Abstract
The main focus of our study is to improve environmentally friendly polymer blends suitable for biomedical applications. A bio-based blend of Polylactic acid (PLA) and Polybutylene adipate co-terephthalate (PBAT) was prepared via melt blending, offering a sustainable, better-mixing and cost-effective approach. However, the inherent phase incompatibility of the blend decreases its mechanical properties. To overcome this, epoxidized linseed oil (ELO) was introduced as a compatibilizer to enhance interfacial adhesion between PLA and PBAT. The blends were systematically characterized using various techniques. Fourier Transform Infrared Spectroscopy (FTIR) and Field Emission Scanning Electron Microscopy (FESEM) confirmed improved interfacial adhesion, while mechanical testing demonstrated significant enhancements in tensile strength and impact resistance, with elongation at break increasing to 56.61%. Differential Scanning Calorimetry (DSC) analysis further revealed an increase in the melting point, indicating enhanced thermal stability. Additionally, biological evaluations, including hemocompatibility and serum stability, were conducted. The in vitro cell proliferation and adhesion tests were performed using L6 cells, and the polymer samples demonstrated excellent biocompatibility. The results suggest that the optimized PLA/PBAT blend exhibits superior hemocompatibility, serum stability, cell adhesion, and proliferation, making it a promising candidate for biomedical applications.
Keywords
Introduction
Environmental pollution, including soil, water, and air contamination, has become a global concern, with the excessive consumption of synthetic plastics being a major contributor. As a sustainable alternative, bio-based plastics offer an eco-friendly solution to mitigate these issues.1,2 Various bio-based plastics are available, such as PLA, PBAT, Polycaprolactone (PCL), Polybutylene Succinate (PBS), and Poly (butylene succinate-co-adipate) (PBSA), among others.2–5 Among these, PLA is utilized mainly due to its cost-effectiveness, ease of processing, and environmental benefits. It finds applications across multiple industries, including packaging, automotive, and industrial manufacturing.6–11 Furthermore, its biocompatibility makes it suitable for various biomedical applications, such as sutures, wound healing, tissue engineering, scaffolds, and medical implants.12–17 Despite its benefits, PLA is limited by its low ductility, limited toughness, and reduced heat distortion temperature, which hinder its wider application. To address these issues, blending PLA with other biopolymers offers a viable solution. Melt blending is considered the most efficient and industrially viable approach for polymer blending.18–21 In this study, PBAT was selected as a blend component due to its superior flexibility, which complements PLA’s brittleness.1–3,9 The primary obstacle in melt blending PLA with PBAT is their poor interfacial compatibility, which results in distinct phase separation due to inadequate adhesion. To enhance compatibility, a third component, known as a compatibilizer, is often introduced. Various oil-based compatibilizers are available, and their selection depends on the intended application. For biomedical applications, it is essential to use bio-based compatibilizers to maintain biocompatibility.22–25 Several studies have explored the use of epoxidized vegetable oils as compatibilizers to improve PLA/PBAT blend properties. Epoxidized soybean oil (ESO), for instance, has been utilized to enhance mechanical properties like tensile strength, elongation at break, and impact resistance.1,23 Similarly, epoxidized canola oil (ECO) has been employed to improve interfacial adhesion, leading to significant increases in mechanical performance. One study reported a 62 and 106% increase in impact strength with the addition of 20 and 30 wt.% PBAT, respectively, however, the sample with 30 wt.% PBAT was identified as the optimized blend for 3D printing applications. Therefore, the addition of ECO to the blend not only enhances interfacial compatibility but also leads to improved mechanical properties and printability. 17 Likewise, researchers have used epoxidized and melanized cottonseed oil as compatibilizers, reporting notable improvements in mechanical and thermal properties, with MCSO exhibiting superior performance. 