Abstract
Nanotechnology use in advanced wound dressings is massively increasing in the prevention and treatment of bacterial infections with anti-inflammatory effects. This research aimed to prepare nanofibers of fluticasone/cefadroxil loaded in chitosan/polyvinyl alcohol (PVA) (FCCPNFs) by electrospinning. FTIR, XRD, SEM and DSC were performed to analyze the successful drug loading, crystalline and amorphous nature, structural properties and thermal integrity of the nanofibers respectively. In pharmaceutical analysis zeta size and zeta potential, swelling behaviour, antibacterial activity and potential of wound healing were ascertained. FTIR spectra revealed the compatibility between polymers and loaded drugs. XRD exhibited the semi-crystalline nature of cefadroxil monohydrate and the crystalline nature of chitosan, PVA, fluticasone, and CFM. SEM analysis revealed the continuous fibrous network of FCCPNFs. DSC study confirmed the thermal stability of FCCPNFs. The zeta size and zeta potential of FCCPNFs were <300 nm and 21.2 ± 5.37 mV respectively. The swelling study exhibited the exudate absorption capacity of FCCPNFs. A drug release study showed the burst release of the drugs. FCCPNFs exhibited antibacterial activity against Staphylococcus aureus. In vivo and histopathological studies substantiated the rapid wound healing by FCCPNFs compared to negative control (untreated) and positive control (Quench). The chemical and pharmaceutical analysis confirmed the successful synthesis of stable FCCPNFs with good antibacterial potential against Gram-positive bacteria and propitious wound-healing.
Introduction
Nanostructures have mounted copious options to formulate advanced transdermal drug delivery systems to overwhelm the obstacles to using these systems. Immense loading capacity and ease of utilization of natural and synthetic-based polymeric nanofibers presented a distinctive structure for drug delivery applications. 1 The low toxicity, biocompatibility and good biodegradability of natural polymeric nanofibers have gained importance from researchers by utilizing their physical, biological and chemical features over synthetic nanofibers. The Electrospinning technique has gained importance due to its multifaceted and easy use in forming composite films, blended and net fibers. This system is comprised of a syringe pump, voltage supplier and collector. There are various varieties of spinnerets and collectors in electrospinning machines. When there is a less amount of solute and solvent then tube-less spinneret will be used over clip spinneret that can hasten the long-term processes. Electrospun nanofibers have provided high drug loading capacity and 100% encapsulation efficiency in transdermal drug delivery systems. 2
Skin serves as a primary defense in all creatures and is susceptible to injury due to continuous exposure to the external environment. 3 Burn is the most serious and painful type of injury and is at the fourth number after falls, ferocity and road accidents. 4 Skin burns are frequently escorted by infection, edema, slow regeneration of tissue and extreme scar formation resulting in delayed wound healing. 5 The severity of the skin burn depends on the depth of the wounds categorized as partial-thickness, superficial and full-thickness wounds and there is a slow healing process in the latter two categories. 5 Necrotic tissue in burn wounds may activate reactive oxygen species and produce excessive inflammation by boosting regional inflammatory cells. Moreover, the burn scar’s necrotic tissue is a foremost environment for bacterial reproduction and colonization resulting in delayed recovery. 6
Wound healing is a sequenced process consisting of inflammation, proliferation, matrix synthesis, deposition of collagen and re-modeling to protect from infection. 7 Tissue remodeling and retaining of inflammatory cells are the two major overlapping phases in the healing process. 8 There is an acute need to formulate a multi-purpose wound dressing that not only prevents and treats infected burn wounds but also suppresses the production of reactive oxygen species. The development of advanced wound dressings is necessary due to the drawbacks of existing options, that is cotton-based dressings and allografts, which often fail to contribute enough moisture balance, antimicrobial potential, and sustained drug release. 9 Electrospun nanofibers have attracted attention to transdermal drug delivery because of their excellent porosity, adjustable mechanical features, and the potential to load drugs effectively. 10 Chitosan-based nanofibers, in particular, have demonstrated excellent biocompatibility, biodegradability, and inherent antimicrobial properties, making them ideal candidates for wound healing applications. 11 The integration of antibiotics and anti-inflammatory drugs, such as cefadroxil and fluticasone, within polymeric nanofibers enhances their antibacterial efficiency and accelerates tissue regeneration, addressing critical challenges in burn wound management. 12 Given the increasing prevalence of antibiotic-resistant bacterial infections, there is a pressing need for innovative wound care strategies that combine biocompatible polymers with potent antimicrobial and anti-inflammatory agents to promote rapid healing.
