 1s can be deconvoluted into four signals: 397.8 eV (attributed to iminic-N), 398.6 eV (attributed to pyridinic-N), 399.6 eV (attributed to pyrrolic-N) and 400.8 eV (attributed to charged nitrogen atoms N+)48–50 In the case of PPy-CoTPPS (Figure 7(e)), the signal of the pyridine nitrogen was clear, occupying 7.6% of the total sum of the deconvolution areas, while in the case of PPy-TPPS (Figure 7(b)) the contribution of pyridine nitrogen signal was 0.0%. This indicates the presence of a Co-N bond in the PPy-CoTPPS electrode and thus confirms metal coordination.49,51 On the other hand, the high-resolution Co 2p is comprised of two main signals (Figure 7(f)): 781.1 eV (assigned to Co 2P3/2) and 796.5 eV (assigned to Co 2P1/2), and three satellite signals at 784.6, 785.6 eV and 801.4 eV. 52 Since the main signal of 781.1 eV can be deconvoluted into two peaks: 778.5 eV (attributed to Co3+) and 781.0 eV (attributed to Co2+), 49 it can be confirmed that cobalt was successfully introduced into the PPy-CoTPPS electrode and that the oxidation state of cobalt in the coating is 2+. These signals were not observed in the PPy-TPPS electrode (Figure 7(c)).
From the photoelectrochemical characterization (Figure 8), it is clear that the presence of cobalt in the coatings influences the total current and photocurrent obtained. Thus, the PPy-CoTPPS photoelectrode presented a photocurrent of 0.030 mA/cm2, double that obtained with the PPy-TPPS photoelectrode. In the case of the total current (current under dark plus current under illumination), PPy-CoTPPS presented 0.062 mA/cm2, which implies an increase of 74% with respect to the total current of the PPy-TPPS. In terms of stability, at 14000 s the total current of PPy-TPPS decreased by 8.7%, for PPy-CoTPPS the decrease was 10.5%, which could be due to a weakening of the adhesion of the polymeric coatings to the substrate. Regarding the quantification of the hydrogen peroxide produced, it was found that in 3 h (under constant illumination) the PPy-TPPS allowed us to obtain 0.45 mg/L of H2O2, while the PPy-CoTPPS allowed us to obtain 0.17 mg/L. This indicates that the presence of cobalt in the polymeric coating does not favor the oxygen reduction via two-electron route equation (1). Nonetheless, chronoamperometric characterization shows that the presence of cobalt catalyzes the oxygen reduction, which likely proceeds via a four-electron transfer route, producing water instead of hydrogen peroxide. We performed EIS measurements to confirm the role of cobalt in the coating (Figure S6) and the experimental data were fitted to the previously used equivalent circuit (Figure 6(b)). In this case, we found that Rct for PPy-CoTPPS was 1305 Ω, lower than that of PPy-TPPS (1665 Ω). Therefore, cobalt is indeed catalyzing the oxygen reduction reaction. Our results are in agreement with other theoretical and experimental studies, where some cobalt porphyrins have been reported to have selectivity for the 4e– ORR pathway.53,54 In the case of Xue S. et al., the authors reported using DFT calculations that some cobalt porphyrins exhibit a higher affinity for the HOO* intermediate, which prevents the formation of H2O2 (Figure 9). This means that for these porphyrins, the overpotential of the 4e- ORR pathway is lower than that of the 2e- pathway.
54
Chronoamperometry characterizations under illumination. 0.1 M Na2SO4 as electrolyte and −0.4 V versus Ag/AgCl as applied potential. Illustration of the oxygen reduction reaction on photoelectrodes of PPy-TPPS and PPy-CoTPPS. * stands for an active site on the electrode.

Conclusions
Photoelectrodes of polypyrrole doped with the anionic porphyrin meso-tetra-(4-sulfonatophenyl)-porphyrin (TPPS) were electrochemically synthesized on FTO and characterized for the oxygen reduction reaction. It was found that the coatings obtained at different synthesis potentials (0.65, 0.70, 0.75 and 0.80 V vs Ag/AgCl) follow an instantaneous nucleation with three-dimensional growth during the initial stage of the polymerization. In addition, it was evident the formation of polypyrrole microstructures (microwires) on the surface of the electrode when the electric charge supplied was larger than 10 mC/cm2. With regard to the photoelectrochemical performance, the results indicate that the current associated with the oxygen reduction reaction depends on the thickness of the coating. Thus, an increase in the thickness leads to obtaining a larger photocurrent attributed to a higher absorption of light, however, it is known that the reaction occurs at the interface where the semiconductor/oxygen/water meet, so after a certain point, no improvement is achieved by increasing the amount of the coating. On the other hand, it was shown that the insertion of cobalt into the photoelectrodes contributes to improving the performance of the coatings for the oxygen reduction reaction, which is reflected in higher currents. However, our results suggest that the mechanism does not occur through a two-electron pathway since in 3 h of the photoelectrochemical process the coating with cobalt produced 62% less H2O2. In view of the above, it could be interesting to explore the combination of polypyrrole-based cathodes with photoanodes that allow achieving larger reduction currents.
Supplemental Material
Supplemental Material - Photoelectrochemical production of H2O2 using polypyrrole doped with meso-tetra-(4-Sulfonatophenyl)-porphyrin
Supplemental Material for Photoelectrochemical production of H2O2 using polypyrrole doped with meso-tetra-(4-Sulfonatophenyl)-porphyrin by Jhon Puerres, Pablo Ortiz, María T. Cortés in Polymers and Polymer Composites.
Footnotes
Acknowledgments
The authors thank the Department of Chemistry and the Department of Chemical Engineering of the Universidad de los Andes for their support. JP acknowledge the support received from the CEIBA Foundation, the Department of Narino (Colombia) and the Universidad de La Salle (Colombia). MTC thanks the Faculty of Science (INV-2025-213-3345). The authors acknowledge the instruments and scientific and technical assistance of the MicroCore Microscopy Core at the Universidad de los Andes. The authors would like to thank the “Laboratorio XPS-UdeA” for the XPS technical service. All authors acknowledge financial support provided by the Vice Presidency of Research & Creation publication fund at the Universidad de los Andes.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Universidad de los Andes (INV-2025-213-3345).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.