21 Additionally, studies have investigated the influence of processing parameters on PLA-based 3D-printed models using fused deposition modeling (FDM) with varying infill patterns, densities, and layer thicknesses. The results showed that increasing the infill density significantly improved tensile and compressive strength by up to 60 and 65%, respectively. The optimal combination 80% infill density, a gyroid infill pattern, and a 0.3 mm layer thickness yielded the highest tensile and compressive strengths of 44.03 and 45.23 MPa, respectively. 24 Similarly, another study investigated the mechanical and physical properties of 3D-printed PLA prototypes by varying six key parameters. The findings revealed that increasing the infill density from 20 to 100% enhanced strength from 4 to 56.5 MPa. Additionally, reducing the layer thickness from 0.3 to 0.15 mm significantly diminished dimensional accuracy, reducing deviation from 1.65 to 0.446%. 25
While extensive research has been conducted on enhancing the mechanical strength of PLA/PBAT polymer blends, limited studies have explored their potential for biomedical applications. This study aims to bridge this gap by developing PLA/PBAT blends tailored for biomedical use, including applications in scaffolds, implants, and disposable medical devices. The primary objective is to optimize the blend composition with varying ratios of epoxidized linseed oil (ELO) to enhance interfacial adhesion and mechanical properties. Furthermore, the biological properties of these blends will be evaluated through in vitro assays to assess their suitability for biomedical applications. The novelty of this research lies in utilizing ELO as a compatibilizer, optimizing blend compositions with minor content of ELO, and conducting comprehensive biological evaluations to establish the material’s potential in biomedical applications.
Experimental investigation
Materials required
Biopolymers, including PLA (2003D) and PBAT (PART-F) pellets, were procured from Nature Tech India Pvt. Ltd., Chennai. Linseed oil was sourced from Green Leaf Industries, Surat. Hydrogen peroxide solution was purchased from Thermo Fisher Scientific India Pvt. Ltd., Mumbai and Formic acid from SD Fine Chem Limited, Mumbai.
Preparation of epoxidized linseed oil (ELO)
Approximately 200 g of linseed oil was placed in a clean round-bottom flask (RBF), and formic acid (78.6 g) was added dropwise at room temperature. Subsequently, 173.2 g of aq. hydrogen peroxide (30%) solution was added into the RBF using an addition funnel over a period of 1 h. The reaction proceeded in an oil bath at 60°C for about 6 hours. During the oxidation process, the double bonds between carbon atoms were converted into epoxy groups, resulting in the formation of epoxidized linseed oil (ELO). Once the reaction completed, the crude product was separated via solvent extraction, and the ELO was purified using a rotary evaporator.
Preparation of polymer blend samples
The biopolymers were initially dried in a vacuum oven overnight at 50°C. The PLA (matrix) to PBAT (minor dispersed phase) blending ratio was maintained at 70 wt.% / 30 wt.% across all blend systems, as this ratio provided a good balance between the two polymers with enhanced properties, as reported in the literature.
17
ELO was incorporated into the blend at concentrations of 0.5, 1.0 and 1.5% of the total blend weight. The reaction scheme is shown in Figure 1. Reaction scheme representing the compatibilization process of PLA/PBAT with ELO through chemical interaction.
Melt blending was performed using a rotating twin-screw extruder (TSE) [Make: Deesha Impex, Model No: DI-TSE-10-2020] at a screw speed of 60 rpm. A consistent temperature profile was maintained from hopper to die (190°C–195°C–200°C–205°C–210°C). The extruded blends were water-cooled, pelletized, and vacuum-dried overnight at 60°C to eliminate surface moisture ahead of further processing.
For injection molding (Make: Deesha Impex, Model: IM-D30), the temperature settings were adjusted to 190°C–200°C–210°C–215°C from the hopper to the injection nozzle. The injection pressure of 60 bar was maintained, while the temperature of the mold was controlled at 30 ± 5°C. To ensure proper solidification, a 20 s of cooling time was applied. The molded samples were designated as follows: PLA/PBAT (70/30), PLA/PBAT/ELO (70/30/0.5%), PLA/PBAT/ELO (70/30/1.0%), and PLA/PBAT/ELO (70/30/1.5%). For biological analysis, PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) pellets were processed into films using compression molding (Make: Deesha Impex, Model: IM-D30). The material was pressed at 195°C for 3 min under 20 bar pressure.