Fluticasone Propionate (FTP) a class III synthetic fluorinated corticosteroid is used to treat cutaneous wounds owing to its good patient acquiescence and anti-inflammatory properties. 13 FTP was used in in vivo experiments in 2003 and 2017 in double-blind randomized groups with marvellous results in skin treatment. 14 FTP is available in marketed cream and ointment products with a concentration ranging from 0.005 to 0.05%. 15 Chitosan formed by deacetylation of chitin, is a non-toxic, biodegradable and biocompatible linear polysaccharide. Chitosan possesses unique features that make it a suitable candidate for a wide range of applications including drug delivery, tissue engineering and antimicrobial protection. The unique properties of chitosan, combined with the advanced drug delivery capabilities of biopolymer conjugates, make these systems a promising platform for the development of innovative and effective pharmaceutical formulations. 15
Cefadroxil is effective against gram positive cocci including staphylococcus aureus, anaerobic Streptococci and streptococcus pneumonia and gram negative rods Escherichia coli and Klebsiella pneumonia and is widely used in topical preparations for wound healing. 16 Polyvinyl alcohol (PVA) is a crystalline, biocompatible and biodegradable semi-synthetic polymer and can be merged with natural polymers to fabricate nanocomposite fibers. 17 PVA has unique features of non-carcinogenicity and effortless workability. 18
Montmorillonite is a yellowish-white colored nano clay used as a nanofiller to improve the mechanical properties of nanocomposite fibers. 19 Previous researchers formulated MMT based CG/PU/PAN/Ne nanofibers by electrospinning technique. These nanofibers showed high porosity and fast wound healing owing to a quick release of the drug in 2-3 h. 20 Nanofibers of PVA/MMT containing root extracts of Berberis lycium exhibited pronounced antibacterial activity against Staphylococcus aureus than Pseudomonas aeruginosa and promoted efficient wound healing. 10 Zn/Ag nanoparticles were incorporated in chitosan nanofibers, resulting in significant wound healing when the wound was scratched after 24 h of treatment. 21 A previous study described the use of Bromelain as a debriding agent for the preparation of chitosan nanofibers containing bromelain 2% and 4%. Chitosan 2% w/v bromelain showed lesser toxicity and was more coherent in healing burned skin than 4% w/v bromelain. 11 The zone of inhibition of CFX-loaded CPNFs, free CFX, and blank CPNFs was 15.11 ± 1.67 mm, 4.01 ± 1.15 mm and 1.4 ± 0.97 mm respectively, against S. aureus. 22 Chit/PEO/SiO2/Cip nanofibers were formulated and showed the capacity of these to release drug until the 13th day and biocompatibility for 72 h with efficient antimicrobial activity against both E. coli and S. aureus. 23
This pioneered study aimed to develop cost-effective, biodegradable and efficient antibacterial drug delivery system of FCCPNFs for effectual wound healing.
Materials and methods
Materials
All materials utilized in this study were of analytical grade and were used without further purification. Chitosan (medium molecular weight, 85% degree of deacetylation), Montmorillonite (MMT, nanoclay, purity ≥ 98%) and glycerol (GC) were acquired from Sigma-Aldrich (International Laboratory, USA). Polyvinyl alcohol (PVA, Mw = 89,000–98,000, 99% hydrolyzed) was purchased from Merck Chemicals (Germany), Cefadroxil Monohydrate (CFM, purity ≥ 99%) by Obsons Pharmaceutical, Lahore, Pakistan, Fluticasone Propionate (FTP, pharmaceutical grade, purity >98%) from Saffron Pharmaceuticals (Pvt) Ltd, Islamabad, Pakistan and Glacial Acetic Acid (≥99.7% purity) by Daejung laboratory chemicals Co., Ltd (Korea).
Staphylococcus aureus (SA) a clinically isolate strain, were collected from the burn unit of Jinnah Hospital (Lahore).
Solution preparation
Chitosan solution (2%) was made by dissolving 1.7 g of chitosan in distilled water (12 ml) having few drops of dilute glacial acetic acid solution (1% v/v) under constant stirring at a temperature of approximately 50 ± 2°C for about 2 h. On the other hand, 1.7 g of PVA was dissolved in distilled water (12 ml) to prepare PVA solution (w/w) by stirring at 340 rpm at 50°C for about 12 h. Chitosan solution was combined with PVA solution with continuous stirring for about 5 h at 80 ± 2°C. Both solutions were employed in equal volumes to maintain 1:1 ratio between the quantities of polymers.
Composition of different formulations due to varied quantities of CFM and FTP.

Development of nanofibers
The polymer solution was primed in a 5-mL syringe having an 11.99 diameter fastened on the equipment. The solution was pumped at a controlled rate of 400 µL/h through a nozzle set to a high voltage of 14–15 kV. The distance of 14 cm between the needle and the aluminium collector was maintained throughout the procedure. The temperature and relative humidity were of 20°C–25°C and 35–40% respectively. The fabricated fibers were collected on aluminium foil enfolded in the static collector plate and allowed to air dry.
Chemical characterizations
Scanning electron microscope
The fabricated electrospun fibers were investigated for morphological attributes using (SEM, JSM 5500, JEOL, Japan). Pieces of 2 cm2 of aluminium foil holding electrospun nanofibers were sputter-coated with a thin layer carbon for 60 s using sputter coater (JSM 5500, JEOL, Japan) to improve conductivity and imaging quality. The coated samples were then examined under the SEM at an accelerating voltage of 4 kV. Image J software was used to analyze the 3D structure of the nanofibers. 14
Fourier transform infrared spectroscopy
The Fourier transform infrared spectra of nanofibers were acquired by an FT/IR-6600 type A instrument operated at a wavenumber range of 4000–500 cm−1 to analyze the existence of different functional groups, intermolecular interaction, compatibility between drug-polymer excipient and polymer excipient. 5
X-ray diffraction
X-ray diffraction analysis was performed to ascertain the crystalline and amorphous nature of the ingredients. X-ray diffractometer system X-600 was adjusted at 2θ mode with an x-ray source of copper K-alpha radiation wavelength of 1.540 Å. The analysis was performed on all FCCPNFs and pure drugs within a 2θ range of 10° to 60°, employing a step size of 0.013° and an acquisition time of 30 s per step with an applied current of 40 mA and voltage of 45 kV. 20
Differential scanning calorimetric (DSC) analysis
The thermal stability of the synthesized FCCPNFs was examined by using a DSC-modules Q5000 series thermal analyzer (TA Instruments, West Sussex, UK). The 10 ml/min flow rate of nitrogen was kept constant with every rise in temperature of 10°C/min up to 500°C after each precise weight. 24
Particle size analysis
A Zeta sizer was used to measure the particle size of the optimized nanofibers. NF suspension was made by pure and filtered water added into the cuvette cell. Data acquisition was initiated by inserting the cuvette cell into the instrument. The analyzer calculated the particle size based on well-established theories followed by measuring the Brownian motion of the particles using Dynamic Light Scattering (DLS). 25
Pharmaceutical evaluations
Swelling ratio
Percentage swelling describes the dual parameters of the nanofibers with drug release being the former and exudate management capacity being the latter. The swelling ratio of the FCCPNFs was evaluated by dipping them in distilled water and weighed at foreordained periods of 10, 20, 30, 60 min, 1.5, 2, 3, 4, 6, 8, 12, 16, 20 and 24 h. The experiment was performed in triplicates. The formula used to calculate the swelling ratio is stated below.
26
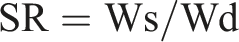
Drug release study
The release pattern of CFM and FTP from FCCPNFs was determined by immersing them in wide-mouthed glass containers containing 250 ml of distilled water. The bottles were kept under constant shaking using a shaking incubator for at least 5 h at 37°C. At predetermined time intervals (i.e., 0, 5, 15, 30, 60, 90, 120, 150, 180, 240 and 300 min) approximately 2 mL aliquot was removed from each container. To maintain sink conditions, a 2 mL aliquot was removed from each container at predetermined intervals and replaced with an equal volume of fresh distilled water. This ensured that the drug concentration remained below saturation, allowing continuous drug release. The collected aliquots were analyzed by spectrophotometer at 246 nm for FTP and 264 nm for CFM. 27
Antibacterial assay
The antibacterial efficiency of formed FCCPNFs was assessed by the disc diffusion method against clinically isolated strains of Staphylococcus aureus. The nutrient agar media was prepared by dissolving 0.0896 g of agar in 40 mL of distilled water. Nutrient agar media, Petri plates and other essential glassware were sterilized at 121°C for 2.5 h in an autoclave. After sterilization, the media was poured into Petri dishes and allowed to solidify. A loop full of S. aureus was evenly spread on nutrient agar media and incubated for 24 h at 37°C. After 24 h each film with 10 mm size was placed on inoculated media and further incubated for 48 h at 37°C. The zone of inhibition was measured in millimeters to check the activity of FCCPNFs against bacterial strains. Results were reported as mean ± standard deviation from three independent measurements. 8
Wound healing study
Protocols for wound healing study on rabbits were approved by the Research Ethics Committee of the Institute of Pharmaceutical Sciences, University of Veterinary and Animal Sciences Lahore. 24 healthy male rabbits of 2–3 kg halved in four groups were used to assess the effectiveness of the FCCPNFs against commercially available Quench cream. At first, the dorsal region of the rabbits was hairless and cleaned with ethanol. Two full-thickness incisions of approximately 0.5 cm in length and 1.6 cm2 in area were made on each rabbit. • Group 1 (control): untreated rabbits. • Group 2: rabbits treated with drug-loaded FCCPNFs. • Group 3: rabbits treated with the commercially available formulation Quench®. • Group 4: rabbits treated with drug-free nanofibers.
The wound size reduction was observed throughout 0, 7, 14 and 21 days, with results represented as the percentage of wound area (cm2). The following mathematical formula will be used to measure the wound area.

Histopathological analysis
On the 21st day of the wound healing study, all the rabbits were sacrificed and wound samples were collected for histopathological analysis. Three skin tissue samples were taken from the wound area of each rabbit, washed thoroughly, immersed in 10% formalin solution and fixed in paraffin. The samples were processed using hematoxylin and eosin (H&E) staining. 29 The stained slides were then analyzed with a high-accuracy digital microscope to assess the signs of wound repair, such as epidermal regeneration, neovascularization, collagen reformation, and fibrosis.
Statistical analysis
Statistical analysis of the results obtained from different experiments was conducted using one-way ANOVA through GraphPad Prism software on the basis of p < 0.05.
Results and discussion
Chemical characterization of nanofibers
Scanning electron microscope (SEM)
Figure 1(a) illustrates the optical microscopic image of FCCPNFs exhibiting a continuous fibrous network. Figure 1(b) exhibited the SEM micrograph which showed the morphology, structure and interconnected network of the nanofibers. The provided images represent a PVA/Chitosan nanofiber film. The macroscopic view (a) shows a thin, flexible, and slightly wrinkled film, indicating successful electrospinning and collection. The SEM image (b) reveals a highly porous, fibrous, and interconnected structure with randomly oriented nanofibers. The rough surface and dark regions suggest pore formation, while lighter areas indicate denser fiber accumulation. The film’s morphology enhances mechanical properties and fluid absorption, making it suitable for wound healing, drug delivery, or tissue engineering applications. The porous structure and biocompatibility of the PVA/Chitosan nanofiber film contribute to its potential in biomedical applications. Rathinavel et al. prepared the PCL/Curcumin nanofiber incorporated in mesoporous silica. The SEM analysis of these nanofibers exhibited the smooth, dense and wheat-like morphology of nanofibers with an average particle size of 32.17 nm.
3
CURCSNPs made by Fahimirad et al. exhibited the uniform distribution of the drugs in nanofibers.
30
Morphological evaluation of the fabricated nanofibers. (a): The optical microscopic image of FCCPNFs exhibiting a continuous fibrous network. (b): SEM micrograph which showed the morphology, structure and interconnected network of the nanofibers.