Characterization
Fourier transform infrared spectroscopy (FTIR)
The prepared sample’s spectra were obtained using a Shimadzu FTIR instrument. Each blend sample was cast into thin films through compression molding and directly scanned in the FTIR system. The instrument used had a resolution of 1 cm−1. Pristine and ELO samples were coated onto the surface of the KBr pellets before scanning.
X-ray diffraction (XRD)
XRD analysis of the prepared samples was conducted using a RIGAKU tabletop X-ray diffractometer equipped with Cu Kα radiation with a wavelength of 1.5406 Å. The samples were scanned over a 2θ range (5 to 75°) at a speed of 5°/min.
Differential scanning calorimetry (DSC)
DSC measurements of the polymeric samples were performed using a PerkinElmer 8000 DSC instrument. Each sample, weighing approximately 3–5 mg, was analysed in a nitrogen environment. The DSC analysis consisted of three cycles: first heating, first cooling, and second heating. In the first heating cycle, the samples were heated from room temperature to 200°C at a rate of 10°C per minute and held at this temperature for 5 min to remove thermal history and eliminate any residual moisture. During the first cooling cycle, the temperature was reduced from 200°C to −20°C at the same rate of 10°C/min. In the final cycle, the second heating, the temperature was once again increased from −20°C to 200°C at a rate of 10°C/min. The thermogram obtained during the second heating cycle was recorded and analyzed to determine the key thermal properties of the blends, including crystallization temperature, melting temperature, glass transition temperature, and crystallinity.
Contact angle measurement
Contact angle measurements were conducted using an Apex Instruments, to evaluate both the permeability and repulsion characteristics of the blends. Prior to testing, all samples were dried at approximately 50°C to remove moisture on the surface. Once dried, the samples were immediately placed uniformly on the instrument for testing. A 5 µL droplet of distilled water was carefully dispensed onto the surface of each blend. After that, it was left for 15 s, and then the contact angle of the droplet was measured at multiple points on the sample using the integrated software.
Mechanical properties
The mechanical properties, including tensile and impact tests, were performed for the prepared blends. Injection-molded samples with dimensions of 116 × 6 × 3.2 mm were tested in accordance with the ASTM D638 standard. A universal testing machine (Deesha Impex-DTD-UTM) was used to measure the tensile strength of the molded samples at a testing speed of 4 mm/min, equipped with a 1 kN load cell. The tensile modulus was determined from the initial linear segment of the stress-strain curve. The impact strength of V-notched samples, measuring 128 × 13 × 3.3 mm was assessed following the ASTM D6110 standard using an impact tester (Deesha Impex-DIIZ). The weight of pendulum is 0.339 kg, and tests were conducted at a velocity of 3.45 m/s. Both tests were conducted five times, and the average values were calculated for subsequent analysis.
Field emission scanning electron microscopy (FESEM)
The internal microstructure of the prepared blends was captured using a Zeiss FESEM instrument. Prior to analysis, all samples were cryo-fractured by immersing them in liquid nitrogen for approximately 10 min, followed by breaking them into two or more parts. To improve conductivity, the cryo-fractured surfaces were coated with gold. High-resolution images of the internal structure were then captured for detailed study.
Hemolytic assay
The hemolysis assay was performed to assess the effects of the blends on red blood cells (RBC). Fresh blood was collected from a donor and mixed with an EDTA solution to prevent clotting. The sample was then mixed with PBS buffer and centrifuged multiple times to remove leukocytes and plasma, isolating the RBCs. To maintain cell lysis, a dextrose-PBS solution was added as a medium. The PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) blend samples, at different concentrations, were then introduced into 2.5 mL of the RBC suspension and incubated at 37°C for 2, 4, and 12 h. This study was approved by the Institutional Human Ethics Committee of Adichunchanagiri University, and informed consent was obtained from volunteers before blood collection. The percentage of hemolysis was determined using a standard formula from the literature. 16 All experiments were performed in triplicate, and the results are presented as means ± standard errors. Data analysis was carried out using Microsoft Excel.