Fourier transforms infrared spectroscopy (FTIR)
FTIR spectroscopy illustrates the compatibility and functional groups of various ingredients in FCCPNFs. Figure 2 exhibits the FTIR spectra of FTP and CFM and their nanofibers. FTIR spectrum of fluticason revealed a sharp peak at near 1750 cm-1 indicating C=O (carbonyl group). C-F (fluorine groups) exhibited peaks between 1400 and 1000 cm-1, while C-H and O-H exhibited their peaks at around 3000 cm-1 and 3500 cm-1, respectively. FTIR spectrum of CFM is also presented in Figure 2. Hydroxyl group (O-H) exhibited a broad peak around 3500 cm-1. Peaks in a range of 3300–3500 cm-1 indicated the presence of N–H group. Carbonyl group (C=O), amine group (C-N), and carboxyl group (C-O) groups showed their characteristic peaks at around 1750–1650 cm-1, 1360–1250 cm-1, and 1300–1000 cm-1, respectively. FTIR spectra of FTP and CFM and their nanofibers.
XRD spectroscopy
XRD of chitosan is shown in Figure 3 which parades a sharp and distinguished peak at 19.98°. Microneedle patches of chitosan containing levosulpride formed by Habib et al. using solvent casting method. XRD of these patches has reported similar kind of results as seen in our study.
31
The X-ray diffractogram of PVA shown in Figure 3 showed two distinct and intense peaks at 2θ values of 19.72° and 40.22°, indicating the crystalline nature of the PVA. Wasef et al. prepared CS/PVA/ZnO nanocomposite films. The diffraction peak of PVA in these films at 2θ values is 19.6° exhibiting identical behaviour of PVA as in our fibers.
7
X-ray diffractogram of CFM shows the sharp, distinct peaks correspond to the specific atomic arrangements and lattice structures at various diffraction angles (2θ) of 10.5°, 14°, 20.1° and 22.9°. The high-intensity peak of CFM exhibited the semi-crystalline phase in nanofibers. Marand et al. formulated nickel oxide nanoparticles (NiONPs) fused with CFM. The XRD of pure CFM depicted peaks at 2θ of 10.1°, 15.5°, 19.7° and 22.9° that complied with our study.
28
The XRD pattern for FTP demonstrated a sharp peak at 9.1, 14, 15.5, 16.5, 20.9 and 26.3 at 2θ angle that represented the specific atomic arrangements and lattice structures within the crystalline FTP. Mehmood et al. formulated Mesoporous silica nanoparticles (MSN) fused with FTP for nasal drug delivery. They mentioned the results of XRD of the FTP at 2θ that matched with the values of our diffractogram of FTP.
26
Figure 3 also depicts the X-ray diffraction (XRD) pattern of drug-loaded nanofibers. The sharp peaks indicate the presence of crystalline phases within the nanofibers sample. PXRD diffractogram of chitosan, PVA, FTP and CFM and their nanofibers.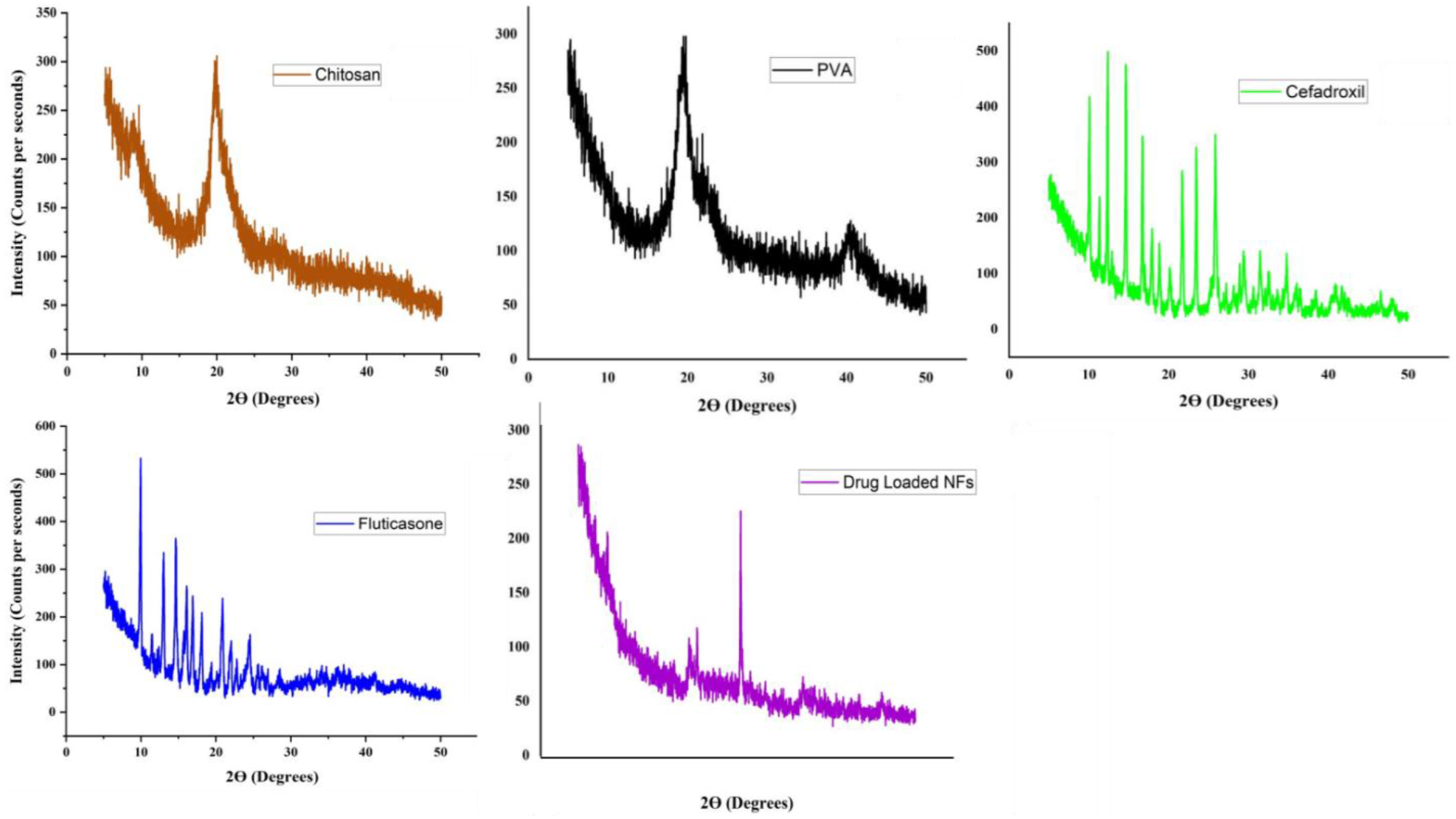
Thermogravimetric analysis
Figure 4(a) shows the thermogram of chitosan that displayed an endothermic peak around 231.22°C. Mukherjee et al., have developed thiolated chitosan-based mucoadhesive films by using a solvent evaporation method for the therapeutic delivery of methylprednisolone in osteoporosis-induced rats. They carried a thermal analysis to verify the thermal behaviour of chitosan that depicted the prominent peak at 220.16°C.