Serum stability test
The stability of the blend samples in biological fluids was assessed using this test. Blood was drawn via venipuncture by a trained phlebotomist and transferred into procoagulant-coated tubes. The tubes were gently tilted to ensure proper mixing of the procoagulant and then incubated overnight at 15°C. The samples were then centrifuged at 3000 rpm to separate the serum, which was further filtered using a 0.4-micron filter. The polymer samples used for this test were PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) films. Before analysis, the samples were weighed and sterilized by washing with 70% ethanol for 5 min, followed by air-drying in a biosafety cabinet. The sterilized samples were then transferred into sterilized tubes containing 1 mL of serum and incubated at 37°C for different time intervals. The samples were reweighed after incubation periods of 7, 14, and 21 days. This study was performed in triplicate, and the results were recorded.
Cell compatibility assay
The in-vitro cell adhesion and proliferation test were performed using PLA/PBAT/ELO (70/30/1.0%) film with L6 cells. These cells were procured from the National Centre for Cell Sciences, Pune, India, and cultured in DMEM supplemented with 10% FBS, 1 mM L-glutamine, 100 μg/mL penicillin, and 0.1 mg/mL streptomycin. Following trypsinization for detachment, the cells were gathered by centrifugation and reconstituted in full DMEM. A cell suspension (1 × 105 cells/mL) was seeded onto 100 mm culture dishes containing PLA/PBAT/ELO (70/30/1.0%) film and incubated at 37°C with 5% CO2 overnight for attachment. Over the next 48 h, cell adhesion and morphological changes were observed, and images were captured using an inverted brightfield microscope.
Results and discussion
FTIR analysis of linseed oil and epoxidized linseed oil
The obtained FTIR spectrum of LO and ELO is shown in Figure 2. The characteristic peaks for the =C–H and –CH=CH– stretching vibrations of pure linseed oil are observed at 3012 cm−1 and 719 cm−1, respectively. The complete disappearance of the carbon-carbon double bond at 3012 cm−1 confirms the epoxidation reaction. Newly appeared peaks at 956 and 826 cm−1 correspond to the vibrations of methine and oxirane groups and appear simultaneously. This confirms that the epoxidation reaction was successful. The presence of epoxy groups and the appearance of hydroxyl groups (around 3467 cm−1) are attributed to the excess hydroxyl groups generated by the ring-opening of some epoxy groups in the epoxidized linseed oil, as previously reported.
23
FTIR spectral image of linseed oil and epoxidized linseed oil.
FTIR spectra of blend samples
Figure 3 illustrates the FTIR spectra of PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) blends, providing insight into their molecular interactions. The absorption peaks observed at 1753 and 1712 cm−1 correspond to the carbonyl groups present in PLA and PBAT, respectively. Additionally, both polymers exhibit O=C−O stretching vibrations, as evident from the peaks at 868.85 and 941.6 cm−1. The presence of C=C bonds, associated with the aromatic ring structure in PBAT, is indicated by the absorption peak at 1450 cm−1. Furthermore, the peaks detected between 2850 cm−1 and 3001 cm−1 are characteristic of the −CH2− functional group, which is commonly found in PLA, PBAT, and ELO within the blend. Notably, the characteristic epoxy group peaks at 826 and 719 cm−1 are absent in the PLA/PBAT/ELO spectrum, suggesting a chemical reaction between ELO and PLA/PBAT, as observed in Figure 1. This interaction leads to the formation of bonds between ELO and the polymers, effectively acting as a molecular bridge between the two polymers and significantly enhancing interfacial compatibility.
1
FTIR spectral image of PLA/PBAT (70/30), and PLA/PBAT/ELO (70/30/1.0%).