32
Figure 4(b) elicits the thermogram of PVA that initially showed an endothermic peak starting around 192°C due to moisture loss, phase transition at 210°C and final degradation at above 400°C indicating the complete combustion of the polymeric structure. Alhazime et al. carried out DSC analysis of PVA–PEG/Co3O4 NCP that decompose at 115°C owing to dehydration in the first stage, followed by removal of the hydroxyl group at 115°C to 250°C and ultimate backbone decomposition at 365°C.
33
The thermogram of CFM depicted in Figure 4(c) showed a definite and sharp peak at 230°C analogous to the melting point of CFM when it changes to its liquid state. The absence of other peaks was evidence of its purity or the presence of a crystalline phase. Zia et al. formulated CMC-PVA-FHNTs nanocomposite hydrogel as a carrier of CFM that illustrated decomposition of CFM at 263°C showing thermal stability of nanocomposite hydrogel.
24
Figure 4(d) displayed the DSC thermogram of FTP. It covers a wide range of temperatures from 150°C to 300°C parades the broad and shallow endothermic curve. This thermal behaviour is a hallmark of the amorphous nature of FTP owing to not having a well-defined melting point. Thermogram for the drug-loaded nanofibers composed of FTP, CFM, Chitosan, and PVA presented in Figure 4(e). The DSC plot exhibits a complex thermal behaviour, with multiple endothermic and exothermic events observed across the temperature range. DSC thermograms of chitosan (a), PVA (b), FTP (c) and CFM (d) and their nanofibers (e).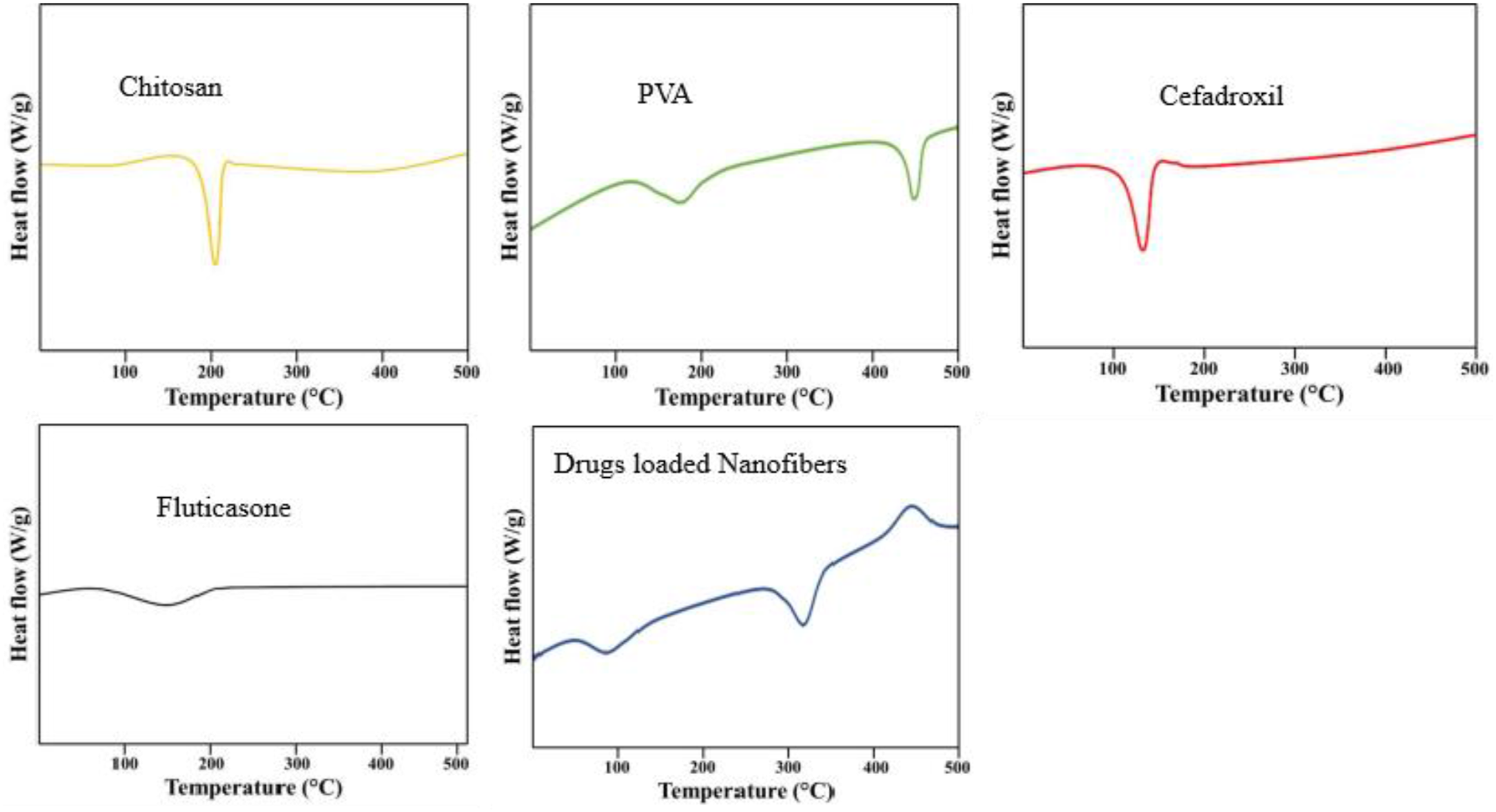