X-ray diffraction study of PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) blends
Figure 4 presents the XRD spectra of PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) blends. The spectra do not exhibit any sharp crystalline peaks. However, broad peaks indicate the semi-crystalline nature of the processed blends. In the uncompatibilized blend, diffraction peaks are observed at 21.7 and 32.4°, corresponding to PLA and PBAT, respectively. The XRD patterns of PLA primarily show diffraction peaks associated with the (110) and (200) lattice planes due to its α-form crystalline structure. For PBAT, the prominent lattice planes correspond to (102) and (100). The intensity of the peaks decreases slightly with the introduction of ELO into the blend system. While PBAT is inherently semi-crystalline, its crystallinity decreases upon the addition of the compatibilizer, as evidenced by the reduced peak intensity, which indicates disruptions in its molecular arrangement. The compatibilizer enhances interfacial adhesion and improves compatibility between PLA and PBAT, leading to better dispersion while simultaneously hindering the self-organization of PBAT chains. Furthermore, the presence of PLA and the compatibilizer restricts the mobility of PBAT polymer chains, preventing effective crystallization and resulting in a semi crystalline structure.
1
X-ray diffraction pattern of PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%).
DSC analysis of PLA, PLA/PBAT (70/30) and PLA/PBAT/ELO’s samples
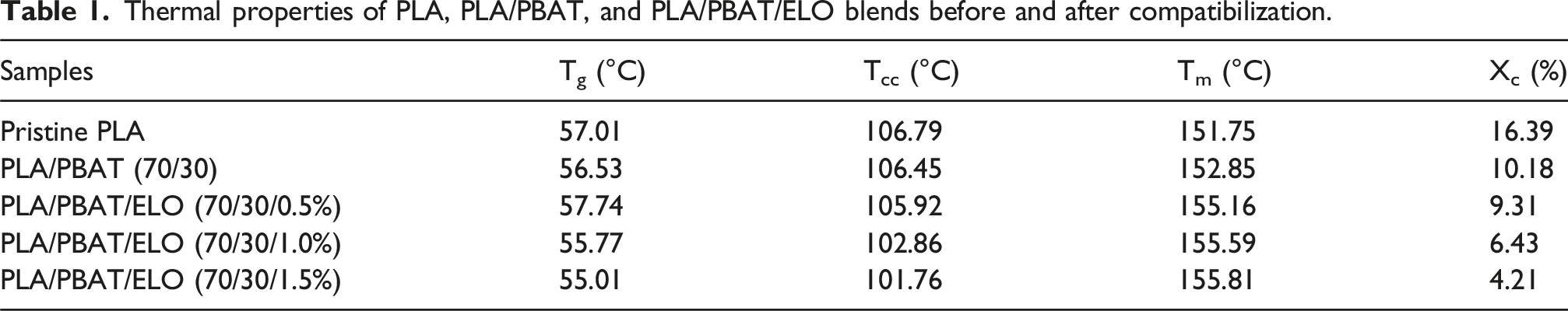
The DSC thermograms of the prepared PLA, PLA/PBAT, and PLA/PBAT/ELO’s blends are presented in Figure 5. This analysis provides key insights into the thermal properties of the samples, including melting temperature (Tm), cold crystallization temperature (Tcc), and glass transition temperature (Tg), which are presented in Table 1. For pure PLA, Tm, Tcc, and Tg, are observed at, 151.74°C, 106.73°C, and 57.79°C, respectively. In the uncompatibilized PLA/PBAT (70/30) blend, these values shift slightly, with Tg at 56.55°C, Tcc at 106.42°C and Tm at 152.67°C. Upon incorporating ELO in varying proportions (0.5%, 1%, and 1.5%), the Tg of the compatibilized samples decreases from 57.74°C to 55.01°C, indicating enhanced compatibility between PLA and PBAT with increasing ELO content. The Tcc of neat PLA is recorded at 106.73°C, with a crystallinity (Xc) of 16.4%. In the PLA/PBAT (70/30) blend, Tcc slightly decreases to 106.42°C, while crystallinity reduces to 10.2%. The addition of ELO further lowers both Tcc and crystallinity, as evidenced by XRD analysis. Meanwhile, the melting temperature of the samples ranges between 151.16°C and 155.81°C with the introduction of the compatibilizer. Additionally, the presence of a double peak in the melting endotherm suggests processes such as recrystallization, re-melting of crystals, and partial melting within the blend.
14
DSC thermogram image of (a) Neat PLA, (b) PLA/PBAT (70/30), (c) PLA/PBAT/ELO (70/30/0.5%), (d) PLA/PBAT/ELO (70/30/1.0%), and (e) PLA/PBAT/ELO (70/30/1.5%). Thermal properties of PLA, PLA/PBAT, and PLA/PBAT/ELO blends before and after compatibilization.
Mechanical properties of polymer blend samples
Mechanical properties of PLA, PLA/PBAT, and PLA/PBAT/ELO samples.

The mechanical properties of the PLA/PBAT blend with epoxidized linseed oil are compared with those from existing studies on epoxidized soybean oil, canola oil, and cottonseed oil in the table below.
Comparison of the mechanical properties of PLA/PBAT/ELO (70/30/1.0%) blends with previously reported studies.

Contact angle measurement of the blend samples
The water contact angle of the blend samples was analysed and the results are presented in Figure 6. In general, materials with a contact angle below 90° are classified as hydrophilic, while those with a contact angle above 90° are considered hydrophobic. The average contact angles measured for PLA, PLA/PBAT (70/30), PLA/PBAT/ELO (70/30/0.5%), PLA/PBAT/ELO (70/30/1.0%), and PLA/PBAT/ELO (70/30/1.5%) were 67.58°, 69.01°, 68.94°, 68.26°, and 67.99°, respectively. The results shows that the water contact angle decreases as the compatibilizer content increases, suggesting an increase in hydrophilicity with higher ELO content. Although epoxidized linseed oil (ELO) is inherently hydrophobic and typically repels water, a reduction in hydrophobicity was observed in our case, suggesting that the epoxidized oil effectively functions as a compatibilizer in the PLA/PBAT/ELO blend. Surfaces with high hydrophilicity support better cell attachment, growth, and development, making them valuable for biomedical scaffolds and tissue engineering. Their ability to attract water enhances protein adsorption, which plays a key role in cell interactions and tissue regeneration. Improved wettability also boosts compatibility with cells, making these materials more suitable for scaffolds, implants and biomedical devices.
15
Contact angle images of water on the surface of (a) Neat PLA, (b) PLA/PBAT (70/30), (c) PLA/PBAT/ELO (70/30/0.5%), (d) PLA/PBAT/ELO (70/30/1.0%), and (e) PLA/PBAT/ELO (70/30/1.5%).
FESEM analysis of the blend samples
Figure 7 presents the FESEM images of the blends, both with and without compatibilization, highlighting the interfacial compatibility between PLA and PBAT. These images offer valuable insight into the internal microstructural arrangement of the blends. In the uncompatibilized blend (Figure 7(a)), we observe droplet structures where the dispersed PBAT phase appears within the continuous PLA matrix. This indicates limited compatibility between PLA and PBAT. However, with the addition of the compatibilizer (ELO) at varying weight ratios (0.5%, 1.0%, and 1.5%), the PBAT droplets progressively decrease in size as the ELO content increases (Figure 7(b)–(d)). This reduction in droplet size signifies improved interfacial adhesion and enhanced compatibility between the two phases. Among the tested formulations, the PLA/PBAT/ELO (70/30/1.0%) blend demonstrates the most effective compatibilization between PLA and PBAT, as evidenced by the uniform distribution and reduced phase separation. Based on these observations, PLA/PBAT/ELO (70/30/1.0%) is considered the optimal formulation for further analysis.
19
FESEM images captured on the fractured surface of (a) PLA/PBAT (70/30), (b) PLA/PBAT/ELO (70/30/0.5%), (c) PLA/PBAT/ELO (70/30/1.0%), and (d) PLA/PBAT/ELO (70/30/1.5%).