Particle size analysis
The size distribution of FCCPNFs measured by the zeta sizer is displayed in Figure 5. The plot exhibits a bimodal distribution, with two distinct peaks: The first peak was centered around 50–100 nm in size representing small polymeric fragments or nanodispersed drug molecules. The second peak, observed around 250–282 nm, corresponds to the larger nanofiber aggregates rather than drug or polymer debris. The presence of this second peak suggests partial agglomeration of nanofibers in suspension, a common phenomenon in electrospun fiber systems. Similar observations have been reported in previous studies
34
where electrospun nanofibers, when dispersed in aqueous media, tend to form small aggregated clusters due to hydrophilic interactions between polymer chains. Size distribution and zeta potential of the FCCPNFs measured by the zeta sizer.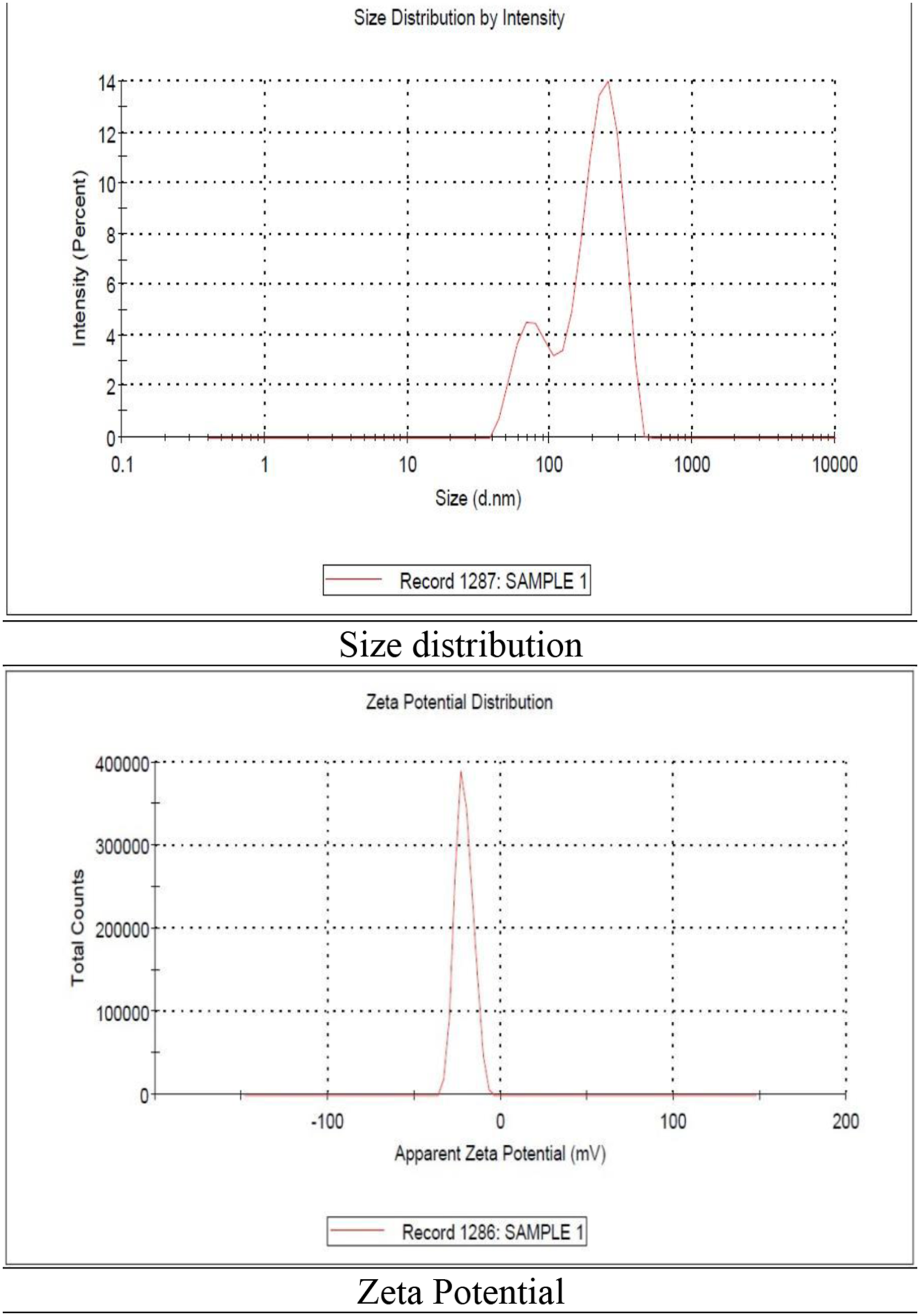
The surface charge of the drug-loaded nanofibers was −21.2 ± 5.37 mV (mV), as depicted in Figure 5. A negative value indicated the affluent blending of PVA and chitosan sufficient to prevent aggregation and long-term stability. Ibrahim et al. found zeta potential of −15.9 ± 4.37 mV of nanocomposite film of PVA/SA/TiO2. 34
Pharmaceutical research
Swelling ratio
The swelling ratio of FCCPNFs was depicted in PBS as shown in Figure 6. Chitosan /PVA nanofibers showed maximum solvent absorption owing to a large number of hydroxyl groups.
35
In the first 20 min F4, F5 and F6 exhibited pronounced solvent absorption and was significantly different p < 0.05. After half an hour the capacity to absorb solvent decreases gradually due to the less availability of hydroxyl groups. Barkhordari et al. observed the increased swelling ratio of Zn/Al-LDH nanocomposite films due to the gel forming capacity of carboxy methyl cellulose.
36
Swelling ratio of the FCCPNFs.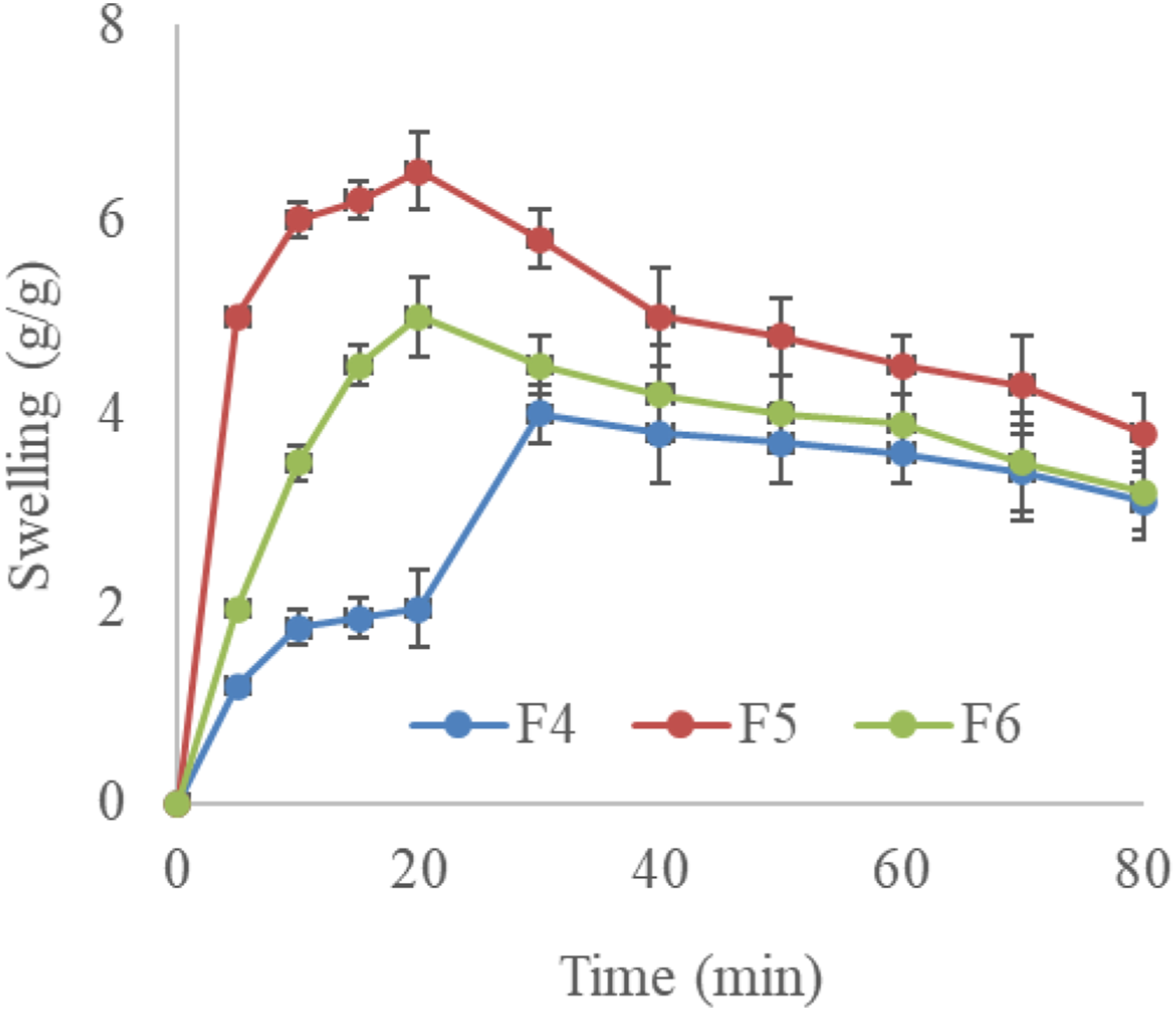
In vitro drug release study
The drug release study of FCCPNFs was found in dissolution analysis in PBS at pH 7.4. Khan et al. formulate CPC/NFs that showed 88.7% release of drug at pH 6.5 (wound pH) and 82% at physiological pH of the skin.