Evaluation of hemolysis in polymer blends
Hemolysis, the phenomenon of RBC breakage resulting in the release of hemoglobin into the surrounding medium, is a key test for assessing the hemocompatibility of materials. In this study, both PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) samples were evaluated, and the extent of hemolysis was determined by measuring the optical density (OD) at 540 nm after incubation periods of 2, 4, and 12 h.
16
As illustrated in Figure 8(a) and (b), the hemolysis percentage observed in our test samples remained minimal, indicating that the materials do not exert a toxic or damaging effect on RBCs. This suggests that our samples do not adversely affect blood cells. The blend samples may possess surface properties that prevent plasma protein absorption and, consequently, do not activate the complement system, which could otherwise trigger hemolysis. The results of this study strongly suggest that PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) exhibit excellent hemocompatibility, making them suitable for various biomedical applications involving direct blood contact, such as medical devices and scaffolds. Hemolysis study of (a) PLA/PBAT (70/30) and (b) PLA/PBAT/ELO (70/30/1.0%).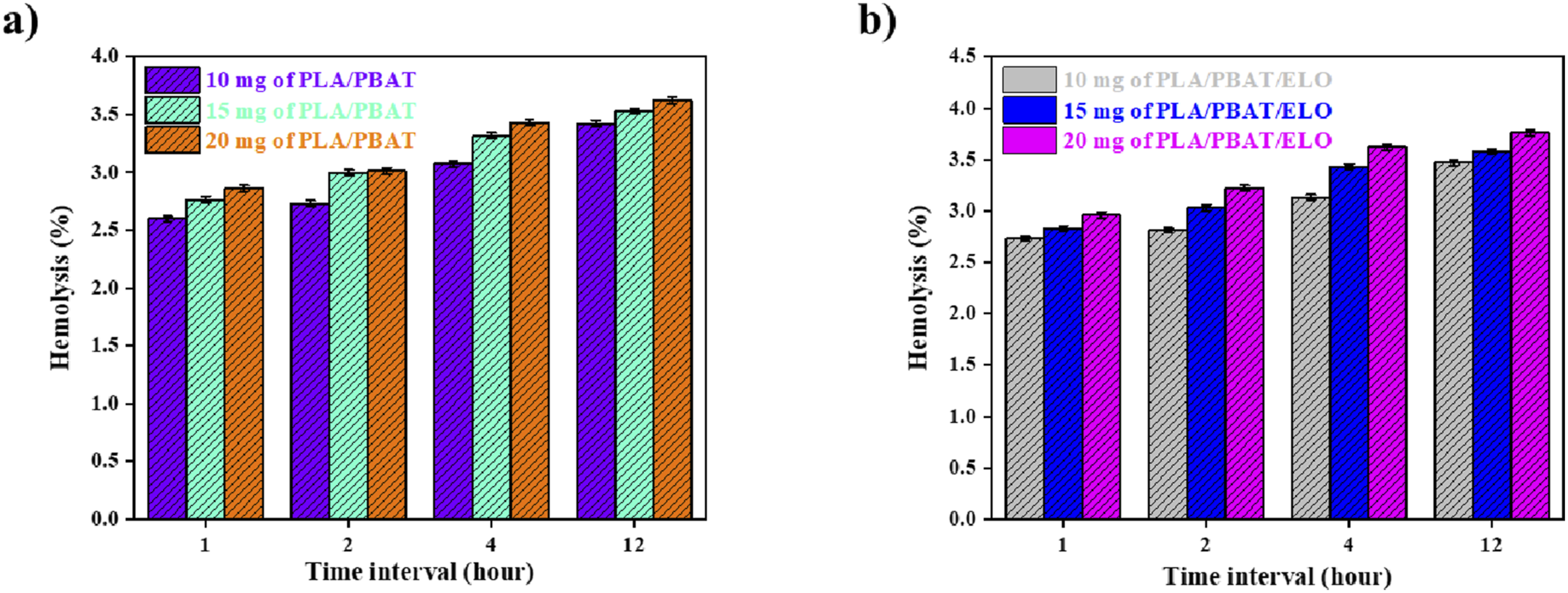
Stability of polymer blend samples in serum
The serum stability test is a crucial method for assessing the durability and performance of biomaterials in biological fluids. Serum, the fluid portion of blood after clotting factors have been removed, contains enzymes and proteins that mimic the in vivo environment and may influence the stability of biomaterials over time. This study is particularly important in biomedical research to ensure the reliability of materials intended for medical applications. In this study, PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) samples were immersed in serum for varying periods. As shown in Figure 9, no significant changes in blend weight were observed, with only slight variations detected. The results indicate that our samples remain stable, do not dissolve or disintegrate in tissue fluids, and exhibit resistance to plasma hydrolytic enzymes. These findings confirm the blend’s stability and suitability for biomedical applications. Serum stability results of (a) PLA/PBAT (70/30) and (b) PLA/PBAT/ELO (70/30/1.0%).
Assessment of cell adhesion, proliferation, and compatibility
The cell adhesion and proliferation tests are commonly used in biomedical research to assess the suitability of materials for biological applications, including implants, scaffolds and drug delivery systems. This test evaluates whether a material supports cell attachment, growth, and multiplication, which are essential for tissue engineering applications. For this test, we cultured L6 cells with the PLA/PBAT/ELO (70/30/1.0%) film sample and incubated overnight. Observations from the study, as represented in Figure 10, reveal that the cells adhered to the sample surface and continued to propagate over prolonged incubation periods, indicating the blend’s biocompatibility and support for cellular growth. The rapid attachment of L6 cells to the blend’s surface suggests that its surface properties are favourable for cellular adhesion, a critical factor for effective tissue integration and material interaction. Additionally, the proliferation of cells on the blend’s surface demonstrates that it provides a conducive microenvironment for cell viability and growth. Furthermore, the cell compatibility results, along with the hemolysis assay and serum stability findings, support the potential use of the blend in applications such as scaffolds, implants, and medical devices. (a), (b), and (c) show microscopic images of L6 cell growth on the blend sample surface, with a scale bar of 200 μm.
Conclusion
In this study, bio-based PLA/PBAT blends were developed through melt blending at a 70/30 wt.% ratio to enhance the ductility of PLA. However, the compatibility between the two polymer phases was poor. To improve interfacial adhesion, epoxidized linseed oil (ELO) was introduced as a compatibilizer in three different concentrations: 0.5%, 1.0%, and 1.5% to the total weight ratio of the blend. FTIR and FESEM analyses confirmed that ELO effectively enhanced compatibility between the PLA and PBAT phases. The FESEM images revealed a reduction in droplet size, indicating improved interfacial adhesion. While the blends were not fully miscible, both analyses suggested they exhibited good compatibility. Mechanical testing, including tensile and notched impact strength tests, demonstrated that the addition of ELO enhanced the mechanical strength of the blends. The formulation containing 1% ELO exhibited optimal elongation up to 56.61% and impact strength 6.46 kJ/m2 without compromising tensile strength, making it the most promising composition for further studies. These blends hold potential for applications in industrial packaging, automotive components, and other sectors. However, the primary focus of this study is to develop these materials for biomedical use. To evaluate biocompatibility, biological tests were conducted on the PLA/PBAT (70/30) and PLA/PBAT/ELO (70/30/1.0%) samples. A hemolysis assay using red blood cells (RBCs) confirmed that the blend did not induce RBC lysis, indicating hemocompatibility of samples. Additionally, serum stability tests were performed by incubating the polymer samples in serum for 21 days. Minimal weight variation was observed, confirming the samples stability in biological fluids. Furthermore, in vitro cell proliferation and adhesion studies were carried out using L6 cells. The results demonstrated excellent cell adhesion and proliferation, reinforcing the blend’s suitability for biomedical applications.
Footnotes
Acknowledgements
The authors express their sincere gratitude to the Center for Research and Innovation (CRI), Adichunchanagiri University, for providing the research facilities. They also thank Mr Byresh Gowda B. K. for his support in conducting the biological tests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Adichunchanagiri University (001/2023).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