16
In our study F5 formulation showed quick release of drugs owing to their solubility in PBS. The release pattern of drugs in F4, F5 and F6 is depicted in Figure 7. Iqbal et al. showed a non-Fickian drug release pattern in CPNFs fused with CFX.
37
In vitro release of drugs from FCCPNFs.
Antibacterial evaluation
Zone of Inhibition of formulations against S. aureus.

Wound healing and histopathological analysis
The wound healing study was performed to check the percentage area of wounds over time. The wound healing process over 21 days is shown in Figures 8–9. The percentage of wound closure after treatment with a commercial antibacterial cream (Quench), CFM and FTP-loaded NFs, and without any treatment was observed at 0, 7, 14, and 21 days. The wound healing rate was 55%, 75%, and 88% and complete closure after 20 days with FCCPNFs owing to efficient activity against S. aureus. The readings were statistically significantly different (p < 0.05). The in-vivo compatibility of CS/PVA and CS/PVA/AB3 was observed in a mouse model by Alizadeh et al. with no obvious inflammatory response and rejection.
9
AgNPs were modified with COS by Loo et al. and the resulting formulation PVA/COS-AgNP exhibited the fastest wound recovery.
15
Shah et al. formulated CS-SER-AgNP that exhibited better angiogenesis and increase collagen formation than Bactigras as a positive control.
10
An OLYMPUS-DP 72 digital camera is employed to capture histopathological photographs, providing insights into the different stages of the wound healing process. Figure 9 represented the histopathological study of the control, standard, and drug-loaded nanofiber (NF). The drug loaded nanofibers exhibited efficient epithelial growth without scar formation. Tissue growth and fast collagen formation were observed previously against the control group with necrotic cells.
16
Wound closure comparison of CPCCNF with Quench and the control. Macroscopic view of the wound healing study and the histopathological investigations of healed tissues.

The comprehensive characterization of FCCPNFs using SEM, XRD, thermal analysis, and FTIR confirmed the successful synthesis and stability of the nanofibers. SEM imaging revealed a uniform, interconnected fibrous network, ensuring efficient drug loading and controlled release. XRD analysis demonstrated the semi-crystalline nature of the nanofibers, 40 indicating the presence of both amorphous and crystalline phases, which play a crucial role in drug dispersion and mechanical strength. Thermal analysis (DSC) further validated the stability of the nanofibers, confirming that the incorporation of Fluticasone and Cefadroxil did not compromise the thermal integrity of the polymeric matrix. FTIR spectra provided insights into molecular interactions, confirming the compatibility of chitosan, PVA, and the loaded drugs without significant chemical alterations. The combined data from these characterizations support the structural robustness, thermal stability, and effective drug encapsulation within FCCPNFs, making them a viable candidate for wound healing applications.
Conclusion
This study successfully developed and characterized Fluticasone/Cefadroxil-loaded chitosan/PVA nanofibers (FCCPNFs) using the electrospinning technique for enhanced wound healing. The fabricated nanofibers exhibited a well-structured, interconnected fibrous network with uniform drug distribution and thermal stability. The in vitro evaluations demonstrated a high swelling ratio, burst drug release within the first 30 min, and significant antibacterial activity against Staphylococcus aureus. In vivo studies confirmed the effectiveness of FCCPNFs in accelerating wound closure, achieving complete healing within 20 days, as further supported by histopathological analysis showing rapid epithelial regeneration and collagen deposition. These findings highlight the potential of FCCPNFs as a promising wound dressing material, offering antibacterial protection, controlled drug release, and biocompatibility. Future research should explore large-scale production, clinical trials, and potential applications in treating chronic and antibiotic-resistant infections.
Footnotes
Acknowledgement
We acknowledge Khadija Amjad for improving the language of this manuscript.
Author contribution
Conceptualization: Saman Rashid, Muhammad Khurram Waqas, Ayesha Tahir, Nihal Abdalla Ibrahim, Bushra Nasir, Abida Kalsoom Khan, Munaza Ijaz.
Data curation: Saman Rashid, Ayesha Tahir, Abida Kalsoom Khan, Munaza Ijaz.
Formal analysis: Saman Rashid, Muhammad Khurram Waqas, Ayesha Tahir, Munaza Ijaz.
Funding acquisition: Munaza Ijaz
Investigation: Saman Rashid, Ayesha Tahir, Abida Kalsoom Khan, Munaza Ijaz.
Methodology: Saman Rashid, Ayesha Tahir, Abida Kalsoom Khan, Munaza Ijaz.
Project administration: Munaza Ijaz.
Resources: Munaza Ijaz.
A: Munaza Ijaz.
Supervision: Muhammad Khurram Waqas, Bushra Nasir, Munaza Ijaz.
Validation: Munaza Ijaz.
Visualization: Munaza Ijaz.
Writing – original draft: Saman Rashid, Muhammad Khurram Waqas, Ayesha Tahir, Khadija Amjad, Bushra Nasir, Abida Kalsoom Khan, Munaza Ijaz.
Writing – review & editing: Saman Rashid, Muhammad Khurram Waqas, Ayesha Tahir, Nihal Abdalla Ibrahim, Bushra Nasir, Abida Kalsoom Khan, Munaza Ijaz.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article this work was supported by the we extend our appreciation to the Deanship of Graduate Studies at Ajman University, Ajman, United Arab Emirates, for their support for publication charges.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All the data has been incorporated into this article.
